Litter quality and decomposition rates of selected species in the Ankasa Conservation Area, Ghana
iForest - Biogeosciences and Forestry, Volume 19, Issue 2, Pages 122-129 (2026)
doi: https://doi.org/10.3832/ifor4671-018
Published: Apr 11, 2026 - Copyright © 2026 SISEF
Research Articles
Abstract
Decomposition is essential for nutrient cycling, soil fertility, and productivity in tropical forest ecosystems. However, there is limited information on these aspects for some tropical forest tree species. The present study compared the decomposition rates of leaf litter of three tree species (Baphia nitida, Dacryodes klaineana, and Gluema ivorensis) and the influence of litter quality across three habitat types in the Ankasa Conservation Area, Ghana. Using the litterbag technique, a total of 432 bags were prepared and placed on the forest floor across the three habitats for 360 days. Litter samples were removed monthly to determine the mass remaining. At the beginning of the study, the litter chemistry of each species was determined using standard spectrophotometric methods. Differences in litter constituents and decomposition rates in the three species and habitats were evaluated using analysis of variance. Pearson product-moment correlation was used to explore the relationships between litter mass loss and the initial litter quality. The results showed variations (p<0.05) in litter quality among the three studied species, with nitrogen (4.95%) and lignin (1.63%) contents being highest in B. nitida, whereas phosphorous (0.97%), hemicellulose (31.85%) and C:N ratio (15.98) were more abundant in G. ivorensis. D. klaineana recorded the highest proportion of cellulose (65.3) and potassium (2.53). Decomposition rates averaged between 1.99 g year-1 (in D. klaineana) to 2.35 g year-1 (in B. nitida), although no statistical differences (F[2]=0.252, p=0.0778) were found among the species. Habitat type and duration significantly influenced the decomposition rate (p > 0.05).
Keywords
Nutrient Cycling, Litter Chemistry, Ecosystem Function, Tropical Rainforest
Introduction
Litter decomposition is an important ecosystem process that contributes to forest productivity and regeneration dynamics ([19], [6]). Litter decomposition remains the principal conduit for energy conversion and maintenance of global cycles of nitrogen, phosphorus, and carbon ([3], [16]). It also plays a key role in improving nutrient availability, soil fertility, maintaining plant growth and development, accelerating soil organic matter formation, and increasing net carbon storage in forest land.
Multiple factors, at both spatial and temporal scales, influence litter decomposition ([6], [35]). At the landscape or regional level, litter decomposition is controlled largely by climatic factors (e.g., rainfall, temperature, humidity) and litter quality ([20], [8]). By increasing soil microbial activities ([62]) and fauna abundance ([2]), higher temperature and rainfall, for instance, promote litter decomposition and nutrient release ([11]). Conversely, at the local level, the initial litter content, composition, and density of decomposing biota, as well as the distance to neighbouring species, modulate litter decomposition ([41], [7]). Studies by Mori et al. ([38]) indicate that diverse species or mixtures decompose faster than litter from single species due to the availability of diverse food resources for a wide range of soil micro-organisms involved in decomposition. The soil physicochemical properties such as texture, bulk density, pH and nutrient status, are also documented to play a critical role in litter decomposition by enhancing litter porosity, permeability and activities of soil organisms ([35]) Other factors such as species diversity ([38]), topography ([59], [37], [14]), forest successional age ([53], [17]) and site disturbance ([44], [29]) also affect litter breakdown locally. In humid areas, litter quality traits such as N, C, C/N ratio, P, K, lignin, and cellulose best predict decomposition rate ([1], [48]). A low C/N ratio and high N content promote rapid litter decomposition, whereas a higher proportion of lignin and phenols slows the process ([56], [41], [51]). These variations in litter traits or qualities can alter litter decomposition rates across many ecosystems, including those with similar ecological conditions ([63]). Differences in plant physical and chemical characteristics, including leaf toughness, specific leaf area, leaf carbon content, leaf tannin and phenolic concentrations, and micronutrients (calcium, magnesium, and manganese), can influence the decomposition rate by influencing the activity of the soil microorganism community and habitat conditions ([35]). In the tropics, the rate of litter decomposition may also be sensitive to a wide range of habitat conditions, including climatic factors (rainfall and temperature), soil moisture, land-use changes, plant species abundance, and litter production ([9], [37]). These factors can alter the microenvironmental characteristics of the forest floor by improving soil conditions, soil biota populations, and decomposition processes ([47]). Hence, investigating the complex process of litter decomposition and the effects of litter quality is key to understanding litter decay rates, soil fertility, nutrient cycling, and productivity in the tropics.
The Ankasa Conservation Area (ACA) has received global attention, being one of the wildlife protected reserves in West Africa ([5]) and the only wet evergreen forest in Ghana ([32]). This reserve is also known for serving as habitat for a rich diversity of species and for supporting human wellbeing through the provision of essential ecosystem services ([43]). These contributions, along with those from other forests worldwide, are widely seen as avenues to achieve the Paris Climate Agreement and the Sustainable Development Goals (e.g., 3, 13, 15 - [61]). Like many tropical forests, the ACA exists on nutrient-poor soils ([32]), making litter input an important ecological process for maintaining soil fertility ([27]) and survival of other heterotrophic communities ([46]). While acknowledging the preponderance of literature on litter decomposition in the temperate climates and some tropical forest ecosystems ([60], [23], [64], [13]), there is a dearth of information on litter decomposition in the Ghanaian tropical rainforest ecosystem. In particular, no studies have been published on the decomposition rates of the investigated plant species in the reserve. In this study, a comparative analysis of leaf litter quality and decomposition rates for the three most important species in the ACA reserve ([43]) was conducted to determine how differences in litter quality and habitat influence decomposition rates across the different habitat types of the conservation area. Considering the complexity of this rainforest and the anticipated diverse litter inputs, data on decomposition rates of tree species that characterise this reserve can provide detailed information on nutrient cycle, energy flow, seedling regeneration, and productivity of tropical forests in the face of ongoing environmental change ([41], [7]). Given their taxonomic distinctiveness and potential variation in their physical and chemical characteristics, it was hypothesised that litter decomposition rates would vary with species identity. Secondly, the considerable variations in microenvironmental conditions, including soil physicochemical properties and topography, were expected to influence litter decomposition rate in the reserve.
Materials and methods
Description of study area
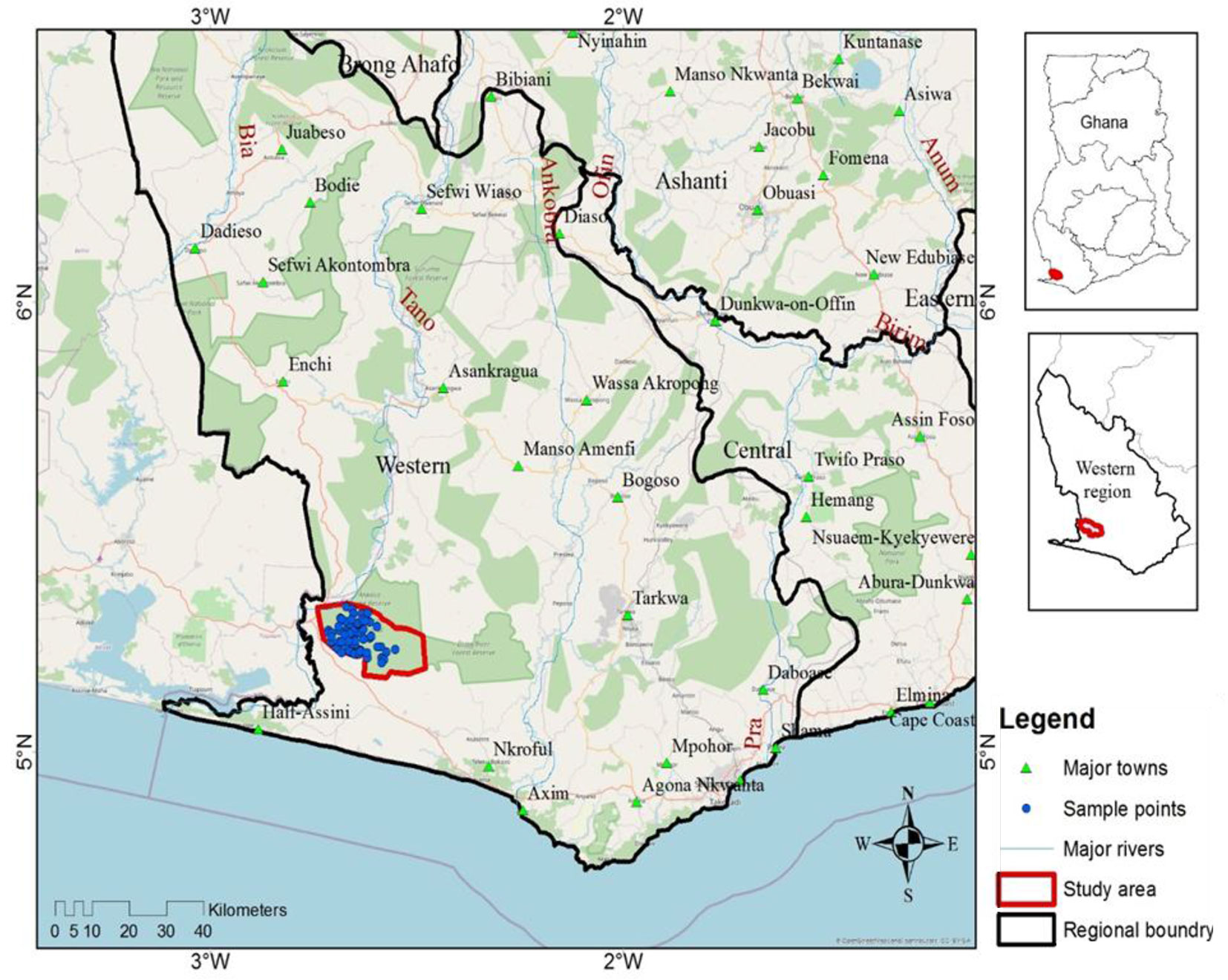
The ACA is located in the southwestern part of Ghana, near the border with Côte d’Ivoire. The Conservation Area lies between latitude 05°09′ and 05°25′ N, and longitude 02°29′ and 02°45′ W (Fig. 1). The ACA covers an area of 509 km2, comprising the Ankasa Resource Reserve (67%) in the south and the Nini-Suhien National Park (33%) in the north ([32]). As in many tropical forests, the ACA has a history of anthropogenic disturbance. Between 1934 and 1975, the southern portion was used for selective logging or timber production ([36]). However, the Conservation Area was designated a protected area in 1976 (Wildlife Reserves Regulations, L.I. 1085) to conserve water and enhance suitable climatic conditions for agricultural purposes. Apparently, the Nini-Suhien National Park, located in the northern portion, has experienced little or no human interference and remains the largest remnant “virgin rainforest” of Ghana ([36]). Consequently, the ACA is considered by some authors as the most species-rich, with tree species richness exceeding 300 per hectare ([43]). Studies by Owusu et al. ([43]) classify ACA in three distinct forest communities or habitat types: Bucket Tray - Lophira Tree (BT-LT), Bamboo Cathedral - Big Tree Area (BC-BGA), and Dadwen - Elubo Camp (DW-EC). These habitat types differ in their disturbance history, species composition, and topography. The landscape has a mean elevation of 90 m. a.s.l. but peaks (102.82 m), at DW-EC. This is followed by BC-BGA (89.43 m), and BT-LT (77.76 m). Tree height averages 27.96, 31.58, and 28.53 m for BT-LT, BC-BGA, and DW-EC, respectively. Mean tree occupancy is in the descending order of BT-LT (321 m2 ha-1) > BC-BGA (271 m2 ha-1) > DW-EC (202 m2 ha-1). Tree species richness and diversity, respectively, are highest (18 and 2.51) at DW-EC and lowest (14 and 1.19) at BC-BGA ([43]). Historically, the three habitats had been exposed to varied intensities of anthropogenic disturbance. Both BT-LT and BC-BGA host the Ankasa River and a network of streams. The BT-LT was exposed to minimal anthropogenic disturbance in the past due to its proximity to the Ankasa management camp. The vegetation of BT-LT is dominated by Gluema ivorensis, Pentadesma butyracea, and Corynthes pachyceras.
Fig. 1 - Map of the Western Region of Ghana with the location of the Ankasa Forest Reserve (marked in red) and the distribution of the sampling plots (blue dots).
The BC-BGA, located in the central portion of the reserve, was the most disturbed habitat, as it has been used for logging, human habitation, and agricultural activities ([43]). The most important tree species of this habitat include Picralima nitida, Funtumia elastica, Dacryodes klaineana, and Tabernaemontana africana. The DW-EC is at the reserve periphery, and it is surrounded by a number of subsistence farms. Dominant species of this habitat include Strombosia pustulata, Strombosia glaucescens, and Dacryodes klaineana. The reserve is also known for its large populations of large wild mammals, including the forest elephant (Loxodonta cyclotis), bongo (Tragelaphus eurycerus), and leopard (Panthera pardus), as well as many iconic wild birds. However, due to decades of continuous poaching for bushmeat, the populations of several mammals, reptiles, and birds are declining.
Annual rainfall in the reserve is bimodal, ranging from 1700 mm to 2000 mm. The major rainy season occurs between May and July, and the minor one between September and October. Mean monthly temperature varies from 24 to 28 °C ([32]). Although the soil is highly acidic (pH 3.5-4.0) and is largely classified as an oxisol ([54]), physicochemical characteristics vary by habitat type. For instance, recent soil analysis revealed that BT-LT has the highest TN but the lowest conductivity and available P among the habitats. On the other hand, pH, C, and available P were dominant at DW-EC.
Litter collection
Leaf litter of the selected species was collected in early January 2023 from the three habitat types, namely BC-BGA, BT-LT, and DW-EC, as previously determined by Owusu et al. ([43]). Freshly fallen leaves of B. nitida, B. klaineana, and G. ivorensis, being the dominant species, were collected ([43]). The leaf litter was used in the study because it is the most abundant (~70%), easily decomposable, and the major source of nutrients and energy in tropical forests ([62]). Two sets of litter were collected simultaneously for the study: one for analysis of initial litter quality and the other for investigation of mass loss and decomposition rates. A total of 36 leaf litter samples (i.e., 3 species × 4 replicates × 3 habitats) were collected for the analysis of the initial litter content. For the litter decomposition studies, 432 samples (i.e., 4 replicates of species per plot × 3 species × 3 habitats × 12 months) were obtained for the litter mass loss experiment across the reserve. The former was dried in the open air (at ambient temperature) and subsequently oven-dried at 65 °C for 48 hours. The latter was placed on the forest floor.
Determination of the litter constituents
Litter was analysed to determine the chemical composition and quality. Total Nitrogen (TN) content was determined using the Micro-Kjeldahl method, which involves oxidation with sulphuric acid and subsequent dissolution in hydrogen peroxide. The C content was analysed using the dry ashing method, in which 1 g of a powdered litter sample was ignited at 550 °C for 6 hours in a muffle furnace. The ash-free mass was computed to be 50% of the total mass. K content was determined by flame photometry after digesting the plant samples with tri-acid (H2SO4-HClO4-HNO3) mixture. The litter material was also analysed for acid detergent fibre (ADF) fractions; lignin and cellulose contents were obtained by stepwise washing of the fibre with neutral detergent ([58]). For cellulose, a known mass (1 g) of each litter was placed in a beaker containing 100 ml of acid-detergent solution, then 2 ml of decahydronaphthalene was added, and the mixture was boiled for 60 minutes. The contents of the beaker were filtered, then washed with hot water and acetone. The residual acid detergent fibre (ADF) was subsequently dried at 100 °C and weighed. The weight loss corresponded to the cellulose content of the litter. The lignin content was obtained following hydrolysis of the litter sample with 72% H2SO4. The residue (i.e., lignin content) was filtered, dried, and then weighed.
Litterbag preparation and decomposition experiment
Decomposition of the three studied leaf litter (B. nitida, D. klaneana, and G. ivorensis) was carried out using the litterbag technique in fenced plots. Each bag, measuring 20 × 20 cm, with a mesh size of 2 × 2 mm, was filled with 10 g of recently fallen leaves. This relatively smaller mesh size was chosen to prevent vertebrate interference while enhancing microbial action on the litter. Four replicate plots were established in each habitat for the study. These plots were separated by at least 100 m to account for spatial variation within the habitats. Of the 432 litter samples, three samples, each representing the species under investigation (B. nitida, D. klaneana, G. ivorensis), were randomly removed from each of the four plots across the three habitats monthly (i.e., 4 replicates, 3 species, 3 habitats) for a year. Upon removal, the bags were carefully cleaned to remove soil and other foreign materials. The litter was dried in the open air and then oven-dried at 75 °C for 24 hours prior to determining the mass loss. Mass loss was computed as the difference between the initial mass (10 g) and the oven-dried weight of the litter (expressed in percentages). The decomposition constant or decay rate coefficient (k) that estimated the disappearance of leaf litter on an annual basis was calculated using the negative exponential decay model of Olson ([42]) - eqn. 1):
where X0 is the original amount of litter, Xt is the amount of litter remaining at time t, and k is the decomposition constant.
Statistical analysis
Differences in initial litter chemistry and decomposition rates among the three studied habitats were evaluated using two-way analysis of variance (ANOVA). Prior to this, the data were subjected to the Shapiro-Wilk normality test, which indicated that they were normally distributed. Tukey’s honestly significant difference (HSD) post-hoc test was used to test for differences in initial litter content and decomposition rate among habitat types. Pearson’s product-moment correlation was used to explore the relationships between litter mass loss during the year and the initial concentrations or ratios of chemical constituents. Graphical presentation and analysis of the data were done using GraphPad Prism™ v. 9.5. All statistical analyses were performed using a significance level of 0.05.
Results
Initial litter chemistry
The study revealed marked variations in the chemical composition of leaf litter for the three studied species (Tab. 1). TN was highest in B. nitida (4.95%) and least in G. ivorensis (3.28%). On the other hand, G. ivorensis recorded the highest phosphorus content (0.97%) in its leaf litter compared to B. nitida and D. klaineana (0.51% and 0.28%, respectively). A similar pattern was observed for hemicellulose, which ranged from 7.00% to 31.85% among the species. D. klaineana recorded more potassium (2.53%) than G. ivorensis and B. nitida, which had corresponding values of 1.25% and 0.82%. Unlike other litter parameters, there was no statistical difference in the C content among the studied litter, which ranged from 50.74% in B. nitida to 52.29% in G. ivorensis. The analysis further revealed variations in the acid detergent fibre content of the litter. Higher lignin content was observed in B. nitida (1.63%) and G. ivorensis (1.53%) than in D. klaineana (0.93%). The cellulose content was 65%, 64%, and 43.74%, respectively, for D. klaineana, B. nitida, and G. ivorensis.
Tab. 1 - Initial litter quality parameters (mean percent ± SE) of the three studied plant species in the Ankasa Conservation Area, Ghana (n = 36). (TN): total nitrogen.
| Parameter | B. nitida | D. klaineana | G. ivorensis | p-value |
|---|---|---|---|---|
| TN | 4.95 ± 0.02a | 4.34 ± 0.80a | 3.28 ± 0.07b | 0.0103 |
| P | 0.51 ± 0.04a | 0.28 ± 0.2b | 0.97 ± 0.04c | <0.0001 |
| K | 0.82 ± 0.06a | 2.53 ± 0.03b | 1.25 ± 0.02c | <0.0001 |
| C | 50.74 ± 1.42a | 51.43 ± 0.67a | 52.29 ± 0.13a | 0.1659 |
| C/ N ratio | 10.26 ± 0.33a | 12.09 ± 2.06b | 15.98 ± 0.40c | 0.0028 |
| Lignin | 1.63 ± 0.01a | 0.93 ± 0.01b | 1.56 ± 0.05c | <0.0001 |
| Hemicellulose | 16.47 ± 0.54a | 7.00 ± 0.41b | 31.85 ± 1.41c | 0.0416 |
| Cellulose | 63.70 ± 0.42a | 65.30 ± 0.11b | 43.74 ± 0.40c | <0.0001 |
Litter mass loss and decomposition rate
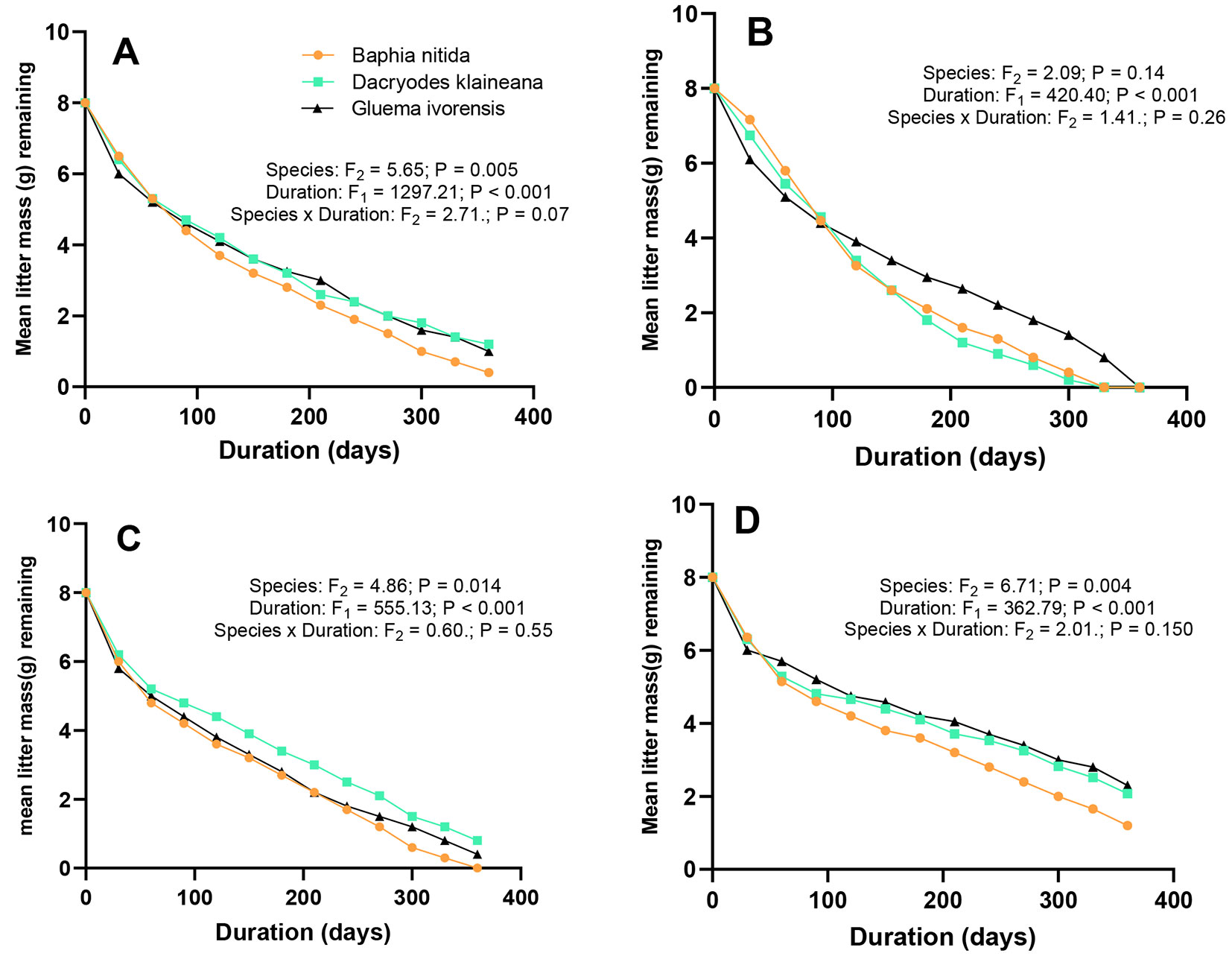
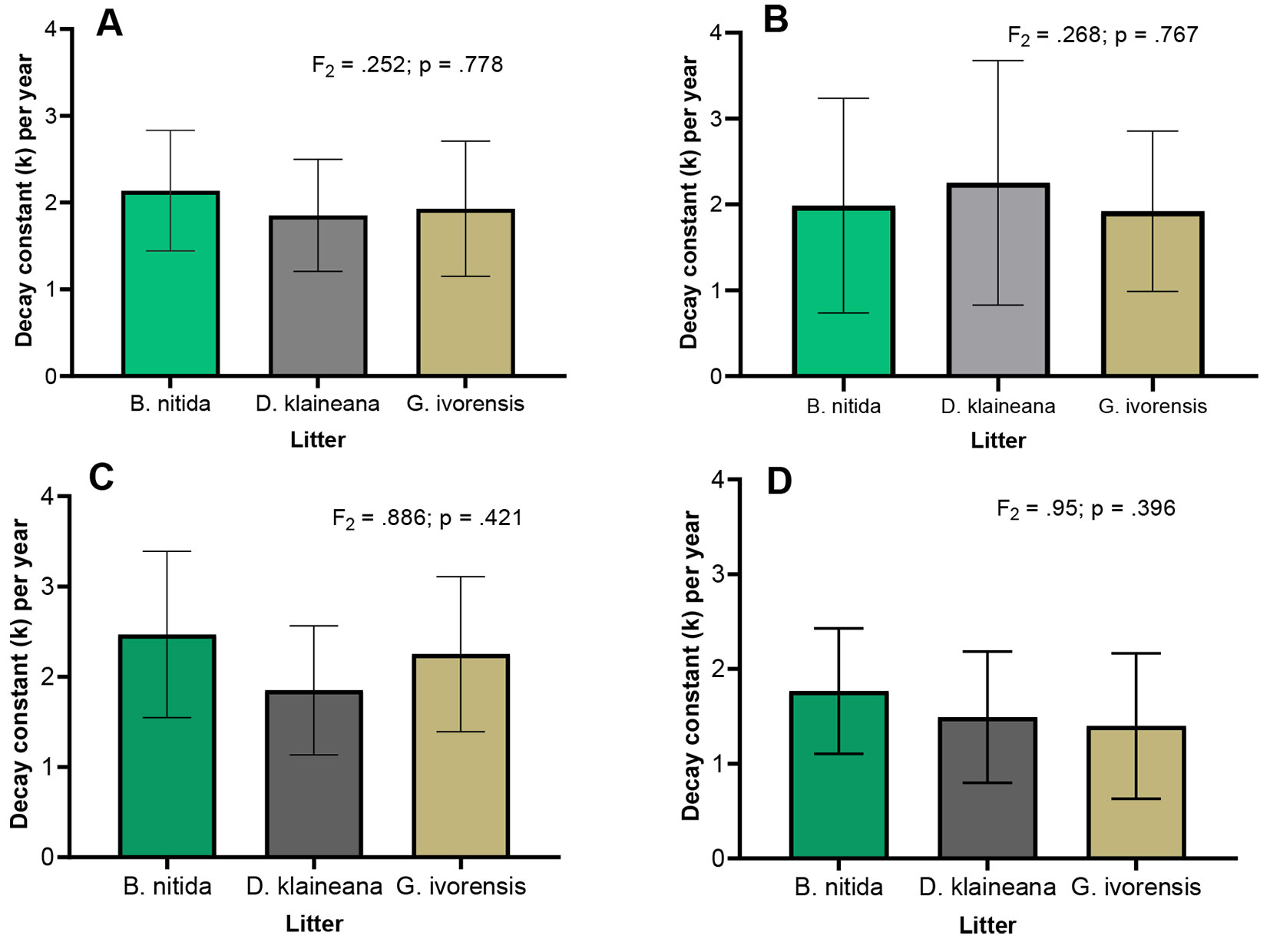
Mass loss was faster in the initial stages (up to 3 months) compared to the latter stages. For instance, B. nitida, G.ivorensis, and D. klaineana respectively lost 56%, 54%, and 53% within the first 90 days (3 months) of the experiment, versus 11%, 8%, and 1% in the later stages (between 300-360 days or 10-12 months). Beyond this period, the decomposition continued, but at a reduced rate, until the end of the experiment. Results of the ANOVA revealed that the mass remaining only varied with habitat type. There was no significant interaction between the habitat and species (Fig. 2). The post-hoc analysis indicated that only DW-EC and BC-BGA differed in their mass remaining or loss. The decay constant k differed by habitat type, with the DW-EC generally recording the highest k (2.05 g yr-1) for the species and the lowest (1.68 g yr-1) at a BC-BGA. There was a wide variation in k of DW-EC and BT-LT, but not BC-BGA. At the habitat level, B. nitida recorded significantly higher k (i.e., lower mass remaining) within both the DW-EC and BT-LT compared with G. ivorensis and D. klaineana. No statistical differences were found between the litter of the two species or among the species at BC-BGA. The decay constant k varied only with species type when the data from the three habitats were pooled (Fig. 3).
Fig. 2 - Remaining mass (%) of the three studied species in the Ankasa Conservation Area. (A) combined sites; (B) BC-BGA (Bamboo Cathedral - Big Tree Area); (C) BT-LT (Bucket Trail - Lophira Tree); (D) DW-EC (Dadwen - Elubo Camp). Mass remaining varied significantly with habitat type (F[2] = 43.35, p < 0.001), and displayed strong interactive effects of habitat and duration (F[2] = 17.69, p < 0.001) as well as habitat and species (F[2] = 3.158, p = 0.017) when data were pooled for all species and habitats. No interactions were found between species and duration (F[2] = 2.71, p = 0.07).
Fig. 3 - Decomposition rate constants for the studied species in the Ankasa Conservation Area: (A) combined sites; (B) BC-BGA (Bamboo Cathedral - Big Tree Area); (C) BT-LT (Bucket Trail - Lophira Tree); (D) DW-EC (Dadwen - Elubo Camp). k varied significantly with habitat type (F[2] = 4.366, p = 0.015), but no interactive effects was found between species and habitat (F[4] = 0.729; p = 0.574).
Relationship between initial litter chemistry and mass loss
We found a weak correlation between initial litter quality and mass loss in the reserve when the data were combined (Tab. 2). There was no significant relationship between the initial litter quality and the mass loss in the reserve. A similar trend was observed at the habitat level, except at BT-LT, where mass loss correlated positively with potassium (r = 0.26) and C (r = 0.28) but negatively with N (r = -0.23) and lignin (r = -0.24).
Tab. 2 - Pearson product-moment correlation coefficients (r) between initial litter chemistry and mass loss in the Ankasa Conservation Area. Analysis was performed by pooling data for all three species studied. BC-BGA (Bamboo Cathedral-Big Tree Area). BT-LT (Bucket Trail-Lophira Tree) and DW-EC (Dadwen-Elubo Camp).
| Nutrient | Combined Sites | BC-BGA | BT_LT | DW_EC | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| r | t-value | p-value | r | t-value | p-value | r | t-value | p-value | r | t-value | p-value | |
| TN | -0.08 | -1.45 | 0.15 | -0.05 | -0.54 | 0.59 | -0.23 | -2.42 | 0.02* | 0.02 | 0.23 | 0.82 |
| P | 0.07 | 1.35 | 0.18 | 0.08 | 0.82 | 0.41 | 0.05 | 0.49 | 0.62 | -0.02 | -0.16 | 0.87 |
| K | 0.05 | 0.97 | 0.33 | -0.08 | -0.80 | 0.42 | 0.26 | 2.75 | 0.01* | -0.02 | -0.21 | 0.83 |
| C | 0.07 | 1.27 | 0.21 | 0.09 | 0.95 | 0.35 | 0.28 | 2.95 | <0.01* | -0.07 | -0.77 | 0.45 |
| C:N ratio | -0.00 | -0.03 | 0.98 | 0.07 | 0.75 | 0.45 | 0.01 | 0.06 | 0.95 | -0.05 | -0.54 | 0.59 |
| Lignin | -0.06 | -1.07 | 0.28 | 0.06 | 0.64 | 0.53 | -0.24 | -2.55 | 0.01* | 0.02 | 0.19 | 0.85 |
| Hemicellulose | 0.03 | 0.52 | 0.60 | 0.16 | 1.64 | 0.11 | -0.04 | -0.40 | 0.69 | -0.04 | -0.49 | 0.63 |
| Cellulose | 0.07 | 1.29 | 0.20 | -0.08 | -0.87 | 0.39 | 0.07 | -0.69 | 0.49 | -0.05 | -0.48 | 0.63 |
Discussion
Variations in litter chemical composition
Litter chemistry is a key factor in determining decomposition rates and nutrient cycling in many ecosystems. In tropical forests with diverse species, litter chemistry is likely to vary considerably among plants, and this can exert significant effects on biogeochemical dynamics ([26], [12]). The current study recorded substantial differences in litter composition, suggesting a highly diverse litter input in the reserve. The higher content of TN, P, C, and cellulose suggests that the litter provides an adequate energy source for microbial activity during decomposition ([62]). N and P are key factors that regulate nutrient status, species diversity, and community structure in tropical forest ecosystems ([3]). Like other constituents, the proportions of N differed among the studied litter. The 5% TN recorded in B. nitida was comparable to the estimated %TN in decomposable organic matter for plant growth and development ([10]), confirming the link between the aboveground productivity and nutrient levels in forest soils. This value was higher than the average N content (1.9%) of trees in mature forests in Ghana ([39]) and elsewhere (e.g., Lama forest reserve - [31]). This implies that a higher abundance of B. nitida, for instance, can improve soil nutrient levels in the reserve. P is a limiting nutrient for plant growth in tropical forests, thus its content in the soil can have implications for nutrient availability, ecosystem productivity, and carbon balance in the tropics. The lack of variation in C content among the leaf litter of the three studied species, perhaps, reflects their common photosynthetic carbon assimilation. C/N ratio is a key predictor of biological processes, including photosynthesis and nitrogen fixation. The C/N ratio range recorded in this study (10-15) is consistent with the threshold (< 20) for quality litter. Despite its early loss or removal, potassium presence could be attributed to its high solubility and mobility in water ([4]). However, its distribution might not have influenced the decomposition process, as it was removed early ([6]). Perhaps this finding underscores the dominant role of the decomposer community (biological factor) during the early stages of decomposition ([28], [11]). Lignin is one of the important determinants of decomposition rates during the latter stages ([33]). The lignin content of the litter, ranging from 0.93% to 1.65%, was below the value of 15%-40% estimated by Esperschutz et al. ([25]). This relatively low content may increase the rate of decay in the reserve. Further, cellulose and hemicellulose are highly degradable carbohydrates that constitute 10%-50% and 20%-30% of litter, respectively ([34]). Thus, the relatively wide range of these carbohydrates (44%-65%) found in the studied litter may confer greater decomposition potential, as they are the primary targets of soil microorganisms ([62]).
Litter decomposition rates
Decomposition of litter in ecosystems plays an important role in sustaining the productivity and functionality of terrestrial ecosystems ([30]). Consistent with several studies ([3], [52]), a two-stage decomposition process, comprising an initial phase associated with rapid digestion and release of water-soluble compounds, and a relatively slower phase involving recalcitrant compounds such as lignin was observed during the current study. The decay constant k recorded in this study is in line with the reported range (0.032 to 3.734 g yr-1 - [63]) for litter decomposition rates in terrestrial ecosystems of West Africa, and with the mean rate (k = 1.87) from Indonesian natural forests during the wet season ([57]). On the contrary, this value was higher than 0.35 and 0.15 obtained from the secondary forests of Ghana ([21]) and a mixed deciduous forest of Northern Thailand ([49]), respectively. Songwe et al. ([55]) reported a relatively higher k (2.23) for selected trees from the Bakundu forest reserve. The relatively low decomposition rate may be related to high moisture or rainfall, resulting in leaf turgidity (toughness) prior to detachment and subsequent decomposition ([56]). Despite being in the same climatic zones, k varied across habitat types, emphasizing the significant control of local microsite conditions on litter decomposition ([6]). The higher k obtained from the DW-EC could be associated with higher species diversity or richness than in the other two habitats ([43]). On the other hand, given the intensity of the previous disturbance and its low nitrogen and organic matter contents, the lower k at BC-BGA was expected. Other factors likely contributing to the lower k at the BC-BGA, might include the slow pace of species recovery relative to the other habitats and high soil moisture. The lack of variation in k among the species suggests that multiple factors, rather than litter chemistry, control k in wet tropical forests, such as Ankasa. The significant variations in k within DW-EC and BT-LT reflect differential responses or interactions of habitats with the selected litter. Apart from the differences in topographical features and species abundance ([43]), changes in soil properties and composition of soil fauna can play a crucial role in determining the rate of decomposition at the habitat level ([45], [40]). Among others, soil fauna can influence litter decomposition by enhancing water infiltration, substrate availability, and affinity for microorganisms, soil nutrient addition via their excreta and dead tissue, as well as releasing soil enzymes to aid diverse energy provision for the decomposers ([22], [51], [15]). Nonetheless, variation in k among species when the species data were combined could be attributable to aggregate effects of the litter in the reserve.
The effects of litter chemistry on litter mass loss
Litter chemistry exhibited a weak relationship with mass loss in the reserve. This observation is plausible, given the complexity of the interactions between environmental factors (e.g., soils, topography) and species or their traits in most tropical rainforests ([18]). For a complex forest like ACA, mass loss may be correlated not only with initial litter quality but also with other local factors. The absence of significant correlation within the two habitats (DW-EC and BC-BGA) could be attributed to differences in habitat conditions. N and P are the most common limiting nutrients for forest productivity, despite their roles in plant cellular structures, nutrient cycling, and other biological processes ([24]). In the present study, mass loss correlated negatively with nitrogen content at BT-LT. This is not surprising, given the higher nitrogen content of the soil. The positive correlation between k and mass loss reflects the solubility and nutrients mobilisation within the habitat. Similarly, the negative correlation with lignin content implies that higher lignin content slows k ([33]).
Conclusion
The study investigated how variations in litter quality and habitat types affect decomposition rates within the Ankasa Conservation Area, Ghana. A wide variation in litter chemical composition and quality was found among the studied species. While the decay constant (k) varied among the habitat types as expected, it was similar among the studied species. The results support the hypothesis that k would vary among habitat types, but the same could not be established for the species. Differences in local conditions across habitats contributed to the effects of habitat variation on decomposition rates. Conversely, the similar decomposition rates among the species may result from the dominant influence of local climatic conditions, such as high rainfall and soil moisture content, which seem to mask the effects of species. Further, the few significant relationships recorded between the initial litter chemistry and k suggest that other factors, such as habitat-level conditions (e.g., topography, soil), may be more important than litter quality in influencing decomposition and biochemical cycling within this humid tropical forest ecosystem. In general, our findings confirm the complex interactions between environmental factors and species in driving decomposition within wet evergreen tropical forest ecosystems.
References
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Authors’ Info
Authors’ Affiliation
Department of Environmental Science, C. K. Tedam University of Applied Science and Technology, Navrongo (Ghana)
Alexander K Anning 0000-0002-9195-9924
Department of Theoretical and Applied Biology, Kwame Nkrumah University of Science and Technology, Kumasi (Ghana)
Corresponding author
Paper Info
Citation
Owusu G, Anning AK (2026). Litter quality and decomposition rates of selected species in the Ankasa Conservation Area, Ghana. iForest 19: 122-129. - doi: 10.3832/ifor4671-018
Academic Editor
Lorenzo Mw Rossi
Paper history
Received: Jun 21, 2024
Accepted: Oct 29, 2025
First online: Apr 11, 2026
Publication Date: Apr 30, 2026
Publication Time: 5.47 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2026
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 759
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 170
Abstract Page Views: 332
PDF Downloads: 219
Citation/Reference Downloads: 0
XML Downloads: 38
Web Metrics
Days since publication: 47
Overall contacts: 759
Avg. contacts per week: 113.04
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
(No citations were found up to date. Please come back later)
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Review Papers
Recent insights in soil nutrient cycling: perspectives from Pinus and Eucalyptus forest studies around the world
vol. 17, pp. 394-404 (online: 20 December 2024)
Research Articles
No home-field advantage in upper Andean tropical forests despite strong differences in site environmental characteristics
vol. 17, pp. 286-294 (online: 27 September 2024)
Research Articles
Contribution of legume and non-legume trees to litter dynamics and C-N-P inputs in a secondary seasonally dry tropical forest
vol. 15, pp. 8-15 (online: 13 January 2022)
Research Articles
Effects of stand age on litter quality, decomposition rate and nutrient release of Kazdagi fir (Abies nordmanniana subsp. equi-trojani)
vol. 13, pp. 396-403 (online: 03 September 2020)
Research Articles
Fine root production and distribution in the tropical rainforests of south-western Cameroon: effects of soil type and selective logging
vol. 3, pp. 130-136 (online: 27 September 2010)
Research Articles
Mid-rotation fertilization and liming of Pinus taeda: growth, litter, fine root mass, and elemental composition
vol. 14, pp. 195-202 (online: 24 April 2021)
Research Articles
Effects of arbuscular mycorrhizal fungi on microbial activity and nutrient release are sensitive to acid deposition during litter decomposition in a subtropical Cinnamomum camphora forest
vol. 16, pp. 314-324 (online: 13 November 2023)
Research Articles
The manipulation of aboveground litter input affects soil CO2 efflux in a subtropical liquidambar forest in China
vol. 12, pp. 181-186 (online: 10 April 2019)
Research Articles
Net ecosystem production of a tropical secondary forest in Jengka, Pahang, Malaysia
vol. 18, pp. 54-60 (online: 04 April 2025)
Research Articles
Soil fauna communities and microbial activities response to litter and soil properties under degraded and restored forests of Hyrcania
vol. 14, pp. 490-498 (online: 11 November 2021)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword