The maternal environment of European beech (Fagus sylvatica L.) affects intrapopulation variability in seed traits and germination
iForest - Biogeosciences and Forestry, Volume 19, Issue 2, Pages 114-121 (2026)
doi: https://doi.org/10.3832/ifor4806-018
Published: Apr 10, 2026 - Copyright © 2026 SISEF
Research Articles
Abstract
Interpopulation variation was investigated using seed samples originating from twenty-six European beech (Fagus sylvatica L.) populations across the Balkan Peninsula, a part of the species’ distribution range characterized by high ecological heterogeneity in key climatic factors, such as temperature (5.8-10.6 °C), precipitation (648-1632 mm), and elevation (185-1410 m a.s.l.). The statistical significance of intrapopulation differences was confirmed by analysis of variance (ANOVA) for all seed traits analyzed: seed weight (g), length (mm), width (mm), thickness (mm), eccentricity and flatness indices, and germination capacity (%). Multivariate principal component analysis (PCA) was applied to examine seed traits in relation to environmental variables of the maternal site, such as mean temperature and precipitation in September and October (the seed maturation period), revealing distinct patterns of relationships among the variables studied. Seed traits were significantly positively correlated with mean temperatures of the maternal site in September and October, indicating that temperature during the seed-filling period affects seed mass. Germination capacity was associated with precipitation during the same period, though the correlation coefficient was not statistically significant; a shorter vector length in the PC biplot suggests a weaker contribution to population separation. Elevation of the site of origin showed a significant negative correlation with temperature, precipitation, and seed traits. Agglomerative hierarchical clustering analysis identified three distinct population clusters. Higher temperature and precipitation values did not necessarily result in higher seed trait values or higher germination percentages. The population with the highest seed mass exhibited the lowest germination capacity (32%) during seed maturation under the lowest precipitation. Conversely, the population characterized by the lowest seed mass showed a higher germination rate of 68% in environments with high precipitation. These results provide valuable insights into the reproductive ecology of European beech, suggesting that other factors beyond those analyzed here may have a more substantial influence on seed germination. The variation in seed traits across habitats that are either drier and hotter or colder and wetter, along the elevation gradient of the studied populations, paves the way for future research and breeding efforts to enhance the species’ survival and reproductive success amid anticipated climate change scenarios.
Keywords
Seed Traits, Seed Germination, European Beech, Fagus sylvatica L., Environmental and Genetic Variation, Southeast Europe
Introduction
One of the primary goals of ecological research is to understand the influence of primary climatic factors on species distribution and their specific functional adaptations that facilitate survival. Studies conducted along geographic latitude-elevation gradients with pronounced ecological heterogeneity in temperature and precipitation can help predict vegetation response to future climate scenarios ([26]). Climatic factors - especially precipitation and temperature - play a crucial role in plant propagation and adaptive capacity ([4]). Rapid climate changes harm forest ecosystems, as evidenced by quantitative and qualitative indicators of global forest growing stock ([1]). Provenance tests conducted across various locations have shown that rising temperatures and reduced precipitation negatively impact the growth and health of species in forest ecosystems. The adaptations of forest ecosystems to climate change are determined by the genetic resources of species ([11]). The adaptability of plant species allows them to thrive and spread across diverse habitats with varying environmental factors ([31]).
Environmental variations driven by climate change may influence critical life stages of plant species, including flowering, seed production, and germination, which affect the species’ life cycle. Variation in seed traits, at both spatial and temporal scales, maintains polymorphism in life-history strategies within populations. In this context, the environmental fitness will increase due to selection ([13]).
Knowledge of seed reproductive traits is crucial for the development and implementation of effective species conservation and management plans ([44]). Seed germination capacity and morphology represent significant adaptive traits of a species ([6]). Seed morphology and dry mass are considered key traits for assessing population adaptive potential ([27]). The timing of germination is determined by the interaction between heterogeneous environmental conditions and genetic correlations with other seed characteristics ([13]). Seedlings from seeds with higher dry mass are characterized by faster growth and a higher survival rate ([33]). Also, the final weight and size of seeds in many woody species are strongly influenced by precipitation ([12]) and mean temperatures during the period of filling ([30]). Air temperature and precipitation during the ripening of beech seeds (September and October) affect dormancy and the percentage of germination capacity ([4]).
Beech (Fagus sylvatica L.) is one of the most important tree species in European forests. It is sensitive to spring frosts and extended drought periods ([36]), making it highly vulnerable to predicted climatic scenarios ([34]). Projections based on IPCC scenarios indicate that areas macroclimatically suitable for beech will decrease dramatically in the coming decades ([8]). It is anticipated that beech will face drastic reductions in its range and even local extinction in more xerothermic habitats and at lower elevations (Mazza et al. 2024); therefore, new, resilient sources of seed should be identified ([42]). However, it must be noted that statistical models of rather pessimistic scenarios use climate data without accounting for the biological properties of forest tree populations, such as their adaptability, longevity, and regeneration ([8]). The endangerment of beech populations, especially in the continental and southern parts of the current range, is closely related to the degree of genetic variability of these populations ([8]). Populations in the southern part of the distribution range are likely to suffer the most in the near future due to the impacts of climate change ([26]).
Seed traits such as mass, size, and shape, as well as germination capacity, may represent adaptive responses to specific environmental conditions (e.g., dry or moist habitats). For instance, seeds originating from drier regions may exhibit greater mass or thicker seed coats as a mechanism of protection against desiccation, thereby improving germination success when moisture becomes available ([3]).
It is hypothesized that the observed variation in seed morphological traits and germination capacity among European beech populations across the Balkan Peninsula is significantly influenced by environmental factors, particularly temperature, precipitation, and elevation, during the seed maturation period. Specifically, populations from warmer and wetter environments are expected to exhibit larger seed size, while germination capacity may show a more complex relationship with precipitation and other ecological factors.
The primary objectives of this study were to: (i) assess the extent of interpopulation variation in seed morphological traits and germination capacity among European beech populations across the ecologically diverse habitats of the Balkan Peninsula; (ii) investigate the relationships between seed traits and key environmental variables of the maternal site of origin - namely temperature, precipitation, and elevation - during the seed maturation period (September and October); (iii) determine the influence of ecological gradients, particularly climate and elevation, on the reproductive characteristics of European beech seeds; (iv) identify potential population clusters based on similarities in seed traits and environmental conditions of the site of origin using multivariate and cluster analyses.
Material and method
Study habitats and species
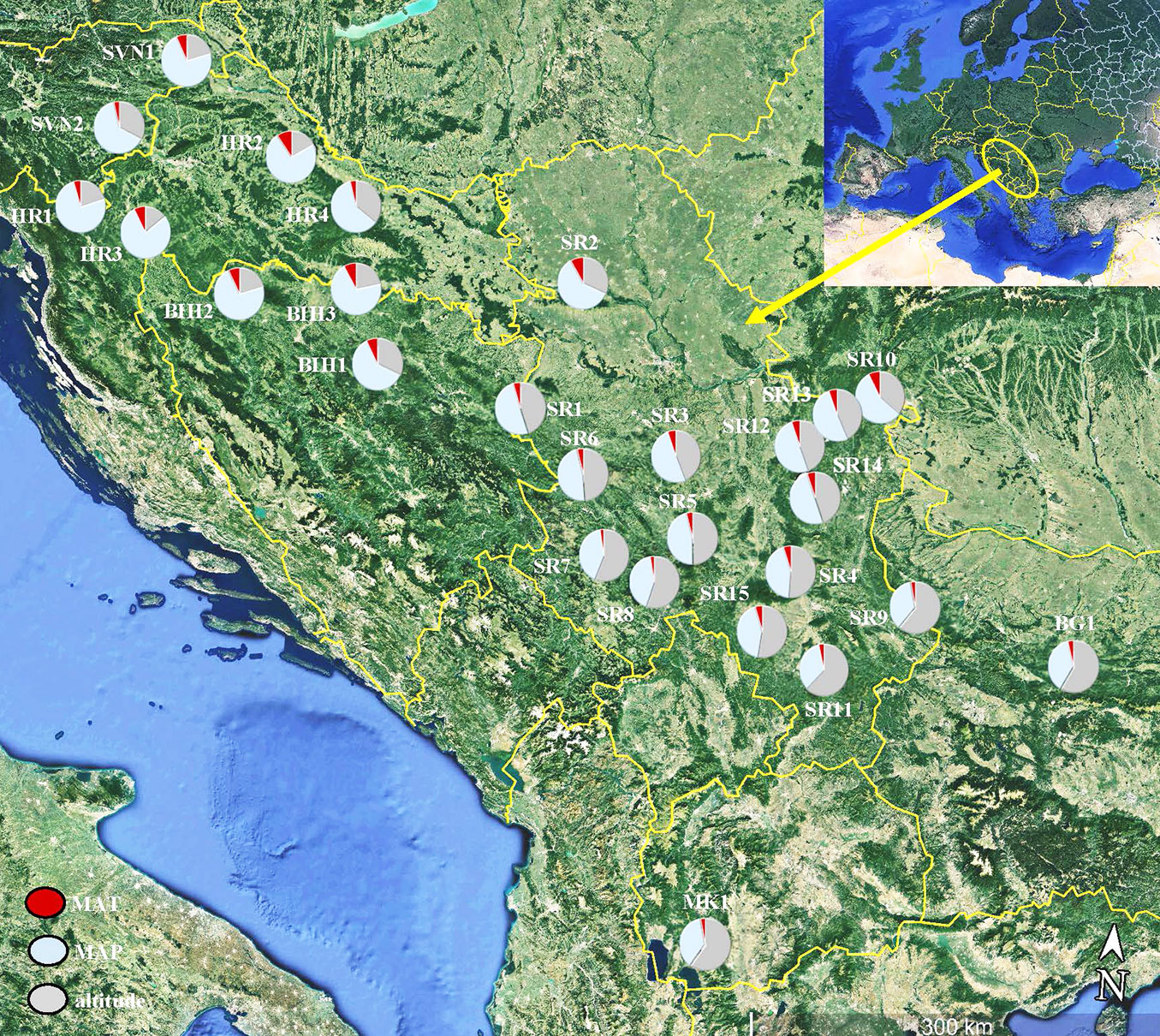
The research was conducted on seed samples from 26 beech populations across the Balkan Peninsula (Fig. 1, see also Tab. S1 in Supplementary material). Basic geographic information on populations and climatic factors was obtained using the Climate EU v. 4.63 software package, available at ⇒ http://tinyurl.com/ClimateEU ([18]).
Fig. 1 - Map of twenty-six populations of European beech (Fagus sylvatica L.) sampled in the Balkan Peninsula. The pie charts represent the three environmental characteristics at the sampled maternal locations: elevation (in grey), mean annual temperature (MAT - in red) amd mean annual precipitation (MAP - light blue).
Trait analyses
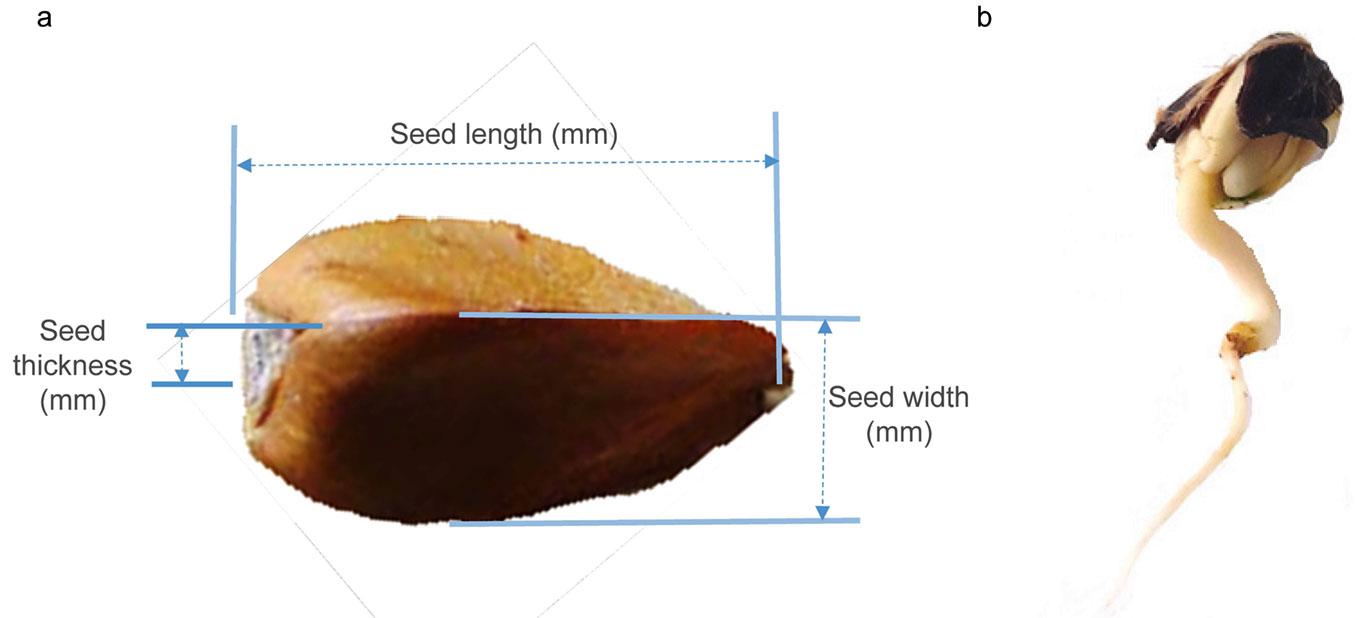
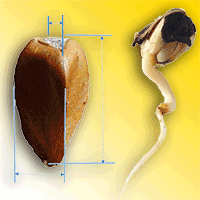
In autumn 2018, approximately 1 kg of visually healthy seeds was collected from each population. Seed material was collected from at least 30 trees per population, spaced more than 50 meters apart to minimize the likelihood of sampling genetically related individuals. The collected material was then processed in the Institute of Forestry’s laboratory in Belgrade. The seeds were mechanically cleaned of impurities, and the empty seeds were removed with an air current. Cleaned seeds were disinfected with 35% H2O2 solution for 2 minutes. Morphological properties of seeds and germination capacity were analyzed (Fig. 2a, Fig. 2b). Random samples of 50 nuts were taken from each population for morphometric analysis. The length, width, and thickness of the seeds (in mm, Fig. 2a) were determined using a digital caliper with a precision of 0.01 mm. Thereafter, the seeds were dried at 130 °C for 1 hour, and the dried seed weight (in g) was measured using an electronic scale with an accuracy of 0.01 g.
Fig. 2 - Seeds of European beech (Fagus sylvatica L.) with indication of the measured traits (a). (b) A seed after germination (radicle length > 3 mm).
Seed shape indices were calculated based on the measured values of morphological traits: (i) eccentricity index (EI), which represents a ratio of the larger and smaller axes of a seed (i.e., seed length and width): EI = length/width; (ii) Flatness index (FI) based on the relationship of three basic axes of a seed: seed length, width, and thickness; (iii) FI = [(length + width)/2] × thickness - a range of values around 1 indicates a spherical seed, while flat seeds have values closer to 2.
Average germination capacity (GERM) was determined at the population level, using a sample of 4 × 50 seeds per population. The germination capacity was assessed according to the ISTA protocol ([20]). The seeds were placed in 15 cm diameter petri dishes, each containing two filter papers for germination. The seeds producing a radicle at least 3 mm long were recorded as germinated.
Statistical analyses
All statistical analyses were performed using the SAS/STAT statistical package ([38]). Mean values for all analyzed beech seed traits were obtained using the MEANS procedure. Estimates of the statistical significance of populations as sources of phenotypic variation in traits conditioned by environmental differences were obtained using analysis of variance (ANOVA) with the PROC GLM procedure in SAS. Analysis of variance was applied to the seed traits, including length, width, thickness, mass, seed shape, flatness, and eccentricity indices. Scheffé’s post hoc test was used to determine specific differences in population means across all analyzed traits.
Multivariate Principal Components Analysis (PCA) was applied to investigate patterns of variance in the data (Pearson-type analysis). Biplots were used to visually analyze the relationships between seed traits (weight, in g; length, in mm; width, in mm; thickness, in mm; and germination, in %), elevation, and environmental variables such as the mean values of MAT and MAP during September and October, the seed maturation months (MATSO and MAPSO). The arrow length in the biplot indicates the variable’s loading to the principal components, while its direction shows the correlation with the component and other variables. Arrows pointing in the same direction indicate a positive correlation between the variables, while those pointing in opposite directions indicate a negative correlation.
Agglomerative hierarchical clustering was performed to assess similarity among the analyzed variables. Pearson’s correlation coefficient was used as the similarity measure, while the Unweighted Pair-Group Method with Arithmetic Mean (UPGMA) served as the agglomeration method. This approach enabled the identification of structural relationships and grouping patterns, presented as a dendrogram. Cluster analysis grouped populations based on seed traits, elevation, and environmental characteristics, the mean values of mean temperature and mean precipitation in September and October (MATSO and MAPSO, respectively) based on the correlation coefficients and the pair-group average method. The two multivariate analyses and graphic representations were performed using XLSTAT, an add-in software package for Microsoft Excel®.
Results
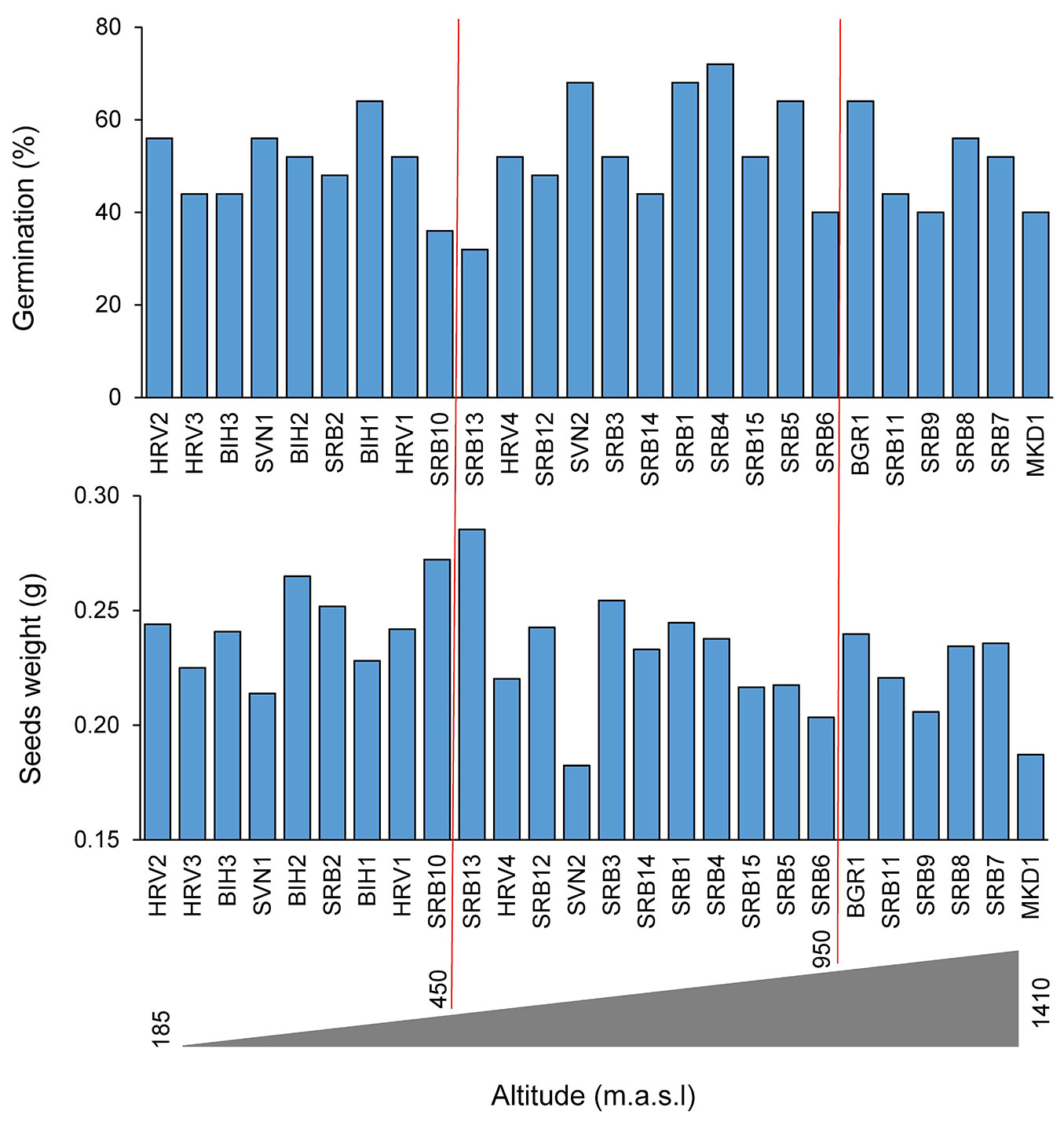
The average seed length (mm) ranged from 15.0 mm, recorded for the population SVN2 (located on the lowest elevation of 285 m a.s.l.), to 18.5 mm, which has been recorded for population SRB13 (1080 m a.s.l.). Additionally, the same population had the lowest seed width value (SVN2, 9.21 mm), while the highest values were recorded in populations SRB13 (11.09 mm) and SRB12 (11.14 mm). The population SRB12 was located at the highest elevation (1420 m). The seed thickness was also the largest for SRB13 (8.32 mm) and the smallest for population MKD1 (6.93 mm), which is located at a lower elevation (435 m a.s.l.). The pattern of seed mass values was the same as for length and width. The smallest seeds from the population SVN2 had the smallest mass (0.18 g), while the largest mass was recorded for population SRB13 (0.29 g - Fig. S1 in Supplementary material). Eccentricity and flatness indices, which describe the seed shape, were closer to 2, ranging between 1.61 and 1.76, compared to 1.68 and 1.94, respectively. The obtained values indicate that the sampled seeds were eccentric (EI) based on the relation between seed length and width, and more flattened (FI) than spherical (Fig. S1).
The percentage of germination was the lowest for population BIH2 (32%) and the highest for population HRV1 (72%). The values of environmental factors were similar (elevation: 650 vs. 825 m a.s.l.; MAT: 8.3 vs. 8 °C; MAP: 764 vs. 704 mm, respectively) (Fig. 3, Fig. 4; see also Tab. S1 in Supplementary material). According to the results of Scheffe test for seed length, there were seven significantly different groups for population SRB13 (mean seed length: 18.49 mm) and SVN2 (15.00 mm), for seed width ten groups (SRB12 11.14 mm - SVN2 9.21 mm), for seed thickness nine groups (SRB13 8.32 mm - MKD1 6.93 mm), for seed mass 19 groups (SRB13 0.28 g - SVN2 0.18 g), and EI three groups (SRB15 1.94 - SVN2 1.68).
Fig. 3 - Mean weight (g) and germination (%) of seeds from twenty-six sampled populations of European beech (Fagus sylvatica L.) along the elevation range from 185 to 1410 m a.s.l. in the Balkan Peninsula.
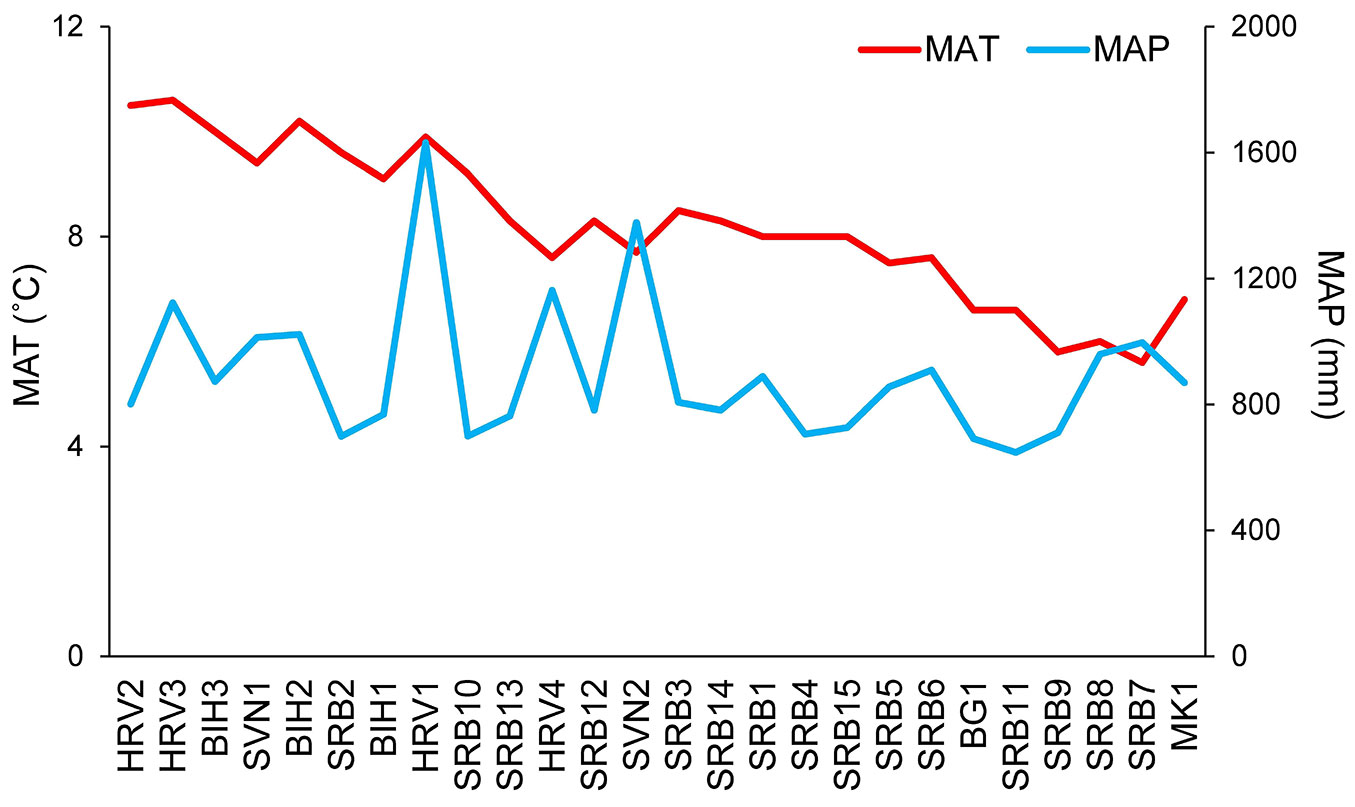
Fig. 4 - The mean annual temperature MAT (°C - red line) and mean annual sum of precipitation MAP (mm - blue line) for twenty-six sampled populations of European beech (Fagus sylvatica L.).
The statistical significance of differences between populations was obtained for all the analyzed traits (p < 0.001) based on the application of one-way ANOVA (Tab. 1). The relationship of germination capacity and seed mass did not show any significant correlation (Pearson’s r = 0.088, p = 0.6680) for any analyzed population.
Tab. 1 - Results of one-way ANOVA with population as a source of variation for the analyzed characteristics of seeds (length, width, thickness, weight, eccentricity, and flatness indices) in the twenty-six sampled populations of European beech (Fagus sylvatica L.). (***): p < 0.001.
| Variable | Parameter | Population | Error |
|---|---|---|---|
| - | df | 25 | 1274 |
| Seed length | MS | 36.58 | 1.56 |
| F | 23.41*** | - | |
| Seed width | MS | 13.63 | 0.91 |
| F | 14.95*** | - | |
| Seed thickness | MS | 6.8 | 0.63 |
| F | 10.83*** | - | |
| Seed weight | MS | 0.03 | 0 |
| F | 12.56*** | - | |
| Eccentricity index | MS | 0.08 | 0.03 |
| F | 3.04*** | - | |
| Flatness index | MS | 0.14 | 0.03 |
| F | 4.79*** | - |
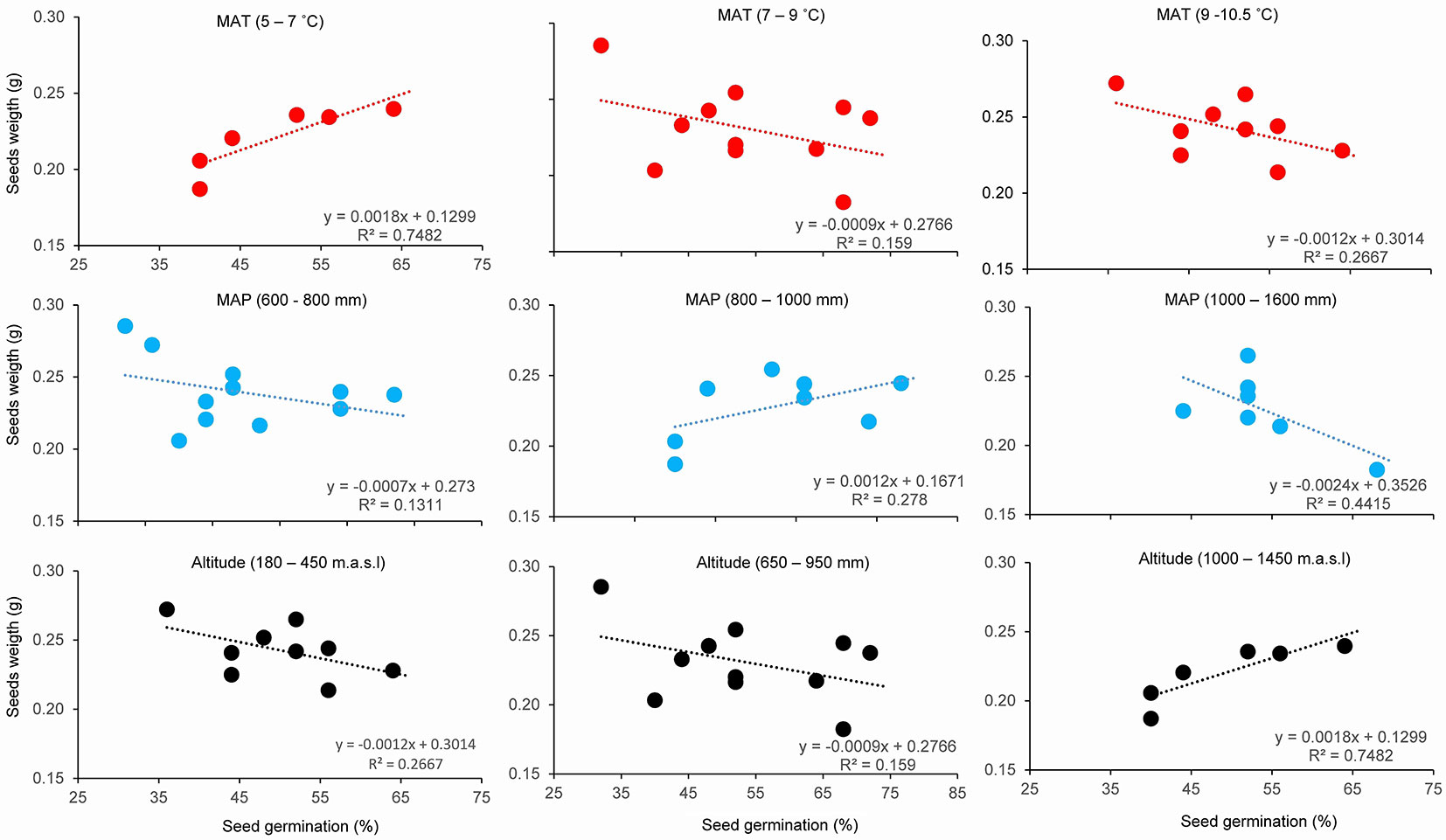
Pattern of correlations between seed mass and percentage of germination changed with increasing temperature, precipitation, and elevation of population sites (Fig. 5). At temperatures below 7 °C, heavier seeds had higher germination percentage compared to higher temperatures, where the correlation of these two seed traits was reversed. Heavier seeds had higher germination percentages in the precipitation range of 800-1000 mm and at elevations from 1000 to 1450 m a.s.l. (Fig. 5).
Fig. 5 - Relationship between seed weight (g) and seed germination (%), in three ranges (columns) of mean annual temperature (MAT, °C - red dots and lines), mean annual sum of precipitation (MAP, mm - blue dots and lines), and elevation (m above sea level - black dots and lines).
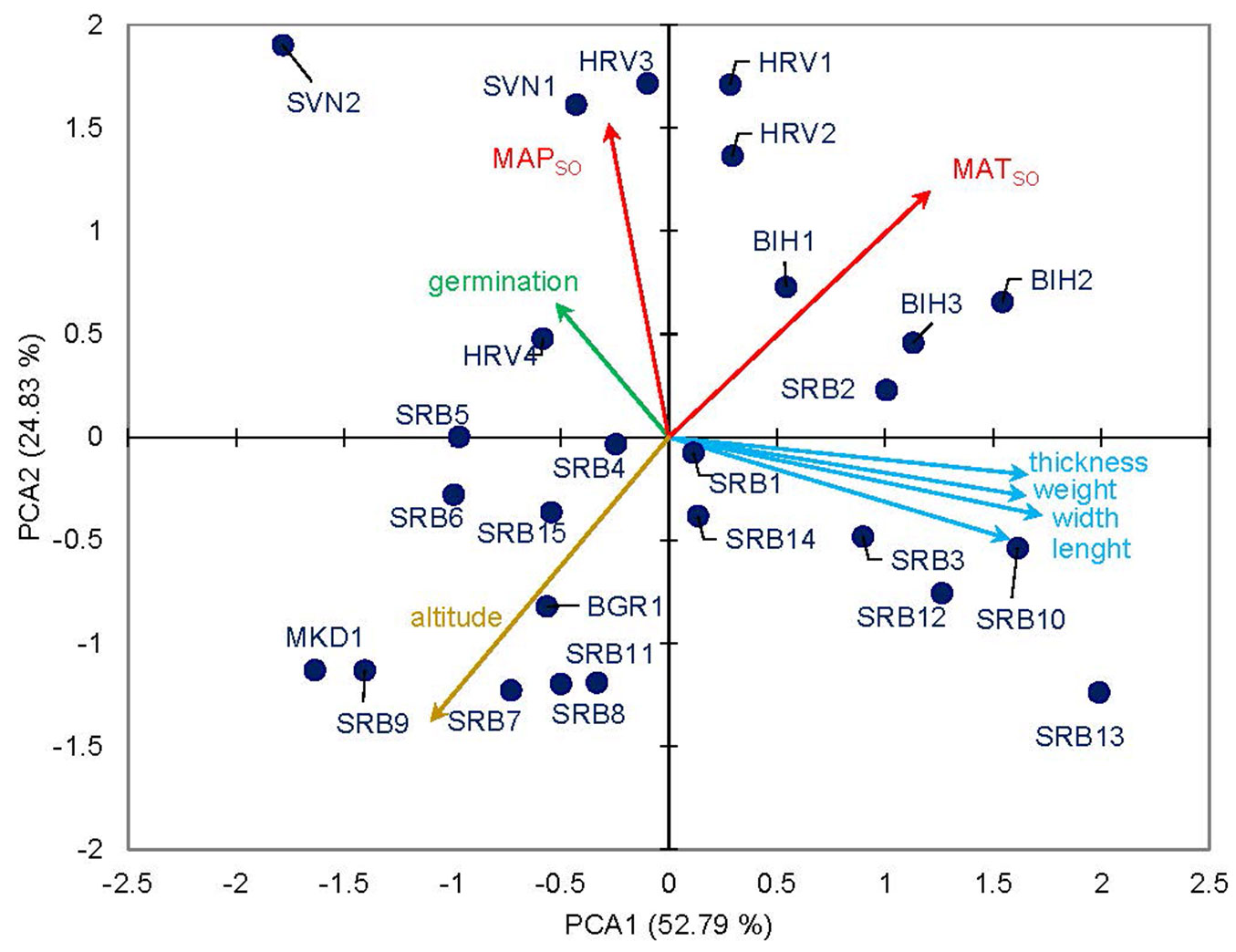
The results of the multivariate principal component analysis, based on eigenvectors and the biplot of seed traits (germination capacity and seed dimensions) in relation to environmental factors across all populations, revealed patterns among the examined variables. The first two principal components accounted for 77.62% of the total variation among populations, with the first component explaining 52.79% and the second 24.83% (Fig. 6). Along the PC1 axis, all populations from Serbia (except SRB2), as well as those from North Macedonia and Bulgaria, were distinctly separated from the others. This separation is primarily due to their lower mean annual temperature and precipitation in September and October (MATSO and MAPSO), as well as their higher elevations (Tab. S1 in Supplementary material). Environmental variables, specifically elevation and MATSO, exhibit a negative correlation, as indicated by the PCA-derived correlation matrix. All analyzed seed traits and MATSO are positively correlated, suggesting that temperature during the seed maturation period significantly influences seed development.
Fig. 6 - Biplot of the first two components (PC1 and PC2) from Principal Component Analyses (PCA) for the twenty-six sampled populations of European beech (Fagus sylvatica L.). The diagram illustrates the relationships among seed traits (weight, length, width, thickness, and germination) and environmental characteristics of the original population (MATSO, MAPSO: mean values of temperature and precipitation for September and October, respectively) and elevation.
Germination capacity appears to be influenced by MAPSO; however, the correlation coefficient was not statistically significant. This is reflected in the shorter vector length in the biplot (Fig. 6), indicating a weaker contribution to population differentiation. Elevation was negatively correlated with both seed traits and germination capacity, as well as with other environmental variables.
According to the second principal component, MAPSO contributes most significantly to the differentiation among populations (Fig. 6).
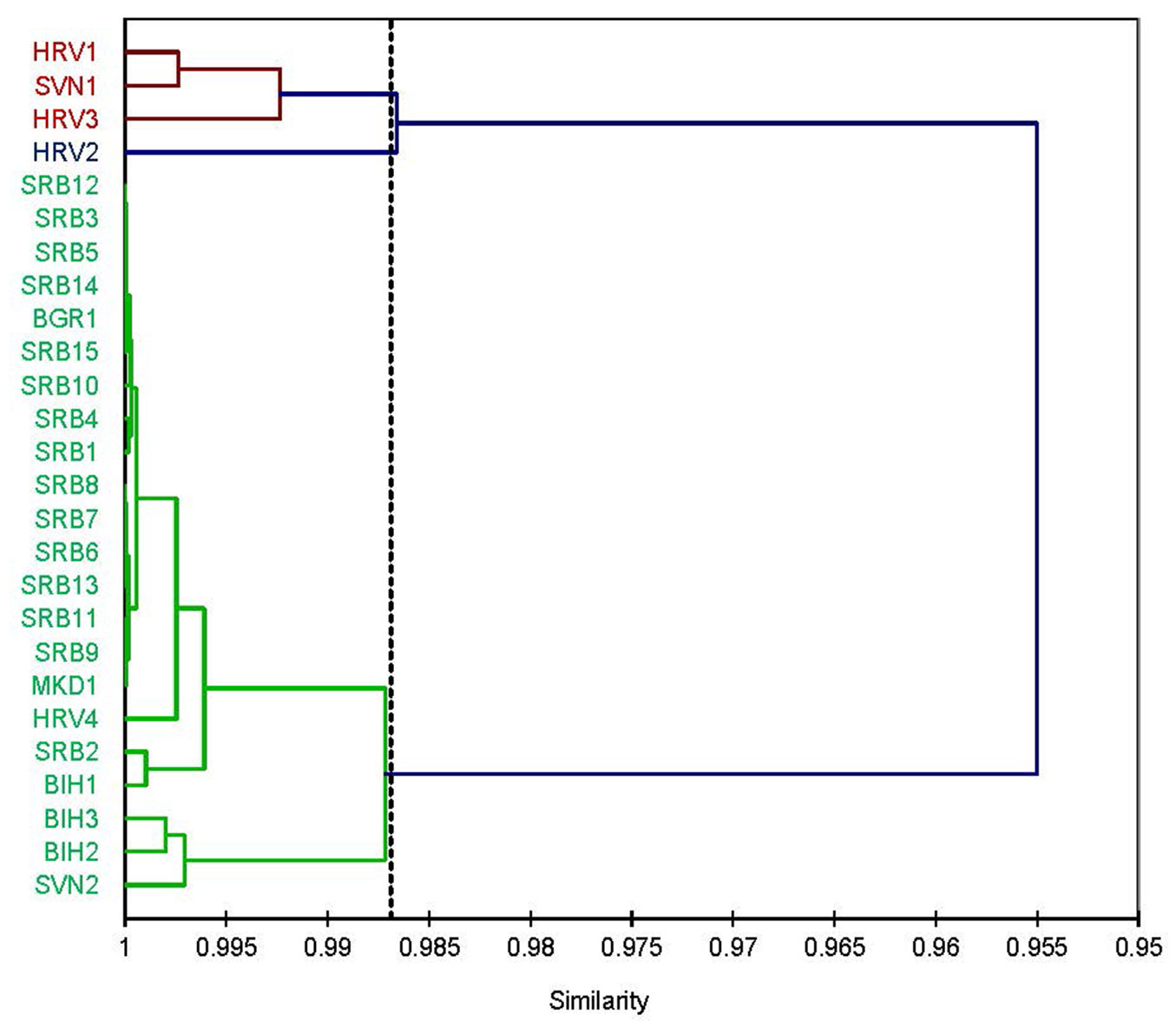
Based on germination percentage, seed traits (weight, width, length, and thickness), and environmental factors (MATSO, MAPSO, and elevation), agglomerative hierarchical clustering identified three distinct clusters. The variance for optimal population classification was 22.31% between clusters and 77.69% within clusters.
The first cluster comprised three populations: HRV1, SVN1, and HRV3. The second cluster included a single population, HRV2. The remaining 22 populations formed the third cluster (Fig. 7). This clustering was influenced primarily by differences in environmental factors, as population HRV2 was located at the lowest elevation (185 m a.s.l.) and exhibited the highest temperatures (MAT: 10.5 °C; MATSO: 16.45 °C), as well as slightly above-average precipitation values during the seed maturation period (MAPSO). Seed traits were close to average, except for germination percentage, which was somewhat higher than the mean (56% vs. 51.53%, respectively).
Fig. 7 - Hierarchical clustering dendrogram based on Pearson’s correlation and Unweighted Pair-Group Method with Arithmetic Mean linkage, illustrating the relationships among the sampled populations of European beech (Fagus sylvatica L.) in the Balkan Peninsula, according the seed traits (seed weight, length, width, thickness) and environmental characteristics of the original population (MATSO, MAPSO: mean values of temperature and precipitation for September and October, respectively) and elevation.
The second cluster, comprising populations HRV1, SVN1, and HRV3, was located at lower elevations (with HRV3 at the lowest among the three) and characterized by higher precipitation (MAP and MAPSO) and higher MAT and MATSO values. Germination percentage was above the average for SVN1 (56% vs. 51.53%, respectively), and slightly above average for the other two populations. Other seed traits were similar to the population mean (Fig. 7, Tab. S1).
Discussion
In changing environmental conditions, as a consequence of various stressors, populations of woody species face challenges to their survival ([17]). Studies of inter- and intrapopulation (genetic) variability estimated the response of forest ecosystem species to different environmental stressors ([1]). Over the last few decades, climate change has led to water shortages and a steady increase in temperature, making the preservation of genetic diversity a necessary condition for adaptation to altered environmental conditions ([37]). A decrease in genetic variability across generations is directly linked to forest decline and reduced population size ([23]). The risk of population decline due to drought is higher along the southern (marginal) borders than in the center of the species distribution ([21]). The studied beech populations are situated at the edge of the species’ southern distribution area. They comprise regions in the eastern part of the Alps (Slovenia), the Dinaric region (Croatia, Bosnia and Herzegovina, and part of Serbia), the Rhodope Mountains (Bulgaria), and the Carpathian Mountains ([45]).
Knowledge of the morphological and biological properties of beech seeds from different sites enables improved restocking and sustainability of beech stands ([16]). Studies on the variability of seed morphometry can also provide preliminary insights into the genetic variability of the studied populations and contribute to improving the production of high-quality reproductive material in beech ([35]) under new climatic conditions. In this study, the observed differences in seed morphology and germination capacity among populations suggest the differentiation of adaptive traits in the studied area. Seed morphology differed significantly between the studied provenances. The largest and heaviest seeds were measured in the population of Mali Pek, part of the Carpathian-Balkan Mountains, while in the population of Gorjanci (the Alpine part of the Dinarides), the seeds were the smallest and lightest. Morphometric research by Yilmaz ([47]) on 14 different provenances of the Oriental beech (Fagus orientalis Lipsky) revealed significant variability within and between populations. Studies on seed length and width of Croatian beech provenances showed that the genetic variability within populations is minimal, while the analysis of variance determined that the populations are statistically different from each other ([14]). Also, morphological trait analyses of beech seeds from eight Serbian provenances ([35]) and from northeastern Greece ([44]) showed high interpopulation variability.
Elevation is an ecological factor that affects multiple environmental variables, including temperatures, length of the growing season, and the intensity of UV radiation. At higher elevations, seed morphology is often characterized by more compact shapes with lower flatness index (FI) values, which may provide enhanced protection for the embryo under stressful conditions ([5]). In contrast, in lowland areas, flatter seed shapes may enhance wind dispersal efficiency and soil contact, both of which are important for successful germination ([32]). Our study revealed statistically significant differences in seed morphological traits among populations, indicating the influence of environmental gradients related to elevation.
Beech can withstand harsh cold in winter, but it is vulnerable to spring frost and extended summer droughts ([36]). This sensitivity to drought makes it highly vulnerable to predicted climate change, emphasizing the need to study reproductive ecology ([34]). Reproductive ecology traits related to seeds, such as germination time or seed size, have high significance, and understanding patterns of diversity in these traits is crucial for formulating successful conservation and management plans ([44]). Reproductive traits, including seed morphology and germination capacity, are key factors in species’ adaptive potential ([6]). The dry weight of seeds and size are considered closely related adaptive traits at the population level ([27]). Tree populations producing seeds with higher dry weight yield seedlings with greater strength, larger growth, and better survival ([33]). Climate factors, particularly precipitation and temperature, play a crucial role in plant reproductive adaptation ([4]). The amount of precipitation during seed filling affects seed mass and size ([12]). Other studies have also found a positive correlation between seed size and the annual precipitation amount ([39]). In addition, mean monthly temperatures during the growth period can affect reproductive traits ([9]). Temperature, as an environmental pressure factor, determines the depth of hibernation, the timing and uniformity of germination, and thereby affects the adaptability of populations ([25]). Differences in temperature between provenances affect seed size ([40]).
In this study, we determined that average beech seeds length and width was 16.75 and 10.17 mm respectively, seed mass amounted to 0.24 g, germination capacity was the smallest for the population BIH2 (32%) with low elevation, high temperature, and precipitation, and the largest for the population HRV1 (72 %) with the most significant amount of precipitation during the whole year and during the seed growing period.
Our findings are very similar to those reported in studies conducted in the Balkan Peninsula area. In Croatian provenances, the average seed length and width were smaller, at 14.89 mm and 9.53 mm, respectively. The absolute mass was 258 g, and the germination capacity was 12% ([16]). In the territory of Velebit, seed length/width was 15.42/8.14 mm, mass 0.24 g, absolute mass 118.9 g, germination percentage was 37% ([10]); Croatian provenances had 15.90/8.43mm ([14]), seeds of eight Serbian provenances were somewhat larger (17.15/9.7 mm), their mass was 0.24 g, absolute mass 253 g and 78% germination capacity percentage ([35]). In the territory of northern Greece, the seed length and width were 16.74 and 9.61 mm, respectively ([44]). According to Smelkova ([41]), the weight of 1000 seeds, i.e., the absolute mass of beech seeds, was 234 g, and 70% germination capacity.
Beech seed production is irregular with significant variations between years ([19]). Seed masting (crop years) is strongly influenced by annual climate conditions and occurs irregularly at intervals ranging from three to fifteen years. Understanding fructification mechanisms is crucial for improving knowledge of stand conditions, assessing the current and future adaptability of forest ecosystems, and developing management strategies in the context of climate change ([46]). Fructification positively influences plant reproductive success by stimulating regeneration, which in turn determines species survival and plays a key role in sustainable forest management ([24]).
The relationship between crop years and climate has been studied and modeled by various authors, and it has been proven that crop years are caused by specific climate conditions in previous years ([43]). Climatic factors directly influence the growth and reproduction phases, as well as flowering and pollination, and indirectly through the utilization of available resources ([2]). The correlations between diameter increment and production of beech seeds in relation to climatic conditions support the hypothesis of the distribution of assimilates.
The climate characteristics in the years preceding fruiting are crucial for the reproductive ecology of beech. A hot and dry summer in the year preceding fruiting has a positive influence on seed crop occurrence ([15]). A summer drought one year before flowering can be associated with an increased C/N ratio, which in turn may be responsible for flower formation. A wet summer two years prior to the seed crop can be considered a secondary factor in crop occurrence, as nutrient reserves accumulate then ([22]). Low temperatures and increased water availability two years before seed masting have been shown to promote fructification. A significant positive relationship was observed between seed crop and spring temperature and light availability two years earlier ([29]). Since temperature, radiation, and drought are highly interconnected, the availability of solar radiation can positively influence the accumulation of reserve nutrients two years prior to seed production ([2]).
Selective pressures are a limiting factor, especially at low elevations with drier, hotter climatic conditions, which increase mortality. Across the distribution range of tree species, the climatic characteristics of elevation and latitude gradients that drive plant survival, as well as the prediction of vegetation responses to future climate change scenarios, are the primary goals of ecological studies ([26]). Tree distribution tends to retreat toward higher, colder areas. The success of germination has increased in hotter regions, but survival has decreased, which prevents beech from thriving in hot and dry areas ([28]). Our research revealed that larger seed mass leads to higher germination percentages both at lower mean annual temperatures and moderate precipitation at higher elevations. The obtained pattern of seed mass and germination capacity may be a hint of acclimatization of this species and its survival under drought and high-temperature conditions. Local adaptation enables better fitness components in populations originating from maternal environments to which they are adapted. The plasticity of early life phases and local adaptation, as well as germination rates and the successful regeneration of young trees, enable the expansion and preservation of the range in response to climate change, including rapid global warming ([28]). Adaptation strategies for germination and seedling growth to environmental conditions vary between populations and provenances, and the physiological mechanisms underlying these adaptations are highly complex.
When conducting a breeding program, it is essential to consider the diversity of ecotypes in relation to specific combinations of climatic factors. The selection of suitable beech provenances for translocation and afforestation in new areas is crucial ([44]). The maternal environment is a key source of phenotypic plasticity. To date, the variability of seeds, germination, and other adaptively important characteristics between maternal environments has been investigated in a limited number of studies. Variability in phenology, including fructification, germination, and seedling growth, across the species’ natural range, helps improve understanding of local adaptation during the most vulnerable stages in response to climate change ([7]).
Conclusions
The variability in seed morphological traits and quality is linked to the conditions of the original maternal environments. Our findings offer valuable insights into the reproductive ecology of European beech in Southeastern Europe, highlighting potential adaptations to naturally drier and hotter, or colder and wetter habitat conditions at the elevation of maternal populations. According to multivariate analysis, elevation and temperature during the seed-filling period (September and October) have the greatest impact on population grouping. Populations with annual temperatures below 7 °C, precipitation ranging from 800 to 1000 mm, and elevation above 1000 m a.s.l. showed a positive correlation between mass and germination percentage, indicating that heavier seeds had higher germination capacity. The variability in maternal environments and reproductive characteristics opens opportunities for further research and breeding programs aimed at enhancing the survival and reproduction of European beech under rapid climate change.
Acknowledgements
This study was supported by the Ministry of Education, Science, and Technological Development of Serbia, Contract numbers 451-03-66/2024-03/200027 and 451-03-66/20 24-03/200007. The work of Gregor Božič was funded by the Slovenian Research and Innovation Agency (P4-0107) and the national research project V4-2222. The results presented in this manuscript are in line with Sustainable Development Goal 15 (Protect, restore and promote sustainable use of terrestrial ecosystems, sustainably manage forests, combat desertification, and halt and reverse land degradation and halt biodiversity loss) of the United Nations 2030 Agenda.
Author’s Contribution
VP conceived the idea and designed the study; VP, MI, BC, GB, and VA conducted fieldwork; VP and AL were responsible for project management and research; DM and VP wrote the manuscript; DM performed statistical analyses, data curation, and visualization of results. All authors have read and agreed to the published version of the original manuscript.
Vladan Popović and Danijela Miljković are co-first authors of this work.
References
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Aleksandar Lučić 0000-0002-4473-1791
Department of Genetics, Plant Breeding, Seed and Nursery Production; Institute of Forestry, Kneza Višeslava 3, 11030 Belgrade (Serbia)
Department for Genetics, Forest Tree Breeding and Seed Science, Croatian Forest Research Institute, Cvjetno naselje 41, 10450 Jastrebarsko (Croatia)
Faculty of Forestry, University of Banja Luka, Bulevar vojvode Petra Bojovića 1A, 78000 Banja Luka, Republic of Srpska (Bosnia and Herzegovina)
Department of Forest Physiology and Genetics, Slovenian Forestry Institute, Večna pot 2, 1000 Ljubljana (Slovenia)
University Ss. Cyril and Methodius, Hans Em Faculty of Forest Sciences, Landscape Architecture and Environmental Engineering, 16 Makedonska brigada 1, 1000 Skopje (North Macedonia)
Department of Evolutionary Biology, University of Belgrade, Institute for Biological Research “Siniša Stanković”, National Institute of the Republic of Serbia, Bulevar despota Stefana 142, 11060 Belgrade (Serbia)
Corresponding author
Paper Info
Citation
Popović V, Lučić A, Ivanković M, Cvjetković B, Božič G, Andonovski V, Miljković D (2026). The maternal environment of European beech (Fagus sylvatica L.) affects intrapopulation variability in seed traits and germination. iForest 19: 114-121. - doi: 10.3832/ifor4806-018
Academic Editor
Rafael Da Silveira Bueno
Paper history
Received: Jan 26, 2025
Accepted: Apr 09, 2026
First online: Apr 10, 2026
Publication Date: Apr 30, 2026
Publication Time: 0.03 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2026
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 795
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 204
Abstract Page Views: 326
PDF Downloads: 226
Citation/Reference Downloads: 0
XML Downloads: 39
Web Metrics
Days since publication: 48
Overall contacts: 795
Avg. contacts per week: 115.94
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
(No citations were found up to date. Please come back later)
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Seed germination traits of Pinus heldreichii in two Greek populations and implications for conservation
vol. 15, pp. 331-338 (online: 24 August 2022)
Research Articles
Patterns of genetic diversity in European beech (Fagus sylvatica L.) at the eastern margins of its distribution range
vol. 10, pp. 916-922 (online: 10 December 2017)
Research Articles
Delineation of seed collection zones based on environmental and genetic characteristics for Quercus suber L. in Sardinia, Italy
vol. 11, pp. 651-659 (online: 04 October 2018)
Editorials
Workshop COST E52 “Evaluation of beech genetic resources for sustainable forestry”
vol. 2, pp. 104 (online: 10 June 2009)
Technical Reports
Conservation and use of elm genetic resources in France: results and perspectives
vol. 13, pp. 41-47 (online: 03 February 2020)
Review Papers
Genetic diversity and forest reproductive material - from seed source selection to planting
vol. 9, pp. 801-812 (online: 13 June 2016)
Research Articles
Seedling emergence capacity and morphological traits are under strong genetic control in the resin tree Pinus oocarpa
vol. 17, pp. 245-251 (online: 16 August 2024)
Research Articles
Patterns of genetic variation in bud flushing of Abies alba populations
vol. 11, pp. 284-290 (online: 13 April 2018)
Research Articles
Comparison of genetic parameters between optimal and marginal populations of oriental sweet gum on adaptive traits
vol. 11, pp. 510-516 (online: 18 July 2018)
Research Articles
Seed trait and rodent species determine seed dispersal and predation: evidences from semi-natural enclosures
vol. 8, pp. 207-213 (online: 28 August 2014)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword