Dynamics of humus forms and soil characteristics along a forest altitudinal gradient in Hyrcanian forest
iForest - Biogeosciences and Forestry, Volume 14, Issue 1, Pages 26-33 (2021)
doi: https://doi.org/10.3832/ifor3444-013
Published: Jan 10, 2021 - Copyright © 2021 SISEF
Research Articles
Abstract
Humus forms are good indicators of environmental conditions and thus important in forest ecological processes. Altitudinal gradients are considered as natural laboratory for evaluating soil ecological processes and humus form distribution. The objective of this study was to evaluate the macromorphology of humus forms along an altitudinal gradient (0-2000 m a.s.l.) covered with plain forest, mixed and pure forests and forest-grassland ecotone, in Alborz Mountains in northern Iran. In total, 225 humus profiles were evaluated. Forest stand variables including tree density, basal area, crown density, and height, forest floor and soil physico-chemical properties along with biological features were measured. We found that altitudinal gradients influence both humus forms distribution and soil properties but with different mechanisms. While soil properties (i.e., temperature, pH, CaCO3, soil N content, soil C/N and microbial biomass N) were significantly correlated with altitude, the forest floor properties were more influenced by tree species composition. Particularly, the abundance of Mull was decreased in plain mixed forests compared to mountain pure forests, whereas the frequency of Amphi was increased. Moreover, Oligomull and Leptoamphi were abundant in mixed beech forests, while Eumacroamphi, Eumesoamphi and Pachyamphi were only observed in pure beech forests. Such a distribution influenced soil fertility where higher values of nitrogen (N), microbial biomass nitrogen (MBN) and pH were observed at lower altitudes under mixed forests compared to pure forests at higher altitudes.
Keywords
Altitude Gradient, Plant-humus-soil Relationships, Humus Systems, Soil Microbial Biomass
Introduction
Forest humus is an indicator of the existing environmental conditions ([25]), because it is the result of complex interactions between stand species composition ([20]), soil properties ([26]), soil micro- and macro-organisms’ activities and environmental factors ([4]). Since humus forms show specific morphological patterns (layering and structure - [16]), they are useful tool for assessing the health status of forests and the overall soil fertility ([27]).
The current classification systems enabled to distinguish five humus systems and sixteen humus forms in terrestrial ecosystems ([16], [36]). Humus forms can be directly identified in field without the need for expensive laboratory tools ([36]).
According to Zanella et al. ([35]), temperature, precipitation and vegetation composition are the three most important factors affecting biological degradation of organic residues and contributing in the formation of different humus forms. On the other hand, altitude through changes in temperature and precipitation, affects the distribution of forest species, forest floor quality and quantity ([7]), soil characteristics ([26]), microorganism types and activities ([37], [33]), thus contributing in humus forms ([3], [27]). Altitudinal gradients are considered as natural laboratories for evaluating soil ecological processes ([20], [9]). Understanding the complex interactions between soil and plant communities along altitude gradients can be used for the prediction of soil microbial activity and forest floor decomposition ([9], [33]).
The natural broadleaf forests in northern Iran are similar to those in central Europe, northern Turkey and the Caucasus. In these forest ecosystems, composition of tree species changes with elevation ([6]). Due to their unique topographical conditions compared to the oldest forest in Asia, Alborz mountains offers the potential to assess changes in forest types and humus forms with altitude ([24]). So far, few studies investigated the pattern of humus forms, forest floor features and soil microbial biomass along altitudinal gradients ([7], [30]).
In this study, we described humus morphology, forest floor, soil quality, microbial biomass carbon (MBC) and nitrogen (MBN) in five different forest types along an altitudinal gradient from 0 to 2000 m a.s.l. (i.e., plain forest, low, medium and high mountainous mixed and pure forests, and forest-grassland ecotone). We hypothesized that: (1) increased beech species abundance and decreased soil temperature along altitudinal gradient strongly affect the pattern of humus forms and organic layer thickness; (2) forest floor and soil characteristics change with altitude and soil fertility; (3) specific soil chemical and biological features correlate with humus forms and vegetation characteristics.
Material and methods
Site description
With an area of about 14,000 hectares, the Vaz catchment forests are located in the northern Alborz mountain, beside the Caspian Sea, in northern Iran (36° 16′ N, 52° 48′ E - Fig. S1 in Supplementary material). The study area was located along an altitude gradient 0-2000 m a.s.l. Forest vegetation in this area largely depends on altitude and therefore five different forest types could be distinguished ([18]): (1) plain mixed forests (PMF - 0 m a.s.l.): Ironwood (Parrotia persica C.A. Meyer), Oak (Quercus castaneifolia C. A. M.) and Hornbeam (Carpinus betulus L.); (2) low mountainous mixed forests (LMMF - 500 m a.s.l.): Beech (Fagus orientalis Lipsky), Ash (Fraxinus excelsior L.), Parrotia persica C. A. Meyer, Acer (Acer velutinum Boiss) and Carpinus betulus; (3) middle mountainous mixed forests (MMMF - 1000 m a.s.l.): Beech (Fagus orientalis Lipsky), Ash (Fraxinus excelsior L.), Parrotia persica C. A. Meyer, Acer (Acer velutinum Boiss) and Carpinus betulus; (4) high mountainous pure forests (HMPF - 1500 m a.s.l.): Fagus orientalis Lipsky; (6) forest-grassland ecotone (F-GE - 2000 m a.s.l.): Hawthorn (Crataegus sp.), Pear (Pyrus communis L.), Apple (Malus communis L.), Barberry (Berberis crataegina), Maple-AC (Acer campestre L.).
The mean annual temperature at PMF, LMMF, MMMF, HMPF and F-GE are 19.2, 16.3, 14, 11.6, and 8 °C, respectively. For every 1000 m increase in altitude, an average 3-5 °C decrease in temperature has been recorded. The mean annual precipitations are 898, 843, 805, 746 and 844 mm in PMF, LMMF, MMMF, HMPF and F-GE, respectively ([17]). About 35-45% of the rainfall occurs in autumn (from September to November), 18-35% in winter (December to February), and the rest (10-20%) in summer (June to August; Noushahr city meteorological station, 1977-2010 - Fig. S2 in Supplementary material). Based on World Reference Basis (WRB) and USDA Soil Taxonomies, plain forest soils were classified as Cambisols (Inceptisol), low and medium altitudes as Luvisols (Alfisols), and higher altitudes as Phaeozems (Mollisols) and Cambisols, developed on dolomite limestones belonging to the upper Jurassic and lower Cretaceous period ([18], [14]).
Experimental design, tree investigation, humus identification, forest floor and soil sampling
At each altitude (0, 500, 1000, 1500 and 2000 m a.s.l.), three 1-ha plots with at least 1500 m distance were delimited. Elevation at each plot was recorded using a Garmin™ model GPSMAP® 60Cx (Olathe, KS, USA). Aspect values were assigned using angles from 0 to 360° given by a pocket compass. In each plot, three random subplots (400 m2) were chosen for sampling. All living trees were counted at each subplot. The diameter at breast-height (DBH, 1.3 m) and total height (> 1.3 m) of all living trees were measured with a diameter tape and Impulse® 200 Laser Hypsometer (Laser Technology Inc., Centennial, CO, USA), respectively (Tab. 1).
Tab. 1 - Mean ± standard error (n=9) and Pearson’s correlation coefficients (Rcor) of vegetation characteristics along the altitudinal gradient in Hyrcanian forests. (PMF): plain mixed forests; (LMMF): low mountainous mixed forests; (MMMF): middle mountainous mixed forests; (HMPF): high mountainous pure forests; (F-GE): forest-grassland ecotone. Different letters indicate significant differences (p<0.05) among altitude levels after Tukey HSD test. (*): p < 0.05; (**): p < 0.01.
| Variables | Altitudinal gradient (m a.s.l.) | ANOVA results | Rcor | |||||
|---|---|---|---|---|---|---|---|---|
| 0 | 500 | 1000 | 1500 | 2000 | F test | P-value | ||
| Forest type | PMF | LMMF | MMMF | HMPF | F-GE | - | - | - |
| Tree density (n ha-1) | 263.8 ± 15.6 cd | 405.6 ± 57.4 bc | 175.0 ± 16.1 d | 586.1 ± 64.5 a | 558.3 ± 34.8 ab | 17.53 | <0.001 | 0.54** |
| Tree basal area (m2 ha-1) | 31.5 ± 6.1 abc | 39.3 ± 10.9 ab | 60.6 ± 10.8 a | 17.5 ± 2.8 bc | 2.9 ± 0.3 c | 8.52 | <0.001 | -0.39** |
| Tree crown density (%) | 82.3 ± 1.9 a | 76.9 ± 2.6 ab | 76.8 ± 2.9 ab | 68.4 ± 1.1 b | 44.7 ± 2.2 c | 44.71 | <0.001 | -0.79** |
| Mean tree height (m) | 16.7 ± 0.5 c | 21.1 ± 0.9 b | 26.6 ± 0.5 a | 17.3 ± 0.44 c | 5.2 ± 0.3 d | 187.68 | <0.001 | -0.52** |
| Beech density (n ha-1) | - | 230.5 ± 49.6 b | 91.7 ± 13.8 bc | 586.1 ± 64.6 a | - | 43.99 | <0.001 | - |
| Oak density (n ha-1) | 33.3 ± 9.32 | - | - | - | - | - | - | - |
| Hornbeam density (n ha-1) | 22.2 ± 10.6 | - | 36.1 ± 8.5 | - | - | - | - | - |
| Maple-AV density (n ha-1) | - | 2.8 ± 2.8 | 33.3 ± 11.1 | - | - | - | - | - |
| Ash density (n ha-1) | - | 97.2 ± 36.4 | - | - | - | - | - | - |
| Alder density (n ha-1) | 2.8 ± 2.8 | - | - | - | - | - | - | - |
| Iron wood density (n ha-1) | 194.4 ± 22.4 a | 75.0 ± 17.7 b | 8.3 ± 2.3 c | - | - | 39.80 | <0.001 | - |
| Populus density (n ha-1) | 11.1 ± 4.39 | - | - | - | - | - | - | - |
| Hawthorn density (n ha-1) | - | - | - | - | 344.4 ± 33.1 | - | - | - |
| Pear density (n ha-1) | - | - | - | - | 94.4 ± 33.1 | - | - | - |
| Apple density (n ha-1) | - | - | - | - | 63.9 ± 22.9 | - | - | - |
| Barberry density (n ha-1) | - | - | - | - | 27.8 ± 11.4 | - | - | - |
| Maple-AC density (n ha-1) | - | - | - | - | 27.8 ± 10.6 | - | - | - |
| Soil texture | clay | clay / clay loam | clay / clay loam | clay / clay loam | clay loam | - | - | - |
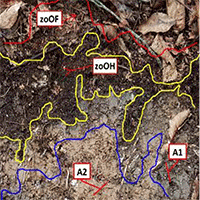
The experiment was conducted during April 2018. Humus profiles (Organic: OL, OF, OH; and organic-mineral: AH) and diagnostic horizons were described and sampled at the corners and at the center of each sub-plot using a metal frame (30×30 cm). The morphological characteristics of each humus profile were described according to Zanella et al. ([36]). The basilar elements of the adopted humus classifications are reported in Tab. S1 (Supplementary material). Humus layer thickness (HLT) was also measured with a tape from the forest floor surface to the top of the mineral soil. The earthworm ecological groups (i.e., Epigeic, Anecic and Endogeic) were also identified ([8]). Forest floor samples including OL and OF layers were finely mixed before sampling. To remove soil, the forest floor samples were soaked gently in tap water for a few seconds (this is not recommended for samples dominated by OH layers) and then dried at 70 °C for 48 h. Dried forest floor samples were finely grounded/homogenized with an electric mixer and analyzed.
Top mineral soil samples (depth 0-10 cm) were collected after removal of the organic layers. Using a standard soil auger (5 cm inner diameter). Soil temperature (ST) was measured at a depth 0-10 cm with a portable temperature probe (model TA-288). Since no rainfall occurred during the sampling time, temperature was quite constant during the day.
To determine microbial biomass, the soil samples were immediately transferred to sterile bags, placed in a cooled and insulated container, transferred to the laboratory and stored at 4 °C. Soil samples used for physico-chemical analyses were air-dried and passed through a 2-mm sieve. In total, 225 samples were analyzed in this study (5 altitude levels × 3 plots × 3 subplots × 5 profiles). The soils and forest floors collected from five elevation level were mixed and the mean of the humus layers and percentage of humus form were used to compute humus layer thickness and humus form classification, respectively.
Laboratory analysis of forest floor and soil physico-chemical and biological properties
Forest floor carbon (FFC) and nitrogen (FFN) contents were determined through dry combustion and semi micro-Kjeldahl techniques, respectively ([12]). Soil texture was determined using the Bouyoucos hydrometer method ([11]). Soil moisture (SM) was measured after drying the soil samples in an oven at 105 °C for 24 h. Soil pH was measured in a ratio of 1:2.5 (M/V) of soil/water using an Orion™ Analyzer Model 901 pH meter (ThermoFischer Scientific, Waltham, MS, USA). Calcium carbonate (CaCO3) content was determined by the neutralization titration method. Soil organic carbon (SOC) and soil nitrogen (SN) contents were determined based on the modified Walkley-Black ([1]) and semi Micro-Kjeldahl methods ([12]), respectively. The microbial biomass carbon (MBC) and microbial biomass nitrogen (MBN) were assessed through the fumigation-extraction method with a conversion factor of 0.45 for microbial C and 0.54 for microbial N ([13], [29]).
Statistical analysis
The normality of data was checked by the Kolmogorov Smirnov test (P >0.05), and the homogeneity of variances was tested using the Levene’s test (P >0.05). One-way analysis of variance (ANOVA) was performed to analyze differences in vegetation properties, humus layer thickness (HLT), forest floor, soil physical, chemical and biological properties along the altitudinal gradient. Means were compared using Tukey HSD post-hoc test. Abundance of humus systems and forms in relation to the altitudinal gradient was tested by Fisher’s exact test. Pearson’s correlation analyses were performed to correlate the vegetation variables and forest floor and soil characteristics across the altitudinal gradient. For non-normally distributed data, the Spearman’s correlation analysis was performed. All statistical analyses were conducted using SPSS® v. 16 (IBM Corp., Armonk, NY, USA). Multivariate correlations were analyzed using factor analysis based on principal components analyses (PCA) performed by the software PC-Ord v. 5.0 ([23]).
Results
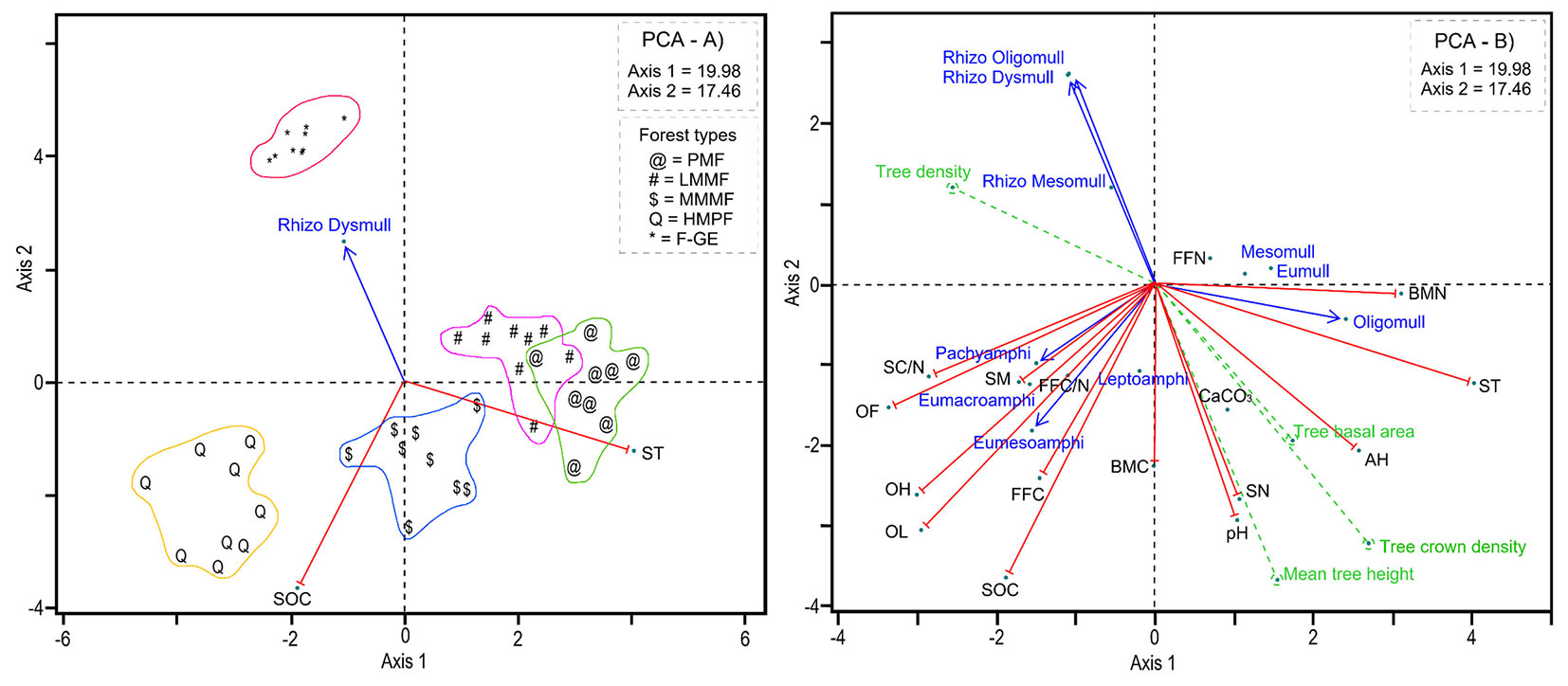
PCA revealed significant changes in all studied soil and humus characteristics along the altitudinal gradient (Tab. 2, Fig. 1A-B), with greater than 45 percent of variations being explained. The left side of the PC axis 1 reflects low quality of forest floor (i.e., high FFC, FFC/N and thickness), and soil (i.e., higher values of SOC and C/N) which resulted in the formation of Amphi humus forms under high mountainous pure forests (Fig. 1B). The right side of PC axis 1, instead, corresponds to conditions with higher forest productivity (tree basal area, tree crown density and mean tree height), improved forest floor (i.e., N) and soil characteristics (N content, MBN, pH and CaCO3). In these conditions, the frequency of the mull humus forms was higher (plain mixed and low mountainous mixed forests - Fig. 1B). Middle mountainous mixed forests showed intermediate conditions with regard to the forest floor and soil properties and thus a strong relationship with leptoamphi humus and MBC (Fig. 1B). In addition, the forest-grassland ecotone with Rhizo-Mull humus forms did not show any relationship with soil properties (Fig. 1B).
Tab. 2 - Correlation of vegetation, humus forms and soil features with PCA components. (*): p < 0.05; (**): p < 0.01.
| Features | PC1 | PC2 | Features | PC1 | PC2 |
|---|---|---|---|---|---|
| Tree density | -0.56 ** | 0.25 | Eumacroamphi | -0.33 | -0.20 |
| Tree basal area | 0.39 * | -0.40 | Eumesoamphi | -0.35 * | -0.37 * |
| Tree crown density | 0.60 ** | -0.67 ** | Pachyamphi | -0.38 * | -0.25 |
| Mean tree height | 0.34 * | -0.76 ** | FFC | -0.32 | -0.50 ** |
| OL | -0.65 ** | -0.63 ** | FFN | 0.16 | 0.07 |
| OF | -0.75 ** | -0.32 | FFC/N | -0.24 | -0.23 |
| OH | -0.67 ** | -0.50 ** | SM | -0.35 * | -0.26 |
| AH | 0.57 ** | -0.43 * | ST | 0.89 ** | -0.25 |
| Eumull | 0.33 | 0.04 | pH | 0.23 | -0.61** |
| Mesomull | 0.25 | 0.03 | CaCO3 | 0.20 | -0.32 |
| Oligomull | 0.54 ** | -0.09 | SOC | -0.42 * | -0.75 ** |
| Rhizo Mesomull | -0.12 | 0.25 | SN | 0.24 | -0.55 ** |
| Rhizo Oligomull | -0.24 | 0.54 ** | SC/N | -0.63 ** | -0.23 |
| Rhizo Dysmull | -0.24 | 0.54 ** | MBC | -0.004 | -0.47 ** |
| Leptoamphi | -0.04 | -0.22 | MBN | 0.70 ** | -0.02 |
Fig. 1 - Principle component analysis (PCA) based on the correlation matrix to identify the relationships between forest types, humus forms, forest floor and soil properties (A-B). (PMF): plain mixed forests; (LMMF): low mountainous mixed forests; (MMMF): middle mountainous mixed forests; (HMPF): high mountainous pure forests; (F-GE): forest-grassland ecotone.
As expected, canopy composition, stand features, humus forms and their characteristics changed with altitude (Tab. 1, Tab. 2). In the plain forest, ironwood was the dominant species followed by oak and hornbeam. However, ironwood density decreased with altitude and this species was totally absent at altitudes above 1500 m a.s.l. At intermediate altitudes (500 and 1000 m), beech, ash and maple were the dominant species, while at higher altitude (1500 m) only beech was present. Above 2000 m a.s.l., hawthorn was the most common species. Total tree density significantly increased with altitude (R = 0.54, p < 0.01), while basal area (R = -0.39, p < 0.01), crown density (R = -0.79, p < 0.01) and mean tree height (R = -0.52, p < 0.01) decreased.
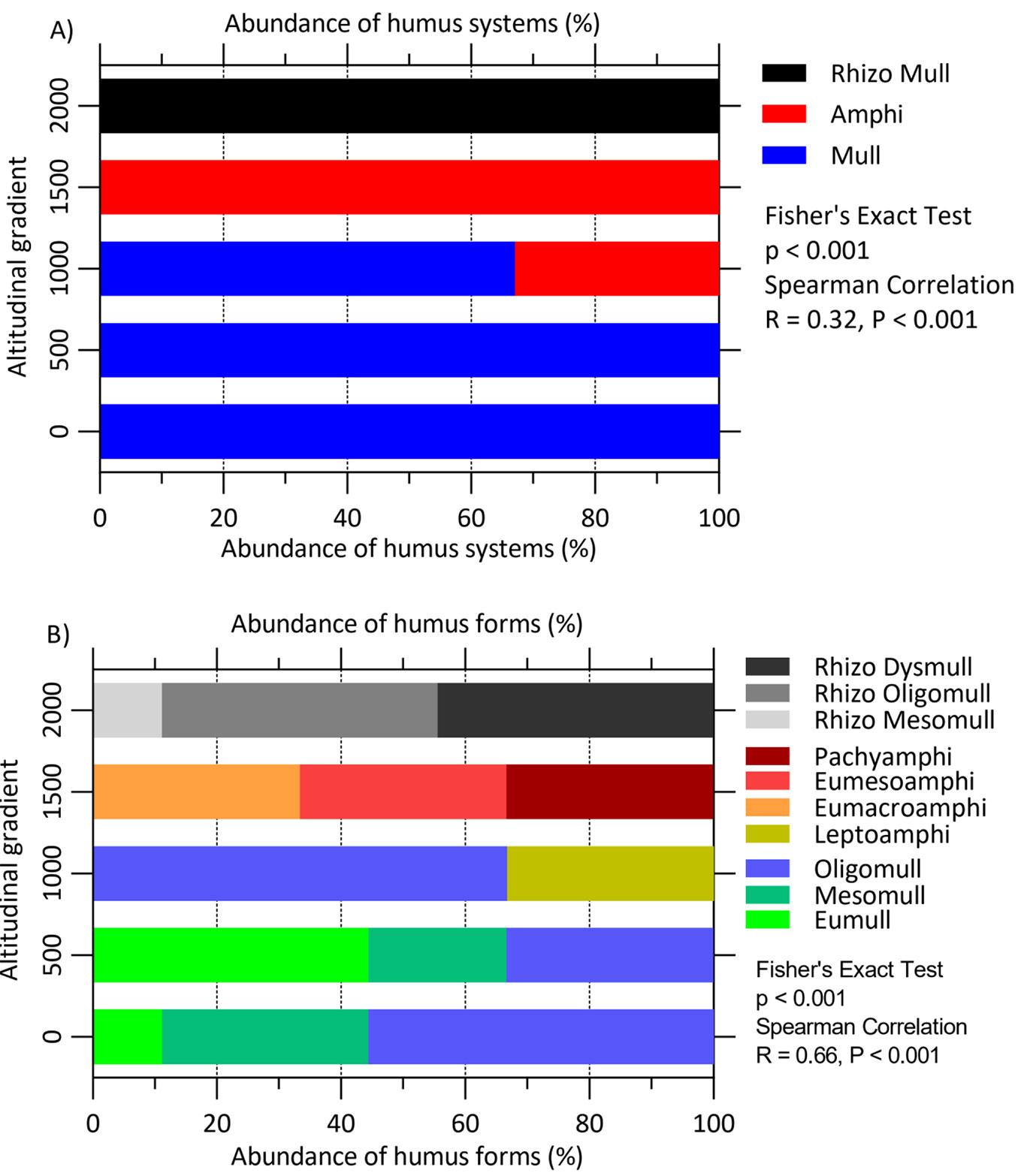
Altitudinal gradient significantly affected the abundance of the humus systems (p < 0.001) and humus forms (p < 0.001 - Fig. 2A). Mull was the dominant system below 1000 m a.s.l.; Amphi appeared at 1000 m and dominated at 1500 m; Rhizo Mull was dominant under F-GE at 2000 m a.s.l. Oligomull was the most common form at 0 and 1000 m and Eumull at 500 m (Fig. 2B). At higher altitudes, no dominant humus form was detected. In fact, Eumacro, Eumeso and Pachyamphi humus forms were equally represented at 1500 m, while at 2000 m Rhizo Mesomull, Rhizo Oligomull and Rhizo Dysmull were equally abundant (Fig. 2B).
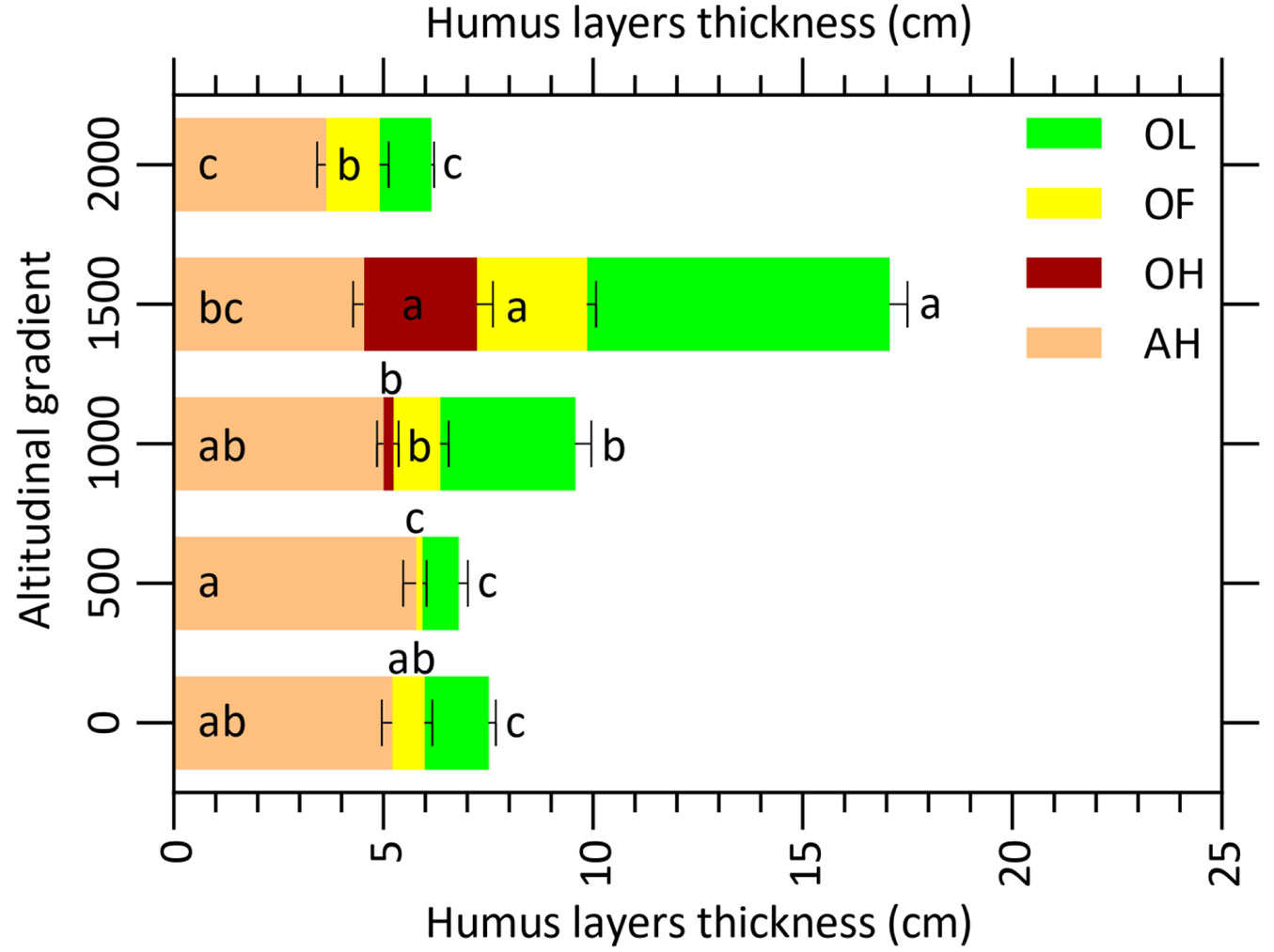
The thickness of the organic layers including OL, OF, OH significantly increased with altitude (R = 0.36, p < 0.05; R = 0.53, p < 0.01; R = 0.36, p < 0.05; respectively), whereas the organic-mineral thickness (AH) decreased (R = -0.62, p < 0.01 - Tab. 3, Fig. 3). The thickness of OL and OF at 1500 m was approximately 2.5 times greater than that at the other altitudes (p < 0.001); the highest thickness of OH was recorded at 1500 m a.s.l., while this layer was not observed at 0, 500 and 2000 m altitudes (Tab. 3, Fig. 3).
Tab. 3 - One-way analysis of variance (ANOVA) and Pearson’s correlation coefficients (Rcor) of forest floor and soil characteristics along the altitudinal gradients. (*): p < 0.05; (**): p < 0.01.
| Humus and soil properties |
Variables | Abbr. | F test | P value | Rcor |
|---|---|---|---|---|---|
| Humus layers thickness | Organic litter (cm) | OL | 81.86 | <0.001 | 0.36* |
| Organic fragmentation (cm) | OF | 21.73 | <0.001 | 0.53** | |
| Organic humus (cm) | OH | 40.81 | <0.001 | 0.36* | |
| Organic-mineral layer (cm) | AH | 11.32 | <0.001 | -0.62** | |
| Forest floor properties | Forest floor carbon (%) | FFC | 3.74 | 0.012 | 0.093 |
| Forest floor nitrogen (%) | FFN | 15.84 | <0.001 | -0.09 | |
| Forest floor C/N | FFC/N | 5.80 | <0.001 | 0.11 | |
| Soil physical properties | Soil moisture (%) | SM | 3.24 | 0.021 | 0.25 |
| Soil temperature (°C) | ST | 219.97 | <0.001 | -0.94** | |
| Soil chemical properties | Soil pH | pH | 75.08 | <0.001 | -0.51** |
| Calcium carbonate (%) | CaCO3 | 16.04 | <0.001 | -0.45* | |
| Soil organic carbon (%) | SOC | 30.25 | <0.001 | 0.18 | |
| Soil nitrogen (%) | SN | 11.11 | <0.001 | -0.39** | |
| Soil C/N | SC/N | 19.14 | <0.001 | 0.53** | |
| Soil biological properties | Microbial biomass carbon (mg kg -1) | MBC | 24.22 | <0.001 | -0.007 |
| Microbial biomass nitrogen (mg kg -1) | MBN | 44.08 | <0.001 | -0.70** |
Fig. 3 - Organic (OL, OF and OH) and organic-mineral (AH) humus layers thickness along the altitudinal gradient. Error bars indicate standard error (n = 45).
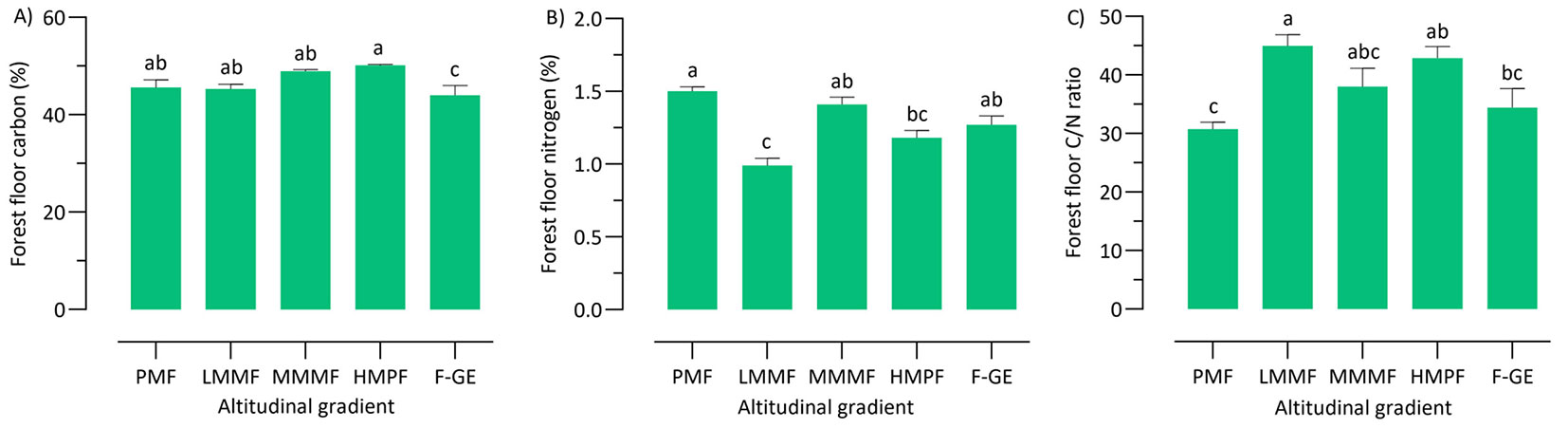
No clear relationship was observed between forest floor properties (C, N and C/N ratio) and altitude (Tab. 3), though some significant differences among forest types were detected (Fig. 4). The highest value of forest floor C was found in high mountainous pure forests (Fig. 4A), while the forest floor N was significantly higher at the plain mixed forests (Fig. 4B). The plain mixed forests, however, showed the lowest forest floor C/N ratio (Fig. 4C), while low and high mountainous mixed forests showed the highest forest floor C/N ratio (Fig. 4C).
Fig. 4 - Mean floor carbon (A), nitrogen (B), and C/N ratio (C) among the altitude levels. Different letters indicate significant differences (p < 0.05) according to the ANOVA and Tukey HSD test. Error bars indicate standard error (n=9). (PMF): plain mixed forests; (LMMF): low mountainous mixed forests; (MMMF): middle mountainous mixed forests; (HMPF): high mountainous pure forests; (F-GE): forest-grassland ecotone.
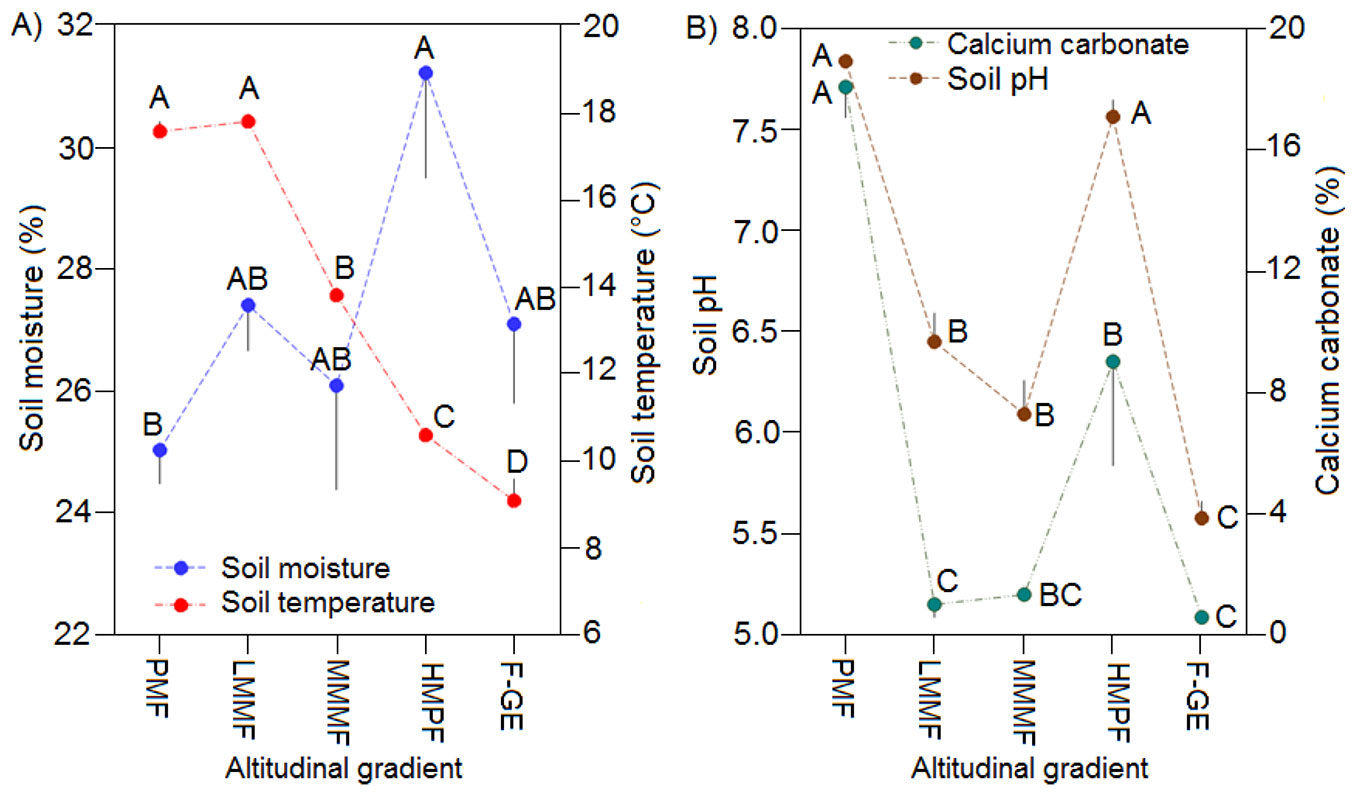
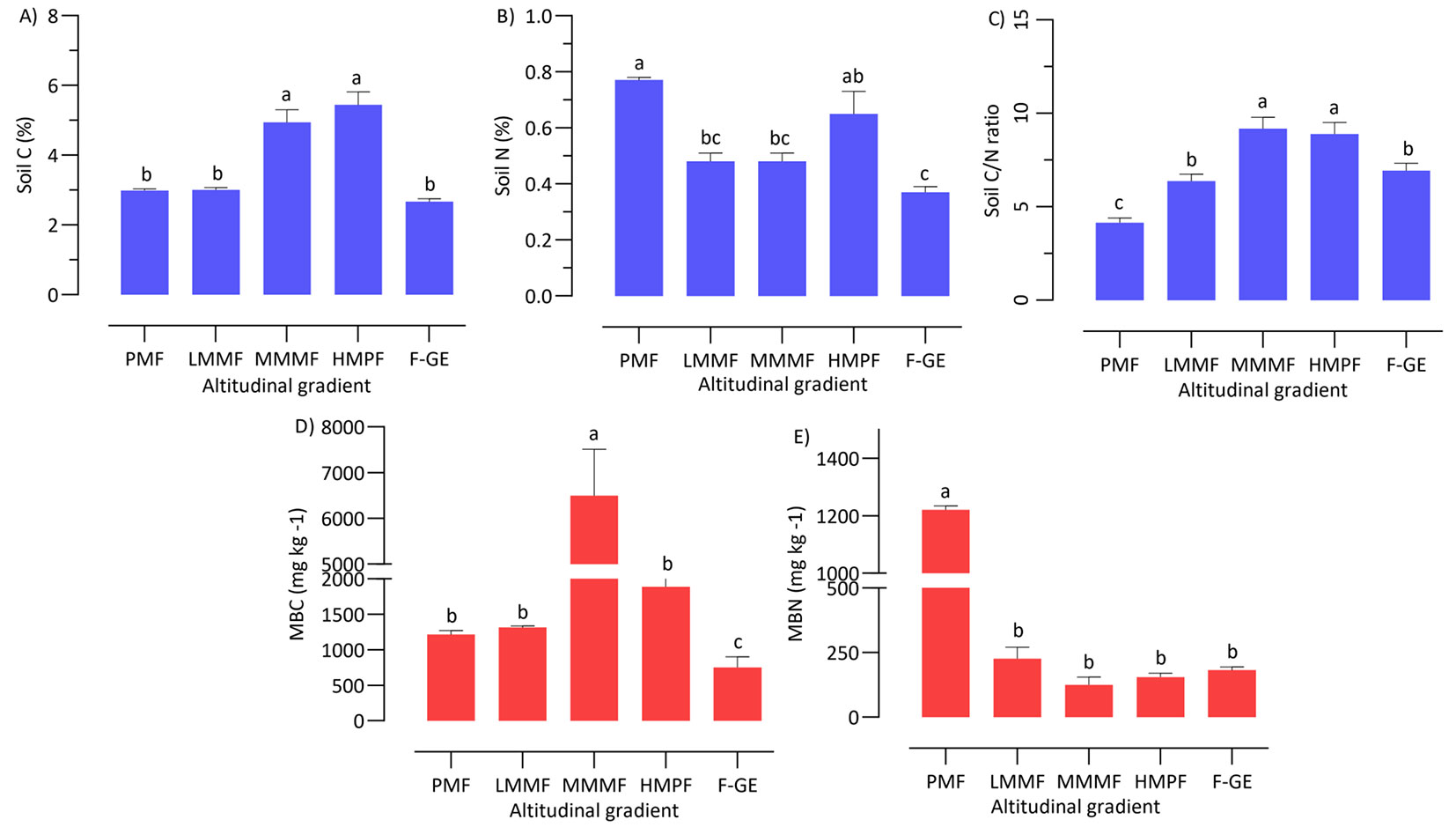
Soil properties, however, significantly changed with altitude (Tab. 3, Fig. 5). Soil temperature differed among forest types and decreased with altitude (R = -0.94, p < 0.01). The lowest soil moisture was measured in plain mixed forests, while the highest was recorded in high mountainous pure forests (Fig. 5A, Tab. 3). On the contrary, soil pH (R= -0.51, p < 0.01), calcium carbonate (R = -0.45, p < 0.01), soil N (R = -0.39, p < 0.01) and microbial biomass nitrogen (R = -0.70, p < 0.01) decreased with altitude, while soil C/N ratio increased (R = 0.53, p < 0.01). The highest soil pH and CaCO3 concentration were observed in plain mixed forests and high mountainous pure forests and the lowest in forest-grassland ecotone (Fig. 5B). The highest concentrations of soil C, C/N ratios and BMC values were measured under high and middle mountainous forests, whereas plain mixed forests showed the highest soil N concentrations and BMN values (Fig. 6).
Fig. 5 - Mean soil moisture and soil temperature (A), pH and CaCO3 (B) among the altitudes. Different letters indicate significant differences (p < 0.05) based on ANOVA and Tukey HSD test. Error bars indicate standard error (n=9). (PMF): plain mixed forests; (LMMF): low mountainous mixed forests; (MMMF): middle mountainous mixed forests; (HMPF): high mountainous pure forests; (F-GE): forest-grassland ecotone.
Fig. 6 - Mean soil carbon (A), nitrogen (B), C/N ratio (C), BMC (D) and BMN (E) along the altitude gradient. Different letters indicate significant differences (P < 0.05) according to the ANOVA and Tukey HSD test. Error bars indicate standard error (n=9). (PMF): plain mixed forests; (LMMF): low mountainous mixed forests; (MMMF): middle mountainous mixed forests; (HMPF): high mountainous pure forests; (F-GE): forest-grassland ecotone.
Discussion
We showed that there exist significant associations between altitudinal gradient and forest characteristics including tree composition, stem density, tree height in Hyrcanian forest in northern Iran, consistently with findings of other studies previously published ([24], [5]). The distribution of humus forms also changed with altitudinal gradient. Particularly, Mull humus forms decreased with altitude, while Amphi forms increased. With respect to the forest type, our results showed that Oligomull and Leptoamphi were dominant in mixed beech forests, while in pure beech forests Eumacroamphi, Eumesoamphi and Pachyamphi were the dominant forms. Previous study by Waez-Mousavi ([30]) also reported that Mull and Amphi are the most dominant humus systems in the Hyrcanian forests. In mixed beech stands, Waez-Mousavi & Habashi ([31]) reported the dominance of Mull and Amphi humus systems.
Both environmental conditions and tree species composition influence humus formation and its characteristics. A significant change in soil temperature, moisture and species composition was noted in our altitudinal gradient. Previous study revealed that a decrease in mean temperature associates with a decline in Mull humus form and an increase in Amphi humus ([26]). In agreement to our finding in plain mixed forest, Ponge et al. ([26]) noted that Mull systems are more frequent at higher tree species diversity and under rich trophic conditions. In contrary, under low tree species richness and in colder environments, Moder and Amphi humus systems with OF and OH layers are dominant ([4]). Labaz et al. ([20]) showed that Amphi humus forms can be found in cold conditions where organic matter decomposition is slower. Previous study by Waez-Mousavi & Habashi ([31]) indicated that Mull humus forms are abundant under forest types with higher floor quality and decomposition rate, while Amphi and Moder humus forms are observed under beech forest type with low floor quality (high C and low N - [6]). Mull humus forms are biologically active (Endogeic and Anecic with high activity) with fine-granular structure, which have low SOC content compared to humus forms with OF and OH layers ([16], [20]). Moder forms are abundant in beech dominated forest with low soil pH (≈ 5.5), while Mull forms are absent in non-beech stands ([5]). The increased Amphi humus form under pure beech forest (1500 m) could likely be due to high soil pH (> 7.5) resulted from high CaCO3concentrations ([21]). The CaCO3 concentration has probably a positive impact on the forest floor decomposition rate and soil microbial activity ([15]) and could likely facilitate the transition from Moder to Amphi form ([20], [10]).
Previous studies showed that climatic (moisture and temperature) and biotic factors (species type and richness) are important factors influencing humus accumulation ([35], [20], [4]). In our study, the thickness of OL, OF, OH layers significantly were increased, while that of AH was decreased. The more favorable conditions for organic matter decomposition in plain mixed forests (i.e., high temperature, good soil moisture and high litter quality) is likely the cause ([27]). Similarly, Bonifacio et al. ([10]) also showed that the OH layer thickness in beech forests with a low litter quality is higher than in hornbeam, maple and ash forests ([20]). Thus, the higher OH layer thickness at 1500 m a.s.l. found in this study can be attributed to the low temperature in this elevation level, which slow down mineralization rates ([4]), decrease litter quality under beech ([6]) and higher soil moisture ([35]).
The chemical composition of humus and soil are the result of the interaction of many factors including topography, climate, tree cover and soil microbial communities ([26]). Shedayi et al. ([28]) showed that altitude has a low impact on soil organic carbon and nitrogen, while vegetation cover explains most of the measured variations. Bayranvand et al. ([6]) reported that, although tree species affect soil chemical properties (i.e., pH, C, and N content), earthworm and microbial activity were mostly controlled by climate. Our results support the idea that soil properties including temperature, pH, CaCO3, soil N content, soil C/N and microbial biomass N are significantly correlated with altitude, while most forest floor properties are not directly influenced by temperature, but affected by tree species composition. In fact, litter quality influences both decomposition rates and the dynamic of nutrient mineralization ([22]). Previous studies have argued that higher forest floor N concentrations are associated with faster litter decomposition rates ([19], [22]). A decrease in forest floor quality (high C content and high C/N ratio) was reported to associate with a higher humus layer thickness and a decreased decomposition rate in beech dominated forests at high altitudes ([7]). In fact, beech litter is known for having a high lignin/N ratio and a relatively low contents of basic cations and N ([10]). In agreement with our findings, low humus layer thickness is related to high quality floors in maple, ironwood, alder and hornbeam ([19], [7]). Forest floor C/N ratio and N content are the two most important factors influencing litter decomposition and nutrient release ([22]).
Badía-Villas & Girona-García ([4]) reported that forest floor N in mountain forest in Spain is decreased during shift from Mull to Amphi forms with increasing elevation. Ponge et al. ([26]) measured lower C content in Mull than in other humus forms. It could be speculated that Mull forms decompose faster and introduce more N into the soil. Zanella et al. ([35]) argued that forest floor and soil C/N ratios in Mull are usually lower than in Amphi and the C/N is an important indicator for the decomposition rate and nutrient cycling in the humus and soil.
Our data also showed a significant decrease in soil MBN under different canopy compositions along the elevation gradient. MBN was significantly higher in mixed forest types (i.e., PMF) at the lowest elevation than in pure stands at the highest elevation levels (i.e., HMPF). This may be the result of a greater and a more diverse litter input in stands with a higher species richness or diversity ([32]). Many investigations have also documented that soil microbial community structure is primarily driven by soil pH and C/N ratio as the altitude increases. Thus, higher levels of pH, such as those at low elevations, may be related to increased microbial biomass and bacterial diversity ([33]). Higher levels of soil temperature and N content, such as those at low elevations, may contribute to a larger microbial biomass ([33], [9], [15]).
Beech litter quality, FFC accumulation and lower earthworm activity are main factors affecting soil quality in this forest system. PMF was correlated with Mull humus and higher forest floor and soil quality (high FFN and SN; low FFC and SN). In this condition, tree species composition along with high biological and microbial activities (i.e., high temperature and soil water content) speed up organic matter decomposition ([34]). Mull humus forms are nutrient rich systems with fast nutrient cycling ([2]) which are associated to high earthworm activity and microbial biomass. In hornbeam and maple trees (MMMF) forest systems, higher forest floor quality and improved soil fertility support larger biological activities than in pure beech forests ([19]).
Conclusion
Altitudinal gradient is a key factor determining the distribution of humus forms. Soil properties (temperature, pH, CaCO3, N content, C/N and MBN) were significantly correlated with altitude, while forest floor properties were more influenced by tree species composition. Our data suggest that the abundance of Mull forms decrease from plain mixed forests to high mountain pure forests, whereas the frequency of Amphi humus forms increase. On the other hand, Oligomull and Leptoamphi are more abundant in mixed beech forests, while Eumacroamphi, Eumesoamphi and Pachyamphi are observed only in pure beech forests. In addition, plain mixed forests typically have higher quality of both forest floor (i.e., N) and soil (i.e., pH, CaCO3, soil N content, soil C/N and MBN) than high mountainous pure one, while middle mixed forests show intermediate characteristics.
Abbreviations
OL: Organic litter; OF: Organic fragmentation; OH: Organic humus; AH: Organic-mineral layer; FFC: Forest floor carbon; FFN: Forest floor nitrogen; FFC/N: Forest floor C/N; SM: Soil moisture; ST: Soil temperature; pH: Soil pH; CaCO3: Calcium carbonate; SOC: Soil organic carbon; SN: Soil nitrogen; SC/N: Soil C/N; MBC: Microbial biomass carbon; MBN: Microbial biomass nitrogen.
Author contributions
MB and MA conceived and designed the experiment. MB performed the experiment. MB, GA and GS-J carried out the statistical analysis. MB, JG, GA and GS-J contributed to the data analysis and data interpretation. MB, JG, GS-J and GA wrote and edited the manuscript.
Acknowledgements
This research was funded by a grant provided by Tarbiat Modares University (TMU) and Iran National Science Foundation (INSF). We would like to thank Mr. J. Eslamdoust, A. Khodadust, MR Soleimani, E. Sharifi, M. Mohammadi, and A. Daryaei for their assistance during field samplings. We also thank M. Naeiji and M. Haghdoust for their assistance during laboratory analysis of the soil samples. The authors also express their sincere appreciation to Augusto Zanella for helping with morphological identification of humus forms and Björn Berg for reviewing the manuscript.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Moslem Akbarinia
Faculty of Natural Resources and Marine Sciences, Tarbiat Modares University, 46417-76489, Noor, Mazandaran (Iran)
Microbial Biotechnology Department, Agricultural Biotechnology Research Institute of Iran (ABRII), Agricultural Research, Education and Extension Organization (AREEO), Fahmideh Blvd., P.O. Box:31535-1897, Karaj (Iran)
Human Genetics Research Center, Baqiyatallah University of Medical Sciences, Tehran (Iran)
Department of Agri-food, Environmental and Animal Sciences, University of Udine, v. delle Scienze 206, 33100 Udine (Italy)
Corresponding author
Paper Info
Citation
Bayranvand M, Akbarinia M, Salehi Jouzani G, Gharechahi J, Alberti G (2021). Dynamics of humus forms and soil characteristics along a forest altitudinal gradient in Hyrcanian forest. iForest 14: 26-33. - doi: 10.3832/ifor3444-013
Academic Editor
Maurizio Ventura
Paper history
Received: Apr 09, 2020
Accepted: Nov 02, 2020
First online: Jan 10, 2021
Publication Date: Feb 28, 2021
Publication Time: 2.30 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2021
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 38914
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 31899
Abstract Page Views: 3146
PDF Downloads: 3193
Citation/Reference Downloads: 5
XML Downloads: 671
Web Metrics
Days since publication: 1936
Overall contacts: 38914
Avg. contacts per week: 140.70
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2021): 11
Average cites per year: 2.20
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Technical Reports
Evaluating humus forms variation in an unmanaged mixed beech forest using two different classification methods
vol. 5, pp. 272-275 (online: 24 October 2012)
Research Articles
Wood-soil interactions in soil bioengineering slope stabilization works
vol. 2, pp. 187-191 (online: 15 October 2009)
Research Articles
Relationship between microbiological, physical, and chemical attributes of different soil types under Pinus taeda plantations in southern Brazil
vol. 17, pp. 29-35 (online: 28 February 2024)
Research Articles
Effects of altitudinal gradients on leaf area index, soil microbial biomass C and microbial activity in a temperate mixed forest ecosystem of Northwestern Turkey
vol. 10, pp. 334-340 (online: 15 December 2016)
Research Articles
Seasonal dynamics of soil respiration and nitrification in three subtropical plantations in southern China
vol. 9, pp. 813-821 (online: 29 May 2016)
Research Articles
The effect of clear-cut age on soil organic carbon and nitrogen indices in Scots pine (Pinus sylvestris L.) stands
vol. 18, pp. 146-153 (online: 09 June 2025)
Research Articles
Soil fauna communities and microbial activities response to litter and soil properties under degraded and restored forests of Hyrcania
vol. 14, pp. 490-498 (online: 11 November 2021)
Research Articles
Thinning effects on soil and microbial respiration in a coppice-originated Carpinus betulus L. stand in Turkey
vol. 9, pp. 783-790 (online: 29 May 2016)
Research Articles
Soil respiration along an altitudinal gradient in a subalpine secondary forest in China
vol. 8, pp. 526-532 (online: 01 December 2014)
Review Papers
The soil-conscious forestry and the forbidden apple
vol. 17, pp. 252-268 (online: 16 August 2024)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword