First-year seedlings respond to a 21-day summer water deficit: morphological and physiological insights of two populations of five Mediterranean Quercus species
iForest - Biogeosciences and Forestry, Volume 19, Issue 2, Pages 149-156 (2026)
doi: https://doi.org/10.3832/ifor4928-019
Published: Apr 19, 2026 - Copyright © 2026 SISEF
Research Articles
Abstract
In the Mediterranean basin, extreme drought events are expected to become more frequent due to climate change. Consequently, selecting and registering forest reproductive material (FRM) adapted to such conditions is crucial to support effective forest restoration. This study aims to identify whether certain seed sources of Quercus exhibit traits potentially advantageous under ongoing climate change. An initial screening of seedlings from five ecologically diverse species from Puglia (southern Italy) was performed to evaluate early-stage responses to drought conditions simulating extreme Mediterranean summer events. Under semi-controlled conditions in a nursery, we evaluated the effect of water removal for 21 days on the growth, physiological, and metabolomic performance of FRM of two seed sources per five Mediterranean Quercus species. Morphological, physiological (SPAD, chlorophyll fluorescence), and metabolic traits (pigments and malondialdehyde) were measured throughout the experimental period and at its end. All Quercus species and their seed sources survived the water deficit and exhibited positive morphological and physiological responses. The effects of seed source were more pronounced in the “Cerris” section than in the evergreen “Ilex” section, making the former species good candidates for afforestation or restoration in Mediterranean areas and valuable material for further research. The high performance of Q. cerris, a species generally considered less drought-tolerant, highlights the Puglia region as a promising source of southern Mediterranean provenances with enhanced resistance to arid conditions.
Keywords
Seed Sources, Tolerance, Water Deficiency, Seedlings, Morphology, Functionality
Introduction
The Mediterranean basin is recognized as a global biodiversity hotspot, encompassing approximately 10% of the world’s plant species in just 1.6% of Earth’s surface ([31]) hosting over 13.000 endemic plants species (4.3% of global plants) ([32]). This region is facing significant threats from climate change, with models projecting robust declines in winter precipitation of up to 40% ([45]). The Intergovernmental Panel on Climate Change (IPCC) has identified this region as a “climate change hot spot”, anticipating increases in the frequency and intensity of climatic events, including mean warming and heat extremes ([1]). In addition to prolonged and harsher summer aridity, the region now faces unpredictable drought events ([7]) even during traditionally wetter seasons (i.e., fall, winter, and spring), further challenging these ecosystems. These climatic shifts pose serious challenges to Mediterranean ecosystems, particularly forests, which play a crucial role in biodiversity conservation ([40]). Rising temperatures exacerbate the effects of drought on plant mortality, potentially surpassing critical thresholds for the natural regeneration of some species ([15]). Both excessive soil water in winter and summer drought hinder forest seedling establishment, with the temporal and spatial variability of soil moisture significantly influencing regeneration dynamics, as seen in oaks ([47]). Consequently, conserving Mediterranean forest ecosystems is a priority given their high ecological value.
It is postulated that seedlings are more vulnerable to abiotic stress than mature trees; therefore, exacerbated summer drought can have severe implications for forest regeneration, hindering seedlings’ survival and growth. Seedlings, not reaching the deeper soil layers, are more vulnerable and dependent on precipitation, as their water availability is more influenced by the upper soil layers than by periods without precipitation ([33]). Therefore, seedlings might contend with both the direct effects of water deficits during critical growth stages and the unpredictable timing and intensity of summer drought events, which sharply limit water availability. In this context, there is a growing interest in assessing the capacity of natural and artificial regeneration to grow and survive under increasing environmental pressures ([20]). Understanding these dynamics is crucial for mitigating stress and ensuring successful regeneration, especially in endangered areas where the divergent fitness between core and marginal populations plays a key role in preventing biodiversity loss due to climate change.
Identifying more resistant provenances to water deficit is essential for several reasons: (i) it contributes to more effective regeneration in populations that are particularly vulnerable to drought ([38], [13]); these populations may require additional artificial regeneration to ensure long-term forest cover, especially under increasingly arid conditions; (ii) it provides valuable FRM for forest restoration efforts in harsher Mediterranean environments, thereby enhancing the success of restoration projects; (iii) identifying seed sources can help enrich the Italian Base Material National Register (BMNR), making them automatically available across the European Union in accordance with European Directive 105/1999, for projects that may require such provenances to produce climate adaptative FRM ([26], [27]); and (iv) it can support assisted migration strategies, which introduce FRM from more resilient provenances or species to areas where local populations may become maladapted under changing climatic conditions ([42]). The availability of locally adapted, high-performing provenances can help FRM better suit the challenges posed by harsh environmental conditions, particularly for species that must endure extended drought periods during early growth and throughout establishment. This approach is key to enhancing adaptive management strategies and ensuring the long-term success of reforestation and restoration efforts across varying climatic conditions.
Southern Europe is home to a broad diversity of species and ecotypes that have evolved under a range of environmental conditions, including adaptations to water deficits. In southern Italy, the Puglia region (latitude ranging from 41° 54′ N to 39° 48′ N) represents a rare blend of Mediterranean and Eastern vegetation elements, creating a diverse ecological mosaic. Numerous habitats in this region are protected under EU Directive 92/43/EEC. They are characterized by distinctive floristic and ecological features ([14]), and variations in the distribution and ecological optima of diagnostic species further emphasize their unique vegetation ([14]). Here, ecologically distinct Quercus species, with varying degrees of adaptation to environmental stress, coexist within a relatively small area of their natural range and offer a natural laboratory for investigating how different provenances respond to the intensifying challenges of water scarcity. Moreover, the Puglia region is among the least forested in Italy, and here the pressure from climate change is strong ([23]), as in other southern parts of the Mediterranean basin. In this context, the study aims to identify whether certain seed sources exhibit traits potentially advantageous under ongoing climate change. An initial screening was conducted on five ecologically diverse Quercus species from Puglia to evaluate early-stage responses to drought conditions by simulating extreme summer events typical of Mediterranean regions. In accordance with the Italian D. Lgs. 386/ 2003, which implements the European Directive 1999/105/EC, the seed sources are included in the national register as source-identified stands, except for Q. coccifera (L.), whose stands are included in the regional register of Apulia. This practical approach, based on officially recognized seed sources, provides preliminary screening to inform future, more detailed assessments of the adaptive potential of these provenances under changing climatic conditions. The seedlings belonged to a cluster of Mediterranean oak species, one deciduous, Quercus cerris (L.), two semi-deciduous, Quercus trojana Webb, Quercus ithaburensis subsp. macrolepis (Kotschy) Hedge & Yalt, and two evergreens, Quercus ilex (L.) and Quercus coccifera subsp. calliprinos Webb. Q. trojana and Q. ithaburensis subsp. macrolepis have the western limit of their range in Puglia. The selected species belong to the subgenus “Cerris”, but the deciduous and semi-deciduous species, Q. cerris. Q. trojana and Q. ithaburensis subsp. macrolepis belong to the “Cerris” section, while the evergreen Q. ilex and Q. coccifera subsp. calliprinos belong to the “Ilex” section ([12] ). The two sections are characterized by genetic and physiological differences: species of the “Ilex” section are sclerophyllous Mediterranean woody species with functional traits well adapted to drought ([35]). The species of the “Cerris” section could better face wetter conditions, allowing faster recovery after drought ([34]).
Materials and methods
Plant material
The seeds of the five studied Quercus species were collected in the fall of 2020 in Puglia, in ten seed sources - two stands per species (Tab. 1). The stands were selected based on recommendations from the Puglia Forest Regional Administration, considering officially registered seed sources at the regional and national levels, and including additional candidate stands identified for potential registration. The seeds were collected from 8 mother trees, spaced at least 50 m apart, and each tree was equally represented in the sample for each seed source. The study was carried out in the summer of 2021 in a field nursery area at the Department of Agriculture, Food, Environment and Forestry, University of Florence (43° 48′ 30.53″ N; 11° 12′ 01.46″ E), Firenze, Central Italy (annual mean temperature: 19.86 °C; annual mean precipitation: 870 mm).
Tab. 1 - Location, altitude, mean annual air temperature (MAT, °C), total annual rainfall (P, mm), and soil and vegetation type of the seed sources.
| Species | Seed sources | Coordinates | Altitude (m a.s.l.) | MAT (°C) | P (mm) | Soil type | Vegetation type | Hereinafter referred to as |
|---|---|---|---|---|---|---|---|---|
| Quercus cerris | Bosco San Cristoforo | 41°32′13.4″N 15°02′55.5″E | 720 | 14 | 770 | Regosols-Phaeozems | pure stand | San Cristoforo |
| Bosco Difesa Grande | 40°44′55.1″N 16°24′29.1″E | 380 | 15 | 608 | Phaeozems-Kastanozems / Regosols-Calcisols | Mixed stand of Q.cerris, Q. pubescens, and Q. frainetto | Difesa | |
| Quercus ilex | Bosco di Manfredonia- Masseria Scopino | 41°42′36.2″N 15°57′53.7″E | 750 | 16 | 434 | Leptosols/ Luvisols-Phaeozems | pure stand | Masseria |
| Bosco dei Cuturi | 40°20′29.2″N 17°39′20.6″E | 85 | 18 | 523 | Luvisols / Luvisols-Calcisols | pure stand | Cuturi | |
| Quercus ithaburensis subsp. macrolepis | Boschetto delle Vallonee | 39°55′29.9″N 18°22′49.1″E | 70 | 17 | 680 | Leptosols | pure stand | Tricase |
| Bosco Colemi | 40°32′35.7″N 17°56′15.7″E | 52 | 17 | 429 | Luvisols | pure stand | Colemi | |
| Quercus trojana | Masseria Palesi | 40°40′06.2″N 17°18′20.1″E | 420 | 15 | 836 | Regosols-Phaeozems | Mixed stand of Q. ilex, Q. trojana, and Q. pubescens | Palesi |
| Bosco Mesola | 40°50′59.5″N 16°46′26.2″E | 430 | 15 | 732 | Luvisols-Phaeozems | pure stand | Mesola | |
| Quercus coccifera subsp.calliprinos | Foresta Mercadante | 40°53′06.1″N 16°42′36.3″E | 420 | 17 | 783 | Luvisols-Phaeozems | Mixed stand of Q. pubescens and Q.coccifera | Mercadante |
| Bosco Occhiazzi | 39°59′45.2″N 18°13′32.8″E | 127 | 17 | 632 | Luvisols | Mediterranean scrubland with Q. coccifera | Occhiazzi |
Experimental conditions
From October 2020 to April 2021, the acorns were stored at 3 ± 0.5 °C in moist sand to simulate normal overwintering and to prevent acorn germination before the beginning of the experiment. In early April, acorns whose weights were within ±5% of the mean weight for each species were placed in wet sand at 18 °C. After 1 week of pre-germination, 350 randomly selected acorns with radicles <5 mm long were selected. The procedure was carried out to ensure that only viable acorns were sown. Square-based containers (18 × 18 cm, 23 cm deep) were used and filled with 0/40 peat, 3/8 fine pumice (3:1), and 1.5 kg m-3 calcium carbonate, with the addition of NPK (Mg) 17-09-11 (2) fertilizer with microelements. The containers were spaced 20 cm apart to avoid competition and to ensure full light for all seedlings. The containers were daily irrigated with 500 ml of water to maintain the substrate at the field capacity. Containers were initially watered to saturation, and then the excess water was drained away until the container capacity was reached ([6]). Changes in container weights were associated with changes in available water capacity, namely the amount of water retained in the soil reservoir that plants can remove ([25]).
The water-deficit experiment was performed in July 2021 (four months after seed germination), and 35 seedlings per species and seed source were selected for homogeneous morphology based on visual comparison. At the beginning of the experiment, 5 seedlings per species and seed source were sampled to estimate the plant biomass, in terms of fresh and dry weight ([8]). Thirty seedlings per seed source were selected and randomly assigned as (i) well-watered seedlings (control, C) and (ii) water-stressed, simulating drought, seedlings (treated, T). The treatment consisted of 21 days of water deficit, imposed by withholding irrigation (until the 21st day of treatment, DOT21). During water deficit, containers of T seedlings were covered with a plastic dome during rainy days to avoid water entry. At DOT21, eight seedlings for each species, seed source, and treatment (C and T) were sampled for biomass measurements and biochemical analyses. At DOT 22, the remaining seven T seedlings from each species’ seed source and treatment were rewatered with the same amount of water as the C seedlings and maintained under these conditions until DOT111. During the experiment, the soil volumetric water content (VWC, %) in C and T seedlings was monitored using a soil moisture meter (PCESMM1, PCE Instr. Corp., PCE Holding, Hamburg, Germany). The VWC was measured at DOT6, DOT13, DOT21, and DOT64 in T and C seedling pots.
Growth parameters
During the experiment, three destructive morphological surveys were performed on T and C seedlings at DOT1, DOT21, and DOT111, corresponding to the beginning of the experiment, the end of treatment, and the end of the growing season, respectively. Root collar diameter was determined by a caliper, and stem height was measured with a meter, whilst leaf number was counted, and total leaf area was measured by using the App Easy Leaf Area Free (freely available at ⇒ https://github.com/heaslon/Easy-Leaf-Area). Fresh weight and dry weight of the stem, leaves, taproot, and fine roots were determined using a scale before and after oven-drying samples to constant weight.
Chlorophyll content and fluorescence
The chlorophyll content and fluorescence were measured on seven seedlings for each species, seed source, and treatment at DOT6, DOT13, DOT21 (corresponding to increasing water-deficit duration), and DOT64 (43 days after the end of the stress). The chlorophyll content was measured on the first fully expanded leaf through the transmittance of red (650 nm) and infrared (940 nm) radiation using the SPAD 502 Plus® (Spectrum Technologies, Aurora, IL, USA - [46]). On the same leaf, away from the main leaf vein, the Chlorophyll-a fluorescence was measured by using a portable fluorimeter (Handy-PEA®, Hansatech Instruments, Ltd, UK) at ambient temperature after 20 minutes of adaptation of leaves to the dark. Changes in fluorescence were measured during a light pulse intensity of 3500 µmol m-2 s-1 and a duration of 1 s. Then, the maximum quantum efficiency of PSII photochemistry (Fv /Fm) and the performance index of PSII photochemistry (PI) were calculated.
Pigments concentrations
One leaf from each of three plants in each treatment was collected for pigment concentration measurements. A sample of 0.1 g of fresh leaves was homogenized with 1 ml of 95% Ethanol (Sigma-Aldrich, Milan, Italy). The homogenized sample mixture was centrifuged at 10.000 rpm for 15 minutes at 4 °C, and an aliquot of 0.05 ml was mixed with 9.5 ml of 96% Ethanol. The solution mixture was analyzed for Chlorophyll a (Chl a), Chlorophyll b (Chl b), and carotenoids (Car) content by a Spectrophotometer (Varian Cary Eclipse 50 Scan UV/Visible Spectrophotometer, Agilent, Santa Clara, CA, USA). To calculate the pigment concentration (mg g-1 FW), the following equations for 95% ethanol ([43]) were used (eqn. 1, eqn. 2, eqn. 3):
where Ax is the absorbance at the wavelength x = 470, 649, and 664 nm.
Pigment determination of Q. ilex and Q. coccifera subsp. calliprinos was not performed due to the limited availability of plant material.
Lipid peroxidation
Lipid peroxidation was measured following the method of Hodges et al. ([21]) with modifications. Briefly, leaf samples (0.120 g) were homogenized in 0.6 ml of 0.1 % (w/v) trichloroacetic acid (TCA). The homogenate was centrifuged at 15.000×g for 10 min (4 °C). A 0.5 ml aliquot of the supernatant was mixed with 1.5 ml of 0.5% (w/v) thiobarbituric acid (TBA) in 20 % (w/v) TCA (+TBA). To another aliquot of 0.5 ml, only 1.5 ml of 20% TCA was added (-TBA). The two mixtures were heated at 95 °C for 30 min and then quickly cooled in an ice bath. After centrifugation at 15.000×g for 5 min, the absorbance of the supernatant was recorded at 440, 532, and 600 nm for samples with and without the TBA reaction.
Lipid peroxidation was measured in terms of malondialdehyde (MDA) concentration, a product of lipid peroxidation, applying the following equations ([21] - eqn. 4, eqn. 5, eqn. 6):
where MDA equivalents (MDAeq) are expressed in nmol ml-1.
Statistical analysis
Means and standard errors of the measured parameters were calculated per species, seed source, and treatment. Analysis of variance (ANOVA) at a significance level of 0.05 was performed on morphological and physiological data to assess species responses at specific times throughout the experiment using the software OriginPro® ver. 8 (OriginLab Corporation, Northampton, UK). Two-way ANOVA was performed for nondestructive sampling parameters (Fv/Fm, SPAD values, stem height, root collar diameter, stem increment, leaf number, PI) to identify differences across treatments and seed source. Two-way ANOVA was also performed to assess differences between treatment and seed source effects on destructive sampling (biomass and total leaf area, chlorophylls and carotenoids concentrations, MDA) at DOT21. Significant effects were evaluated with the least significant difference (LSD) post hoc test.
Relationships between biomass and PI and between height increment and Fv/Fm were calculated at the species level and tested for significant differences between regression coefficients across phylogenetic sections of Quercus (significance values are shown in Tab. S1-Tab. S5 in Supplementay material). Statistical analysis was performed using the software OriginPro® v. 8 (OriginLab Corporation, Northampton, UK)
Results
The intensity of the water deficit was defined as the difference between the VWC values of T and C seedlings. In more detail, the VWC in T pots was 25%, 50%, and 70% lower than in C pots at DOT6, DOT20, and DOT21, respectively. At the end of the experiment, none of the species or seed sources considered exhibited seedling mortality. The growth, physiology, and metabolic parameters were monitored during the experiment, revealing statistically significant species-specific behaviors, as described below. Moreover, the two seed sources considered for each species showed significant differences in seed stands/provenances across traits.
Quercus cerris
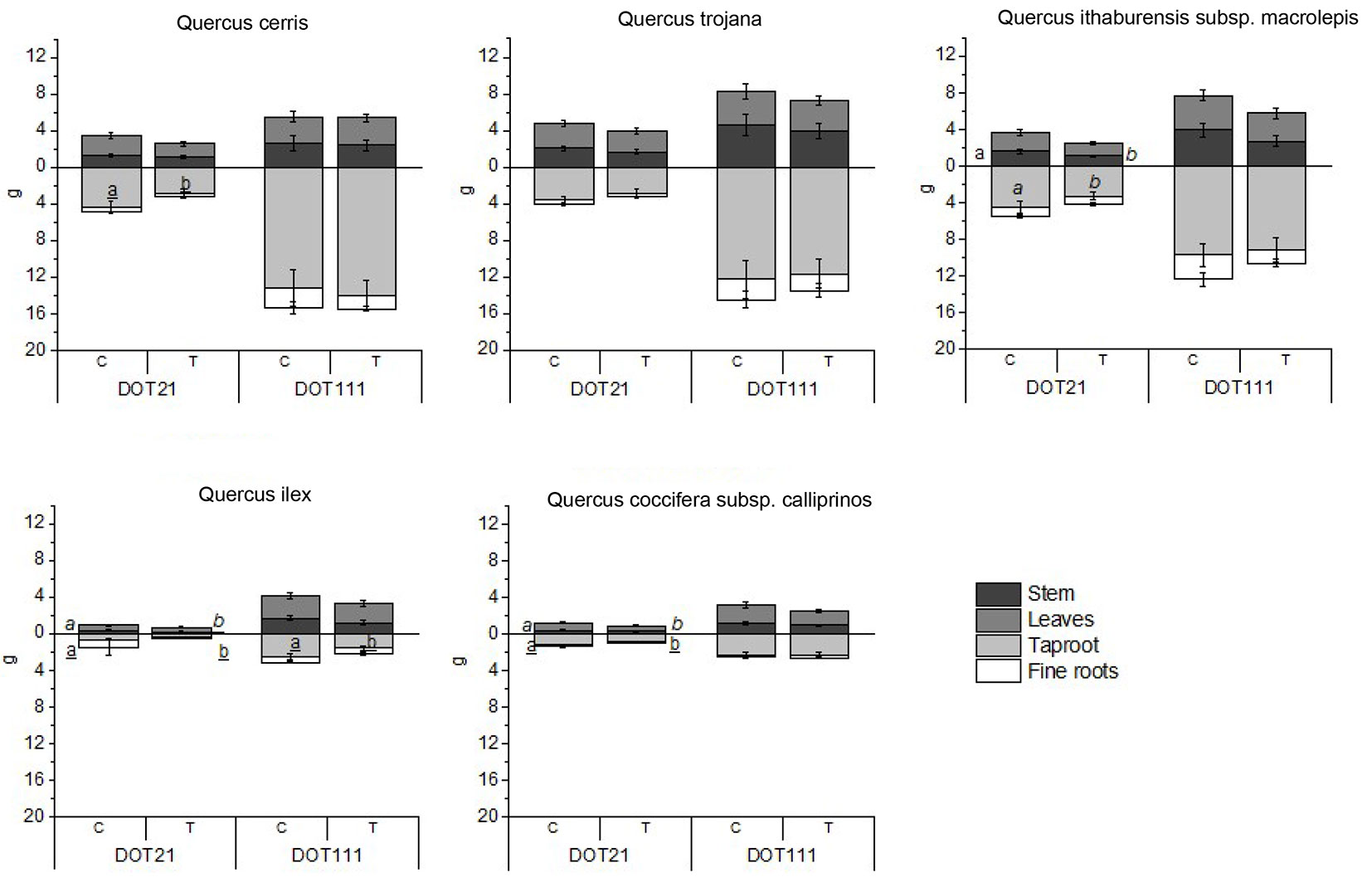
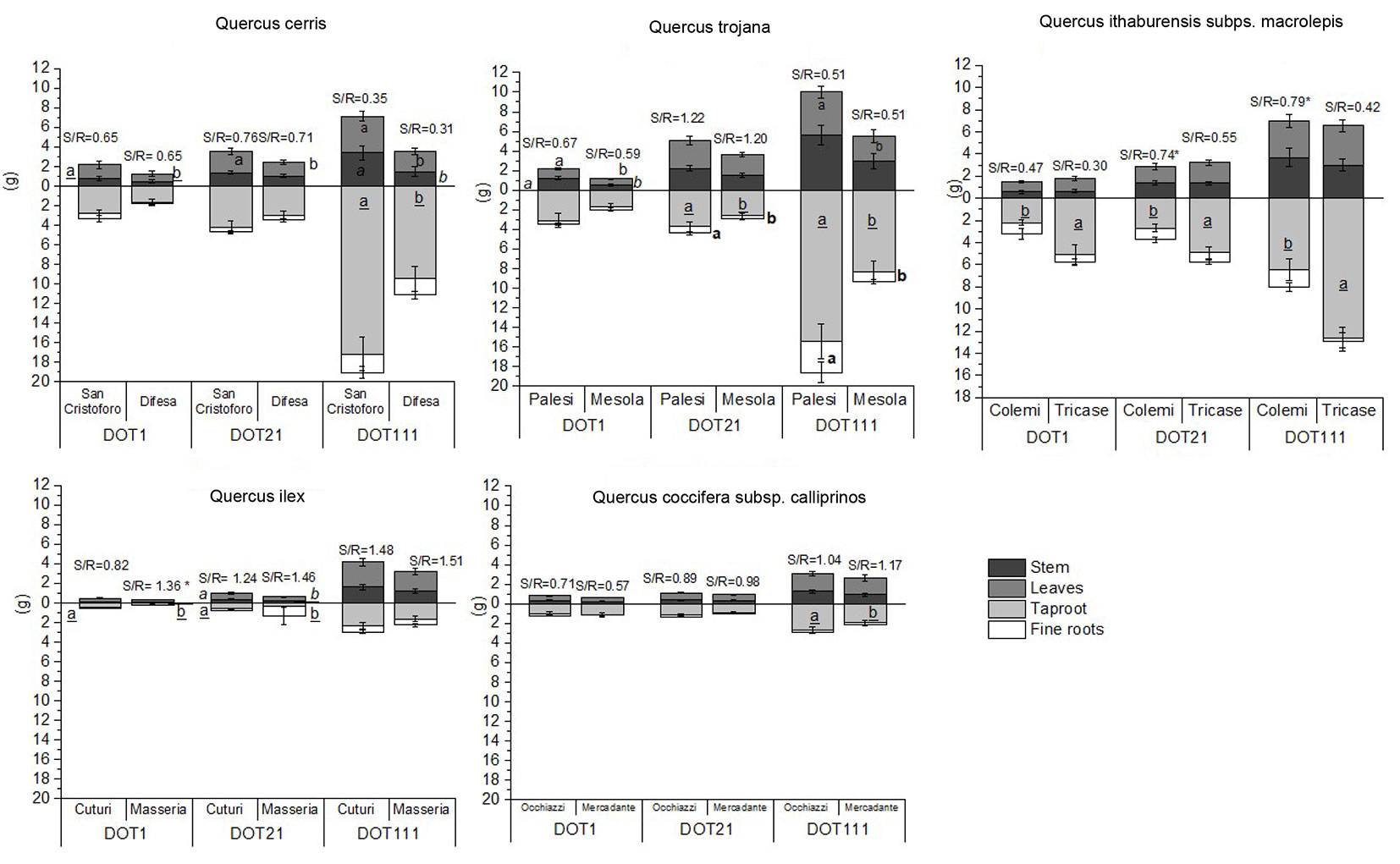
Tab. S1 (Supplementary material) shows the results concerning Q. cerris. At the end of the treatments (water deficit - DOT 21), T seedlings showed lower values of taproot dry weight and total biomass than C ones (Fig. 1). Furthermore, a significant increase of SPAD at DOT13 in T seedlings was recorded compared to C ones (Tab. S1). At DOT64, T seedlings showed lower Fv/Fm values than C seedlings (Tab. S1). At DOT13, Q. cerris showed higher performance index (PI) values in T seedlings, while at DOT64 the PI was higher in C seedlings (Tab. S1). At DOT21, C seedlings showed higher values of chlorophyll a and carotenoids than T ones (Tab. S1). Observing the seed sources, Q. cerris “San Cristoforo’’ showed higher biomass for taproot (DOT1 and DOT111), leaves (DOT21 and DOT111), and stem (DOT111) than “Difesa” (Fig. 2). At DOT6, “San Cristoforo” showed higher values of Fv/Fm than “Difesa’’ (Tab. S1), whereas at DOT21, “Difesa” showed higher concentrations of the pigments and MDA than “San Cristoforo” (Tab. S1).
Fig. 1 - Mean biomass (± standard error) (g, DW) of stem, leaves, taproot, and fine roots of Q. cerris, Q. trojana, Q. ithaburensis subsp. macrolepis, Q. ilex, and Q. coccifera subsp. calliprinos. For each species, the graphs show the mean values for the two days (DOT21: post water removal; DOT111: end of growing season), highlighting the significance of the treatment (C: control; T: treatment).
Fig. 2 - Mean biomass (± standard error) (g, DW) of stem, leaves, taproot, and fine roots of Q. cerris, Q. trojana, Q. ithaburensis subsp. macrolepis, Q. ilex, and Q. coccifera subsp. calliprinos. For each species, the graphs show the mean values over the three days (DOT1, DOT21: post-water removal; DOT111: end of growing season) to highlight the significance of the seed source.
Quercus trojana
As shown in Tab. S2 (Supplementary material) at DOT21, root collar diameter values were lower in T seedlings. Q. trojana showed lower values of SPAD at DOT6 and higher values at DOT21 in T than C seedlings (Tab. S2). At DOT 21, the C seedlings showed higher concentrations of chlorophyll a and carotenoids than the T ones (Tab. S2). Regarding seed sources and not treatment conditions, Q. trojana showed the higher dry weight of leaves (DOT1 and DOT111), stem (DOT1), taproot, fine roots, and total biomass (DOT21 and DOT111) in “Palesi” than “Mesola” (Fig. 2). Lower values of the leaf number were in “Mesola” at DOT1, DOT21 and DOT111 (Tab. S2), and lower values of root collar diameter at DOT111 than in “Palesi” seedlings (Tab. S2). “Mesola” showed higher values of SPAD at DOT6 and DOT13 (Tab. S2), and higher levels of the pigments at DOT21 than “Palesi” (Tab. S2).
Quercus ithaburiensis subsp. macrolepis
Tab. S3 (Supplementary material) details the results of Q. ithaburiensis subsp. macrolepis. The C seedlings had higher values of the dry weight of stem and taproot at DOT21 than the T ones (Fig. 1). This species showed values of Fv/Fm higher at DOT13 in T than C seedlings (Tab. S3). At DOT21, chlorophyll a, carotenoids, and MDA concentrations were higher in the T than in C seedlings (Tab. S3). Regarding seed sources, “Tricase” showed higher values of the tap root dry weight at DOT1, DOT21, and DOT111, lower shoot/root ratio at DOT21 and DOT111, and lower values of root collar diameter at DOT21 than “Colemi” (Fig. 2). At DOT6, SPAD values were higher in “Tricase” than in “Colemi” (Tab. S3). In Q. ithaburiensis subsp. macrolepis, the seed source “Tricase” showed higher values of chlorophyll a than “Colemi” at DOT21 (Tab. S3).
Quercus ilex
Tab. S4 (Supplementary material) shows the results regarding Q. ilex. Lower values of the stem (DOT21) and taproot (DOT21 and DOT111) dry weight were found in T seedlings than in C ones (Fig. 1). At DOT21, the height increment was lower in T than in C seedlings (Tab. S4). At DOT13, T seedlings showed higher values of the performance index (PI) than C seedlings (Tab. S4). By considering the seed sources, a higher shoot/root ratio at DOT1 and a higher stem and taproot dry weight at DOT21 were found in “Cuturi” than in “Masseria” (Fig. 2). Moreover, “Masseria” showed lower values of stem increment at DOT111 (Tab. S4) and lower values of root collar diameter at DOT21 (Tab.S4). At DOT21, “Cuturi” showed higher values of Fv/Fm, and “Masseria” had higher values of PI (Tab. S4).
Quercus coccifera subsp. calliprinos
Quercus coccifera subsp. calliprinos results are shown in Tab. S5 (Supplementary material). Lower values of stem, taproot, and total biomass dry weight were found in T than in C seedlings at the end of the water stress period (DOT21 - Fig. 1). The stem increment was lower in T seedlings at DOT111 (Tab. S5), and a lower root collar diameter was found in T seedlings of Q. coccifera supsp. calliprinos at DOT21 (Tab. S5). The effect of seed sources on Q. coccifera subsp. calliprinos was observed with higher values of tap root dry weight at DOT111 in “Occhiazzi” (Fig. 2), and higher values of PI in “Occhiazzi” at DOT13 than the other seed source (Tab. S5).
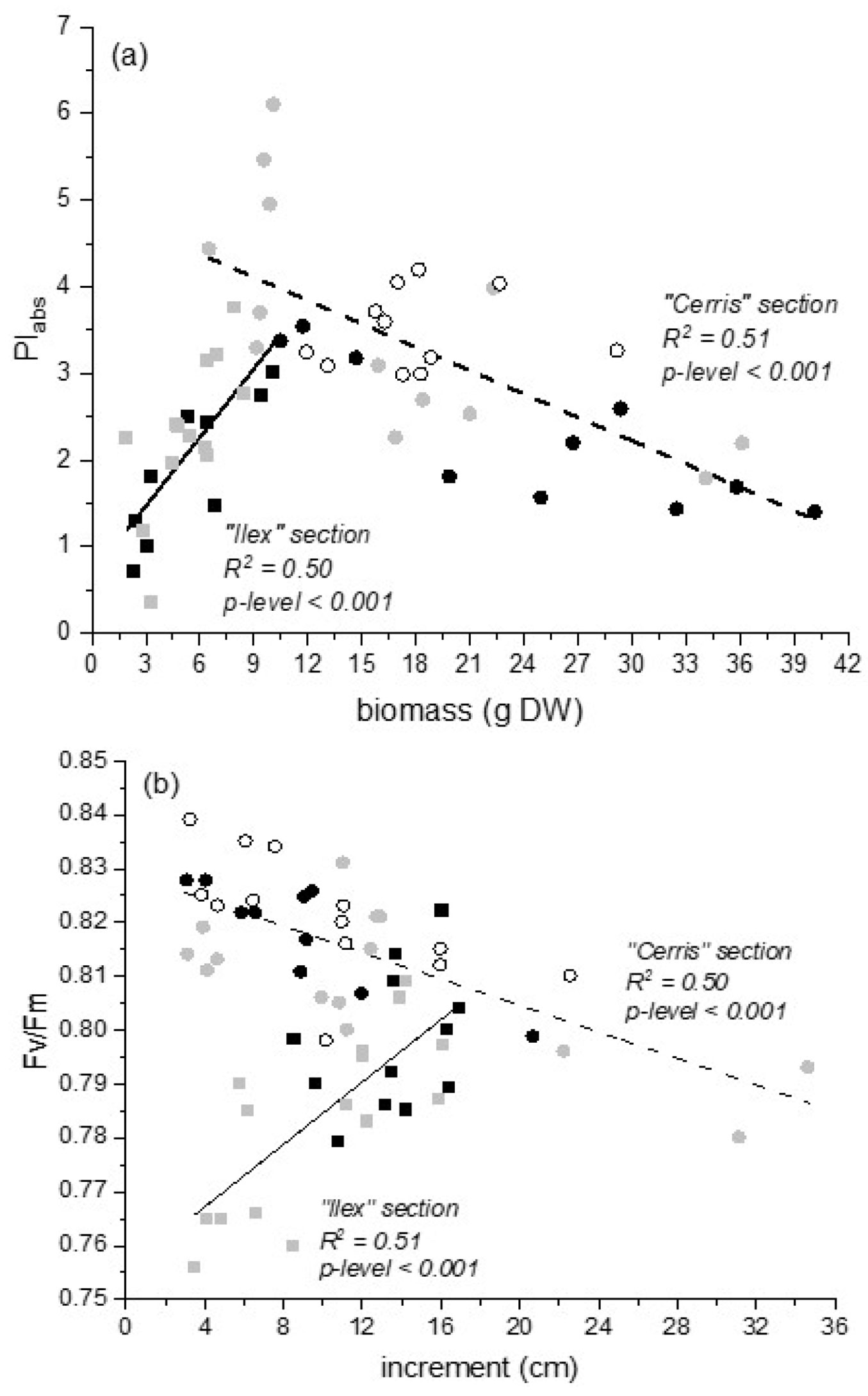
At the end of the recovery (DOT111), the relationships between growth and physiological parameters differed between the “Ilex” and “Cerris” sections. Biomass correlated with PI (Fig. 3a) and increment correlated with Fv/Fm (Fig. 3b) positively and negatively in “Ilex” and “Cerris” sections, respectively.
Fig. 3 - Relationships between the biomass and PI (a) and between the increment and Fv/Fm (b) into two different intrageneric groups of Quercus: “Ilex” intrageneric group - Q. ilex (black squares) and Q. coccifera subsp. calliprinos (grey squares); “Cerris” intrageneric group - Q. cerris (black circles), Q. trojana (grey circles), and Q. ithaburensis subsp. macrolepis (white circles) at the end of the recovery period. Significant correlations are indicated by p-level.
Discussion
Prior studies ([28]) have emphasized species-specific responses to water deficit within the genus Quercus, including Q. ilex, Q. cerris, Q. trojana, and Q. ithaburiensis subsp. macrolepis ([37], [48]). The results showed that the water treatment had limited effects on the studied morphological and physiological traits of Quercus seedlings. Moreover, both stressed and control seedlings showed similar height and biomass growth, with values comparable to those reported in other nursery studies on oak species grown under non-stress conditions ([44], [19], [9]). Photosynthetic activity and pigment content were mainly affected in Q. ithaburiebsis subsp. macrolepis, with low effects in the other species. Generally, biotic or abiotic stresses reduce Fv/Fm values ([37], [48], [39]) and the performance index ([2]), indicating damage to the photosynthetic mechanism and photoinhibition. Therefore, the Fv/Fm results provided insights into seedlings’ ability to tolerate the applied experimental constraints by preserving damage to the light reaction center. Likewise, chlorophyll content, assessed as SPAD values, did not differ under water deficiency, as observed in droughted Q. ilex plants ([18]). Water deficit reduced the concentrations of chlorophylls and carotenoids in Q. cerris and Q. trojana, as reported in previous drought studies ([5], [24]). The opposite behavior was observed in Q. ithaburiensis subsp. macrolepis, in which Chl a, Chl b, and carotenoid concentrations increased under water deficit. The increase in pigment concentrations might be an adaptive response to ensure the stability of functional mechanisms, thereby improving the photosynthetic rate and increasing biomass ([41]). The experimental water-deficit conditions did not result in structural or functional damage, highlighting the plants’ constitutive tolerance. However, we observed an increase in MDA concentrations in Q. ithaburensis subsp. macrolepis, which is consistent with other findings ([16], [22]). The increase of MDA concentrations in Q. ithaburiensis subsp. macrolepis might indicate damage to cell membrane phospholipids through lipid peroxidation induced by ROS accumulation due to the experimental drought conditions.
In this group of deciduous and semi-deciduous species (Q. cerris, Q. trojana, Q. ithaburiensis subsp. macrolepis), belonging to the “Cerris” section, the seed source had a significant effect on the development of functional morphological traits (e.g., biomass, taproot). This was observed at the end of the growing season in Q. cerris, whereas in Q. trojana it appeared at the end of the water-deficit stage and, by the end of the experiment, this affected a greater number of morphological parameters (leaves, taproot, and fine roots). On the other hand, for the evergreen Q. ilex and Q. coccifera subsp. calliprinos, both belonging to the “Ilex” section, the provenance effect was not significant, excluding a few morphological parameters in Q. ilex at the end of the water limitation treatment. However, both species experienced the negative effect of treatment on some functional morphological parameters (e.g., taproot) and, in Q. ilex, also physiological, even though their growth at the end of the season was not affected and resulted in line with values recorded on oak species grown in a nursery in Italy ([30]). Drought can change the dynamics of biomass accumulation in Mediterranean species ([36]); under water stress, lower biomass accumulation is expected in Quercus species, as observed in Q. ilex ([17]), Q. ithaburiensis subsp. macrolepis ([17], [3]), Q. cerris ([10]). Stem height increment was not affected by the treatment except in Q. ilex, where it has been observed as a highly sensitive parameter to a reduced water supply in Quercus species ([49]).
Quercus species displayed different strategies, demonstrating a positive relationship between growth and physiological parameters in the evergreen “Ilex” section and a negative relationship in the “Cerris” section. These findings can help define strategies for Quercus groups that differentiate evergreen species with low biomass growth from deciduous and semi-deciduous species with high biomass accumulation. Investigating chlorophyll fluorescence parameters (maximum quantum yield of photosystem II, Fv/Fm, and performance index based on absorption, PI) revealed similar species clustering within the sections. Healthy plants usually achieve a maximum Fv/Fm value of approximately 0.83 ([11]), whereas lower values have been observed when samples are exposed to biotic or abiotic stress ([39]). All species and seed sources within each species not only survived the water-deficit period but also maintained satisfactory morphological and physiological responses. It is worth noting that this experiment was conducted with seedlings grown in containers, which restricts the volume accessible to their roots; by contrast, seedlings developing roots freely in the soil can explore a larger volume and reach deeper, moister layers. Despite a more pronounced influence of seed sources observed in the “Cerris” section than in the evergreen “Ilex” section, both provide valuable materials for in-depth study and serve as initial indicators of suitable species and provenances for afforestation or forest restoration in the Mediterranean region. The results of the Q. cerris provenances were particularly interesting, as this species is usually considered less resistant to water limitation than the other studied Mediterranean oaks ([4], [29]).
Conclusions
Improving forest resilience to ongoing climatic changes requires identifying promising seed sources that provide seedlings better able to withstand prolonged summer droughts. Selecting FRM with higher drought tolerance represents a crucial step for ensuring the long-term success of afforestation and forest restoration initiatives in Mediterranean environments. The results of this preliminary screening highlight the potential of several Quercus provenances from Puglia as valuable candidates for future studies and for practical use in the production of resilient planting material. Quercus species (Q. cerris, Q. trojana, Q. ithaburensis subsp. macrolepis, Q. ilex, and Q. coccifera subsp. calliprinos) survived the 21-day water deficit. The experimental water deficit induced only limited changes in morphological and physiological traits, suggesting a high level of constitutive tolerance in these seedlings. Species maintained their light reaction center functionality, as evidenced by stable chlorophyll fluorescence (Fv/Fm) values. However, the study revealed distinct survival strategies between the two sections. The “Cerris” section exhibited a negative relationship between biomass growth and physiological parameters at the end of the recovery period. Nevertheless, seed source effects were significantly more pronounced in this section. On the other hand, the “Ilex” section showed a positive relationship between growth and physiology; although they experienced some negative effects on taproot biomass during stress, they recovered fully by the end of the season. Adaptive species-specific responses were observed. Despite being traditionally viewed as less drought-tolerant, Q. cerris from Puglia showed high performance, identifying this region as a promising source for resilient provenances. Whereas Q. ithaburenisis subsp. macrolepis showed a unique adaptive response by increasing pigment concentrations (chlorophyll and carotenoids) under stress to stabilize its photosynthetic mechanism, although it also showed signs of lipid peroxidation (increased MDA levels). Therefore, in terms of practical implications for restoration, the Puglia region, where Eastern and Western Mediterranean vegetation converge, serves as a “natural laboratory” for identifying seed sources that can withstand extreme aridity. The identified seed sources, Forest Reproductive Material (FRM), are valuable candidates for assisted migration and forest restoration projects aimed at increasing resilience to climate change in the Mediterranean basin. These findings support the inclusion of these provenances in the National Register to ensure the availability of climate-adaptive planting material. Further research should deepen the understanding of the physiological and genetic mechanisms underlying drought tolerance to support the selection and registration of well-adapted seed sources for sustainable forest management.
Author Contributions
Conceptualization: B.M., A.M., A.G., and C.C.; methodology: B.M., A.M., A.G., and C.C; investigation: L.M., M.L.T., B.E.M.; resources: A.M., A.G.; data curation: L.M., B.E.M. and M.L.T.; writing - original draft preparation: L.M., B.M., C.C. and B.E.M.; writing - review and editing: L.M., B.M., A.M., F.S., M.L.T., A.G., B.E.M and C.C.; funding acquisition: A.M. and A.G. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by Regione Puglia funding “Individuazione e descrizione di piante madri per la produzione di materiale di propagazione certificato di specie forestali destinate ad interventi di rinaturalizzazione. Impostazione di campi collezione ex-situ di arboreti da seme”, 2019 and 2022.
This work is also supported by a project funded under the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.4 - Call for tender no. 3138 of 16 December 2021, rectified by Decree no. 3175 of 18 December 2021 of the Italian Ministry of University and Research, funded by the European Union - NextGenerationEU; Project code: CN_00000033, Concession Decree no. 1034 of 17 June 2022 adopted by the Italian Ministry of University and Research, CUP B83C22002930006, Project title “National Biodiversity Future Center - NBFC”.
Data Availability Statement
Data are available upon request to the authors.
Acknowledgements
The Authors thank Sofia Martini and Fabio Bandini for their contribution to the lab activities.
Conflicts of Interest
The authors declare no conflict of interest.
References
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Barbara Mariotti 0000-0003-3779-2104
Alberto Maltoni 0000-0002-9057-1266
Claudia Cocozza 0000-0002-0167-8863
Department of Agriculture, Food, Environment and Forestry (DAGRI), University of Florence, Florence (Italy)
Department of Agricultural Science, University of Sassari, Sassari (Italy)
Alessio Giovannelli 0000-0002-7217-7177
CNR - Institute of Research on Terrestrial Ecosystems (IRET), Sesto Fiorentino, FI (Italy)
NBFC - New Biodiversity Future Center, 90133, Palermo (Italy)
CNR - Institute for Sustainable Plant Protection (IPSP), Sesto Fiorentino, FI (Italy)
Corresponding author
Paper Info
Citation
Mondanelli L, Mariotti B, Maltoni A, Salbitano F, Traversi ML, Giovannelli A, Maserti BE, Cocozza C (2026). First-year seedlings respond to a 21-day summer water deficit: morphological and physiological insights of two populations of five Mediterranean Quercus species. iForest 19: 149-156. - doi: 10.3832/ifor4928-019
Academic Editor
Michele Colangelo
Paper history
Received: Jun 18, 2025
Accepted: Feb 24, 2026
First online: Apr 19, 2026
Publication Date: Apr 30, 2026
Publication Time: 1.80 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2026
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 862
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 180
Abstract Page Views: 312
PDF Downloads: 338
Citation/Reference Downloads: 0
XML Downloads: 32
Web Metrics
Days since publication: 50
Overall contacts: 862
Avg. contacts per week: 120.68
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
(No citations were found up to date. Please come back later)
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Growth, morphology, and biomass allocation of recently planted seedlings of seven European tree species along a light gradient
vol. 13, pp. 261-269 (online: 03 July 2020)
Research Articles
Links between phenology and ecophysiology in a European beech forest
vol. 8, pp. 438-447 (online: 15 December 2014)
Research Articles
Seed germination traits of Pinus heldreichii in two Greek populations and implications for conservation
vol. 15, pp. 331-338 (online: 24 August 2022)
Research Articles
Frost hardening in seedlings of four provenances of Norway spruce in response to simulated autumn conditions with decreasing and fluctuating temperatures
vol. 19, pp. 141-148 (online: 19 April 2026)
Research Articles
Tolerance to heavy metal stress in seedlings of three pine species from contrasting environmental conditions in Chile
vol. 9, pp. 937-945 (online: 12 August 2016)
Research Articles
The effect of seed size on seed fate in a subtropical forest, southwest of China
vol. 9, pp. 652-657 (online: 04 April 2016)
Research Articles
Seed trait and rodent species determine seed dispersal and predation: evidences from semi-natural enclosures
vol. 8, pp. 207-213 (online: 28 August 2014)
Research Articles
Drought tolerance in cork oak is associated with low leaf stomatal and hydraulic conductances
vol. 11, pp. 728-733 (online: 06 November 2018)
Research Articles
Assessment of cadmium tolerance and phytoextraction ability in young Populus deltoides L. and Populus × euramericana plants through morpho-anatomical and physiological responses to growth in cadmium enriched soil
vol. 10, pp. 635-644 (online: 01 June 2017)
Research Articles
Matching seedling size to planting conditions: interactive response with soil moisture
vol. 12, pp. 220-225 (online: 25 April 2019)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword