Quantifying the vertical microclimate profile within a tropical seasonal rainforest, based on both ground- and canopy-referenced approaches
iForest - Biogeosciences and Forestry, Volume 15, Issue 1, Pages 24-32 (2022)
doi: https://doi.org/10.3832/ifor3780-014
Published: Jan 27, 2022 - Copyright © 2022 SISEF
Research Articles
Abstract
Quantifying the microclimate of a tall and complex tropical forest is quite challenging because of the difficulty in accessing the canopy. Traditional ground-referenced methods may underestimate the contribution of canopy heterogeneity to structuring the vertical profiles of forest microclimate. The present study examined how the reference height affects vertical variation of microclimate in a tropical rainforest in southwest China, based on both ground- and canopy-referenced approaches. The results show that the canopy-referenced approach yielded a higher model fit than did the ground-referenced method, and only canopy-referenced method could detect two thresholds in the leaf area index at approximately -22.6 ± 2.7 m and -36.6 ± 6.6 m below the canopy top; the higher threshold is consistent with thresholds of the annual mean temperature, the diurnal ranges of air temperature, and the relative humidity in the vertical profile; while the lower threshold is similar with the breakpoints of annual mean relative humidity, the annual ranges of air temperature and the relative humidity along the profile. The discontinuous variance in the microclimatic factors was due to the canopy structure in the vertical profile. Selecting the top of the canopy as the reference height could be a better approach for quantifying the microclimatic profiles in the studied forest, and this approach can improve our understanding of the effects of the vertical stratification of microclimates on species composition and diversity in this forest.
Keywords
Tropical Seasonal Rainforest, Vertical Gradient, Light Environment, Temperature And Humidity, Distance From The Canopy Surface
Introduction
The forest canopy reshapes understory microclimates, and vertical climatic gradients within forests may vary even more sharply than those driven by elevation and latitude ([40]). Although the interactions between forest canopies and atmosphere such as rainfall interception and evapotranspiration at large-scale are relatively well understood, only a few studies were focused on canopy structure and microclimate interactions at smaller scales ([32]).
Although the transmission of radiation affects the microclimate in the forest understory, most previous studies have taken the ground as a reference due to the difficulty of accessing the forest canopy. These ground-level measurements may underestimate the microclimatic edge effects on vertical stratification in forests ([11]). As radiation is attenuated from the top of the canopy to the understory, ground-referenced radiation transfer models limit the estimation of microclimate factors when used in the canopy height model (CHM), although the CHM can extract the vegetation structure at both the plot level and the individual plant level ([35]).
Many studies have suggested that vertical radiation transmission in forest canopies can be described by Beer’s law ([1], [47]), and the transmittance of diffuse light in the forest has an exponential relationship with the aboveground height ([20]). However, at the fundamental biophysical level, Beer’s law describing the radiation absorbed by the canopy was believed oversimplified, particularly in the case of highly heterogeneous canopies and those with significant foliage clumping ([22]), because high rugosity is a common feature of old-growth forests ([34]). The amount of solar radiation received at the canopy floor is largely dependent on the leaf area index (LAI) of the forest, but approximately 50% of the total leaf area in forests has been reported to be concentrated within a depth of 5 m from the top of the canopy ([45]), and the relationship between the LAI and the height of a forest is not linear. The discontinuous profile of the LAI further complicates the transmission of radiation in the understory, especially in tropical forests, which canopy was high and quite heterogeneous.
Radiation reduction in a forest also affects other environmental factors such as air temperature and humidity, and these microclimatic factors may affect the productivity and biodiversity of different height layers. The spatial and temporal variation in air temperature is qualitatively similar to the variation in photon flux density ([2]). In addition, temperature is a key determinant of the rate of metabolic processes and hence has a major impact on carbon balance, as a consequence of photosynthesis and respiration ([22]). Relative humidity (RH), another important factor that is affected by radiation and temperature, determines the water loss from transpiration and affects the photosynthesis and respiration of plants ([22]). Plants grown at high RHs (RH ≥ 85%) frequently show a rapid wilting trait when exposed to lower RHs (≈60% - [16]). The vertical gradient in the species composition of bryophytes is strongly associated with the decreasing gradient of RH and other microclimate factors ([41]); and amphibians prefer shaded, cool, and moist habitats on the forest floor ([3]). However, quantitative studies of species distribution patterns in the vertical gradient are limited, the vertical composition of biodiversity in forests is not well understood, and more accurate microclimate profiles are needed.
In a seasonal rainforest in Xishuangbanna, southwest China, the transmitted radiation that eventually reaches the understory surface was reported to be approximately 7.13% of the total radiation ([48]). Compared to full-light conditions, the total solar radiation, infrared radiation, and daily radiation of visible light into the canopy were all decreased as height descended ([12]), resulting in higher temperature in upper canopy and lower temperature in lower canopy ([46]). However, the heterogeneity of the canopy height in the forest is quite substantial, and the height difference between the emergent layer (Parashorea chinensis, approximately 60 m high) and the upper canopy layer (approximately 30 m high) exceeds 20 m ([4], [6]). We also observed that the transmittance diffuse light varied from 0.3% at 3.7 m to 100% at ≥ 21.2 m based on the recent measurements using a canopy crane in this forest ([10]). Early studies that used the ground as a reference for measuring the variations in vertical microclimate, however, may still be insufficient to quantitatively describe the within-forest vertical profile of microclimate, although these features are very important for understanding and explaining the high biodiversity in the forest.
In the present study, we tried to examine the change in the microclimate ranging from the top canopy to the ground (forest floor), demonstrating the trajectory of top-down variation in the factors. We hypothesized that the height and distance downward to the ground affect the microclimate within the forest and that the major factors of vertical microclimate exhibit discontinuous variance based on the nonlinear attenuation of radiation in the vertical profile. A canopy crane was used to investigate the vertical profiles at the study site, and both the distance to the canopy surface and the distance to the ground of each point at the profile were recorded. By comparing variations in biophysical factors such as the LAI, the diffuse non-interceptance (DIFN), the ratio of red to far-red light (R:FR ratio), the air temperature, and the RH in the profile, we sought to understand the differences between canopy-referenced and ground-referenced methods and to develop an approach for determining the vertical profiles of forest microclimates.
Materials and methods
Overview of the study area
This study was carried out in the crane plot in the tropical seasonal rainforest of Xishuangbanna (101° 34′ 59.1″ E, 21° 37′ 2.6″ N; altitude 653.4 m a.s.l.). This plot was established in December 2014, approximately 300 m from the Xishuangbanna tropical seasonal rainforest 20-ha dynamics plot (XTRDP). The height of the canopy crane (TCT7015-10E, Zoomlion, Changsha, China) is 80 m (approximately 20 m higher than the canopy of emergent trees), and the jib length is 60 m. A long-term monitoring plot of 120 × 120 m was built up around the canopy crane. According to the results of a survey in 2014, there were 6928 individual trees with diameter at breast height (DBH) ≥ 1 cm in this plot, belonging to 55 families and 217 species ([10]). The tree species composition of the crane plot is close to that of the XTRDP ([6]), and P. chinensis has the highest relative importance value (21.73%) in this plot.
Measurement of the vertical light environment
With the assistance of the canopy crane, we set 40 vertical light environmental survey profiles (Fig. S1 in Supplementary material) in three concentric circles with radii of 20, 40, and 56 m respectively. For the two outer circles, the profiles were spaced every 22.5° horizontally; and for the inner circle, the profiles were spaced every 45° horizontally. At each vertical survey profile, the light environment was measured every 5 m of interval, from the top of the tower crane (80 m) down to the ground. The height from the canopy (Hcanopy) and height to the ground (Hground) at each measurement point were recorded. The maximum canopy height at all 40 vertical investigation profiles was between 15.0 and 63.5 m, with a mean (± standard deviation) of 34.8 ± 14.6 m.
Since the actual measurement plane is located at the top of the hanging basket, the lowest measuring position in each profile was approximately 2 m above the ground.
The spatiotemporal distribution of light in the understory is quite heterogeneous, and the measurement of direct light is difficult for long-term sampling ([14]). Indirect measurements, such as the transmittance of diffuse light (presented as the DIFN), are also acceptable for estimating light conditions. The DIFN is a single-value representation of the canopy structure because it combines the LAI and the mean tilt angle of leaves (MTA) into one number ([24]). A previous methodological comparison showed that the DIFN had a good fit with the directly measured long-term photosynthetic photon flux density (PPFD) in the understory of a tropical forest ([14]). In this study, both the DIFN and LAI were measured using the plant canopy analyzer LI-2200C® (LI-COR, Inc., Lincoln, NE, USA). Open sky measurements were conducted at the top of the crane, and each profile was measured at the highest working height of the tower crane with five replicates. A 270° view cap was used to prevent the crane jib from obstructing the view. The repeated measurements of each measuring point were used to adjust the open sky measurements with FV2200® software (LI-COR, Inc., Lincoln, NE, USA) and to estimate the DIFN and LAI. The measurements were repeated five times at each height of each profile.
The leaves of the canopy absorb much more light in the photosynthetically active portion of the spectrum (400-700 nm) than in the near-infrared range (700-1000 nm -[23], [36]), which shifts the ratio of red to far-red wavelengths in the understory ([36]). Occasional light flecks can have a significant effect on the average daily PPFD, but these flecks have little effect on daily R:FR ratios ([43], [7]), which indicates that the R:FR ratio can be used in assessing light levels in the understory ([7]). The R:FR ratio was measured by a SpectroSense2+® spectrometer (Skye Instruments Ltd, Wales, UK). The measurements were repeated five times at each height of each profile, and the mean value was taken as the corresponding light environment at this position.
The measurements of the DIFN, LAI and R:FR ratio were carried out and accomplished in April 2016 from approximately 10:00 to 16:00 on sunny days, when it is in the end of the dry season.
Vertical temperature and humidity profiles
Three vertical profiles were set up, which included three emergent P. chinensis individuals in different directions around the crane (where the maximum heights of the canopy were 56.5, 62.7, and 63.1 m, respectively). The air temperature and RH were recorded by an Onset HOBO U23-001® (ONSET, USA). Sensors were set at following levels: (i) For the individual of 56.5 m: 2 m, 12 m, 22 m, 32 m, 42 m (lower crown), 47 m (middle crown), and 54.5 m (upper crown); (ii) For the individual of 62.7 m: 2 m, 12 m, 22 m, 32 m, 42 m (lower crown), 52.5 m (middle crown), and 58.7 m (upper crown); (iii) For the individual of 63.1 m: 1.5 m, 11.5 m, 21.5 m, 31.5 m, 41.5 m, 51.5 m (lower crown), 55 m (middle crown), and 60.5 m (upper crown).
In total, 22 sensors were installed in the forest to continuously monitor changes in the air temperature and humidity in these four profiles. The measurements were performed every 30 min. A radiation shield was used to protect the sensor from direct sunlight to prevent data drift. This study used data from May 27, 2016 to December 31, 2019 (the data from January to May 2016 and from December 2018 to February 2019 were missing, so they were extrapolated from previous annual means).
Data analysis
Five temperature indices, i.e., annual mean temperature (AMT), monthly mean temperature (MMT), monthly (MDRT) and annual mean diurnal range of temperature (ADRT), and mean annual range of temperature (MART), and five humidity indices, i.e., annual mean RH (AMRH), monthly mean RH (MMRH), monthly (MDRRH) and annual mean diurnal range of RH (ADRRH), and mean annual range of RH (MARRH), were computed to investigate the vertical and temporal changes in the air temperature and humidity. The MMT and MMRH were averaged arithmetically according to the daily mean values (the average values of 48 records per day, and one record per 0.5 h) of the temperature and RH in each month from January 2016 to December 2019. The AMT (or AMRH) was the arithmetical average of 4 × 12 MMT (or MMRH) values. The MDRT (or MDRRH) was the mean value of the diurnal range of temperatures (DRT - or RH, DRRH - the difference between the daily maximum and minimum temperatures or RH) in each month from January 2016 to December 2019, and the ADRT (or ADRRH) was the mean value of 4 × 12 MDRT (or MDRRH) values. The annual range of temperature (ART - or the annual range of RH, ARRH) was the difference between the MMTs (or MMRHs) of the warmest and coldest months within a year, and the MART (or MARRH) was the mean value of the ART (or ARRH) from 2016 to 2019.
Microclimatic factors in the crane plot are summarized in Tab. S1 (Supplementary material). The nonparametric locally estimated scatterplot smoothing (LOESS) model was used to approximate the number and positions of the breakpoints in light-related data between heights, and a piecewise linear regression model (PLRM) was used to estimate the breakpoints for LAI, DIFN, R:FR ratio, AMT, ADRT, MART, AMRH, ADRRH and MARRH in each vertical gradient. The LOESS model was fitted using the statement “loess” in R ([38]), and the PLRM was implemented using the R package “segmented” ([30]).
To estimate breakpoints incorporating linear segments, the PLRM was expressed as follows ([42] - eqn. 1):
where y and x are the values for the response and predictor variables, respectively; α1 and α2 are the breakpoints; and β1, β1 + β2 and β1 + β2 + β3 are the slopes of linear regression models 1, 2 and 3, respectively. The thresholds and slopes for the PLRM are listed in Tab. S2 and Tab. S3 (Supplementary material). R (v. 4.0.5) was used to perform model fitting and to calculate the relevant model parameters ([38]).
Results
Light environment
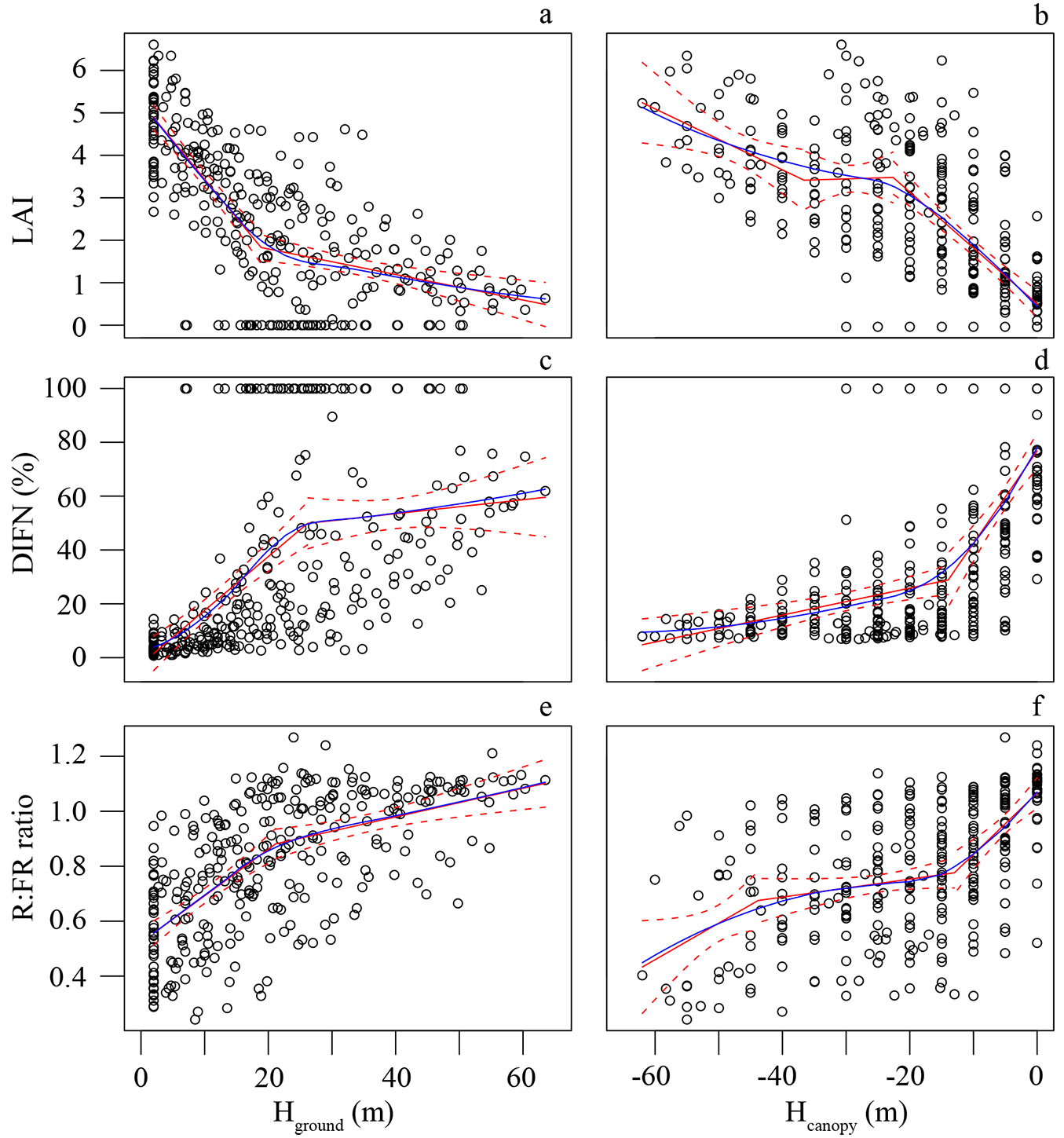
When the ground was used as the reference, the LAI was highest at 2 m above ground (estimate ± SE = 4.81 ± 0.97) and rapidly decreased with decreasing height until a height of 19.0 ± 1.6 m (Fig. 1a). When the height was over 19 m, the slope of LAI vs. height became gentler as the LAI began to stabilize somewhat. When the canopy was used as the reference, the LAI increased dramatically from the canopy surface (0 m) to -22.6 ± 2.6 m below the canopy top. The LAI was relatively stable at Hcanopy values lower than -22.6 ± 2.6 m and higher than -36.6 ± 6.6 m, but then the slope of LAI vs. height became steep again below -36.6 ± 6.6 m (Fig. 1b).
Fig. 1 - Variation in LAI, DIFN and R:FR ratio in reference to Hground (a, c, e) and Hcanopy (b, d, f). The white cycle represents the observed value at the corresponding height. The blue solid line represents LOESS model, the red solid line represents PLRM, and the red dash line represents 95% prediction bands of PLRM.
DIFN gradually increased with increasing Hground, and from Hground = 26.2 ± 3.6 m upward, the slope of DIFN vs. height became flatter (Fig. 1c). When using Hcanopy for reference, however, DIFN increased gently with increasing height from forest floor to -14.0 ± 2.0 m high and rapidly increased from -14.0 ± 2.0 m to the top canopy (Fig. 1d). In addition, DIFN was not saturated at the canopy surface, showing a value of 76.95 ± 24.19% at Hcanopy = 0 m.
The R:FR ratio at 2 m above the forest floor was 0.557 ± 0.173, and it increased with increasing Hground until it slowed down at the height of 21.2 ± 3.3 m (Fig. 1e). In terms of Hcanopy, the curve of the R:FR ratio vs. height exhibited two breakpoints, at -13.0 ± 2.5 m and -43.8 ± 6.8 m in height (Fig. 1f), and the R:FR ratio only showed greater stability between these two breakpoints.
Vertical variation in air temperature
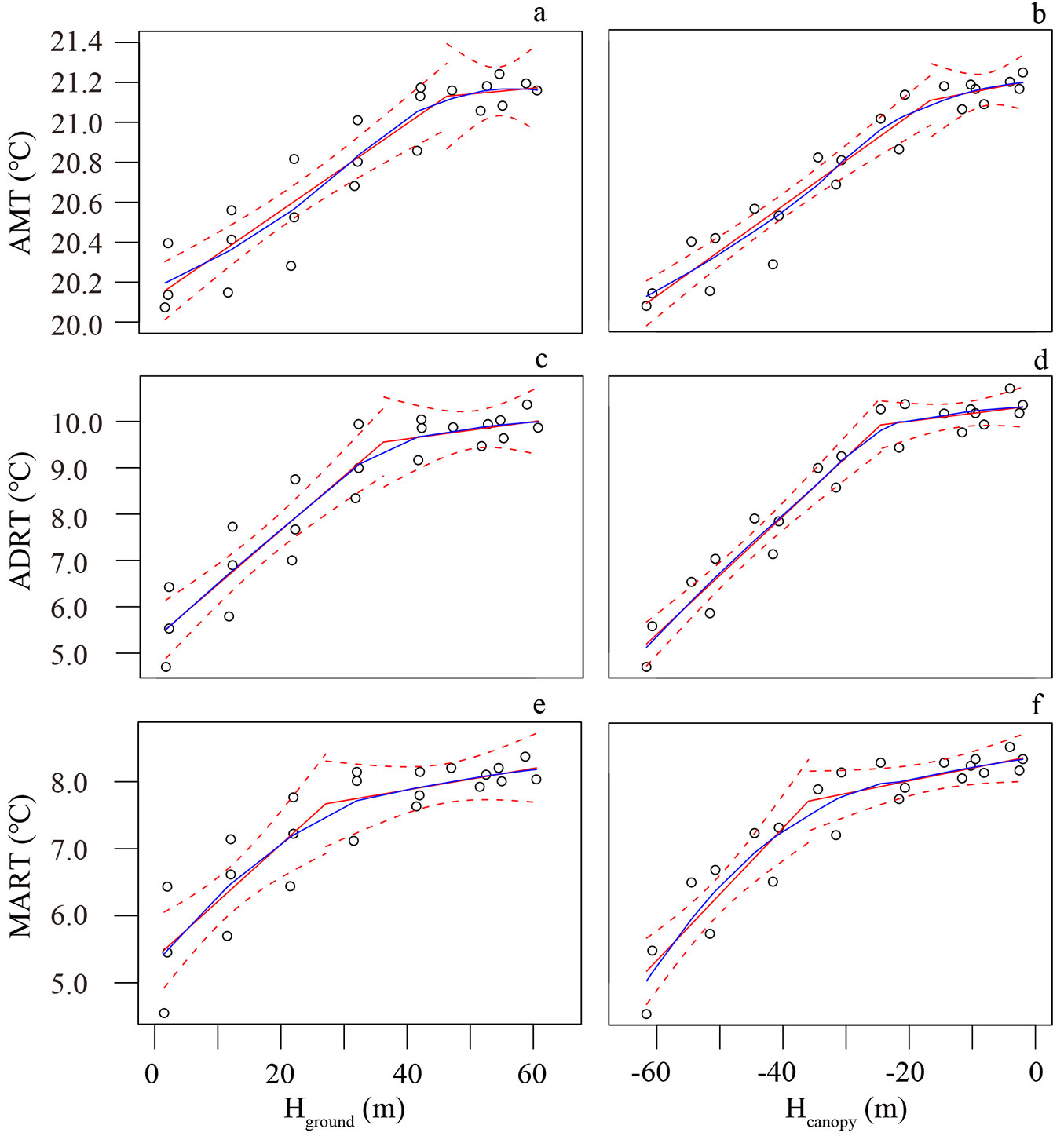
The AMT exhibited a linear increase with increasing height from the ground until the height of 46.2 ± 7.9 m (Fig. 2a) or -16.7 ± 6.5 m under the canopy surface (Fig. 2b), then became relatively stable in the upper canopy. The PLRM exhibited a better fit (r2 = 0.9260) compared to when the ground was used as the reference (r2 = 0.8519).
Fig. 2 - Variation in the AMT, ADRT, and MART with reference to Hground (a, c, e) and Hcanopy (b, d, f). The white cycle represents the observed value at the corresponding height. The solid blue line represents LOESS model, the red solid line represents PLRM, and the red dash line represents 95% prediction bands of PLRM.
The breakpoint where the ADRT slope changed with increasing height appeared at 35.9 ± 5.9 m high (Fig. 2c) or -24.5 ± 3.4 m under the canopy surface (Fig. 2d). The PLRM regression based on Hcanopy (r2 = 0.9490) had a better model fit than the model based on Hground (r2 = 0.8738) for the ADRT.
Regardless of whether the ground or canopy was used for reference, the MART showed a positive relationship with height (Fig. 2e, Fig. 2f), and PLRM suggested a breakpoint at 27.1 ± 6.7 m high or -36.0 ± 4.5 m under the canopy surface. MART displayed a better model fit (r2 = 0.8788) when the canopy-referenced method was used compared to the ground-referenced method (r2 = 0.7707).
Vertical variation in relative humidity
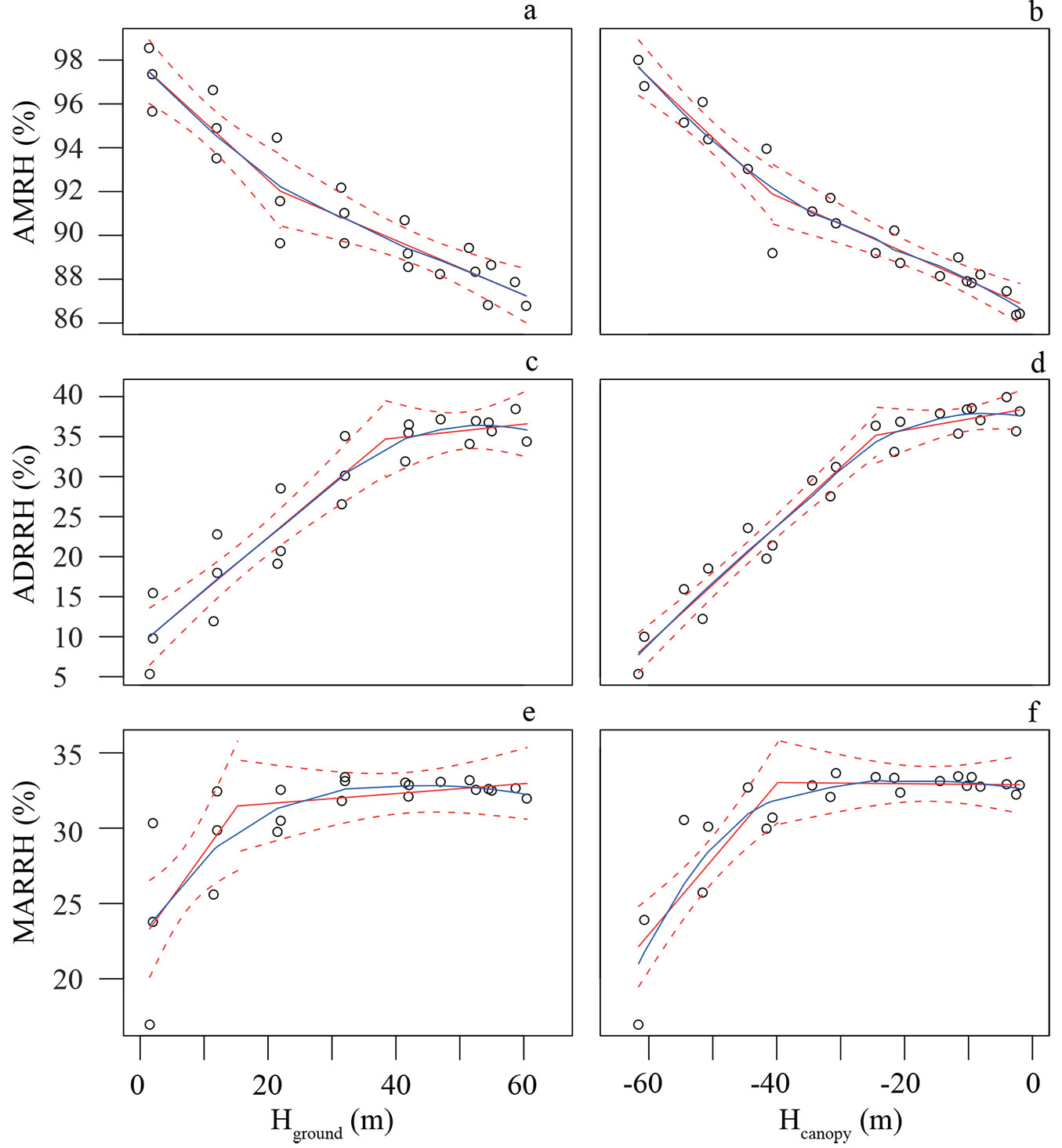
The AMRH in the forest exhibited a negative relationship with height (Fig. 3a, Fig. 3b), and the r2 of the PLRM of AMRH vs. canopy-referenced height (0.9227) was higher than that of the model of AMRH vs. ground-referenced height (0.8712). The breakpoint was -40.7 ± 5.9 m under the canopy surface (22.0 ± 7.8 m high from the forest floor).
Fig. 3 - Variation in the AMRH, ADRRH and MARRH with reference to Hground (a, c, e) and Hcanopy (b, d, f). The white cycle represents the observed value at the corresponding height. The solid blue line represents LOESS model, the red solid line represents PLRM, and the red dash line represents 95% prediction bands of PLRM.
The ADRRH exhibited a positive relationship with the ground-referenced profile from forest floor to 38.4 ± 5.4 m (Fig. 3c), and when the canopy was referenced, the breakpoint was at -24.5 ± 3.5 m (Fig. 3d), beyond which the ADRRH became relatively stable with change in height. Canopy-referenced PLRM still showed better fit (r2 = 0.9563) than did the ground-referenced method (r2 = 0.8846).
The MARRH was low near the ground and increased rapidly as the height increased until 15.2 ± 3.5 m high (Fig. 3e) or -40.0 ± 3.7 m (Fig. 3f) under the canopy surface. The PLRM of the MARRH showed better fit with Hcanopy (r2 = 0.7373) than with Hground (r2 = 0.5732).
Seasonal variations in air temperature
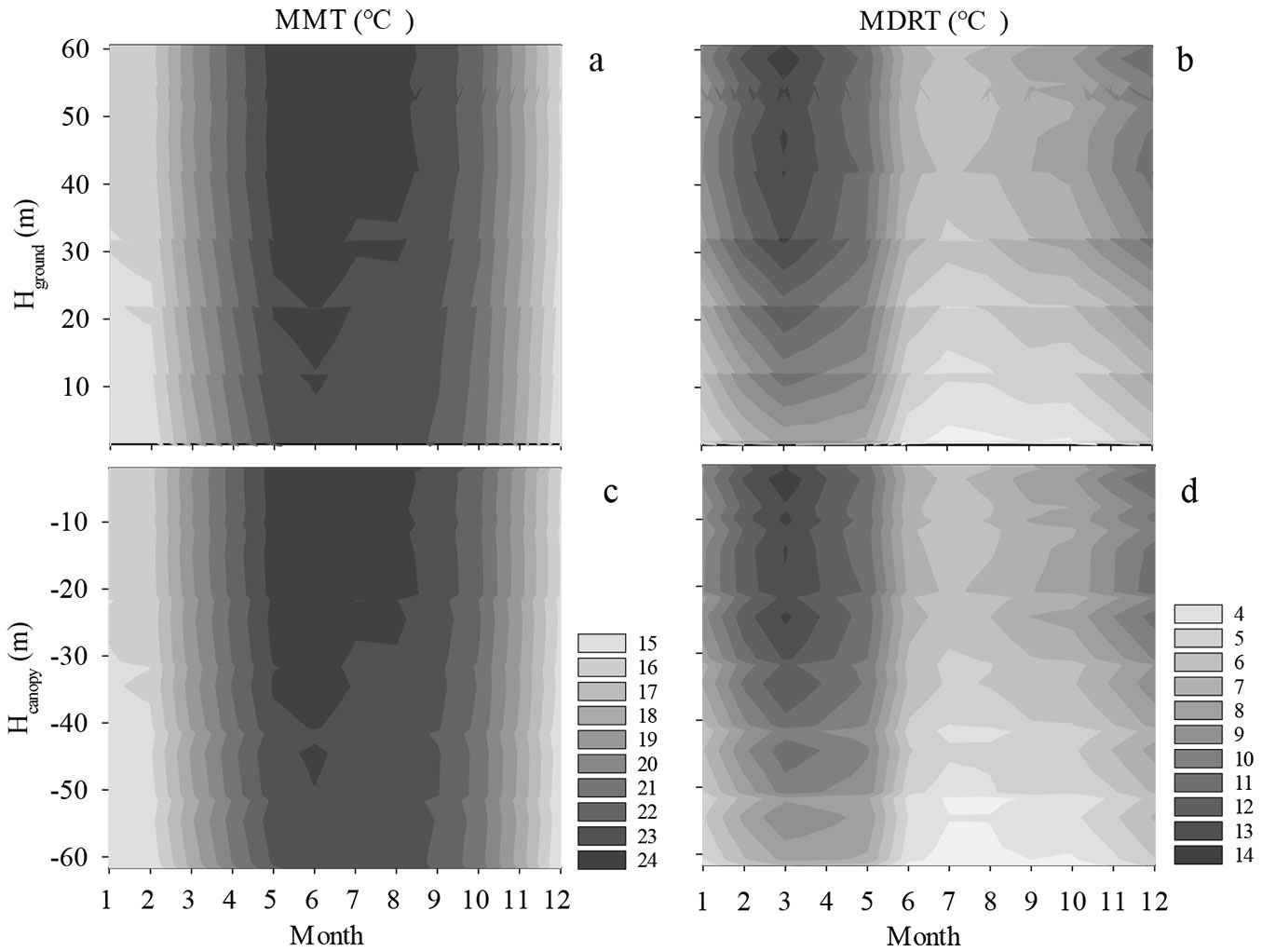
The highest MMT in the forest occurred in June at all height levels. The highest value in the profile (24.7 °C) was recorded at Hground = 52.5 m or Hcanopy = -10 m; the lowest value in the profile (23.6 °C) was recorded at Hground = 1.5 m or Hcanopy = -61.6 m (Fig. 4a, Fig. 4c). In December, the MMT at each height was the lowest. In the vertical profile, the highest value in December (16.1 °C) was recorded at Hground = 54.5 m or Hcanopy = -2 m, and the lowest value (14.9 °C) was recorded at Hground = 1.5 m or Hcanopy = -61.6 m (Fig. 4a, Fig. 4c). The temperature difference between the top and bottom of the canopy was the smallest in July, at 0.8 °C, and the difference was the largest in March, at 1.9 °C.
Fig. 4 - The seasonal variation in the MMT and MDRT for different Hground (a, b) and Hcanopy (c, d) profiles. Light and dark colors represent low temperature and high temperature respectively.
The highest MDRT was observed in March. For the vertical profile, the MDRT near the canopy was higher than that in the understory (Fig. 4b, Fig. 4d). The highest MDRT in March was 14.6 °C and was recorded at Hground = 58.7 m or Hcanopy = -4 m, and the lowest MDRT in March was 7.0 °C and was recorded at Hground = 1.5 m or Hcanopy = -61.6 m.
The lowest MDRT occurred in July. For the vertical profile, the highest MDRT in July was 6.8 °C and was recorded at Hground = 42 m or Hcanopy = -20.7 m, and the lowest MDRT in July was 3.1 °C and was recorded at Hground = 1.5 m or Hcanopy = -61.6 m (Fig. 4b, Fig. 4d).
The lowest difference in the MDRT in the vertical profiles was in July, at 3.6 °C; and the largest difference was in March, at 7.6 °C.
Seasonal variations in relative humidity
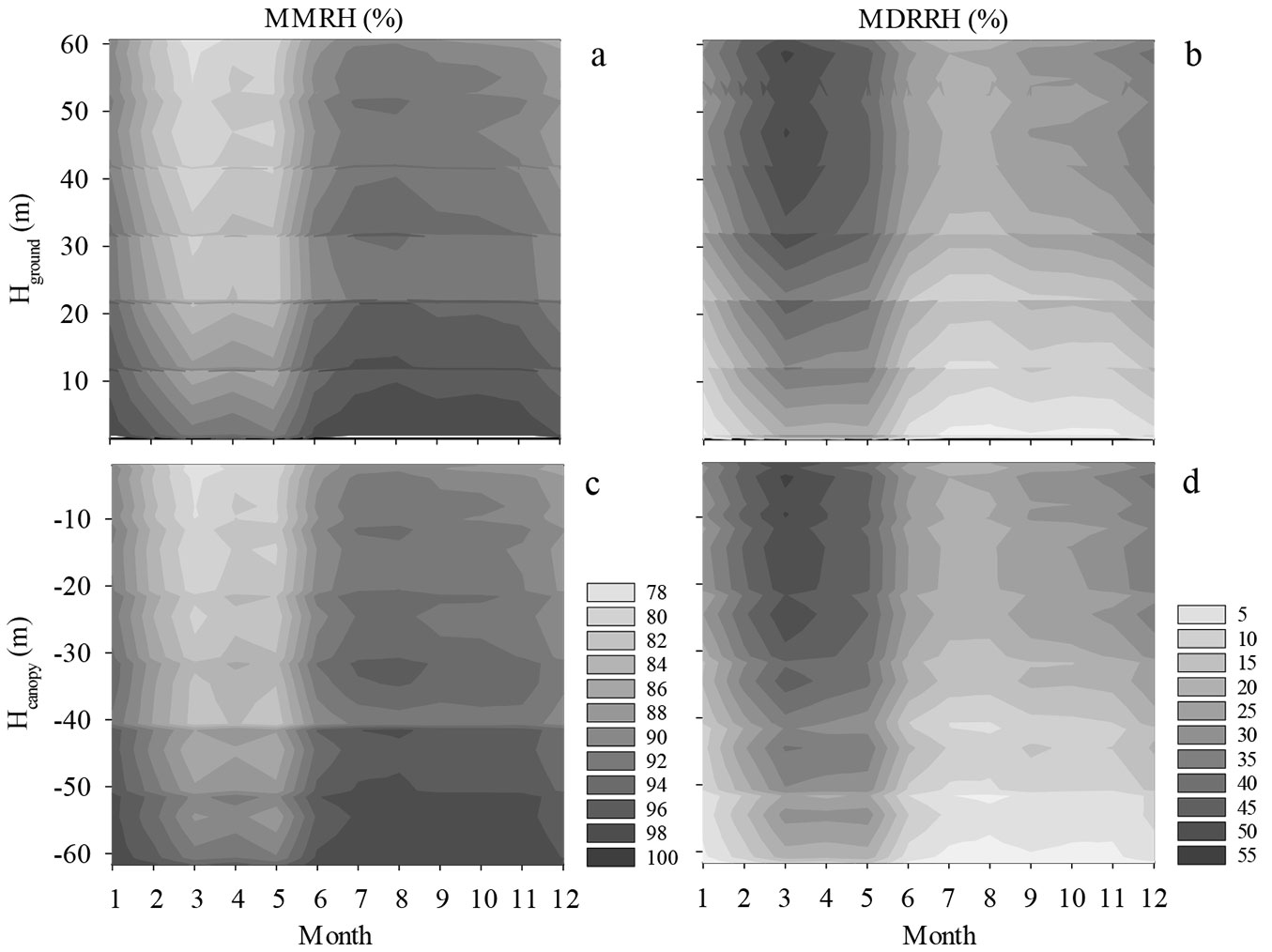
The MMRH peaked in August. The maximum was 100% and was recorded at Hground = 1.5 m or Hcanopy = -61.6 m, and the minimum was 92% and was recorded at Hground = 54.5 m or Hcanopy = -2 m (Fig. 5a, Fig. 5c). The maximum vertical difference in the MMRH was 9% in August, which also represented the least difference for the entire year.
Fig. 5 - The seasonal variation in the MMRH and MDRRH for different Hground (a, b) and Hcanopy values (c, d). Light and dark colors represent low temperature and high temperature, respectively.
MMRH was the lowest in March. The maximum was 96% at Hground = 1.5 m or Hcanopy = -61.6 m, and the minimum was 78% at Hground = 60.5 m or Hcanopy = -2.6 m (Fig. 5a, Fig. 5c). In March, the MMRH difference between the top of the canopy and the ground was 17%, the highest value among all the twelve months throughout the year.
The MDRRH peaked in March (Fig. 5b, Fig. 5d). The maximum was 56% and was recorded at Hground = 58.7 m or Hcanopy = -4 m; and the minimum was 13% and was recorded at Hground = 1.5 m or Hcanopy = -61.6 m.
MDRRH was the lowest in August (Fig. 5b and d). The maximum value in August was 26% recorded at Hground = 58.7 m or Hcanopy = -4 m. The minimum value in August was 1% recorded at Hground = 1.5 m or Hcanopy = -61.6 m.
Discussion
Vertical profile of light environment within the forest
Variations in the LAI are a consequence of canopy structural attributes ([33]). In this study, the LAI and light environment variables (i.e., DIFN and R:FR ratio) seemed to exhibit steeper variances in the upper canopy with the canopy-referenced approach, whereas the ground-referenced method suggested that the variances varied considerably in the lower canopy compared to the upper canopy. Although a similar rapid change based on the ground-referenced method has also been reported for the PPFD ([34]), the ground- and canopy-referenced approaches demonstrated a DIFN profile that basically followed Beer’s law ([1], [47]) and displayed an exponential relationship with height (Fig. S2 in Supplementary material), and the ground-referenced approach was not consistent with the fact that foliage exhibits significant clumping in the canopy ([22]). The difference between the ground-referenced and canopy-referenced methods is probably because the former mixed the effect of crown foliage clumping and canopy height heterogeneity together, and a high rugosity is a common feature of old-growth forests ([34]). The canopy-referenced approaches were more consistent with the attenuation of radiation from the top down in forests and more concerned with foliage clumping of tree crowns without the disturbance of height heterogeneity. Thus, these approaches can better quantify the vertical stratification of microclimates.
The LAI (4.81 ± 0.97) at 1.5-2 m above the ground in this study was similar to LAI values from previous studies in seasonal rainforests in Xishuangbanna (4.26-6.33 - [12]) and close to the values in two tropical rainforests in Brazil (5.07 ± 1.5 and 5.05 - [27], [34]). However, our results show that the LAI decreased rapidly from the canopy surface to -22.6 ± 2.7 m depth. These findings are different from the vertical distribution of the LAI in the Barro Colorado Island tropical rainforests, where the LAI at 5 m below the canopy surface contributes approximately 50% to the understory cumulative LAI ([45]). That is, deeper depth beneath the canopy is needed to achieve a relatively stable LAI in the forests of our study, as a consequence of differences in statures of canopy tree species. The adult tree of P. chinensis was approximately 60 m tall, its crown occupied 28.04% (approximately 15-20 m) of the entire adult tree height (Fig. S3 in Supplementary material), and the emergent layer (mainly P. chinensis) was 20 m higher than the main canopy layer in the local rainforest ([4], [6]); thus, the tree structure affected the vertical variation in the LAI and light environment within the forest.
The canopy-referenced PLRM in this study suggests that the LAI and the R:FR ratio have secondary breakpoints at -36.3 ± 6.6 m and -43.3 ± 6.8 m, which is hardly detectable by the ground-referenced model but more consistent with the multilayer structure of local forest. The tropical seasonal rainforest in Xishuangbanna contains 3-4 tree layers: the emergent layer (tallest trees up to 60 m), main canopy layer (up to 30 m) and understory layer (5-18 m, sometimes can be further divided into upper and lower sublayers - [50]). The coverage of the understory layer is approximately 40% ([50]), and the clutter of understory tree species and treelets would further increase the LAI in this layer. The R:FR ratio also showed a similar breakpoint with canopy-referenced height, which probably because high LAI indicates a greater number of leaves and thus higher absorption of red light ([23], [36]).
The DIFN at 1.5-2 m above ground in this forest was 4.18 ± 3.33%, which was close to that of the light environment at 1 m above the forest floor (5.7%) in a primary moist tropical forest in Brazil ([34]) and was within the normal range of 2-7% on the ground in forests ([1]). Considering the mechanical layout, method in this study did not deliberately avoid canopy gaps, the mean ground DIFN in this study might already indicate the light heterogeneity of the local forest surface in its natural state, including canopy gaps.
The DIFN in this study could not reach saturation on canopy surface (76.95 ± 24.19%), mainly because the canopy in the tropical rainforest was uneven. Even on the top surface of a tree crown, shadow projection from both surrounding emergent trees and topography should be considered in light transmission around the canopy.
In this study, the R:FR ratio at 1.5-2 m above the ground was 0.55 ± 0.18, which was close to the mean value in a wet tropical forest in Costa Rica (0.51 - [7]) but higher than the normal understory level (0.23) reported earlier ([8]). This difference may be due to the small gaps included in this study plot, and environmental factors, including the degree of canopy closure, the canopy structure, and the presence of clouds, could also affect the R:FR ratio ([13]). In the understory, the relationship between the R:FR ratio and the diffuse transmittance is logarithmic ([31], [7]), and LAI calculations also depend on the logarithmic transformation of the DIFN ([44]); as result, the R:FR ratio showed a sharp change from the top surface to a depth of -13.0 ± 2.5 m into the canopy, which was linearly correlated with the variation in the logarithmic DIFN (F = 364.7, r² = 0.5422, P < 0.001 - Fig. S4a) and LAI (F = 341.8, r² = 0.5261, P < 0.001 - Fig. S4b in Supplementary material).
Vertical profiles of temperature within the forest
For both the air temperature and the RH, the r2 values of the PLRMs were all lower when using Hground than when using Hcanopy (Tab. S2 in Supplementary material). Since the canopy is the first interface layer connecting the forest with the external energy environment, the canopy-referenced approach could better describe the attenuation of solar energy after it enters the canopy.
The AMT varied from 21.2 ± 0.04 at the canopy surface to 20.2 ± 0.2 on the forest floor, which is consistent with the assumption that forests reduce the understory air temperature in tropical ([15]) and temperate ([11]) areas. Previous local studies have also reported higher temperatures in the canopy than in the understory ([46]), but these studies failed to provide a continuous temperature variation curve in the vertical profile. Previous studies from another local tropical rainforest have reported that the air temperature increased as the height in the understory increased, and the diurnal range of the air temperature at the canopy layer exceeded that in the understory ([25], [49]). These results indicated that the upper canopy is an “edge” where heat flow degression occurs vertically from the atmosphere above the canopy to the understory ([26]). The AMT and ADRT showed similar breakpoints (-16.7 ± 6.5 m and -24.5 ± 3.4 m) with LAI in the upper canopy (-22.6 ± 2.7 m), and the LAI is the major factor limiting the transmission of heat flow in forests ([26], [11]). Although 41.74% of the LAI was concentrated from 0 m to 15 m below the top of the canopy, the emergent crown of P. chinensis was exposed to full sunlight above the main canopy layer, and the diurnal and annual variations in illumination from all directions ensured that the differences in the air temperature (and RH) were not affected by overhead shading. The turbulence is another reason to keep difference in temperature (and humidity) stable, which could mix warm air in upper canopy, and lead a completely decoupled course of temperature and humidity in the understory ([21], [39], [29]).
Vertical profiles of humidity within the forest
Forest canopies have a strong moderating effect on irradiance and wind speed below and within the canopy, and the air temperature moderately decreases within vegetation. These microclimatic trends enhance the RH in the understory ([16]). The AMRH was 10% higher 1.5-2 m above the ground (97 ± 1%) than in the upper canopy (87 ± 1%) in this study, close to previously reported values for AMRH of 82-85% in the rainforest canopy and 80-92% in the understory ([17]).
The diurnal and annual ranges in RH in the vertical profile are important for the settlement of some specific species, such as epiphytes ([41]) and amphibians ([3]). This is particularly true for the forest in our study, because this region is predominated by a typical tropical monsoon climate with a distinct dry season from November to April yearly ([5]). Although the ADRRH in the upper canopy (higher than -24.5 ± 3.5 m) exceeded 30%, and similar diurnal RH differences have been reported in Amazonian rainforests (RH varies from 70% to 100% - [28]) and in French Guiana (RH varies from 81 to 93% - [19]), the ADRRH decreased to 10 ± 5% at 1.5-2 m above ground. A similar vertical gradient in the diurnal range of the RH in the dry season has been reported in another local tropical seasonal rainforest ([25]).
The PLRM estimated the segment of the ADRRH to be at -24.5 ± 3.5 m in Hcanopy, which was consistent with the crown structure of P. chinensis in the emergent layer (Fig. S3 in Supplementary material). The breakpoints for AMRH, MARRH and MART (-40.7 ± 5.9 m, -39.8 ± 3.7 m and -36.0 ± 4.5 m, respectively) were similar to the breakpoint for LAI in the understory (-36.6 ± 6 m), which may have resulted from high plant density in the understory layer limiting air circulation around plants ([37]), and a valley across the crane plot could compensate for the lack of precipitation in the dry season.
The mean value of the MMRH in the profile was the highest in August and the lowest in March; these extreme values occurred approximately one month later than the annual extreme values of the local precipitation (local precipitation is the highest in July and the lowest in February - [9]). Although the correlation between the mean value of the MMRH in the profile and the precipitation in the same month was not high (PCC = 0.376, P > 0.05 - Tab. S4 in Supplementary material), the mean value of the MMRH in the profile was closely correlated with the precipitation of the previous month (PCC = 0.651, P < 0.05), which may be due to the lag effect of local forests on hydrological processes. The soil in local tropical forests has a good water conservation capacity, which significantly increases and delays the base flow ([18]), and this hysteresis effect in local hydrologic process is also embodied in the soil water content and RH in the understory.
Conclusions
When the canopy was used as the reference, the r2 values for all microclimatic factors exceeded those for the ground-referenced model, which indicates that the canopy-referenced approach can better describe the microclimatic variation in the vertical profile within the forest, especially when the canopy structure is heterogeneous and the vertical variation in micrometeorological aspects is nonlinear. The canopy-referenced approach avoided interference from canopy height heterogeneity and was more consistent with the LAI and the foliage distribution in the canopy. This approach therefore exhibited a better fit when used to model the light attenuation process in the vertical profile of the forest.
At least two breakpoints were observed in vertical microclimate profiles of local forests, at approximately -20 m (for AMT, ADRT and ADRRH) and -40 m (for AMRH, MARRH and MART) under the canopy, which corresponded to the transition points for LAI in the vertical profile. At the first breakpoint (approximately -20 m), crown structure of canopy species led to a sharp change in light environment variables such as the DIFN and the R:FR ratio, and microclimatic factors such as the annual mean temperature and the diurnal ranges in temperature and RH; however, turbulence kept the annual ranges in temperature and RH high but stable in the emergent layer. At the second breakpoint (approximately -40 m), the high plant density in the understory layer further increased LAI, limited air circulation around plants, and maintained a low but stable environment in terms of the annual mean RH and the annual ranges in temperature and RH. These discontinuous variations in microclimatic factors can be better observed with the canopy-referenced approach. Canopy-referenced microclimatic profiles may improve our understanding of vertical variation in species composition and biodiversity in tropical forests with high canopies.
List of abbreviations
The following abbreviations have been used throughout the paper:
- LAI: leaf area index
- DIFN: diffuse non-interceptance
- MTA: mean tilt angle of leaves
- R:FR: red to far-red light ratio
- PPFD: photosynthetic photon flux density
- AMT: annual mean temperature
- MMT: monthly mean temperature
- MDRT: monthly mean diurnal range of temperature
- ADRT: annual mean diurnal range of temperature
- ART: annual range of temperature
- MART: mean annual range of temperature
- RH: relative humidity
- AMRH: annual mean relative humidity
- MMRH: monthly mean relative humidity
- MDRRH: monthly mean diurnal range of relative humidity
- ADRRH: annual mean diurnal range of relative humidity
- ARRH: annual range of relative humidity
- MARRH: mean annual range of relative humidity
Acknowledgements
MC, XD, and YD conceived of the ideas and designed the experiment; YD wrote the first draft of the manuscript; JD, WZ, and SY collected and analyzed the data; ZT and QS contributed considerably to the revisions of the manuscript. We thank Dr. Jin Wu for his valuable suggestions on our manuscript and Mr. Hanxiang Yan, Mr. Mingzhong Liu and Mr. PinPing Zeng for their assistance with the field work. We thank the National Forest Ecosystem Research Station at Xishuangbanna, Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences, for assistance in routine maintenance and technical support in the crane plot. We also thank the Management Bureau of Xishuangbanna National Nature Reserve for permission to conduct field work in the nature reserve. This work was supported by National Science Foundation of China (grant no. 32061123003 and no. 41771099) and the Chinese Academy of Sciences 135 Program (grant no. 2017XTBG-T01) and the Field Station Foundation of the Chinese Academy of Sciences.
References
Online | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Jinlong Dong
Wenfu Zhang
Shengdong Yuan
Qinghai Song
Xiaobao Deng
Min Cao
CAS Key Laboratory of Tropical Forest Ecology, Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences, Mengla, Menglun 666303, Yunnan (China)
Xiaobao Deng
Min Cao
Center for Plant Ecology, Core Botanical Gardens, Chinese Academy of Sciences, Mengla, Menglun 666303, Yunnan (China)
Department of Environmental Sciences, Hainan University, Haikou 570228 (China)
Corresponding author
Paper Info
Citation
Deng Y, Dong J, Zhang W, Yuan S, Tan Z, Song Q, Deng X, Cao M (2022). Quantifying the vertical microclimate profile within a tropical seasonal rainforest, based on both ground- and canopy-referenced approaches. iForest 15: 24-32. - doi: 10.3832/ifor3780-014
Academic Editor
Tamir Klein
Paper history
Received: Feb 10, 2021
Accepted: Nov 26, 2021
First online: Jan 27, 2022
Publication Date: Feb 28, 2022
Publication Time: 2.07 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2022
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 37099
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 30529
Abstract Page Views: 3393
PDF Downloads: 2581
Citation/Reference Downloads: 14
XML Downloads: 582
Web Metrics
Days since publication: 1556
Overall contacts: 37099
Avg. contacts per week: 166.90
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2022): 1
Average cites per year: 0.25
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Analysis of canopy temperature depression between tropical rainforest and rubber plantation in Southwest China
vol. 12, pp. 518-526 (online: 09 December 2019)
Research Articles
Canopy temperature variability in a tropical rainforest, subtropical evergreen forest, and savanna forest in Southwest China
vol. 10, pp. 611-617 (online: 17 May 2017)
Research Articles
Distribution of juveniles of tree species along a canopy closure gradient in a tropical cloud forest of the Venezuelan Andes
vol. 9, pp. 363-369 (online: 08 December 2015)
Research Articles
Spatial heterogeneity of light and tree sapling responses in the understory of disturbed montane forests
vol. 8, pp. 448-455 (online: 08 October 2014)
Research Articles
Intra-annual tree growth responds to micrometeorological variability in the central Amazon
vol. 14, pp. 242-249 (online: 21 May 2021)
Research Articles
Net ecosystem production of a tropical secondary forest in Jengka, Pahang, Malaysia
vol. 18, pp. 54-60 (online: 04 April 2025)
Research Articles
The estimation of canopy attributes from digital cover photography by two different image analysis methods
vol. 7, pp. 255-259 (online: 26 March 2014)
Research Articles
How environmental factors condition natural regeneration in the altitudinal gradient of a montane rainforest
vol. 17, pp. 132-139 (online: 04 May 2024)
Research Articles
Identification and characterization of gaps and roads in the Amazon rainforest with LiDAR data
vol. 17, pp. 229-235 (online: 03 August 2024)
Research Articles
Temporal patterns control carbon balance in forest and agricultural tropical peatlands in North Selangor, Malaysia
vol. 14, pp. 362-369 (online: 10 August 2021)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword