Frost hardening in seedlings of four provenances of Norway spruce in response to simulated autumn conditions with decreasing and fluctuating temperatures
iForest - Biogeosciences and Forestry, Volume 19, Issue 2, Pages 141-148 (2026)
doi: https://doi.org/10.3832/ifor5041-019
Published: Apr 19, 2026 - Copyright © 2026 SISEF
Research Articles
Abstract
The Norway spruce (Picea abies Karst.) is Europe’s most important conifer, both economically and ecologically. In Norway, it dominates regions such as Trøndelag, Eastern Norway, and Helgeland. With climate change leading to warmer and more variable autumn temperatures, understanding how these fluctuations affect the development of frost hardiness in seedlings is critical for successful forestry. This study investigated the autumn frost hardiness of first-year Norway spruce seedlings from four provenances: Undesløs seed orchard (latitude: 60.7° N, elevation: 140 m a.s.l.), L1 (63.3°- 65°N, 0-149 m), M4 (64°- 65°N, 350-449 m), and the long-distance transferred provenance Cv1 (59.1°-60.2°N, 0-149 m). Seedlings were grown under two controlled conditions: (i) a “typical autumn” with decreasing temperatures and (ii) an “atypical autumn” with fluctuating temperatures. In the “typical autumn” setting, day and night temperatures were programmed to decrease gradually over four weeks. In the “atypical autumn” setting, temperatures varied throughout the same period. Frost hardiness was assessed using a -25°C freeze test and quantified via shoot electrolyte leakage (SEL). An index of injury (SELdiff-25) value ≤ 4% indicated sufficient frost tolerance. Results showed that all seedlings from provenances Cv1, M4, and Undesløs developed sufficient frost tolerance. For provenance L1, 19 seedlings per treatment had SELdiff-25 values below 4, while one seedling in each treatment showed higher values (5.5 and 5.1, respectively). Negative SELdiff-25 values were observed in some seedlings across all provenances, particularly from Undesløs. The results demonstrate that first-year Norway spruce seedlings can successfully develop frost hardiness under both decreasing and fluctuating autumn temperature regimes. These findings highlight the potential for nursery practices to replicate natural climatic patterns, thereby enhancing seedling frost resilience. However, differences between controlled growth chamber conditions and natural nursery environments must be considered when applying these results to practical forestry. Overall, this study provides valuable insights into the variability in frost hardiness often observed in field trials involving young Norway spruce.
Keywords
Apical Shoot, Climatic Conditions, Cold Hardiness, Conifers, Electrolyte Leakage, Freeze-test, Freezing Tolerance, Picea abies
Introduction
Norway spruce (Picea abies [L.] Karst) is the most important conifer species in Europe, both economically and ecologically ([3], [8]). In Norway, it accounts for approximately 44% of the growing stock (volume inside bark) in forests ([24]). Most spruce forests in Norway are regenerated by planting, and robust seedlings are essential for successful establishment. Each year, nearly 40 million Norway spruce seedlings are planted for forest regeneration, and about 7.2 million cubic meters of timber are harvested. In 2024, Norwegian nurseries supplied approximately 47.6 million seedlings, of which Norway spruce comprised 92% ([6]). In Trøndelag, commercial spruce seedlings are produced by Skogplanter Midt-Norge (SPMN).
Frost hardiness refers to the minimum subfreezing temperature a seedling can withstand without sustaining damage ([10]). Developing frost hardiness is critical in nursery culture because seedlings lacking adequate hardiness exhibit lower survival rates after field planting ([12]). The onset and development of frost hardiness in conifer seedlings depend on several factors, including seed origin ([5]), photoperiod, and temperature ([4], [10], [13], [23]).
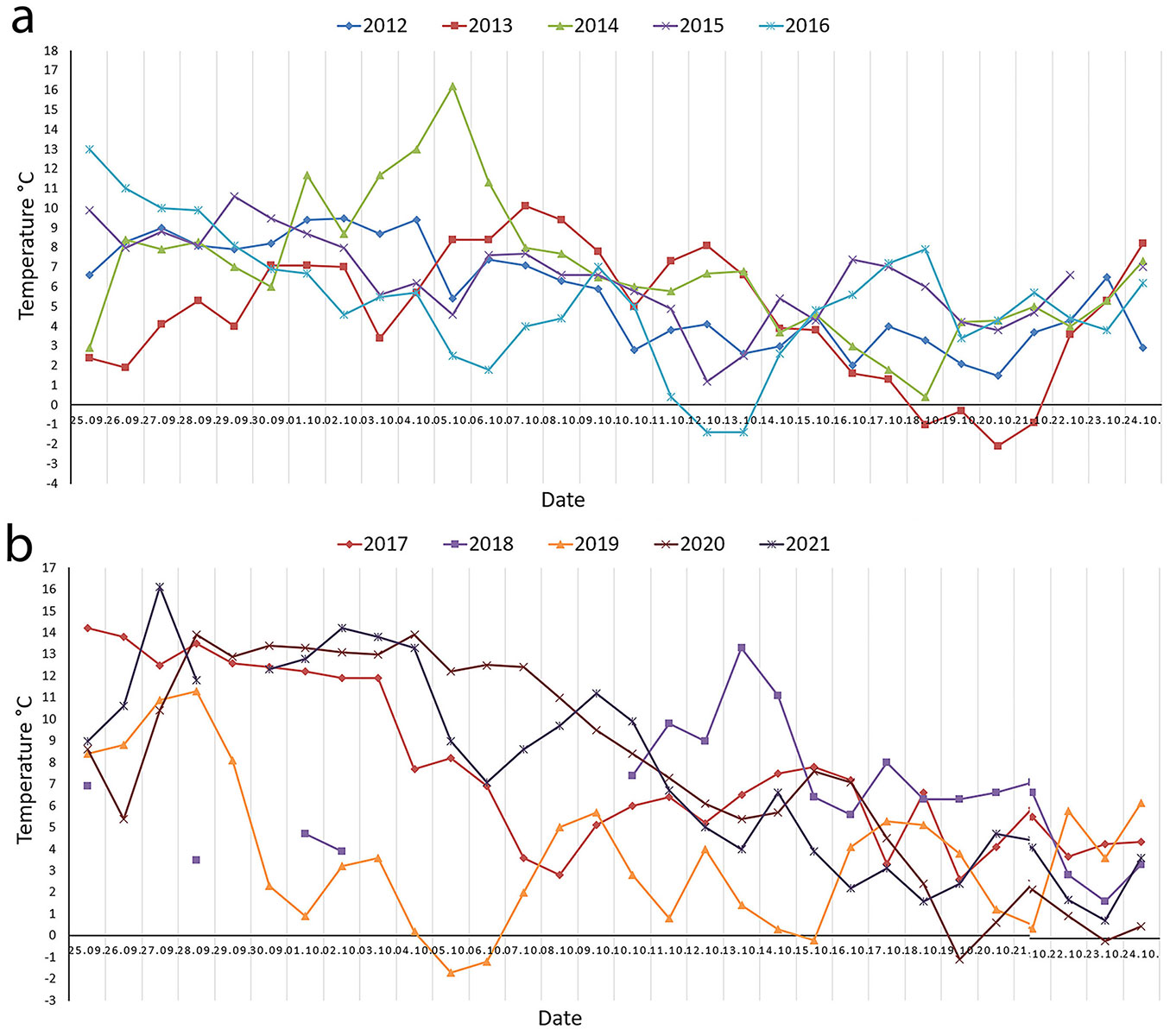
Norway’s annual temperature is projected to rise by approximately 3.4 °C by the end of the century if greenhouse emissions keep increasing ([18]). Between 1901 and 2024, the mean temperature increased by about 1.4 °C, while precipitation rose by 21%. Recent weather patterns near Kvatninga (Central Norway) during September and October have shown considerable variability in air temperature (Fig. 1). For instance, during the last week of September in 2014, temperatures ranged from 2.9 and 8.4°C (Fig. 1a), whereas in 2017, they ranged from 12.4 and 14.2 °C (Fig. 1b). Similarly, during the first week of October in 2012, temperatures ranged from 5.4 to 9.5 °C (Fig. 1a), while in 2020, they ranged from 12.2 and 13.9 °C (Fig. 1b). These fluctuations may influence frost hardiness development in seedlings from different provenances. However, limited information exists on how variable autumn temperatures affect the development of frost hardiness under standard nursery practices in seedlings from different provenances.
Fig. 1 - Daily mean air temperature (°C) recorded by meteorological stations near SPMN, Kvatninga (Central Norway) during the autumn season (from the end of September to the end of October). (a): From 2012 to 2016; (b): from 2017 to 2021. Meteorological data were obtained from MET Norway weather stations: Namsos Airport (2012) and Overhalla-Skogmo (2012-2021), available at ⇒ https://seklima.met.no/. Data was unavailable for these dates: in 2015 (23/10); in 2021: (29/09); and in 2018 (26/09 - 27/09, 29/09 - 30/09, and 03/10 - 09/10). The discontinuous temperature curve in 2018 is due to missing data for 26 and 27 September, 29 and 30 September, and from 3-9 October.
The main aim of this study was to evaluate differences in autumn frost hardiness among seedlings from four provenances: Undesløs seed orchard, L1 (recommended for the lowland areas of Mid-Norway), M4 (recommended for higher elevations in Mid-Norway), and Cv1 (recommended for southern Norway). We examined their response to two distinct autumn temperature regimes: decreasing temperatures and fluctuating temperatures. Our hypotheses were: (i) seedlings grown under decreasing temperatures will exhibit greater freeze tolerance than those grown under fluctuating temperatures; and (ii) frost hardiness development will vary among provenances originating from different latitudes and altitudes.
Material and methods
Seed material
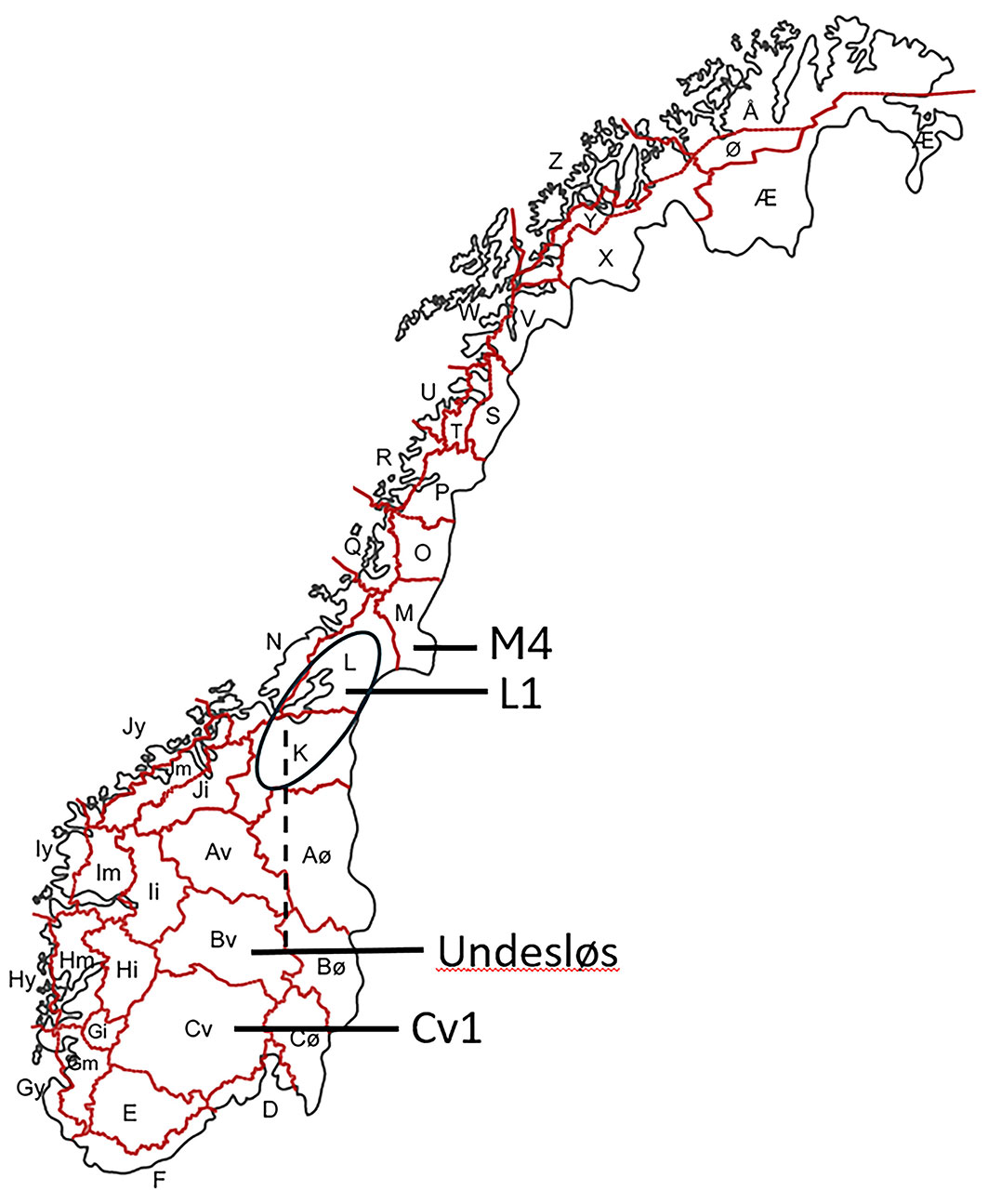
Seeds from four provenances (see Tab. 1) were obtained from the Norwegian Forest Seed Center, Hamar, Norway. For the northern regions of Trøndelag county, the recommended unimproved forest-stand seed sources (provenances) are L1 for lowland and M4 for higher elevations. L1 originates from forest stands located between 63.3° and 65° N, at elevations of 0-149 meters a.s.l. (Fig. 2). In contrast, M4 is collected between latitude 64° and 65° N at elevations of 350-449 meters a.s.l. (Fig. 2). Undesløs seed represents the improved seed source from the breeding program orchard (Norwegian Forest Seed Center). This orchard is assembled with tested and selected parents from the lowland areas between 63° and 65° N. Its southern seed orchard (location at 60.7° N) implies some pollen contamination from the surrounding forest. Additional seed sources for the region may include long-distance transfers of provenances from latitudes around 59-60° N, as previously suggested by Liziniewicz et al. ([15]). This is exemplified in the present study by Cv1, which originates from 59.1° to 60.2° N at elevations of 0-149 meters a.s.l.
Tab. 1 - An overview of seed material of different provenances. Mean annual temperature (MAT) and temperature sum >5 °C (ddeg) are estimated from interpolated 1×1 grid data obtained from the Norwegian Meteorological Institute for the period 1961-1990.
| Seed lot | Provenance | Place name | Crop year |
Latitude (N) |
Altitude (m a.s.l.) |
MAT (°C) |
ddeg (>5°C) |
Provenance description |
|---|---|---|---|---|---|---|---|---|
| F06-046 | Undesløs (seed orchard) |
Østre Toten | 2006 | 60.7° | 140 | 3.4 | 1100 | Seed orchard for lower elevations in Trøndelag, Norway |
| F07-002 | M4 | Lierne | 2006 | 64°-65° | 350-449 | 1.2 | 670 | The Northern (Trøndelag) natural stand provenance begins growth early, terminates early, and develops frost hardiness earlier than L1, Cv1, and Undesløs |
| F07-071 | Cv1 | Drangedal/Kragerø | 2006 | 59.1°-60.2° | 0-149 | 5.0 | 1300 | Southern low-elevation natural stand provenance: growth starts moderately late and terminates late |
| F07-073 | L1 | Steinkjer | 2006 | 63.3°-65° | 0-149 | 4.3 | 960 | Northern (Trøndelag) lower elevation natural stand provenance, adapted to Steinkjer |
Fig. 2 - Origin of provenances Cv1, L1, M4, and Undesløs. M4, L1, and CV1 were collected from natural stands within the seed zones shown on the map. The provenance Undesløs is a seed orchard located in seed zone Bv, fairly well isolated from surrounding pollination, and the parents originate from seed zones K and L.
Plant cultivation and fertilization
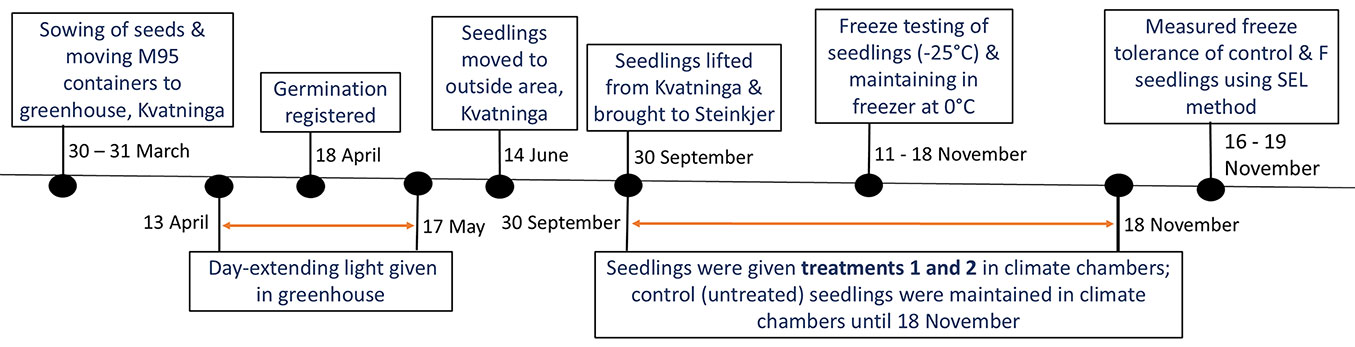
The experiments were conducted at the forest nursery (SPMN), Kvatninga, Overhalla (64.47° N, 11.65° E), both in the greenhouse and an outdoor area. Fig. 3 presents the experimental timeline. Seeds were sown on March 30, 2022, in base-fertilized Sphagnum peat mixed with 20% perlite (pH 4.5), supplied by Svenarums Torvprodukter AB, Sweden. Seeds were sown in multipot plastic containers, each pot having a volume of 50 cm3 corresponding to a seedling density of 791 m-2. One seed was placed in each pot. After sowing, a layer of perlite was added to the top surface of the plugs to protect the seeds, improve aeration, ensure good drainage, and reduce moisture evaporation. On March 31, the containers were transferred to the greenhouse, where they received extended day-length lighting from April 13 to May 17, between 23:00 and 03:00. On June 14, the containers with seedlings were relocated to an outdoor area at Kvatninga (see Fig. 3). Fig. S1 (Supplementary material) shows the daily mean and total air temperatures recorded between May 31 and September 30.
Fig. 3 - Experimental timeline, including the date of seedling lifting, application of autumn conditions with decreasing and fluctuating temperatures (Treatment 1 and Treatment 2) in two separate growth chambers, freeze-testing of seedlings at -25 °C, and assessment of freeze tolerance in both control and freeze-tested (F) seedlings using the SEL method.
Seedlings were watered once or twice per week from early April through the end of September. Fertilization consisted of a mixture of nutrient solutions: Pioneer NPK(Mg) Makro Blå® 14-3-23(3) (10% w/v), with a pH of 4.9 and an electrical conductivity (EC) of 1.2 µS cm-1, and Pioneer Micro® with iron (1.5% v/v). While the seedlings were grown outdoors (June 14 to August 12), they received six applications of Calcinit (10% w/v). Additionally, in June, seedlings were treated twice with Acadian, a biostimulant derived from Ascophyllum nodosum, at a concentration of 3.0 mL L-1. Throughout the growth period, light intensity was measured both inside the greenhouse and outdoors using the SpectraPen mini-SM® 110/UVIS (Photon Systems Instruments, PSI, Drasov, Czech Republic). Measurements were taken on randomly selected dates.
Seed germination was recorded on 18 April. The germination rate for provenance Cv1 was 89 %, for provenances M4 and Undesløs it was 84 %, and for provenance L1 it was 80 % (data not shown).
Growing conditions in growth chambers
On September 30, the multipot container with seedlings was transferred from the outdoor environment to growth chambers (SGC Standard Growth Chamber, Weiss Technik GmbH, Heuchelheim near Gießen, Germany). Each multipot container was placed on a tray inside the growth chambers. Using the S!MPATI® software (Weiss Technik - ⇒ https://weiss-na.com/product/simpati/), we simulated two distinct autumn conditions: Treatment 1, “typical autumn with gradually decreasing temperature”, and Treatment 2, “atypical autumn with fluctuating temperature” (see Tab. S1 and Tab. S2 in the Supplementary material). The autumn conditions were based on daily mean air temperatures recorded at meteorological stations near SPMN, Kvatninga, during the autumn period (September 25 - October 24) from 2012 to 2021 (Fig. 1). Treatment 1 (Tab. S1) represented a four-week autumn period (late September to late October) with a gradual decrease in day and night temperatures. In contrast, Treatment 2 (Tab. S2) mimicked the fluctuating day and night temperatures observed at Kvatninga during the same period (Fig. 1). For example, during the last week of September (25.09 - 30.09), temperatures varied considerably across years: in 2014, they ranged from 2.9 to 8.4 °C (Fig. 1a); in 2020, from 5.4 to 13.9 °C; and in 2021, between 9.0 and 12.3 °C (Fig. 1b). Similarly, in the second week of October (08.10 - 14.10), temperatures ranged from 2.6 to 6.3 °C in 2012 (Fig. 1a), 2.8 to 7.5 °C in 2017, and 4.0 to 11.2 °C in 2021 (Fig. 1b). In Treatment 1, temperature ranges were as follows: during the first week, 8-14 °C; the second week, 7-12 °C; third week, 6-10 °C; and fourth week, 4-7 °C (Tab. S1). In Treatment 2, temperatures fluctuated more strongly: first week, 7-11 °C; second week, 9-15 °C; third week, 4-7 °C; and fourth week, 4-11 °C (Tab. S2 in Supplementary material).
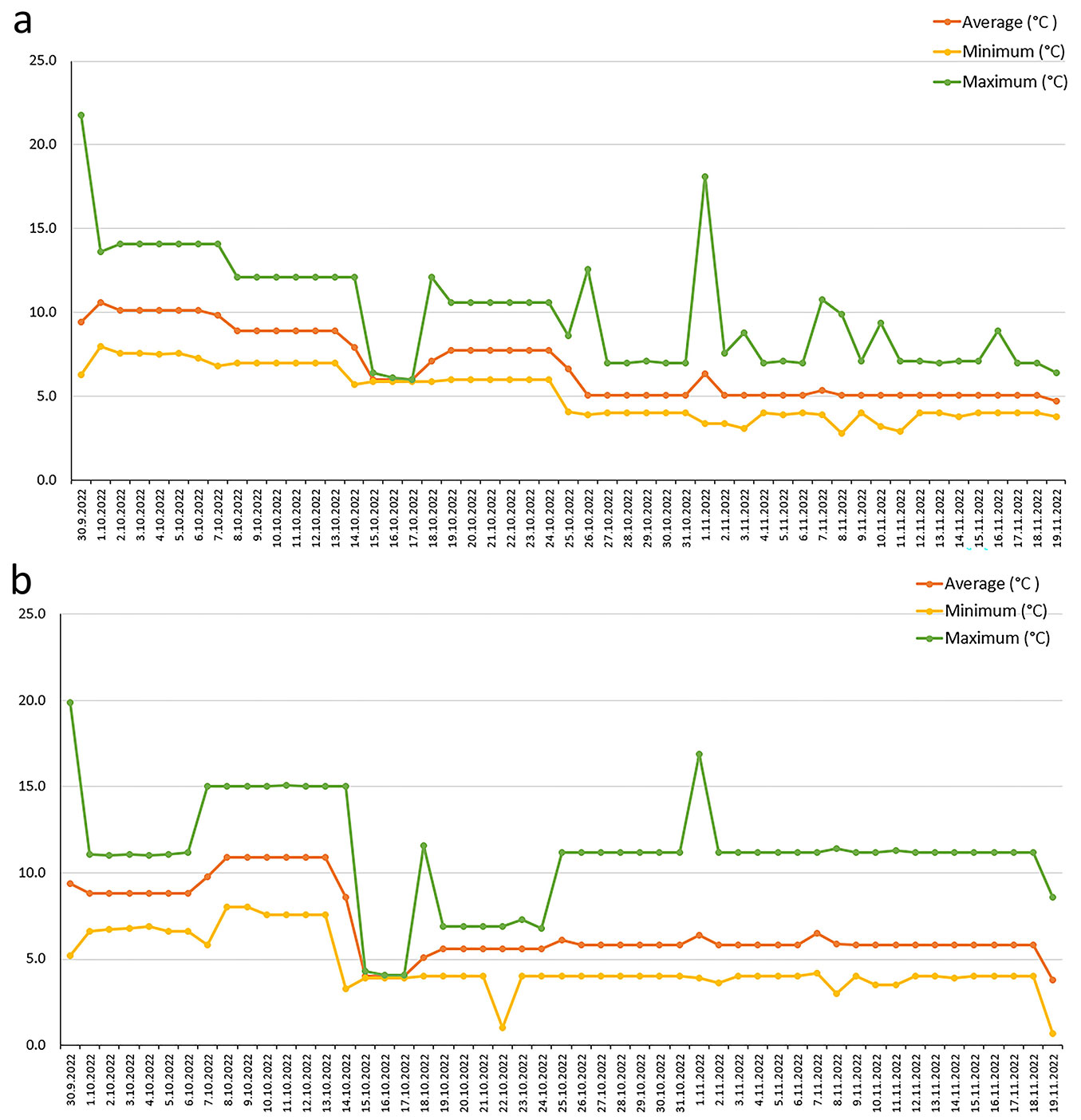
Daily average, minimum, and maximum temperatures from September 30 to November 19 for both treatments are shown in Fig. 4and detailed in Tab. S3 (Supplementary material). The cumulative temperature sums were nearly identical: 348.2 °C for Treatment 1 and 347.2 °C for Treatment 2 (Tab. S3). Due to technical issues, both Treatments were extended to 6-7 weeks instead of the planned 4 weeks. This extension was achieved by maintaining the environmental conditions from week 4 for an additional 10-11 days (see Fig. S2 and Fig. S3 in Supplementary material).
Fig. 4 - Average, minimum, and maximum temperatures (°C) recorded for each date from September 30 to November 19 (see also Tab. S3 in Supplementary material). (a): Temperatures recorded in growth chamber 1, where the Treatment 1 program was applied. (b): Temperatures recorded in growth chamber 2, where the Treatment 2 program was applied.
In both chambers, light intensity from cool and warm white lights was reduced weekly: 70% in week 1, 60% in week 2, 50% in week 3, and 40% in week 4 (Tab. S1, Tab. S2). Day length was shortened from 12 hours to 9 hours over four weeks, decreasing by one hour each week, while night length increased from 12 to 15 hours over four weeks. Relative humidity was maintained at 80% in both chambers (Tab. S1, Tab. S2). On October 18, due to issues with the S!MPATI software, the program for week 3 had to be restarted (see Fig. 4). Watering was done by adding 2 L of spring water to each tray on four occasions: September 30, October 18, November 1, and November 7. Changes in humidity and temperature were recorded on these dates, coinciding with the opening of the growth chambers for watering. These events resulted in a temporary drop in humidity and a rise in temperature (Fig. 4, see also Fig. S2 in Supplementary material).
The day-night cycles of temperature, light, photoperiod, and humidity recorded in two growth chambers from September 30 to November 19 are shown in Fig. S2 and Fig. S3. Light intensity in each chamber was measured using the SpectraPen mini-SM (Photon Systems Instruments). By adjusting the cool and warm white lights to 70%, seedlings received approximately 240 µmol m-2 s-1 of photon flux density (PFD) during the first week. This intensity was gradually reduced to 60% (~200 PFD µmol m-2 s-1) in the second week, 50% (~160 PFD µmol m-2 s-1) in the third week, and 40% (~120 PFD µmol m-2 s-1) in the fourth week.
Seedling freezing test
The experiment was designed to evaluate the effects of provenance, autumn temperature conditions (Treatments 1 and 2), and replication on seedlings’ frost hardiness. On November 11, seedlings from each provenance and treatments were divided into four groups (Tab. 2). Control seedlings from Groups 1 and 3 remained in the growth chambers until November 19. Seedlings from Groups 2 and 4 were frozen at -25 °C in a laboratory freezer, following established protocols ([9]). Frost hardiness was assessed by the extent of cellular damage following exposure to subzero temperatures. Seedlings were placed in polystyrene trays and covered with perlite to protect their roots, with a Styrofoam plate placed underneath. Using a programmable laboratory freezer (ZLN-T 300 Smart PRO®, POL-EKO, Poland), the temperature was gradually decreased from room temperature (21 °C) to 0 °C at a rate of 2 °C per hour. This temperature was maintained for three hours before being raised back to 0 °C at the same rate. During the freezing process, the needles were misted for ten seconds upon reaching 0 °C. After the freezing cycle, the seedlings were held at 0 °C in the same freezer for measurement of shoot electrolyte leakage (SEL), alongside the control (unfrozen) seedlings in growth chambers, as shown in Fig. 2. The temperature cycle recorded during the freezing test is shown in Fig. S4 (Supplementary material).
Tab. 2 - Seedling groups from both treatments for freeze testing at -25 °C.
| Provenance | Treatment 1 | Treatment 2 | ||
|---|---|---|---|---|
| Group 1 (Control) | Group 2 (freezing test) | Group 3 (Control) | Group 4 (freezing test) | |
| Cv1 | 20 | 20 | 20 | 20 |
| L1 | 20 | 20 | 20 | 20 |
| M4 | 20 | 20 | 20 | 20 |
| Undesløs | 20 | 20 | 20 | 20 |
Freezing tolerance with electrolyte leakage
The SEL of each individual seedling was assessed using the method described by Lindström et al. ([14]). This method is based on the principle that the primary site of injury in damaged tissues is the cell membrane. Loss of membrane integrity leads to diffusion of cytoplasmic electrolytes into water when tissues are immersed ([10]). SEL was determined from apical shoots of 20 control and 20 freeze-tested seedlings per provenance and from each treatment (Tab. 2). The upper 3 cm of each apical shoot was excised, washed once with spring water, and rinsed three times with deionized water. Each shoot was then immersed in 40 ml of deionized water in a 60 ml plastic bottle. Samples from control (unfrozen) and freeze-tested (-25 °C) seedlings were prepared in parallel. Bottles were shaken on an orbital shaker at ~150 rpm for 24 h in darkness at ~21 °C. Electrical conductivity (EC) was measured using an HQ 1140 conductivity meter with temperature compensation. We recorded EC for unfrozen control samples (ECcontrol) and freeze-tested samples (ECfreezed-25°C). Subsequently, all samples were autoclaved at 110 °C for 20 min at 1.2 bar, then cooled overnight at room temperature. A final EC measurement (ECboiled) was then taken. Natural cell leakage (SELcontrol) was calculated as the ratio of ECcontrol to ECboiled, following Lindström et al. ([14]). The index of injury (SELdiff-25) was calculated by subtracting the SEL value of a control seedling from that of a freeze-tested seedling: SELdiff-25 = SEL-25 - SELcontrol. This calculation was performed for 20 seedlings per provenance for each treatment group.
Data analysis
The data were analyzed using Minitab Statistical Software, v. 20.2 (Minitab LLC, PA, USA). To verify that SEL data met the assumptions of normality, independence, and homoscedasticity, we applied the Anderson-Darling test, Durbin-Watson statistic, and Bartlett’s test, respectively. A probability plot for SEL data indicated an approximately normal distribution. To assess differences between control and freeze-tested seedlings within each provenance and treatment, we conducted a One-Way ANOVA followed by Tukey’s test (p < 0.05). A mixed-effects model (model 1) was employed to evaluate SEL responses to fixed factors: provenance, Treatments 1 and 2, freeze-testing at -25 °C, and interactions between provenance by treatments, provenance by freeze-testing, and treatments by freeze-testing, with replicates treated as a random factor (eqn. 1):
where SELijkl is the observation for the i-th provenance (P) under the j-th treatment (T, 1 and 2), under the k-th freezing test (F, -25 °C and control), in the l-th replicate (R), PT, PF, and TF are the interaction terms, and eijkl is the residual effect, and μ is the average. R and e are considered random effects that are normally and independently distributed with mean = 0 and a variance (NID, σ2).
A mixed-effects model (model 2) was also applied to assess responses of SELdiff-25 to fixed factors: provenance, Treatments 1 and 2, and interaction between provenance by treatments, with replicates treated as a random factor (eqn. 2):
where SELijk is the observation for the i-th provenance (P) under the j-th treatment (T, 1 and 2), in the k-th replicate (R), PT is the interaction term, and eijk is the residual effect, and μ is the average. R and e are considered random effects that are normally and independently distributed with mean = 0 and a variance (NID, σ2).
Results and discussion
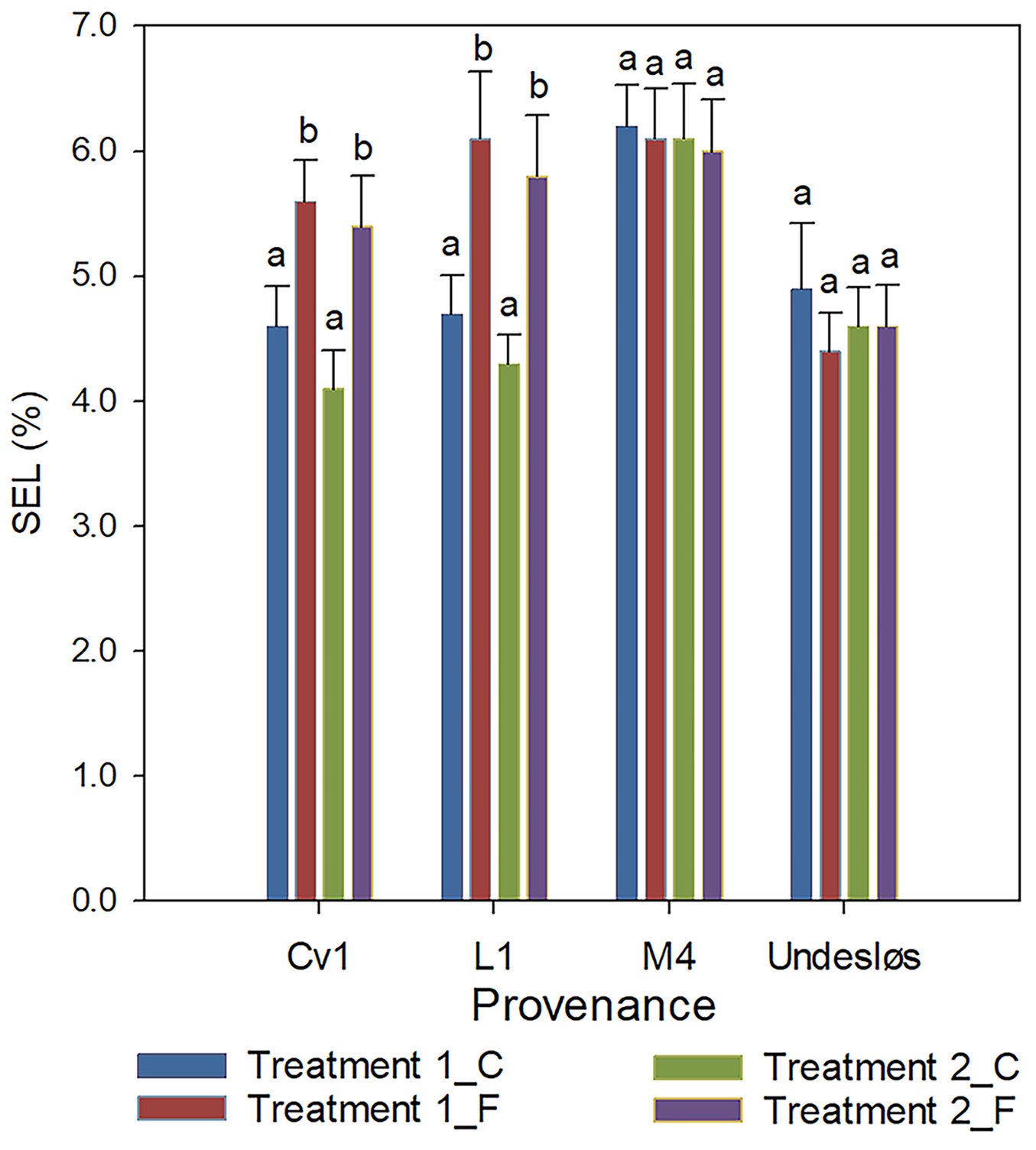
The SEL from the shoots of control (unfrozen) seedlings across four provenances ranged from 4.6% to 6.2% in Treatment 1 and from 4.1% to 6.1% in Treatment 2. SEL from unfrozen shoots is measured to evaluate the real effect of freezing ([14]), as disruption of cell membrane semi-permeability typically leads to increased leakage. The SEL from shoots of freeze-tested seedlings across four provenances ranged from 4.4% to 6.1% in Treatment 1 and from 4.6% to 6.0% in Treatment 2 (Fig. 5). One-way ANOVA analysis revealed that freeze-tested seedlings of provenances Cv1 and L1 exhibited significantly higher SEL values compared to their respective controls (p < 0.001). In contrast, no significant differences were observed between provenances M4 and Undesløs (Fig. 5). Across both treatments, SEL from control seedlings ranged from 4.1% to 6.2% (Fig. 5). These values are notably lower than those reported in previous studies. Sarvaš ([22]) found SEL values of 11%-13% in unstressed Norway spruce plants, while Malmqvist et al. ([16]) observed natural electrolyte leakage ranging from 12.5% to 29.5% across different species and provenances. The relatively low SEL observed in our study suggests that seedlings maintained under the two treatments experienced minimal stress prior to freezing, likely due to controlled climatic conditions.
Fig. 5 - Percentage of shoot electrolyte leakage (SEL) in control (unfrozen) and freeze-tested (-25 °C) seedlings from four provenances of Norway spruce subjected to Treatments 1 and 2. SEL in control (C) and freeze-tested (F) seedlings. Within each provenance, the F seedlings from Cv1 and L1 provenances had significantly higher SEL than control seedlings (as indicated by different letters above the bars), while no significant differences were found for provenances M4 and Undesløs (p < 0.05). Error bars represent 95% confidence intervals (n = 20).
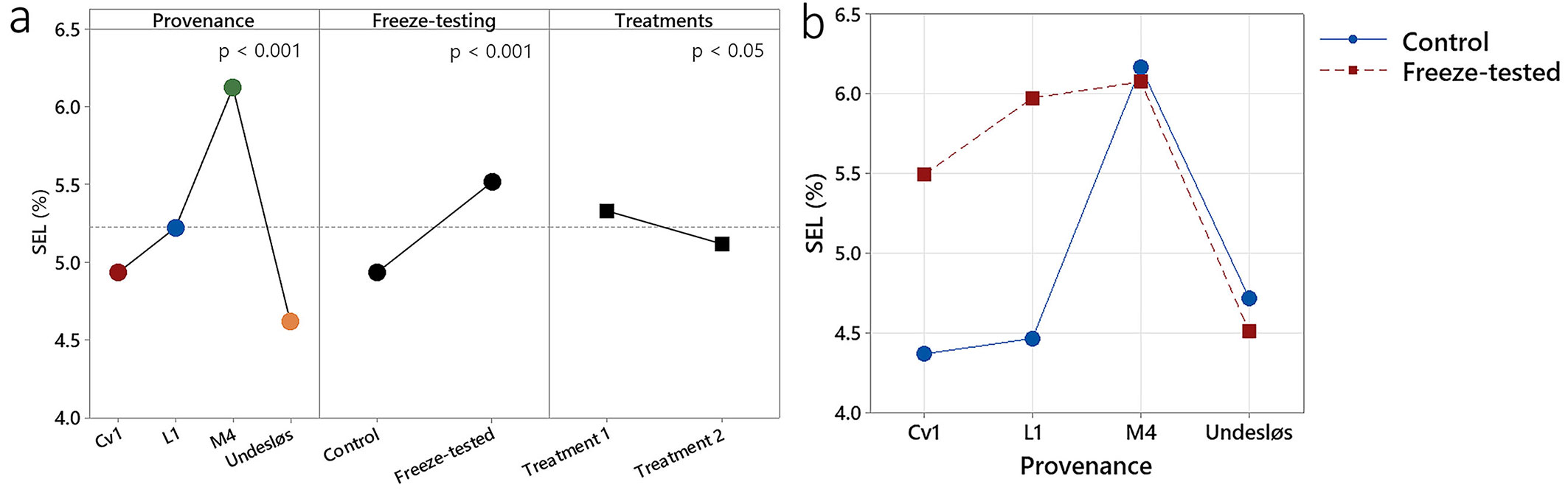
The mixed model analysis showed highly significant effects of both freeze testing and provenance on SEL (p < 0.001). There was also a significant effect of Treatment (1 vs. 2, p < 0.05 - Fig. 6a). Additionally, the interaction provenance × freeze-testing was statistically significant (p < 0.001 - Fig. 6b). All other interactions were insignificant.
Fig. 6 - Effects of provenance, freeze-testing, and treatments on SEL, including an interaction between provenance × freeze-testing. (a): Effects of provenance (p < 0.001), freeze-testing (p < 0.001), and treatments (p < 0.05) on SEL, where the dashed reference line represents the overall mean. (b): Effect of an interaction between provenance × freeze-testing on SEL (p < 0.001).
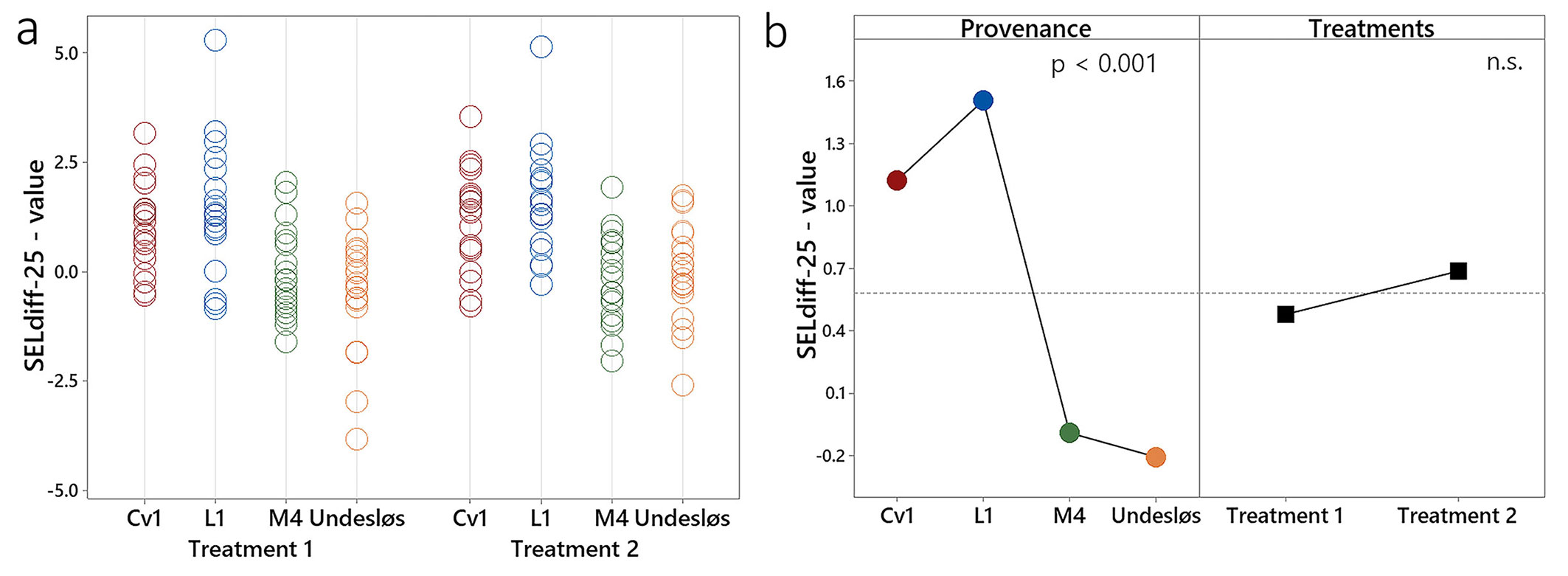
An index of injury (SELdiff-25) was calculated for each of the 20 seedlings per provenance. According to [14], a SELdiff-25 value ≤ 4 indicates sufficient frost hardiness for autumn conditions, allowing seedlings to be placed in freeze storage for planting in the following spring. All seedlings from provenances Cv1, M4, and Undesløs exhibited a SELdiff-25 value < 4 across treatments (Fig. 7a). In provenance L1, 19 seedlings per treatment met this threshold, while one seedling in each treatment showed slightly higher values (5.5 and 5.1). Negative SELdiff-25 values were observed in some seedlings across all provenances. Notably, two seedlings from provenance Undesløs under Treatment 1 exhibited values of -3.0% and -3.8%, and one seedling from Treatment 2 showed a value of -2.6 (Fig. 7a). Provenance had a significant effect on SELdiff-25 (p < 0.001), whereas the effect of Treatment (1 vs. 2) was not statistically significant (Fig. 7b).
Fig. 7 - Index of injury (SELdiff-25) values for twenty seedlings from four provenances of Norway spruce subjected to Treatments 1 and 2. (a): SELdiff-25 values for individual seedlings. (b); Effects of provenance (p < 0.001) and Treatments 1 and 2 on SELdiff-25, where the dashed reference line represents the overall mean. (n.s.): non-significant.
Overall, regardless of autumn conditions applied, nearly all seedlings achieved the target value (SELdiff-25 ≤ 4), except for two seedlings from L1 (Fig. 7a). This demonstrates that simulating autumn climatic conditions in controlled growth chambers for approximately six weeks was effective in inducing frost tolerance in first-year seedlings. Although it is important that seedlings develop sufficient hardiness to tolerate winter conditions, it remains challenging to predict whether the frost hardiness observed in these provenances will be adequate for more extreme winter events, as these results represent only the initial findings from our investigation. The Norwegian Forest Seed Center has used this procedure for several years to assess commercial batches of Norway spruce seedlings from different provenances prior to freeze storage. Historically, these seedlings have generally performed well in the field after planting the following spring, although results vary with planting-site conditions. Moreover, it is worth emphasizing that frost hardiness can be influenced by numerous environmental and management-related factors, including photoperiod, temperature, day length, precipitation, interactions among these factors, storage duration, and lifting dates ([2], [11], [22]). Additionally, Lindström et al. ([14]) and Malmqvist et al. ([16]) have emphasized that assessments of shoot freezing tolerance should be complemented by evaluations of root freezing tolerance. However, in Norway, no established method for measuring root freeze tolerance is currently in use.
Our results showed robust frost tolerance development under simulated autumn conditions and are consistent with findings suggesting that Norway spruce possesses considerable capacity to cope with climatic variation. For example, Esper et al. ([7]) reported that current climate change has not negatively impacted Norway spruce in the European Alps, where the species remains robust within mountain forests. Vacek et al. ([25]) demonstrated that Norway spruce exhibits greater resistance to climatic extremes when grown in admixture stands than in monospecific stands. These studies, together with our findings, suggest that Norway spruce may have substantial potential to adapt to future climatic conditions.
The SELdiff-25 method, widely used to assess the storability of Norway spruce seedlings ([14], [16]), confirmed slight differences in freezing tolerance between treatments (Fig. 7b). This likely reflects the combined influence of low temperatures and photoperiod in inducing frost hardiness in conifers ([4], [10], [19], [23]). A recent work by Riikonen et al. ([20]) indicated that warm spells followed by low temperatures in late autumn had negligible effects on freezing damage in short-day-treated seedlings. The electrolyte leakage method has been used to test frost hardiness in several forest species, including Douglas fir (Pseudotsuga menziesii), Scots pine (Pinus sylvestris), and Red spruce (Picea rubens). Across these studies, freezing temperatures ranged from -3 to -48 °C, with variation in cooling rates, duration of exposure at targeted temperatures, and the plant parts tested (for example, needles, buds) ([1], [16], [17], [26]). Murray et al. ([17]) applied the electrolyte leakage method to quantify frost damage in 1-year-old red spruce seedlings by exposing them to -3 °C, -6 °C, -9 °C, and -12 °C, demonstrating that the conductivity method is a reliable tool for frost damage assessment in this species. As cited by Malmqvist et al. ([16]), Sakai & Weiser ([21]) found coastal Douglas fir to be considerably more frost-sensitive, with a maximum freezing tolerance of approximately -20 °C. Consequently, Malmqvist et al. ([16]) tested shoot freeze tolerance at -20 °C rather than at -25 °C, thereby diverging from the standard method described by Lindström et al. ([14]). Overall, these studies highlight that the shoot electrolyte leakage method can be successfully applied across a range of forest species; however, the appropriate target temperature may differ from the -25 °C threshold used for testing seedlings of Norway spruce.
Conclusions
All twenty seedlings from provenances Cv1, M4, and Undesløs developed sufficient frost tolerance. In contrast, nineteen of the twenty seedlings from the L1 provenance exhibited frost hardiness under each treatment. This study demonstrates that first-year Norway spruce seedlings have the potential to develop frost hardiness when exposed to controlled autumn conditions characterized by both decreasing and fluctuating temperatures. These findings highlight the potential of nursery practices to effectively mimic natural climate patterns, thereby producing seedlings with improved frost resilience. However, it is essential to recognize the differences between controlled growth chamber conditions and natural nursery environments when applying these findings to practical forestry. Future research should aim to conduct these tests earlier (for example, after four weeks) and include a broader range of temperature variations to better understand their impact on seedling frost hardiness. Furthermore, the results contribute to our understanding of frost-hardiness-related variations observed in field trials with young Norway spruce plants. Additional studies should include a wider array of provenances and more contrasting temperature regimes to further elucidate the mechanisms underlying frost hardiness.
List of abbreviations
The following abbreviations are used throughout the paper:
- Electrical conductivity (EC);
- Shoot electrolyte leakage (SEL).
Acknowledgements
The authors thank Senior researcher Tore Skrøppa (NIBIO) for valuable discussions. Bless Kufoalor, Jørgen Lervik, and Øyvind Ness (all three NIBIO), and Guri Molden Kaldahl (Nord University) are kindly acknowledged for their help with the facilities at the Research lab. We thank Erlend Grande, Ove Solbakken (both SPMN), and Lilli Haugdal (ALLSKOG) for their kind help with the various activities at the greenhouse, Torstein Myhre (The Norwegian Forest Seed Center), and Rannveig Jørstad Kristiansen (Skogselskapet) for transporting plants and for helpful discussions. We would also like to thank Diana Floor (The Norwegian Forest Seed Center) for showing the electrolyte leakage method. Microsoft™ Copilot® was used for language improvement.
Authors’ contributions
IA: original draft, conceptualization, methodology, investigation, visualization, data analysis; AS: conceptualization, methodology, data analysis, review & editing; ØME: methodology, review & editing; ISF: acquired funding and project administration, conceptualization, review & editing; JMT: conceptualization, methodology; POG: conceptualization, methodology. All authors approved the final manuscript.
Funding
This research was carried out as part of the project “Robust Forest plants”, funded by Statskog, Skogfrøverket (The Norwegian Forest Seed Center), Skogplanter Midt-Norge AS, Skogplanter ØstNorge AS, and Fagrådet for skogplanteskolene.
References
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Arne Steffenrem 0000-0001-7116-9151
Norwegian Institute of Bioeconomy Research (NIBIO), Skolegata 22, 7713 Steinkjer (Norway)
Jo Magne Tyldum
Skogplanter Midt-Norge (SPMN), Kvatninga 88, 7860 Skage i Namdalen (Norway)
Norwegian Forest Seed Center, Birkebeinvegen 11, 2316 Hamar (Norway)
Corresponding author
Paper Info
Citation
Ahuja I, Fløistad IS, Grande PO, Tyldum JM, Edvardsen ØM, Steffenrem A (2026). Frost hardening in seedlings of four provenances of Norway spruce in response to simulated autumn conditions with decreasing and fluctuating temperatures. iForest 19: 141-148. - doi: 10.3832/ifor5041-019
Academic Editor
Marco Borghetti
Paper history
Received: Nov 17, 2025
Accepted: Mar 14, 2026
First online: Apr 19, 2026
Publication Date: Apr 30, 2026
Publication Time: 1.20 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2026
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 853
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 183
Abstract Page Views: 354
PDF Downloads: 272
Citation/Reference Downloads: 1
XML Downloads: 43
Web Metrics
Days since publication: 50
Overall contacts: 853
Avg. contacts per week: 119.42
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
(No citations were found up to date. Please come back later)
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Relationship between frost hardiness of adults and seedlings of different tree species
vol. 7, pp. 282-288 (online: 01 April 2014)
Research Articles
Links between phenology and ecophysiology in a European beech forest
vol. 8, pp. 438-447 (online: 15 December 2014)
Research Articles
Distribution of the major forest tree species in Turkey within spatially interpolated plant heat and hardiness zone maps
vol. 5, pp. 83-92 (online: 30 April 2012)
Research Articles
Relationships between leaf physiognomy and sensitivity of photosynthetic processes to freezing for subtropical evergreen woody plants
vol. 12, pp. 551-557 (online: 17 December 2019)
Research Articles
Ozone fumigation effects on the morphology and biomass of Norway spruce (Picea abies L.) saplings
vol. 2, pp. 15-18 (online: 21 January 2009)
Research Articles
Assessment of cadmium tolerance and phytoextraction ability in young Populus deltoides L. and Populus × euramericana plants through morpho-anatomical and physiological responses to growth in cadmium enriched soil
vol. 10, pp. 635-644 (online: 01 June 2017)
Research Articles
Coping with spring frost-effects on polyamine metabolism of Scots pine seedlings
vol. 10, pp. 227-236 (online: 27 January 2017)
Research Articles
Drought tolerance in cork oak is associated with low leaf stomatal and hydraulic conductances
vol. 11, pp. 728-733 (online: 06 November 2018)
Research Articles
Tolerance to heavy metal stress in seedlings of three pine species from contrasting environmental conditions in Chile
vol. 9, pp. 937-945 (online: 12 August 2016)
Research Articles
Dynamics of soil organic carbon (SOC) content in stands of Norway spruce (Picea abies) in central Europe
vol. 11, pp. 734-742 (online: 06 November 2018)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword