Wintertime photosynthesis and spring recovery of Ilex aquifolium L.
iForest - Biogeosciences and Forestry, Volume 12, Issue 4, Pages 389-396 (2019)
doi: https://doi.org/10.3832/ifor2983-012
Published: Jul 31, 2019 - Copyright © 2019 SISEF
Research Articles
Abstract
Former studies using the chlorophyll fluorescence technique on evergreen Ilex aquifolium L. showed that its photosynthetic potential for electron transport in winter recovers quickly when exposed to more favorable conditions. Since little is known, however, about its photosynthetic carbon gain in winter, we investigated its leaf gas exchange over an entire winter and spring period. Measurements were made rotationally in the field and in the laboratory to also investigate if I. aquifolium profits from warmer phases during winter in terms of net carbon gain. From the end of autumn until the end of spring, three different climate-driven phases of photosynthetic responses could be distinguished: first, an acclimation phase which lasted until February and was characterized by a gradually decreasing light-saturated gross photosynthesis (Amax(gross)), decreasing apparent quantum yield of CO2-assimilation (ΦΦi) and a decreasing ability of these parameters to recover overnight inside the laboratory. At the same time, maximal quantum yield of PSII (Fv/Fm) could fully regenerate. In this phase, single warmer days had a positive effect on carbon assimilation. Second, a phase of relatively constant but low photosynthesis which was virtually unaffected by temperature, lasting for almost two months occurred. Here, Amax(gross) and Φi had lost their ability to recover from winter conditions in the field, while Fv/Fm was much less affected. I. aquifolium was still able to conduct positive light-saturated net photosynthesis at a leaf temperature of -0.5 °C, but during this time it could not profit from milder temperatures in terms of carbon gain. Third, a phase of increasing photosynthesis (spring recovery) occurred, starting in March when the 5-day average temperature was above 5 °C and radiation in the field increased, and where all parameters slowly recovered from winter depressions. Our findings show that I. aquifolium is photosynthetically active over the whole winter, even at temperatures around 0 °C. In terms of carbon gain, however, I. aquifolium does not profit from warmer phases during winter, despite the fast recovery seen in chlorophyll fluorescence measurements.
Keywords
Chlorophyll Fluorescence, Frost, Leaf Gas Exchange, Photosynthetic Induction, Quantum Yield, Respiration
Introduction
Photosynthesis of broad-leaved evergreens is sensitive to frost events in winter and this might play a role in limiting their distributional range. Frost can reduce the activity of RuBisCO, inhibit photosynthetic electron transport or even cause irreversible damage to the photosynthetic apparatus ([21]). As an Atlantic-Submediterranean species, Ilex aquifolium L. is one of the few native evergreen broad-leaved trees in Central Europe and it is the northernmost widespread, usually growing in the understory of forests. Its distributional range is closely climate-related and relatively similar to that of European beech (Fagus sylvatica): towards south-west Europe (e.g., Spain), it becomes gradually limited by decreasing water availability; there, I. aquifolium primarily grows at higher altitudes and sites with higher humidity ([24], [1]). The northern and eastern distribution is closely related to the 0 °C isoline in January ([16]). As a result of increasing winter temperatures within the last decades, this isoline has shifted more to the north and east and now runs from the southwest coast of Norway via the south of Sweden to northeast Germany. With the isoline shift, I. aquifolium increased its distribution range across northern and eastern Europe ([2], [32], [4]). It is likely that global warming will continue to shift climate zones in the future and, as a consequence, the distribution ranges of species and compositions of forest ecosystems will change ([28], [7]).
The central populations of I. aquifolium, on the other hand, might profit from generally milder temperatures or single warmer days in winter and could potentially become more established in the understory of northern and eastern European forests: their ability to conduct photosynthesis during winter and early spring leads to an ecological advantage over deciduous trees ([30]). Increasing winter temperatures could additionally increase photosynthetic rates and thus further increase the competitiveness of evergreen species. The ability to immediately take advantage of an extended growing season may also prove advantageous for I. aquifolium in the event of climate change.
In winter, however, leaves of evergreen plants may become photoinhibited, as induced by low temperatures in combination with high light intensities, thereby reducing photosynthetic efficiency, a phenomenon known in conifers of the temperate and boreal zones ([12], [26]) and in many Mediterranean evergreens ([11]). Photoinhibition is accompanied by a decrease in maximal potential PSII efficiency (Fv/Fm), as has been shown for I. aquifolium (originating from different habitats and climates) in experiments in winter ([13], [29], [30]). In the experiments of Groom et al. ([13]) and Veste & Kriebitzsch ([30]), however, Fv/Fm could fully recover within hours after plants were exposed to more favorable conditions. These results, obtained by the chlorophyll fluorescence technique, show a high plasticity of the electron transport related part of photosynthesis.
In this study we additionally investigate the potential of I. aquifolium to profit from warmer phases during winter in terms of carbon gain. Objectives were: (i) to investigate photosynthetic performance (CO2 gas exchange) in I. aquifolium during winter and subsequent spring; (ii) to study if a temperature increase in winter has a positive effect on carbon gain, since an increase in Fv/Fm does not necessarily indicate an increase in the rate of photosynthesis.
Material and methods
Plant material and experimental sites
Five saplings of I. aquifolium L. were used in the experiment. The plants were obtained from a nursery and, to the best of our knowledge, wild-types (no varieties) from northwestern Germany. They were about 70 cm in height, potted in 5 l pots in a fertilized white peat substrate with a pH of 4.5. Three of the plants which were used for field measurements were dug into the ground (but remained in their pots for easy transport) at a nearby site in the Botanical Gardens of the University of Hohenheim, Baden-Württemberg, Germany (48° 42′ 35.79″ N, 09° 12′ 39.6″ E; about 373 m a.s.l.). Mean annual temperature is 9.7 °C with a mean annual precipitation of 736 mm (“Klima- und Wetterstation Hohenheim” from 1981-2010). During the experiment, additional microclimatic data were collected using a WatchDog Weather Station (2000 Series®, Spectrum Technologies, Plainfield, IL, USA) directly next to the plant site. Air temperature (°C) and global radiation (Wm-2) were measured in the field at about 1.5 m height in 10 min intervals. Two plants used as references were kept inside the laboratory at room temperature (≈ 21 °C and ≈ 40% relative humidity, RH) and illuminated 9 hours a day with photosynthetically active radiation (PAR) of up to 400 µmol m-2s-1 under an artificial light source (NAV-T®, 400 W, OSRAM, Germany); plants were well-watered.
Measurement approach
Measurements were carried out from November 2013 to July 2014, always following the same procedure (principle shown in Fig. 1): the evening before plants were measured in the field, one leaf of each plant was wrapped in aluminum foil to keep it in darkness so that photosynthesis could not be induced at sunrise. In the subsequent morning hours, photosynthetic induction gain was measured in these darkened leaves (circles on Monday in Fig. 1), followed by a light response curve (see below). These measurements were identically performed on the same leaves and in the same order during the whole experimental period. During measurements of leaf gas exchange, chlorophyll fluorescence (triangles in Fig. 1) was measured in three further leaves of each plant, resulting in nine leaves in total (see below). In the evening, plants were transported from the field into the laboratory to induce temperature acclimation overnight. Thereafter, the same measurement procedure was repeated the following (Tuesday) morning (now in the laboratory) and chlorophyll fluorescence was measured in three leaves of every reference plant (six leaves in total). Afterwards, plants were once again exposed for at least five days to ambient conditions in the field and until the start of the next series of measurements. On the third day (Wednesday), photosynthetic induction gain and light response curves in one or both reference plants were measured in the laboratory, always using the same leaves over the whole experimental period.
Photosynthetic induction
Low photosynthetic induction strongly limits carbon gain ([33]). The faster dark-acclimatized leaves become induced by light, the faster these limits to carbon gain decline. In order to evaluate if low temperatures critically affect this velocity of induction gain in light, the continuous increase of photosynthetic carbon gain following a rectangular step in light was monitored with the portable gas exchange fluorescence system GFS 3000 (Heinz Walz GmbH, Effeltrich, Germany). Once inside the darkened porometer chamber, leaves were allowed to adjust to the measurement conditions for at least 10 min. For leaves measured in the field, leaf temperature (Tleaf) was kept constant at about 1 °C above ambient temperature at the beginning of the experiment to avoid changes in Tleaf during the measurements. In the laboratory, Tleaf was always kept constant at 21 °C and about 65% RH. In both cases, outdoor and indoor, airflow through the cuvette was maintained at 750 µmol s-1 with a CO2 concentration of 390 µmol mol-1. Once CO2 exchange reached steady state, respiration in darkness (RD) was recorded. Thereafter, leaves were illuminated by a single rectangular step of PAR of 500 µmol m-2 s-1 until light-saturated net photosynthesis (Amax) was reached (full induction).
Photosynthetic induction Aind(t) was then calculated according to Chazdon & Pearcy ([8]) as (eqn. 1):
where A(t) is net photosynthesis at time t. From this, times to reach 50% of full induction (t50%A) were evaluated.
Light response of net photosynthesis
Subsequent to the induction measurements, photosynthetic light response curves were measured in these fully induced leaves. When ambient temperatures during induction measurements rose by a few degrees, leaf temperature was re-adjusted (to 1 °C above ambient). After leaves were acclimatized, measurements started at PAR of 500 µmol m-2 s-1, followed by a stepwise decrease. Data were recorded at PAR of 500, 400, 300, 100, 50, 20, 10, 5 and 0 µmol m-2 s-1, allowing for a new steady-state of CO2 exchange (after 2.5 min) between light steps. From light response curves, several parameters were evaluated: light compensation point (Icomp) as the light at which net photosynthesis is zero; light saturation of photosynthesis (Isat) as the light at which 90 % of Amax is reached; apparent quantum yield of CO2 assimilation (Φi) estimated as the slope of the initial linear part of the light response curve (usually between PAR of 10 and 50 µmol m-2 s-1 but above the so-called “Kok effect”); respiration in light (RI) estimated by extending this linear part to the y-axis and Amax(gross) = Amax + |RI|. Since Tleaf could differ by more than 30 °C between winter and spring, Amax(gross) was used to better illustrate the CO2 uptake without the temperature-dependent effects of respiration.
Chlorophyll fluorescence
Maximum quantum yield of PSII (Fv/Fm) in darkened leaves was determined using a MINI-PAM® (Heinz Walz GmbH, Effeltrich, Germany - eqn. 2):
where Fv is the maximal variable fluorescence and Fm and F0 the maximal and minimal fluorescence yields of the darkened samples respectively. Dark acclimation was initiated for at least 15 min prior to the measurements using leaf clips (DLC-8).
Statistics
To evaluate statistical differences between groups of leaves measured in the field and the same leaves measured in the laboratory, paired t-tests were performed with Sigmaplot® ver. 12.0 (Systat Software, San Jose, CA, USA). Because of leaf-specific differences, data have been normalized prior to statistical tests.
Results
Weather conditions during the experimental period
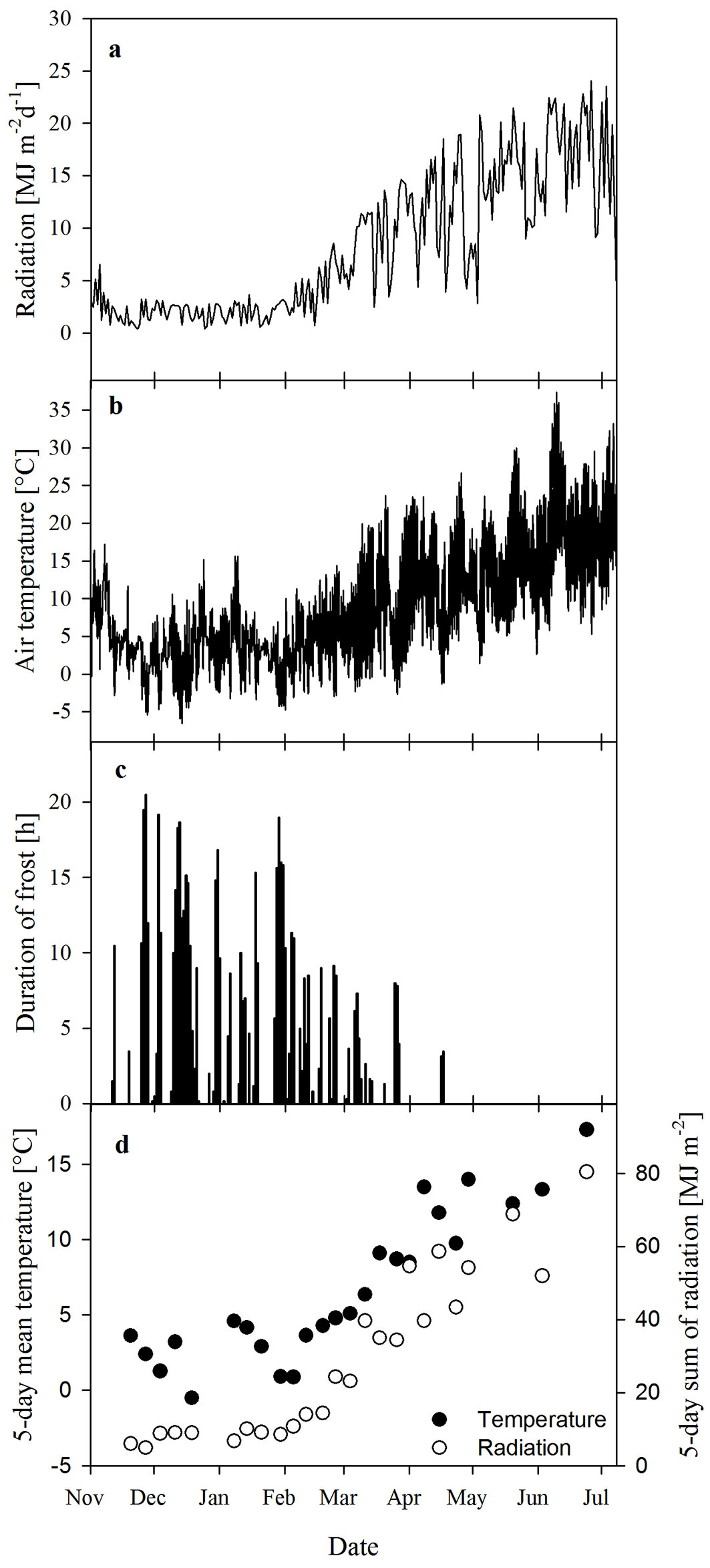
Daily radiation, air temperature and duration of frosts during the period of measurements are shown in Fig. 2a-c. Overall, temperatures were exceptionally mild during the winter of 2013/2014 (fourth warmest winter in Germany since 1881 - [9]), so that no severe frost was observed and frosty periods never lasted for a complete day. Air temperatures ranged from -6.5 °C in December 2013 to 37.4 °C in June 2014. Freezing usually occurred overnight or in the early morning. There were only a few days where temperatures of 0 °C or slightly below lasted during daytime. Fig. 2d shows mean temperatures of the five days (Wednesday to Sunday in Fig. 1) prior to the measurements conducted in the field, as well as the sum of radiation over these five days. The coldest 5-day periods (means < 1 °C) plants were exposed to in the field occurred during mid-December (mean of -0.5 °C) and two periods in series between the end of January and beginning of February (both means ≈ 0.9 °C - Fig. 2d).
Fig. 2 - Climatic conditions at the field site. (a): radiation; (b): air temperature; (c): duration of frost (including 0 °C); and (d): mean temperature and sum of radiation over the five days prior to each set of field measurements.
Respiration and photosynthesis in winter
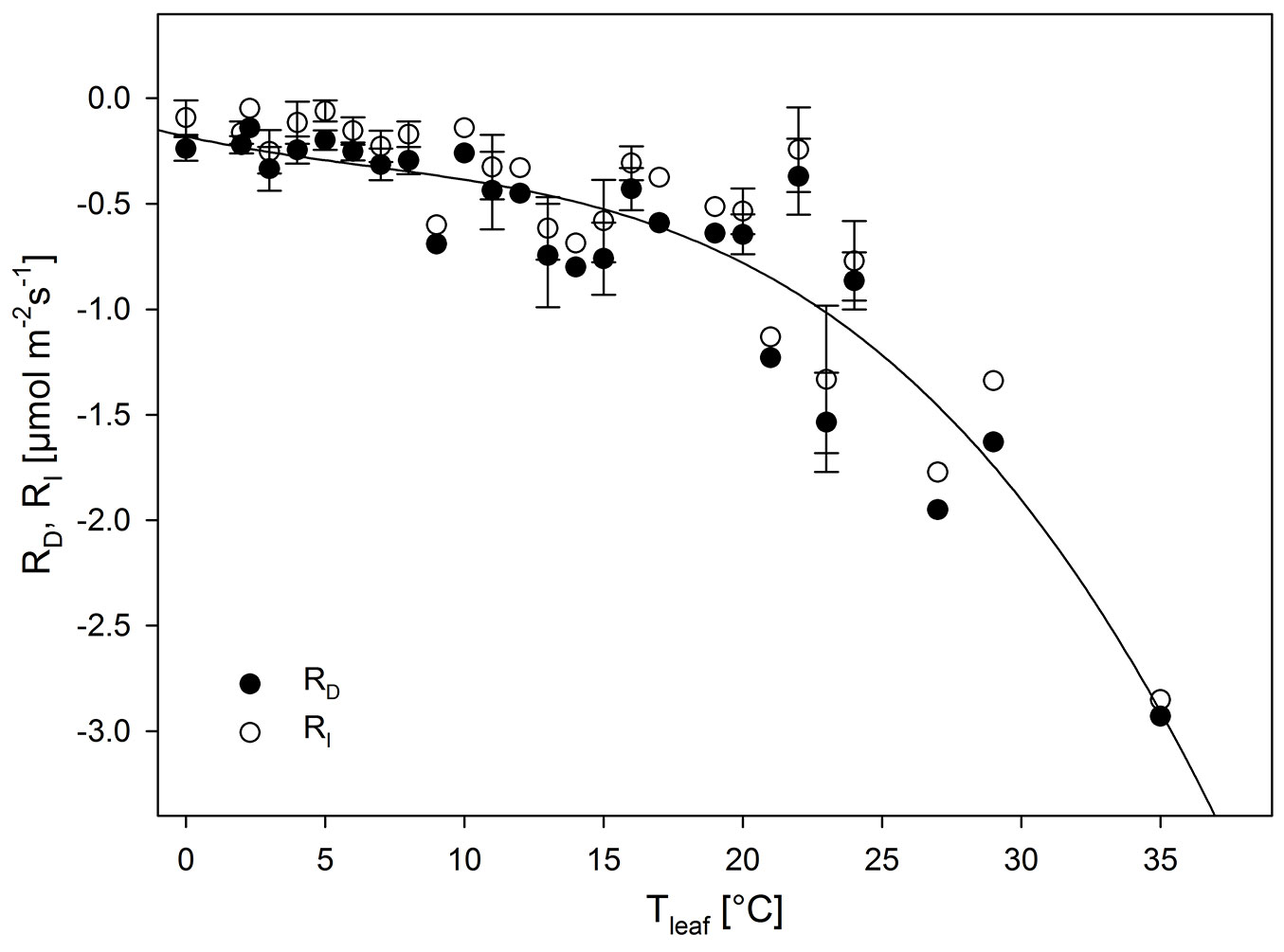
Respiration during winter was relatively low, since RD and RI clearly depended on temperature in leaves of all plants and over the whole period (Fig. 3). From 0 °C to about 10 °C, RD and RI showed the lowest respiration (i.e., least negative values), which did not change significantly within this temperature range (mean values of |RD| at 0.2-0.3 µmol m-2s-1, and |RI| as low as 0.05 µmol m-2s-1). Between 10 °C and about 20 °C there was a clear increase in respiration and, with the beginning of spring, a large increase from 20 to 35 °C.
Fig. 3 - Respiration in darkness (RD) and in light (RI) at different leaf temperatures (Tleaf) as obtained over the whole experimental period (November to July). Values are means ± SE (n = 1-8). RD is also described as the regression: RD = -0.1806 - 0.0291 · Tleaf + 0.0018 · T2leaf - 0.0001 · T3leaf; r2=0.73.
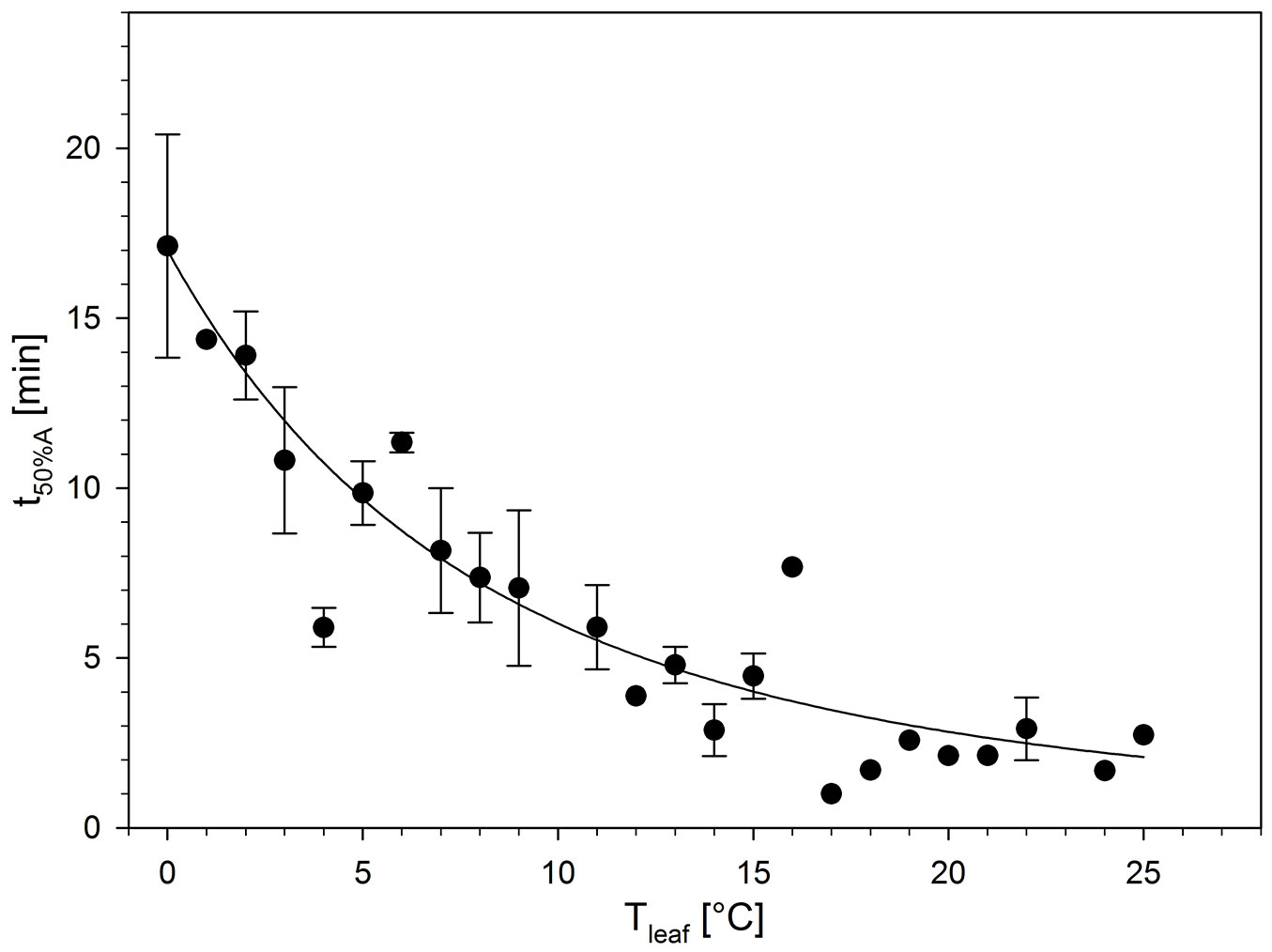
Slower activities at low temperatures could also be observed for the velocity of photosynthetic induction, which depends on the activity of enzymes in the Calvin-cycle and on stomatal opening (see review by [17]). Independent of photosynthetic rates, photosynthesis was induced more quickly with increasing Tleaf (measured as time to reach 50 % of full induction, t50%A) from more than 17 min at 0 °C to less than 3 min at Tleaf above 15 °C (Fig. 4). These temperature-affected induction times are well within the range of other broad-leaved (but deciduous) trees, e.g., Fagus sylvatica and Betula pubescens, observed when stomata did not limit the induction process ([31]). We conclude that in I. aquifolium, even temperatures as low as 0 °C do not critically reduce the velocity of photosynthetic induction in light.
Fig. 4 - Time to reach 50 % of full photosynthetic carbon gain induction (t50%A) in relation to leaf temperature (Tleaf). Values are means ± SE (n = 1-12). t50%A is also described as the regression: t50%A = 0.0233 · exp(352.6577 / (Tleaf + 53.5026)); r² = 0.61.
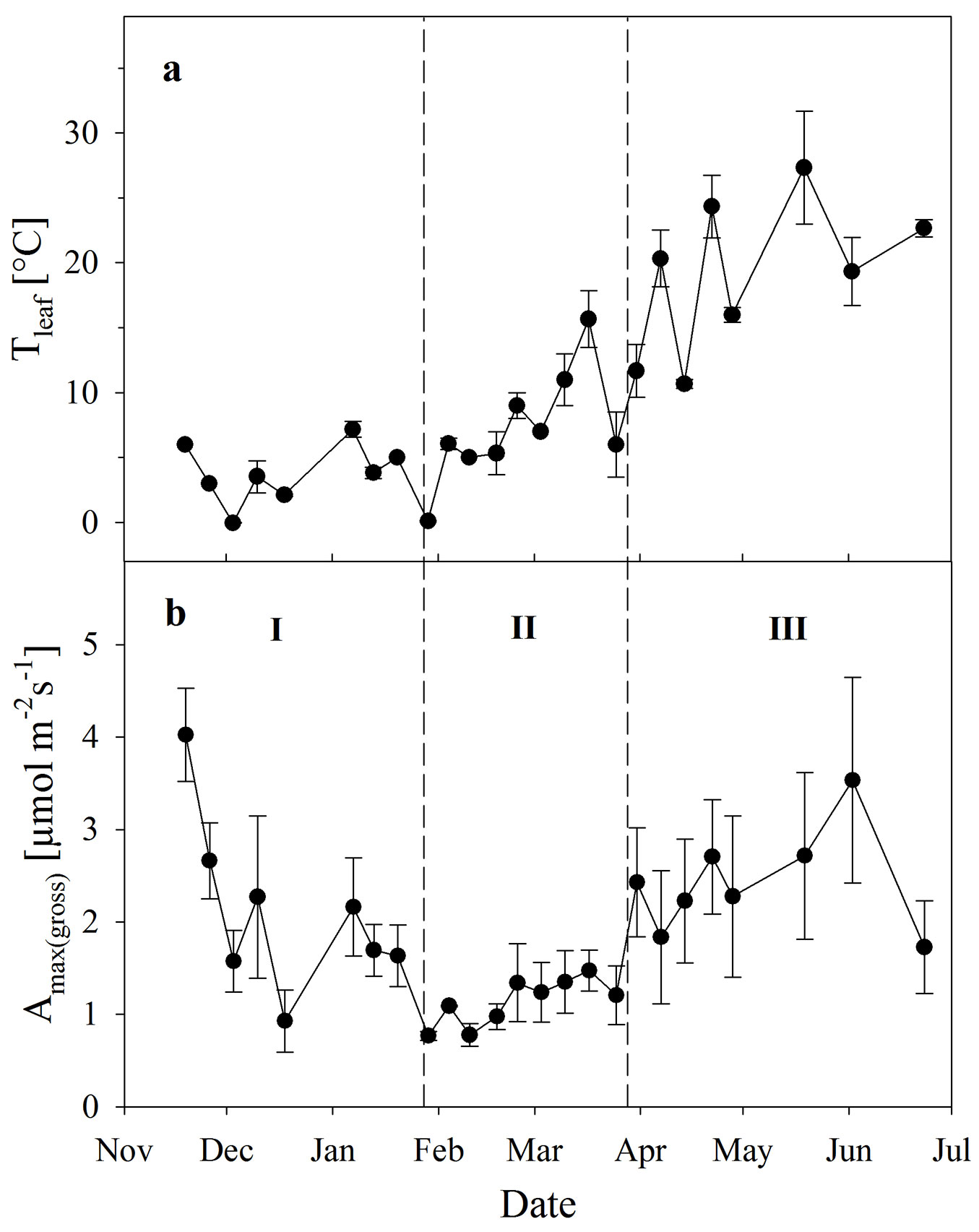
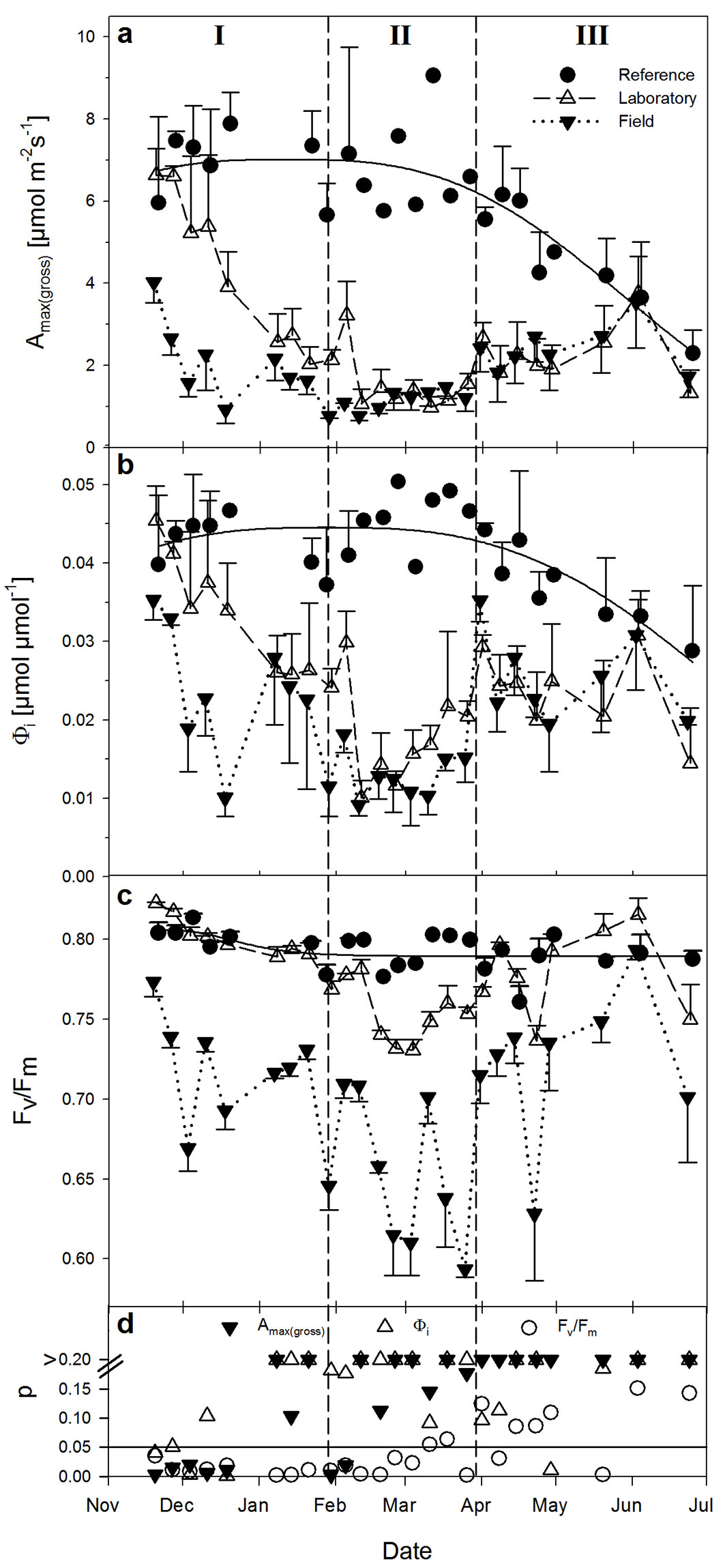
There were no such clear responses to temperature in other photosynthetic parameters (Amax, Icomp, Isat - data not shown). Amax(gross) as measured in the field changed with time and from this, three distinct phases could be visually distinguished (Fig. 5b): phase (I) of generally decreasing photosynthesis, which lasted from the beginning of measurements to February; phase (II) of low photosynthesis lasting from February to April; and a recovery phase (III) from April to June.
Fig. 5 - Leaf temperature (a) and light-saturated gross photosynthesis (b) derived from light response curves as measured in the field (means ± SE, n = 3). I, II, III show different phases of photosynthetic performance based on the magnitude of Amax(gross) (for further explanation see the text).
During phase I, Amax(gross) was not only affected by Tleaf, but very likely also by the acclimation to long-lasting low air temperatures (and frosts) in combination with low radiation (Fig. 2), these being the conditions which induce frost-hardening ([15], [22]). Until about February, Amax(gross) decreased and increased with Tleaf (Fig. 5a, Fig. 5b). Even at a Tleaf of 0 °C (December 3) leaves of Ilex exhibited Amax(gross) of 1.57 ± 0.34 µmol m-2s-1 (mean ± SE, n = 3). Low temperatures (5-day mean temperatures below 4 °C, in one case even -0.5 °C) and likely also the continuously low radiation (Fig. 2d) could have resulted in further frost-hardening of photosynthesis, so that even lower photosynthetic rates than those observed in December resulted for the same Tleaf, e.g., now Amax(gross) was only 0.76 ± 0.05 µmol m-2s-1 (n = 3) at Tleaf of 0 °C (on January 29th).
During the following weeks until April (phase II), Amax(gross) in the field was lower for the same or higher Tleaf compared to phase I, and remained at a relatively consistent low rate despite the increasing temperature (Fig. 5). Although Amax(gross) already decreased during phase I, it is worth noting that phase II started with the longest-lasting cold period in the field (transition from January to February - Fig. 2d).
From April onwards (phase III), when temperature and radiation in the field markedly increased (Fig. 2), photosynthesis gradually recovered so that Amax(gross) in the field clearly increased again with rising temperatures. Although leaves reached photosynthetic rates as high as at the beginning of the experiment (Fig. 5b), this occurred at a higher Tleaf (Fig. 5a), where an even higher photosynthetic rate would have to be expected.
The apparent quantum yield of CO2 assimilation (Φi, Fig. 6b) behaved relatively similar to what was observed for Amax(gross) in the field (Fig. 5 and Fig. 6a). The response of Fv/Fm was also similar, showing lowest values at highest variation during phase II (Fig. 6c). Both light compensation point and light saturation of net carbon gain (not shown) increased with time from an average of about 11 µmol m-2s-1 (Icomp) and 149 µmol m-2s-1 (Isat) in phase I to 27 µmol m-2s-1 and 158 µmol m-2s-1 in phase II and 36 µmol m-2s-1 and 176 µmol m-2s-1 in phase III. Based on the relatively low Icomp during winter, it can be assumed that I. aquifolium potentially exhibited a positive net daily photosynthesis on most winter days. Moreover, even in winter, light-saturation of photosynthesis was reached almost every day. In combination with low respirational losses at low temperatures during nighttime (Fig. 3), one can assume a low but positive carbon balance on the leaf level in this species over the whole winter period.
Fig. 6 - Courses of light-saturated gross photosynthesis (a), apparent quantum yield of CO2-assimilation (b) and maximal quantum yield of PSII (c) over time in leaves of reference plants (closed circles, n = 1-2), in leaves of plants measured in the field (closed triangles, n = 3) and the same leaves measured in the laboratory after one night of acclimatization (open triangles, n = 3); all means ± SE. Graph (d) shows the level of significance (p-value) derived by paired t-tests between leaves measured inside the laboratory and the same leaves measured in the field (as shown in a-c).
Short- and long-term recovery of photosynthesis
In order to quantify the short-term recovery of photosynthesis from ambient winter conditions, plants were always taken into the laboratory for one night after having been measured in the field. After about 13 to 15 h at laboratory temperature, measurements were repeated here and compared to both their photosynthetic performance in the field and that of the reference plants. Although measurements in the laboratory were always performed under the same conditions (Tleaf = 21 °C), all three phases of photosynthetic performance described above could be observed here (Fig. 6a-c).
Phase I: during the first weeks Amax(gross), Φi and Fv/Fm of the same leaves reached significantly higher values in the laboratory (open triangles in Fig. 6a-c) than in the field (closed triangles). Over time, however, Amax(gross) and Φi clearly decreased. At the end of Phase I, significant differences between measurements in the field and laboratory could only be observed for Fv/Fm (Fig. 6d).
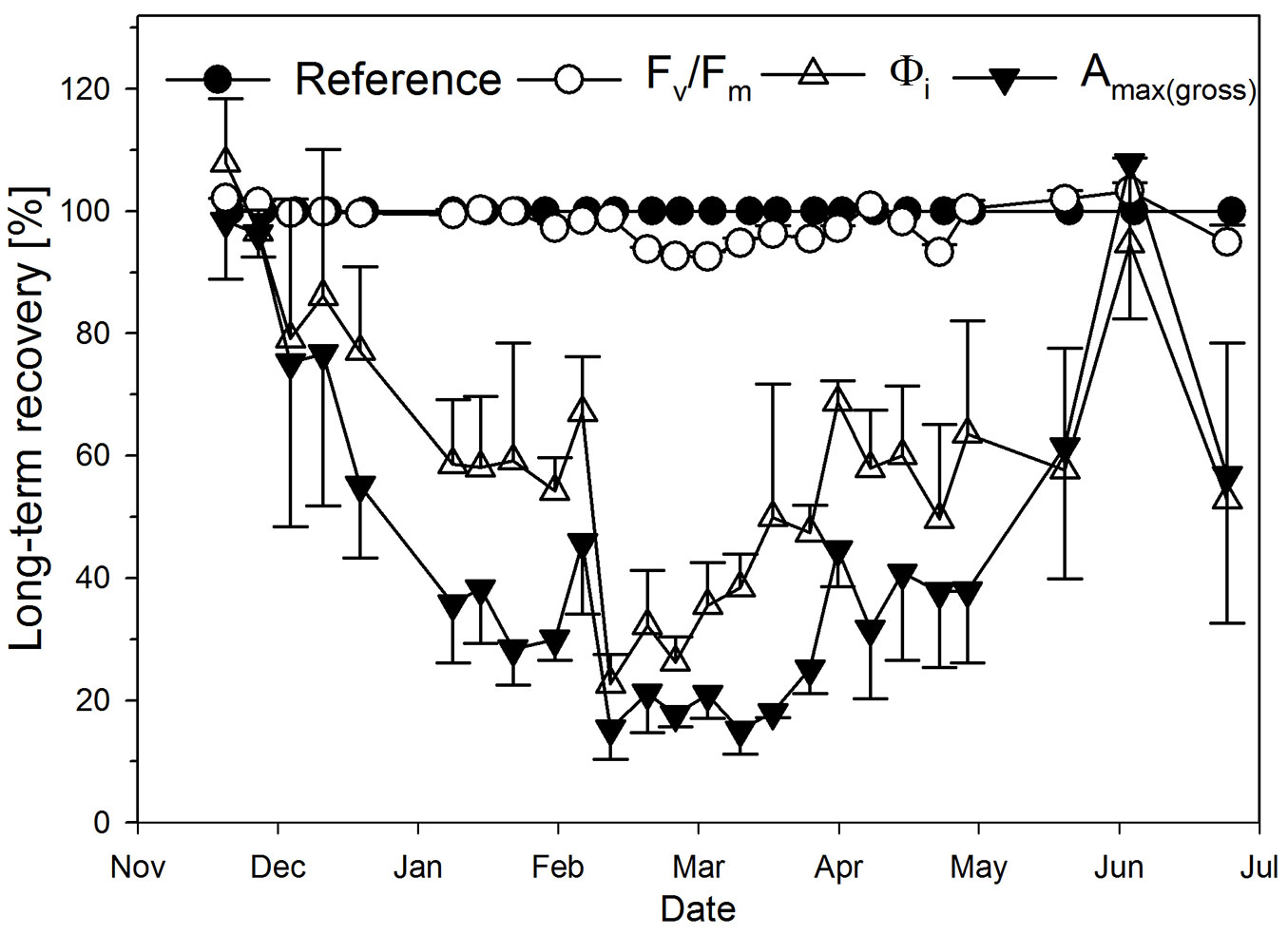
To evaluate the potential of photosynthetic parameters to recover in the long-term (Fig. 7), parameters of field plants measured in the laboratory (open triangles in Fig. 6a-c) were set into relation to that of the reference plants, taking reference values at a given date via regression curves in Fig. 6 always as 100% irrespective of absolute changes over time: within the first two weeks from the beginning of measurements, all photosynthetic parameters were able to fully recover within one night at laboratory conditions. Afterwards, regeneration capability of Amax(gross) and Φi decreased gradually, while Fv/Fm could fully regenerate over the whole of phase I (Fig. 7).
Fig. 7 - Changes in recovery of photosynthetic parameters in the long-term: photosynthetic parameters of field plants which were measured after one night of acclimatization in the laboratory (see open triangles in Fig. 6a-c), in relation to the same parameters of reference plants (always taken here as 100% via the respective regression curves of Fig. 6a-c).
Phase II: the frost-hardening in phase I ([22]) was very likely completed in phase II. Consequentially, all photosynthetic parameters showed lowest rates, both in the field and after short-term recovery in the laboratory (Fig. 6a-c). Testing these variations for statistical significance (Fig. 6d), however, shows a recovery from field conditions for Amax(gross) at the beginning of phase II (closed triangles at p < 0.05), but no longer thereafter. In contrast, the electron transport related Fv/Fm recovered significantly during most of phase II, although not completely (open triangles in Fig. 6c and open circles in Fig. 7). This physiological state lasted for almost two months.
At this point one might question the artificial determination of phases. Based on Amax(gross) one could argue for the beginning of phase II after the last observed recovery in February, whereas our decision was based on the absolute amount of Amax(gross) remaining low from the end of January onwards (Fig. 5b). Nevertheless, results obtained and conclusions drawn would not be affected.
Phase III: with the beginning of spring at the end of March, photosynthesis increased again both in the field and in the laboratory (Fig. 6a-c), thus showing no significant differences between field and short-term recovery over most of the time (Fig. 6d). With time, all parameters gradually recovered from winter conditions (Fig. 7). This long-term recovery of photosynthesis in spring is very likely a consequence of increasing ambient temperature and radiation (Fig. 2a, Fig. 2b, Fig. 2d).
Discussion
Photosynthesis in winter
There are two completely different major traits in plants concerning mechanisms for adjusting photosynthesis to winter conditions, probably as a consequence of different leaf longevities. While herbaceous winter annuals (e.g., winter wheat) adjust their photosynthesis to maximize carbon gain during winter, they retain a higher photosynthetic activity at the risk of (total) leaf damage by frost. In contrast, conifers protect their much longer-lived leaves from frost damage by inducing frost resistance and photoprotection at the cost of drastically decreased carbon gains ([3], [25], [10]). This reduction of photosynthesis during winter is reported in many studies and is therefore quite well-known ([14]). The vast majority of this research, however, has been conducted on conifers, but only rarely on evergreen broadleaved trees. Our results show that down regulation of photosynthesis during winter in I. aquifolium follows the same “strategy” as mentioned above for conifers.
We observed decreasing light-saturated photosynthetic rates in the field from November to February which could not be explained by decreasing temperatures alone (Fig. 5). Also, the ability of photosynthetic parameters (Amax(gross) and Φi) to recover overnight under laboratory conditions declined with time (Fig. 6a, Fig. 6b). This behavior likely resulted from a gradually developing freezing tolerance (frost-hardening) in leaves, as has been shown by Bauer et al. ([3]) for different conifers, and which is induced by long-lasting low temperatures in combination with reduced radiation and a shorter photoperiod ([15], [22]). The assumed process of frost-hardening lasted for more than two months and was completed after the longest-lasting cold period in the field at the beginning of February. As a result of the frost-hardening (and probably of chronic photoinhibition, see below), the following phase (February and March) was characterized by continuously low rates of photosynthetic carbon gain and the lowest ability of all photosynthetic parameters to recover from winter conditions. Nevertheless, even during this time, I. aquifolium exhibited a low but positive net photosynthesis at a Tleaf of around zero with sufficient light.
In Central Europe, I. aquifolium is naturally growing in the understory of deciduous beech forests at reduced light availability during the vegetation period. In this situation, even a low carbon gain over winter (below the now leafless canopies) might be important for the annual carbon gain. For example, Katahata et al. ([18]) reported that carbon gain in the evergreen Daphniphyllum humile (an understory shrub growing in cool-temperate deciduous forests in Japan) during the leafless period in autumn accounted for approximately 49 % of its annual carbon gain. This might be similar in I. aquifolium, since leaves measured here showed relatively high potential carbon assimilation until the end of January (Fig. 5b). However, the gradually increasing inability to regenerate photosynthesis overnight in the laboratory leads to the assumption that I. aquifolium can only slightly profit (in terms of carbon gain) from single warmer days in winter once the frost-hardening is initiated, and it cannot profit at all once frost-hardening is completed. It is even possible that net carbon gain slightly decreases due to increased respiration during warm periods ([6]). Similar results have been recently shown by Bowling et al. ([5]) by use of the eddy covariance method in a subalpine conifer forest in the Rocky Mountains of Colorado, where an increase of photosynthetic carbon gain during favorable days in winter could not be detected when trees were in dormancy.
These findings seem contradictory to chlorophyll fluorescence measurements, which indicate a much better potential for photosynthesis on warm days in winter. From experiments by Groom et al. ([13]) and Veste & Kriebitzsch ([30]) we know that Fv/Fm in I. aquifolium can recover within hours when cold acclimated leaves were transferred to more favorable conditions, thus indicating a high plasticity of the light absorbing part of the photosynthetic apparatus. These results are in agreement with our observations. The above-mentioned authors, however, did not measure leaf gas exchange. From our results we cannot confirm this fast and almost complete recovery overnight for the actual photosynthetic carbon assimilation rate. A longer acclimation time, however, might alter this result. Acclimation at favorable conditions (18 °C) lasting about 3.5 times longer than in our experiment (48 h) increased light saturated rates and apparent quantum yield of photosynthetic O2-evolution in Pinus sylvestris to fully recover from outside winter conditions ([23]). In support of this, Wieser ([34]) showed that CO2 gas exchange in Pinus cembra at an alpine timberline was almost completely suppressed during winter and net photosynthesis increased up to 30% of summer values during an extended warm period.
Photoinhibition
Under high light conditions, especially in winter when temperatures are low, photosynthesis of I. aquifolium (and other plants) may suffer from photoinhibition, as indicated by a reduction in Fv/Fm ([13], [29]). Veste & Kriebitzsch ([30]) have shown that the potential PSII efficiency (Fv/Fm) in I. aquifolium shows a high plasticity during winter and that Fv/Fm could fully recover within two hours when plants were transferred from temperatures close to 0 °C (field) to 20 °C (greenhouse). According to these studies, the Ilex leaves in our study experienced photoinhibition over most of the experimental period when in the field. During phases I and III, Fv/Fm could, however, fully recover overnight after plants were transferred into the laboratory (Fig. 6c), which indicates that certain sections of the photosynthetic apparatus were not suffering longer lasting “stress” over these periods. If Fv/Fm in darkened leaves does not recover after 2 h of acclimation at favorable conditions, then photoinhibition can be seen as chronic ([20]). This was the case here during phase II (February and March), when even after over 12 h at laboratory conditions, Fv/Fm could not fully recover so that a longer lasting PSII depression can be assumed, which is also supported by decreased Φi (Fig. 6b, Fig. 6c).
Spring recovery and leaf aging
For boreal coniferous forests, Suni et al. ([27]) showed that the beginning of spring-recovery of photosynthesis correlates best with a 5-day average of ambient temperatures (between 3.3 °C and 6.5 °C). In our study, spring-recovery of Amax(gross) was most likely initiated at the end of phase II in mid-March when the 5-day average temperature was above 5 °C and radiation clearly increased (Fig. 2d, Fig. 7). Nevertheless, such threshold values remain questionable. Independent of this, the recovery of Fv/Fm and Φi began about 1-2 weeks earlier than that of Amax(gross) (Fig. 7), making a reduction of photoinhibitory stress likely (see above).
Although Amax(gross) clearly rose again with the beginning of spring, leaves exhibited lower photosynthetic rates as compared to the previous autumn. A reason for these lower rates might be that photosynthesis has not yet fully recovered from winter, or leaves measured here during early and late spring could have come close to the end of their life spans. On average, leaves of I. aquifolium have a longevity of two years, and their photosynthetic rates gradually decrease with age ([19]). A gradual decrease of photosynthetic rates over time was also observed in leaves of both reference plants in the laboratory (Fig. 6a) despite continuously favorable conditions.
Conclusion
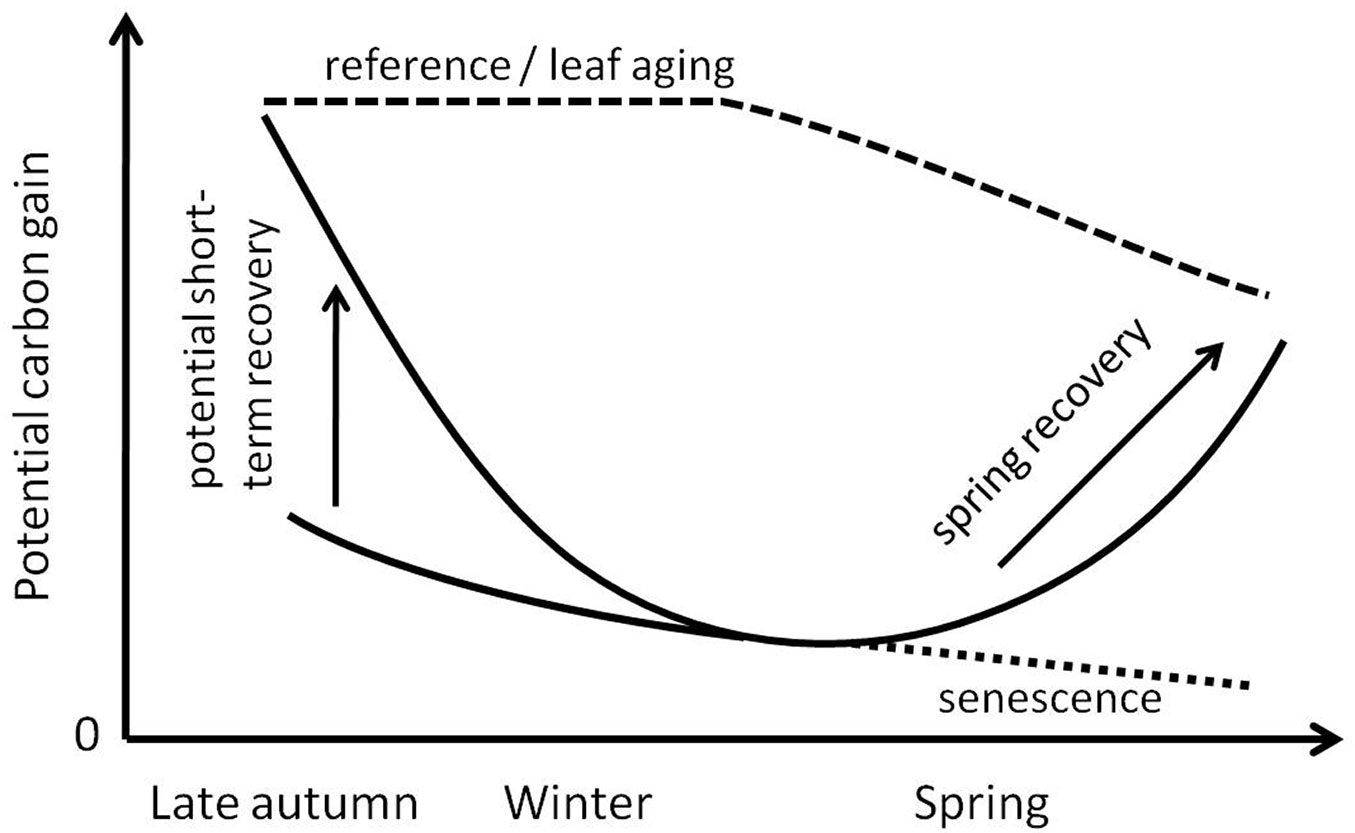
Similar to evergreen conifers, evergreen I. aquifolium down-regulates photosynthesis during winter. During the process of frost hardening, photosynthesis gradually loses its ability to recover under more favorable conditions (this is schematically summarized in Fig. 8). One night of warm temperatures is not sufficient to induce recovery during the hardened phase, where rates of photosynthetic carbon gain stay at about 15% to 20% of those in the pre-hardened phase. Chlorophyll fluorescence measurements indicate a much better recovery potential and implicate higher rates of photosynthetic carbon gain than could be observed, and can, therefore, not reliably predict carbon gain during winter. Obviously the light absorbing part of photosynthesis enabling the early electron transport chain (Fv/Fm) behaves differently from the following biochemistry, especially in the Calvin-cycle (Amax). If not in the complete steady-state, they may behave uncoupled. With regards to carbon gain, I. aquifolium in Central Europe might not profit significantly from single warmer days in winter unless the pre-hardening phase gets extended (e.g., by global warming) or the frost-hardened phase becomes shortened.
Fig. 8 - Simplified scheme of potential carbon gain (Amax(gross), Φi) of I. aquifolium over the time when deciduous trees are leafless, and the capability of the photosynthetic apparatus to recover from low temperatures during a warm day in autumn and winter (short-term recovery), as based on the observations presented in Fig. 6 and Fig. 7.
Acknowledgements
We thank Margaret Eppli (Hohenheim, Stuttgart, Germany) for proofreading the manuscript and correcting the English.
References
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Mona Schloz
Manfred Küppers
Aylin Güney 0000-0002-8955-2770
Maik Veste 0000-0003-2704-2588
Universität Hohenheim, Institut für Botanik 210a, Garbenstraße 30, D-70599 Stuttgart (Germany)
CEBra - Centrum für Energietechnologie Brandenburg e.v., Friedlieb-Runge-Straße 3, D-03046 Cottbus (Germany)
Corresponding author
Paper Info
Citation
Wachendorf M, Schloz M, Küppers M, Güney A, Veste M (2019). Wintertime photosynthesis and spring recovery of Ilex aquifolium L.. iForest 12: 389-396. - doi: 10.3832/ifor2983-012
Academic Editor
Claudia Cocozza
Paper history
Received: Oct 23, 2018
Accepted: May 21, 2019
First online: Jul 31, 2019
Publication Date: Aug 31, 2019
Publication Time: 2.37 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2019
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 46130
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 38593
Abstract Page Views: 3710
PDF Downloads: 2895
Citation/Reference Downloads: 7
XML Downloads: 925
Web Metrics
Days since publication: 2490
Overall contacts: 46130
Avg. contacts per week: 129.68
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2019): 3
Average cites per year: 0.43
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Relationships between leaf physiognomy and sensitivity of photosynthetic processes to freezing for subtropical evergreen woody plants
vol. 12, pp. 551-557 (online: 17 December 2019)
Research Articles
Magnolia grandiflora L. shows better responses to drought than Magnolia × soulangeana in urban environment
vol. 13, pp. 575-583 (online: 07 December 2020)
Research Articles
Calibration of a multi-species model for chlorophyll estimation in seedlings of Neotropical tree species using hand-held leaf absorbance meters and spectral reflectance
vol. 9, pp. 829-834 (online: 17 May 2016)
Research Articles
Gas exchange characteristics of the hybrid Azadirachta indica × Melia azedarach
vol. 8, pp. 431-437 (online: 17 December 2014)
Research Articles
Adaptive variation in physiological traits of beech provenances in Central Europe
vol. 11, pp. 24-31 (online: 09 January 2018)
Research Articles
Day and night respiration of three tree species in a temperate forest of northeastern China
vol. 8, pp. 25-32 (online: 26 May 2014)
Research Articles
Light acclimation of leaf gas exchange in two Tunisian cork oak populations from contrasting environmental conditions
vol. 8, pp. 700-706 (online: 08 January 2015)
Research Articles
Recovery of water potential and leaf gas exchange performance following drought stress in Quercus cerris populations
vol. 19, pp. 94-101 (online: 17 March 2026)
Research Articles
Links between phenology and ecophysiology in a European beech forest
vol. 8, pp. 438-447 (online: 15 December 2014)
Short Communications
Effect of four levels of shade on survival, morphology and chlorophyll fluorescence of Nothofagus alessandrii container-grown seedlings
vol. 8, pp. 638-641 (online: 08 January 2015)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword