Can we expect more from Alnus glutinosa (L.) Gaertn. in a changing climate?
iForest - Biogeosciences and Forestry, Volume 19, Issue 2, Pages 69-76 (2026)
doi: https://doi.org/10.3832/ifor4842-019
Published: Mar 08, 2026 - Copyright © 2026 SISEF
Research Articles
Abstract
Black alder (Alnus glutinosa [L.] Gaertn.) is recognized as a key tree species in moist habitats, riparian ecosystems, and wetland restoration projects. The future success of A. glutinosa establishment is believed to be at risk due to the increasing frequency of high-temperature and drought events associated with climate change. However, some studies have indicated that alder may tolerate greater drought stress than previously assumed. In this study, a series of greenhouse experiments was conducted to assess the sensitivity of black alder to drought during seed germination and seedling establishment in its first year. The results showed that alder seeds can withstand unfavourable germination conditions (e.g., radiation, precipitation, heat) for periods exceeding 16 weeks and still germinate successfully thereafter. Consequently, the expected increase in spring and summer droughts due to climate change will not affect the success of alder germination in years with extended periods of precipitation. Additionally, germination success appears unaffected by the absence of cool spring temperatures. Warmer spring temperatures are necessary for germination and the presence of open mineral soil. Covering dry soil with litter and moss proved problematic, and as drought conditions worsen, there are no safe alternatives to exposed mineral soil. Shading was found to significantly enhance germination success. This allows alder to germinate in a changing climate in shaded locations that remain moist for longer. During germination on open soil, alder seeds did not exhibit specific requirements for soil moisture compared to other deciduous pioneer tree species. Furthermore, drought conditions did not significantly affect growth parameters, including shoot and root growth, root collar diameter, and biomass; however, there was a tendency toward reduced shoot length and belowground biomass under drought stress. Importantly, alder seedlings cannot survive dry periods exceeding 3-6 weeks without soil moisture during spring, summer, or autumn, consistent with other pioneer species such as Betula pendula, Salix caprea, and Populus tremula. These findings support the hypothesis that alder trees are more drought-tolerant than previously thought.
Keywords
Black Alder, Drought Stress, Seed, Germination, Seedling, Radiation, Biomass, Riparian Ecosystem
Introduction
Alnus glutinosa (L.) Gaertn., commonly known as black alder, is a typical pioneer tree species that requires light to sunny conditions for establishment. Alder trees exhibit rapid early growth and a short lifespan, but suffer from competition by other tree species ([6], [18]). Black alder is an important pioneer species in moist habitats and riparian ecosystems, providing habitat for fauna and flora, enhancing soil nutrient levels via nitrogen fixation, stabilizing the soil to protect against erosion, and filtering pollutants from soil and water ([6], [22]). For alder to exert a positive influence on riverbanks, it is essential that it regenerates and establishes successfully in these areas.
Black alder seeds germinate optimally on mineral soil with sufficient soil moisture and high sunlight ([24], [25], [31], [39]). Climate change is leading to declines in cool spring temperatures, a general rise in temperatures, an increase in the frequency of high-temperature events, and reduced rainfall during spring and summer, resulting in frequent and prolonged drought periods ([19]). These changes are also affecting riverbanks and floodplains, causing soil surfaces and upper soil layers to dry out despite the moist environment. This raises the question of how strongly black alder regeneration will be affected by these changing conditions in wetlands.
Certain species necessitate cold temperatures before or during germination, whereas others require warm conditions or a combination of these factors. MacCarthaigh & Spethmann ([23]) recommend a cold treatment of alder seeds lasting three to five weeks at temperatures between 0.5 and 1 °C to promote germination. Thus, the expected decline in cool spring temperatures could negatively affect alder. Alder seeds possess an oily seed coat, a thicker seed testa, and a higher surface-to-volume ratio, which necessitate greater water uptake for swelling and germination, as compared to other pioneer tree species ([25], [11], [38]). Eschenbach & Kappen ([10]) emphasize that alder seeds cannot germinate and establish on dry sites ([22]). According to Gomes Marques et al. ([12]), the future establishment success of Alnus glutinosa is likely to be threatened by climate change. The authors highlight potential challenges, particularly concerning the restoration of riverbanks and disturbed wetland sites ([12]). This raises the question of whether covering or shading the ground could create alternative site conditions that would prolong soil moisture availability, thereby preserving the alder’s germination potential. Additionally, it is relevant to consider the duration for which alder seeds can withstand dry conditions and direct sunlight on the soil surface while remaining viable for germination upon re-watering. Further, it is known that seeds stored at room temperature and high humidity, especially when subjected to repeated swelling and drying cycles due to precipitation, tend to lose their germination capacity more rapidly than seeds stored under dry, cold conditions at freezing temperatures, where metabolic activity and reserve consumption are minimized ([32], [33]).
Zhu et al. ([41]) demonstrated that alder trees may tolerate more drought than previously assumed. Nevertheless, most seedlings of pioneer tree species in temperate forests cannot survive dry periods of more than three weeks ([37]), though there is still a lack of sufficient studies on this topic. Zhu et al. ([41]) wrote: “In the future, more detailed experiments under well-controlled conditions as well as long-term field investigations are recommended to have a deep understanding of the effects of projected drought and heat events on this tree species”.
In this study, we investigated the drought stress sensitivity of Alnus glutinosa during seed germination and seedling establishment within the first year under controlled conditions. Four questions guided this research: (i) Could a reduction in cooling periods in winter and spring have negative effects on the germination of alder seeds in a changing climate? (ii) Are there alternative germination sites for alder, such as shady or covered soils, if dry and sunny mineral soils prove to be poor germination sites as a result of climate change? (iii) Can alder seeds withstand dry conditions and direct sunlight on the soil surface and still germinate later? (iv) How long can alder seedlings survive a dry period in spring and summer-autumn?
The findings from these studies are crucial for understanding the drought stress sensitivity of Alnus glutinosa and assessing its vulnerability to climate change.
Materials and methods
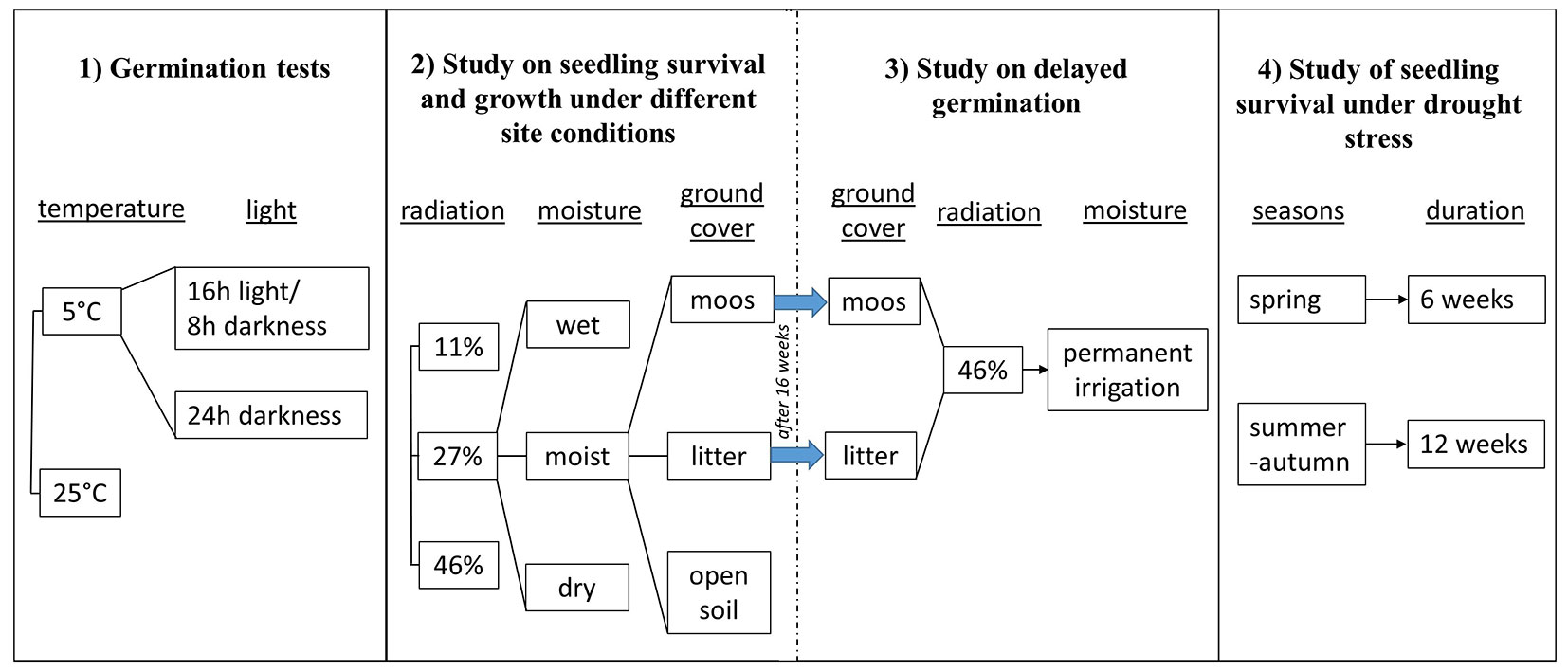
The study consists of four experiments: (1) germination tests, (2) seedling survival and growth under different site conditions, (3) delayed germination, and (4) seedling survival under drought stress (Fig. 1). Only studies 2 and 3 were linked. All other studies were independent and conducted under different conditions, as shown in Fig. 1and described below.
Fig. 1 - Flow chart of the experimental designs for the four experiments: (1) germination tests, (2) seedling survival and growth under different site conditions, (3) delayed germination, and (4) seedling survival under drought stress. The vertical solid lines indicate separate experiments, and a dashed line indicates linked experiments.
The seeds of Alnus glutinosa for all four experiments were obtained from the seed kiln of the state forest enterprises of Saxony in Flöhe, Germany. The seeds were collected in 2019 in hilly locations in Saxony, Germany, and exhibited a germination rate of 71% in 2023.
(1) Germination tests
Germination tests were conducted in a climate chamber to determine whether A. glutinosa seed germination depends on temperature or light availability. High spring or autumn temperatures and the absence of cold periods can influence the germination behaviour of tree species ([23]). Therefore, germination tests following the ISTA ([20]) guidelines were performed in summer and autumn 2023 using Jacobsen seed germination kits with domes. The germination tests included four variants, combining two temperature levels (25 and 5 °C) with two light conditions (16 h light/8 h darkness and 24 h darkness - Fig. 1). Each variant consisted of four replicates ([20]), corresponding to four seed germination kits per variant. Each germination kit contained 100 alder seeds sown on moist filter paper. The germination kits were placed in a climate chamber, and germination was monitored weekly for 3 weeks. Germination was considered successful when seedlings had developed a root, a shoot, and the first leaves.
(2) Seedling survival and growth under different site conditions
A greenhouse experiment was conducted following the study by Tiebel et al. ([37]), to determine whether the seeds of A. glutinosa continue to require high radiation, soil moisture, and open soil for successful germination under climate change, or whether the requirements for germination sites will change. We tested three types (variants) of ground cover, three radiation levels, and three soil moisture levels, with two replicates for each of the 27 treatment combinations established in spring 2023 (Fig. 1). It was hypothesized that shaded conditions maintain higher soil moisture; however, it remains unclear whether this is better achieved through ground cover or shading.
For the preparation of treatment variants, 54 pots were filled with a 1:1 substrate mixture of brown podsol soil from the Tharandter Forest, Germany, and the cultivation substrate “Einheitserde” (Typ ED 73) from Meyer shop, Germany. The ground cover variants included: open soil; a 4-5 cm-high layer of spruce litter; and a 3-5 cm-high layer of moss mixture comprising Pleurozium sp., Polytrichum sp., and Dicranum sp., collected from the Tharandter Forest, placed atop the substrate mixture. The shading treatments consisted of 11% and 27% shade, created with shade mats and controlled using the SKP 210 sensor, which measures photosynthetically active radiation (PAR). Due to the inherent reduction in radiation within greenhouses ([34], [37]), it was not possible to exceed 46% radiation, though alder can tolerate higher radiation levels ([5]). Therefore, the 46% variant was not shaded in the greenhouse and represents the open area radiation variant.
The three soil moisture variants were dry, moist, and wet conditions. The dry variant was maintained above the permanent wilting point at 8-12 vol.%, representing future conditions; the moist variant was at the lower field capacity limit at 15-24 vol.%; and the wet variant was at the upper field capacity limit at 27-35 vol.%, simulating riparian zone conditions.
Each of the 54 pots was sown with 100 untreated A. glutinosa seeds. All pots were randomly arranged within the greenhouse, weighed three times weekly, and irrigated as needed to compensate for weight loss and maintain the designated moisture levels ([37]). The greenhouse conditions ranged from an average daily minimum temperature of 5 °C to a maximum of 36 °C, with relative humidity between 31% and 86%. The seedlings were counted weekly for 16 weeks. At the end of the experiment in August 2023, all seedlings were harvested, thereby destroying the substrate structure within each pot. Measurements of shoot and root lengths were taken using a folding ruler; root collar diameter was measured with a calliper. Seedlings were then oven-dried for 2 days at 105 °C, and their above- and below-ground biomass were determined using a precision scale.
Delayed germination
Following the completion of the previous study (2) on seedling survival and growth under different site conditions, all pots in which no germination had occurred after 16 weeks and the substrate remained intact were subjected to 46% radiation in the cold greenhouse with an automatic irrigation system (Fig. 1). This marked the start of the third experiment. The aim was to determine whether alder seeds could withstand repeated cycles of swelling and desiccation and still germinate successfully weeks later. The study included 36 pots with moss and litter cover. The open soil pots were excluded, as germination was observed in every open soil pot. Only undisturbed pots containing 100 seeds each were included in this delayed-germination assessment. Germination was recorded once a week for the first three weeks. After four weeks, no new germinations were observed. During this period, the average daily minimum and maximum temperatures ranged from 6 to 21°C, with relative humidity between 63% and 85%.
Seedling survival under drought stress
Two drought stress experiments were conducted on April 24, 2023 (spring) and July 31, 2023 (summer-autumn) to investigate the duration of survival of A. glutinosa seedlings under drought conditions when the topsoil dries out. The methodology followed the procedures outlined by Tiebel et al. ([37]). For the spring experiment, four pots (each serving as a replicate) were filled with the described 1:1 substrate mixture of brown podzol soil and “Einheitserde”, and watered to 30-35 vol.%. Similarly, eight pots (eight replicates) were used for the summer-autumn experiment. Each pot was sown with 100 A. glutinosa seeds. Seed germination and seedling development were allowed to proceed for six weeks before watering was discontinued to induce drought stress (Fig. 1).
Temperature and humidity data were the same of the experiment (2) on survival and growth of seedlings under different site conditions. We counted the living seedlings and measured the soil moisture every week. The spring drought stress experiment was concluded once all seedlings had died. The summer-autumn experiment was terminated after 12 weeks of observation in December 2023, as no further seedling mortality occurred and the vegetation period had ended.
Statistical analyses
The Kruskal-Wallis H test was used to analyse the germination test results, as the data were not normally distributed ([40]).
The structure of the experiment (2) on seedling survival and growth under different site conditions corresponded to a full factorial design, with the response variables “surviving seedlings” and “delayed germination” being binomially distributed. Each surviving seedling and germinated seed was considered as a replicate in the analyses ([4], [36]), since seedling survival and seed germination were binary events (absence = 0, presence = 1). Generalized linear mixed models (GLMM) were used to assess the influence of the fixed effects - soil cover (categorical variable), soil moisture (categorical variable), and radiation (continuous variable) - on the response variables ([42]). Pots were included as a random effect, as each of the 100 seeds sown per pot was treated as one observation in the dataset. No overdispersion was observed in the models, as the data consisted of independent binary observations, with the only parameter also determining the variance. The analyses of growth parameters (response variable) - shoot length, root length, root collar diameter, and biomass - in experiment 2 were conducted using linear mixed-effects models (LMMs), with soil moisture and ground cover as categorical variables, radiation as a continuous variable, and pot number as a random effect. The growth data were log-transformed to meet assumptions of normality and homogeneity of variance prior to analysis.
The data on seedling survival under drought stress (4), which were also binomially distributed, were analysed using GLMMs. In this study, soil moisture was treated as a continuous variable, while season was modelled as a categorical fixed effect. The pot number was included as a random effect.
The effects of fixed factors were evaluated using backward selection based on Akaike Information Criterion (AIC) values to identify the optimal model fit ([37]). All modeling was performed using the “lme4” package (version 1.1-35.1) and the “glmmTMB” package (version 1.1.8) in R Studio v. 2022.12.0 ([2], [3], [28]).
Results and discussion
Germination tests
The germination of Alnus glutinosa seeds is closely linked to warm and light conditions. The highest average germination percentage (75%) was observed when the seeds were exposed to 25 °C with a 16-hour light cycle (Fig. S1 in Supplementary Material). These conditions promote good germination success, as supported by previous studies ([24], [25], [31], [9]). When seeds were kept at 25 °C in darkness, the average germination percentage dropped to about 12%, suggesting that light is an important factor for germination. McVean ([24]), Schalin ([31]), Gosling et al. ([13]), and Houston et al. ([18]) also came to this conclusion. However, in our experiment, light had no significant effect on germination (Kruskal test, p-value = 0.369). McVean ([25]) showed that seeds of Alnus glutinosa germinate in the dark. Depending on the seed batch, germination rates in the dark ranged from 25 to 62% ([25]). This indicates that the germination rate of alder seeds may be lower in the dark. Temperature, on the other hand, has proven to be important for germination (Kruskal test, p-value < 0.001). Temperatures of 5 °C did not activate the germination process despite the presence of light. McVean ([24]), Gosling et al. ([13]), and Aniszewska et al. ([1]) showed that germination of black alder seeds typically begins with rising temperatures in late February or early March, around 10-15 °C or 20 °C. Therefore, it can be hypothesized that the expected decrease in cool spring temperatures due to climate change is unlikely to negatively affect the success of Black alder germination.
Seedling survival and growth under different site conditions
During the entire study period, no Alnus glutinosa seeds germinated on moss and litter cover. The results revealed that black alder seeds require exposure to mineral soil to germinate ([24], [25], [31]). This finding was somewhat unexpected, as other species for which litter and moss are not considered safe sites under climate change scenarios, such as Salix caprea and Populus tremula, demonstrated limited germination on these substrates ([37]). A possible explanation is the presence of an oily seed coat in A. glutinosa, which may hinder seed swelling and germination ([24], [25]). On moss and litter, there is no direct contact between seeds and soil ([27], [29]), and the organic layer has a high water-absorbing capacity ([31], [27]). Also, alder seeds and seedlings require approximately one month of high soil moisture and humidity for successful establishment; main humidity levels above 50% are crucial during the first month of germination ([24], [25], [31], [6], [18]).
On open soil, the highest germination rate was observed after four to five weeks, ranging from 12% (23 seeds) to 72% (143 seeds), depending on soil moisture and radiation levels. The oily testa may have contributed to the prolonged germination period, as also reported by previous research ([31], [21]). Gomes Marques et al. ([12]) emphasize that climate change, characterized by increasing temperatures and drought, could lead to extended germination and establishment periods in black alder, a trend consistent with our findings.
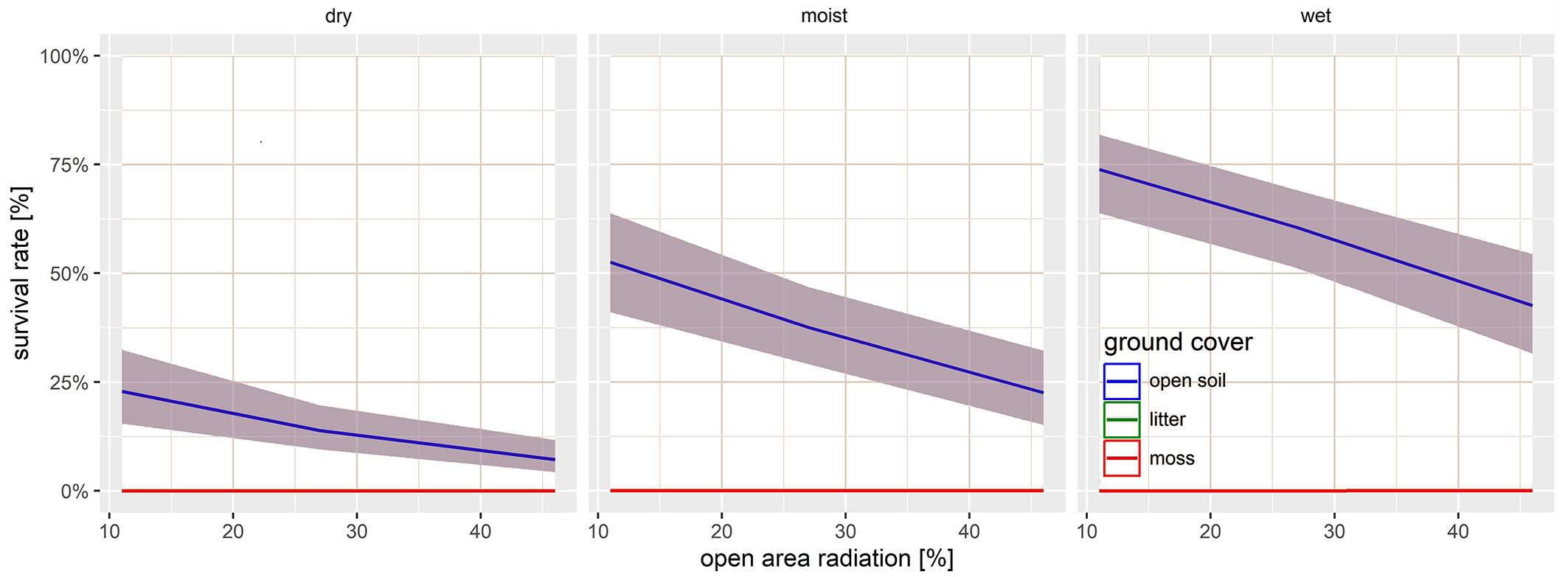
By the end of the study, survival rates ranged from nine to 136 seedlings (Fig. 2). Statistical analysis revealed a significant negative effect of increasing radiation and decreasing soil moisture on seedling survival (Tab. 1). No significant interactions between these parameters were detected. The seedling survival of black alder was significantly constrained on dry, open soil, corroborating the findings of Eschenbach & Kappen ([10]), who reported that alder seeds are unable to germinate and establish in dry sites. The challenges posed by drought and the associated stress responses in seedlings are exacerbated by higher levels of radiation. Our results demonstrated significantly lower survival rates with increasing radiation. This vulnerability to drought stress suggests that successful seedling establishment of Alnus glutinosa may become more difficult under climate change scenarios involving fluctuating water levels, altered watercourses, and reduced precipitation ([13], [22], [12]). It is therefore highly beneficial that alder seedlings can establish in shaded conditions, where moisture retention is often longer. However, it should be noted that this does not imply that the seedlings and mature trees can persist or grow permanently in shaded environments (see experiment 2 - [24], [31]). Black alder is a shade-intolerant tree throughout its entire life span and, like other pioneer tree species, requires light to sunny sites with ± 20% of open area radiation for establishment ([24], [14], [6], [18]).
Fig. 2 - GLMM predictions of the survival probability rate of Alnus glutinosa seedlings on open soil, depending on radiation and volumetric soil water content
Tab. 1 - Results of the survival rate model (GLMM, left) and delayed germination model (GLMM, right) as a function of radiation, volumetric soil water content, and ground cover. The parameter estimation for the final models with the lowest AIC value are shown. The intercept represents shady, wet moss or shady, wet open soil. (SE): standard error; (SD): standard deviation.
| Fixed Effects |
Survival rate | Fixed Effects |
Delayed germination | ||||
|---|---|---|---|---|---|---|---|
| Estimate | SE | p-value | Estimate | SE | p-value | ||
| (Intercept) | 1.456 | 0.302 | <0.001 | (Intercept) | 0.597 | 0.205 | 0.004 |
| moss | -0.274 | 0.000 | 0.999 | litter | 0.447 | 0.147 | 0.002 |
| litter | -0.274 | 0.000 | 0.999 | moist | -0.004 | 0.180 | 0.979 |
| moist | -0.935 | 0.275 | 0.001 | dry | -0.219 | 0.179 | 0.222 |
| dry | -2.249 | 0.289 | <0.001 | radiation | -0.030 | 0.005 | 0.000 |
| radiation | -0.038 | 0.008 | <0.001 | - | - | - | - |
| Random Effects |
Name | Variance | SD | Random Effects |
Name | Variance | SD |
| pot number | Intercept | 0.179 | 0.423 | pot number | Intercept | 0.150 | 0.387 |
The effect of drought stress, in combination with other environmental conditions such as dryness, shading, and soil cover, can be inferred not only through germination success but also from growth parameters, including shoot and root length, root collar diameter, and biomass. The analysis of these growth parameters and their differences provides insights into how trees allocate their growth energy and enables evaluation of environmental factors that affect juvenile growth rates.
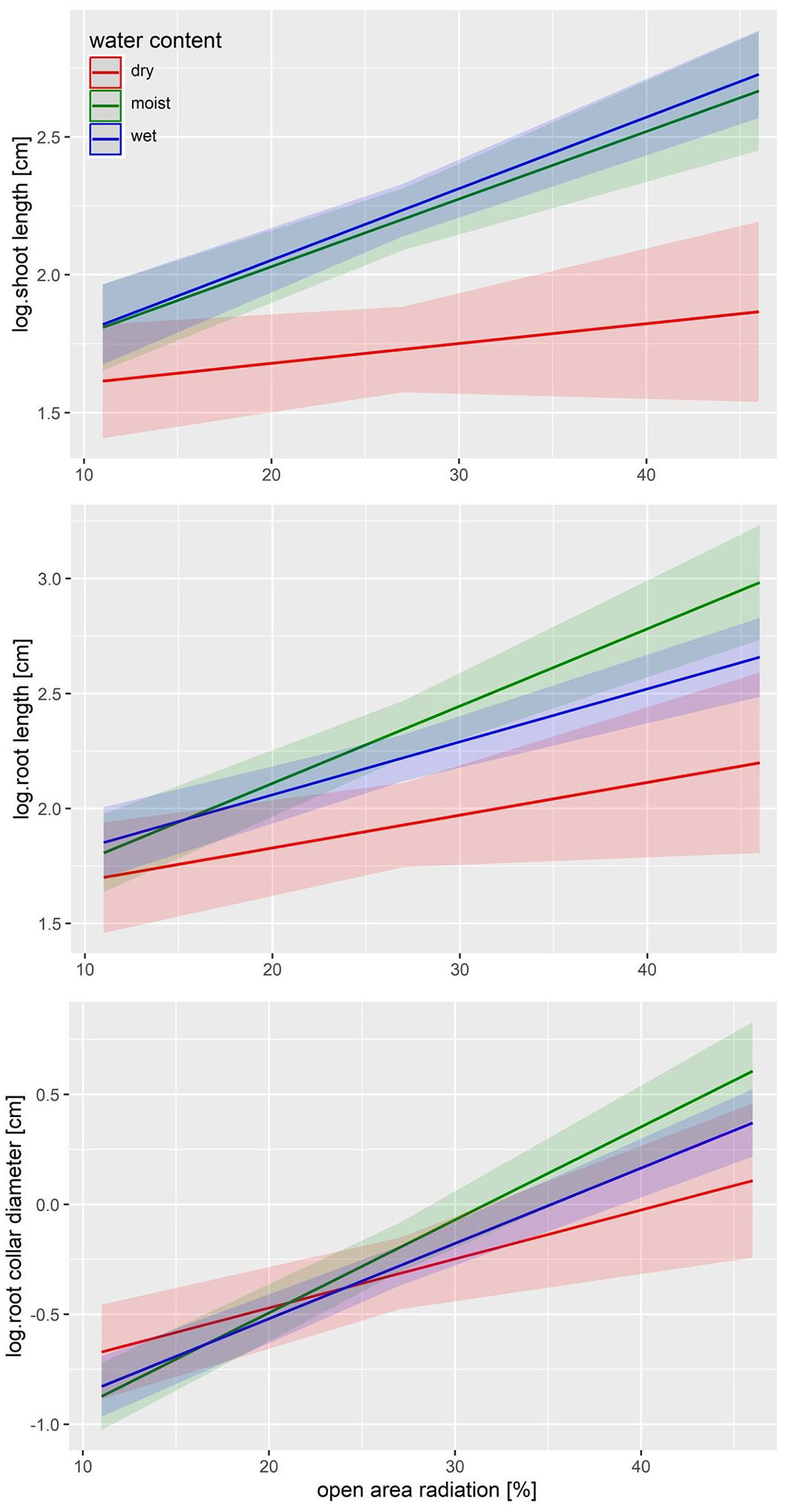
When the black alder seedlings were established on open soil, a significant, positive effect of radiation on shoot growth, root growth, root collar diameter, and biomass was found (Tab. 2, Tab. 3). The mean shoot length and root length of the germinated seedlings on open soil varied from 4.8 to 17.8 cm and 5.7 to 19.6 cm, respectively. The seedlings exhibited an average root collar diameter of 0.5-2.3 mm. The average above-ground biomass ranged from 22 to 548 mg, while the average below-ground biomass varied between 4 and 152 mg. In the first few years, shoots can grow up to approximately 1.5 m per year ([6]). In the present study, an average shoot length of 17.8 cm was achieved after 16 weeks of growth under sufficient moisture and light, which is consistent with the growth rates reported by Lovely ([22]). Radiation levels of 20% lead to stagnating growth rates ([6]), which explains the significantly reduced shoot lengths of 7.1 cm observed in our study at 11% radiation, despite adequate moisture ([22]). The reduced shoot length confirms that alder, as a pioneer tree species, requires sufficient light for optimal growth ([24], [14], [6], [18]). Furthermore, the seedlings showed the lowest biomass yields on dry open soil with 11% radiation, while the highest biomass was recorded on moist, open soil with 46% radiation.
Tab. 2 - LME results for the logarithm-transformed dependent variables shoot length, root length, and root collar diameter of Alnus glutinosa seedlings growing on open soil. The parameter estimates for the final model with the lowest AIC are shown. The intercept represents shady, wet soil. (SE): standard error; (SD): standard deviation.
| Fixed Effects |
Shoot length (cm) | Root length (cm) | Root collar diameter (mm) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Estimate | SE | p-value | Estimate | SE | p-value | Estimate | SE | p-value | |
| (Intercept) | 1.535 | 0.106 | <0.001 | 1.599 | 0.112 | <0.001 | -1.204 | 0.101 | <0.001 |
| moist | 0.004 | 0.159 | 0.979 | -0.162 | 0.172 | 0.368 | -0.134 | 0.154 | 0.409 |
| dry | 0.000 | 0.194 | 0.999 | -0.056 | 0.222 | 0.804 | 0.289 | 0.199 | 0.160 |
| radiation | 0.026 | 0.003 | 0.002 | 0.023 | 0.003 | <0.001 | 0.034 | 0.003 | <0.001 |
| radiation:moist | -0.001 | 0.006 | 0.808 | 0.011 | 0.006 | 0.105 | 0.008 | 0.006 | 0.169 |
| radiation:dry | -0.019 | 0.007 | 0.022 | -0.009 | 0.008 | 0.307 | -0.012 | 0.008 | 0.123 |
| Random Effects |
Name | Variance | SD | Name | Variance | SD | Name | Variance | SD |
| pot number | Intercept | 0.008 | 0.089 | Intercept | 0.006 | 0.079 | Intercept | 0.005 | 0.072 |
| residual | - | 0.351 | 0.592 | - | 0.563 | 0.750 | - | 0.447 | 0.669 |
Tab. 3 - LME results for the logarithm-transformed dependent variables above-ground biomass and below-ground biomass of Alnus glutinosa seedlings growing on open soil. The parameter estimates for the final model with the lowest AIC are shown. The intercept represents shady, wet soil. (SE): standard error; (SD): standard deviation.
| Fixed Effects |
Above-ground biomass (g) | Below-ground biomass (g) | ||||
|---|---|---|---|---|---|---|
| Estimate | SE | p-value | Estimate | SE | p-value | |
| (Intercept) | -4.754 | 0.149 | <0.001 | -7.801 | 0.387 | <0.001 |
| moist | 0.093 | 0.132 | 0.497 | 0.881 | 0.345 | 0.027 |
| dry | -0.183 | 0.167 | 0.289 | 0.354 | 0.399 | 0.387 |
| radiation | 0.067 | 0.005 | <0.001 | 0.100 | 0.011 | <0.001 |
| Random Effects |
Name | Variance | SD | Name | Variance | SD |
| pot number | Intercept | 0.014 | 0.118 | Intercept | 0.224 | 0.473 |
| residual | - | 1.627 | 1.276 | - | 5.298 | 2.302 |
Surprisingly, drought did not significantly impact any growth parameters; however, a clear trend of reduced shoot growth, root growth, root collar diameter, and biomass under drought conditions was observed (Fig. 3). The seedlings of Alnus glutinosa showed significantly higher below-ground biomass in moist soil conditions (Tab. 3), while shoot growth was reduced in dry, open soil conditions as radiation increased (Tab. 2). The higher below-ground biomass indicates that more fine roots were formed with increasing moisture, as root lengths and root collar diameters did not differ significantly. The reduction in shoot growth during drought and under high-light conditions occurs even though the above-ground biomass remains relatively unchanged. This can be explained by noting that black alder does not adapt well to shade, as it does not develop shade leaves ([10], [6]). As a result, leaf biomass remained stable, and shoot length was reduced under drought stress, with no significant effect on overall above-ground biomass. Lovely ([22]) demonstrated that drought can influence sprout growth and root length but does not significantly affect root collar diameter or above- and below-ground biomass. According to Seiler & Johnson ([35]), Eschenbach & Kappen ([10]), Copolovici et al. ([7]), and Lovely ([22]), prolonged drought stress reduces transpiration, photosynthetic, and growth rates in black alder seedlings. In our experiment, the increased radiation appears to largely mitigate the effects of drought, indicating that Alnus glutinosa is less sensitive to drought stress than expected for a riparian tree species. Nonetheless, slow growth caused by limited light or drought can prolong the juvenile phase, thereby increasing the risk of pathogen infection, herbivory, and browsing, as well as mortality from permanent flooding or further drought stress ([22], [12], [26]).
Fig. 3 - LME predictions of the shoot length (cm), root length (cm), and root collar diameter (mm) of Alnus glutinosa seedlings growing on open soil, depending on radiation and volumetric soil water content.
Delayed germination
After 16 weeks without germination in moss and litter pots (experiment 2), germination began just a week after automatic, continuous watering started and was complete in most pots within three weeks (experiment 3). Maintaining consistent moisture levels facilitated the successful germination of alder seeds on moss and litter, aligning with the results of Claessens et al. ([6]). Germination rates ranged from 19 to 64% on moss and from 18 to 78% on litter. In general, the germination percentage of Alnus glutinosa can vary widely, typically between 15 and 80% ([31], [15], [9], [13], [21], [12]).
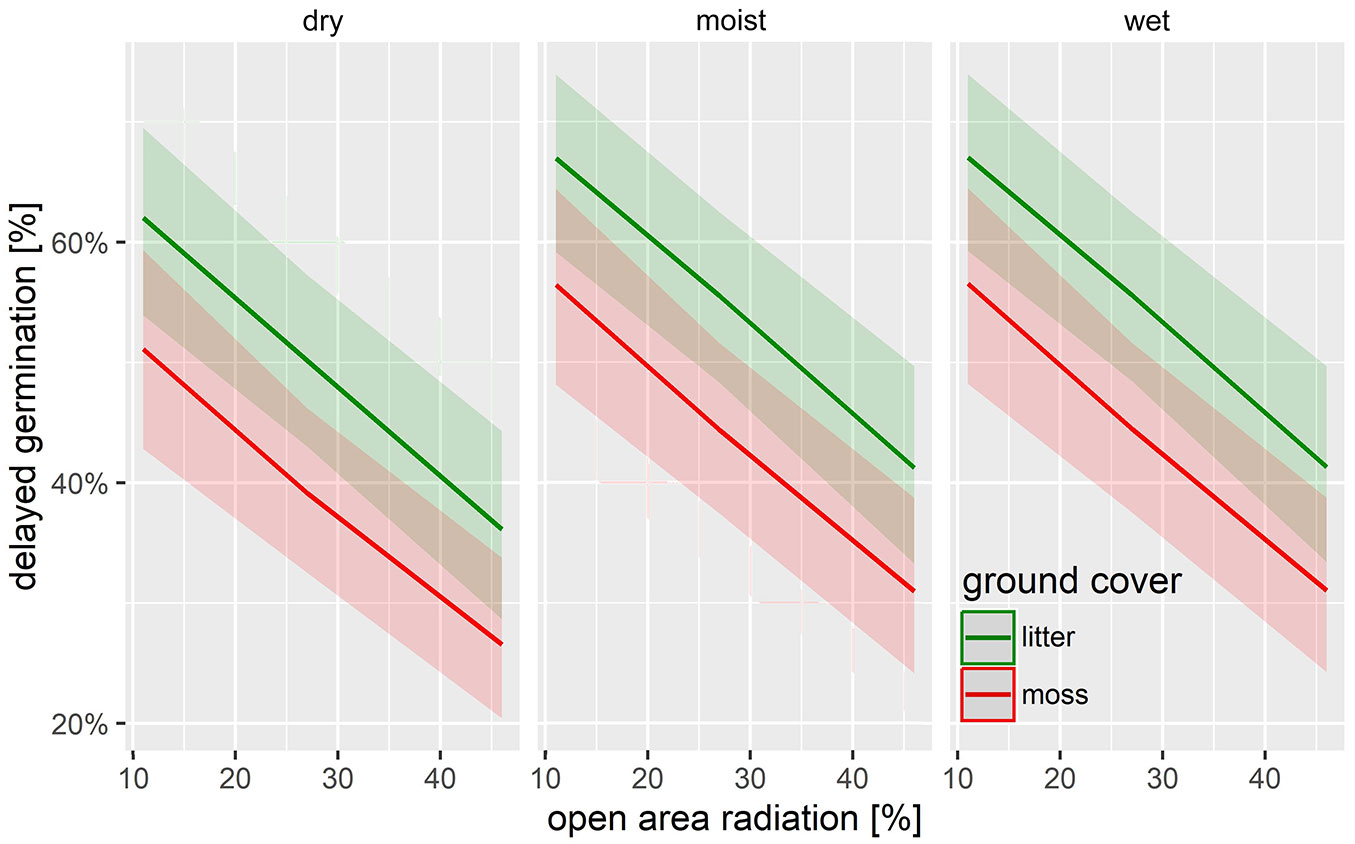
The statistical analysis revealed that the variations in soil moisture conditions (from wet to dry) over the 16 weeks prior to germination, did not significantly influence the success of delayed germination (Tab. 1, Fig. 4). This could be due to the accelerated desiccation of seeds on moss and litter substrates, which determines the lack of contact with soil ([27], [29]), reducing the water absorption by the organic layer ([31], [27]) and ultimately hampering the germination process on these substrates. The previously applied varying radiation levels negatively affected seed germination capacity after the storage period. Only 18-52% of seeds from the 46% radiation variant germinated after 16 weeks. Seeds exposed to continuous shading were protected from damaging UV radiation, whereas seeds exposed to full sunlight experienced persistent UV exposure ([16], [8]). It appears unlikely that the reduction in germination capacity was due to excessive seed desiccation caused by radiation, as low seed moisture content is generally associated with improved storability ([32], [1], [38]). Therefore, we conclude that alder seeds can survive extended periods without germinating, even when subjected to repeated cycles of watering, swelling, drying, and irradiation.
Fig. 4 - GLMM predictions of the delayed germination of Alnus glutinosa seeds depending on radiation, volumetric soil water content, and ground cover.
Seedling survival under drought stress
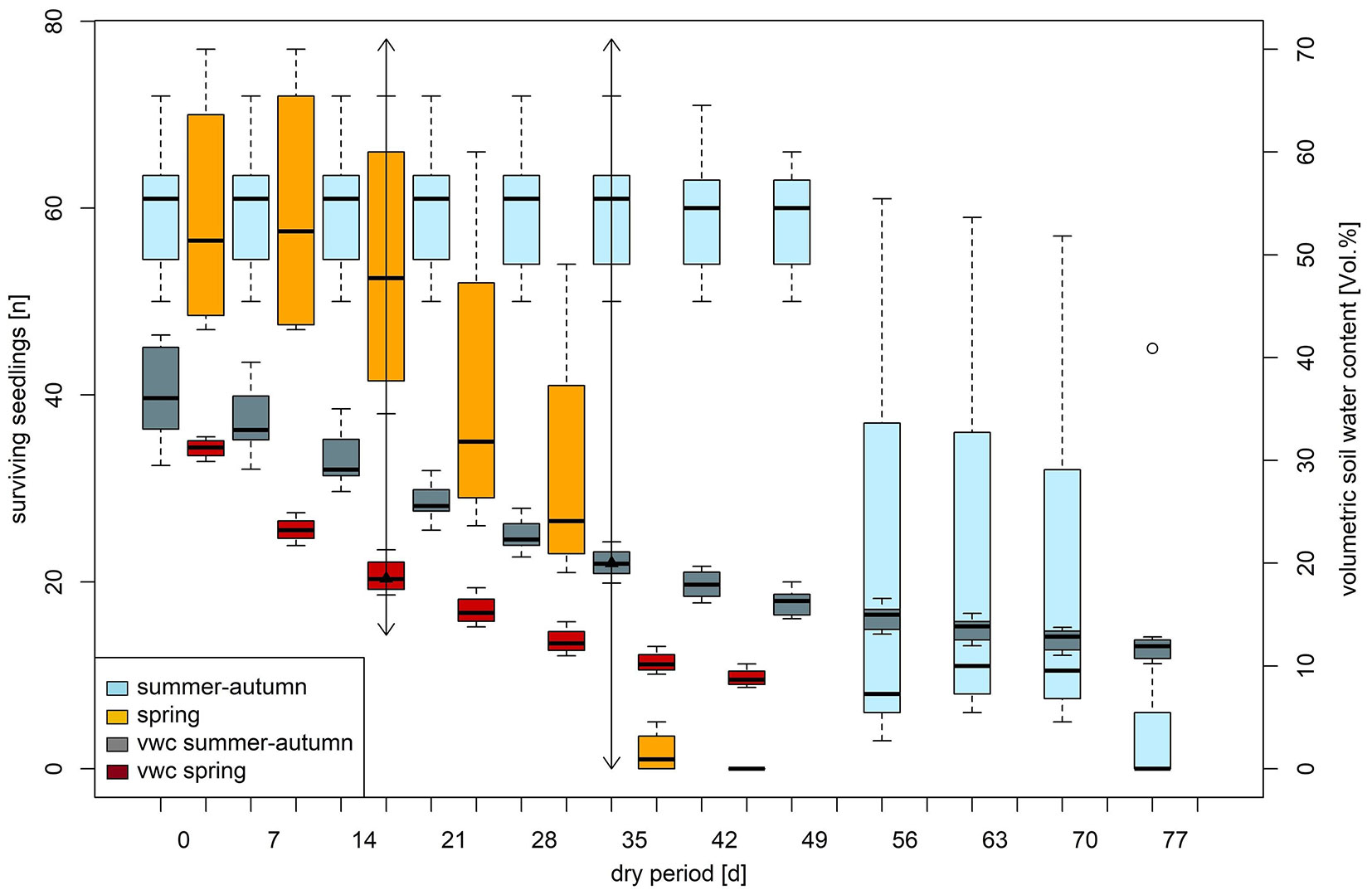
Following a three-week period of drought stress in spring and summer-autumn, during which soil moisture levels fell below the permanent wilting point, a decline in seedlings was observed (Fig. 5). A prolonged drought lasting four to six weeks led to the death of nearly all seedlings. More frequent and severe droughts are expected as a consequence of climate change, and this can often dry out the topsoil in typically moist areas like riverbanks, thus hampering the establishment of black alder seedlings. In contrast, mature trees of A. glutinosa with extensive and deep root systems can generally tolerate drought periods of four to six weeks without significant adverse effects. Old trees are primarily affected when deeper soil layers desiccate, typically after droughts lasting 2-3 months, accompanied by a decline in the groundwater table ([10], [30], [41]). Although Alnus glutinosa can close its stomata during drought conditions ([7]), its stomatal regulation is less efficient compared to species like Fagus sylvatica ([17], [41]). Water consumption remains largely unchanged during drought periods ([10], [6]), making black alder seedlings sensitive to drought stress ([24], [25], [7], [1]). However, alder seedlings are no more susceptible to drought than seedlings of other deciduous pioneer tree species like Betula pendula, Populus tremula, and Salix caprea, which also hardly survive drought periods longer than three weeks ([37]).
Fig. 5 - Effect of decreasing cumulated volumetric soil water content (vwc) on the seedling survival (n) of Alnus glutionsa over time (dry period) in spring and summer-autumn. The arrow indicates the moment when the measured soil moisture has fallen below the permanent wilting point.
The comparison of the impacts of drought stress across seasons revealed that spring drought had a significantly greater effect on seedling mortality than summer-autumn drought. At the onset of the drought stress experiments in both seasons, there were approximately 60 seedlings. The analysis revealed a significantly lower survival rate over time during the spring study. After stopping the irrigation in spring, the soil moisture reached the permanent wilting point after two weeks, and four weeks later, all Alnus glutinosa seedlings had died (Fig. 5). In contrast, during the summer-autumn study, the permanent wilting point was reached after five weeks without irrigation, with the first seedlings dying after an additional three weeks. After six weeks of severe drought, an average of only 7% of the seedlings remained alive in December 2023. For both drought stress studies, a positive influence of soil moisture on the survival rate could be proven (Tab. 4). This positive effect was even more pronounced in the spring study (Tab. 4). In addition, the seedlings were also exposed to intense radiation and high temperatures, which further increased environmental stress on the plants ([7], [30]). In autumn, these factors are significantly less intense and do not contribute additional stress to the seedlings. We hypothesize that the observed seedling mortality from autumn until December was primarily due to the continued photosynthetic activity of the alder leaves.
Tab. 4 - Results of the seedling survival (GLMM) during drought periods in spring and summer-autumn for Alnus glutionsa as a function of soil water content and period. The parameter estimates for the final model with the lowest AIC are shown. The Intercept represents the drought period in the summer-autumn. (SE): standard error; (SD): standard deviation.
| Fixed Effects | Estimate | SE | p-value |
|---|---|---|---|
| (Intercept) | -1.834 | 0.147 | <0.001 |
| soil moisture | 0.077 | 0.003 | <0.001 |
| spring | -1.216 | 0.270 | <0.001 |
| soil moisture:spring | 0.058 | 0.007 | <0.001 |
| Random Effects | Name | Variance | SD |
| pot number | Intercept | 0.094 | 0.307 |
Conclusion
Black alder (Alnus glutinosa) is an important tree species in riparian ecosystems, which are affected by heat waves and drought periods driven by climate change. In view of climate change, the most significant challenge for the species is the drying of the topsoil, which negatively affects the germination and establishment of alder trees. Nevertheless, this study also identified strengths of alder that give hope in the context of climate change.
The germination success of Alnus glutinosa does not appear to be hindered by the absence of cool spring temperatures. However, the more frequent and intense spring and summer droughts is not expected to limit successful germination to years with prolonged periods of rainfall. It has been demonstrated that alder seeds can survive unfavourable germination conditions for over 16 weeks and still germinate successfully afterward. Alder is typically regarded as a species characteristic of riparian ecosystems ([41]). Nonetheless, it has moisture requirements during germination similar to those of other pioneer tree species such as Betula pendula, Salix caprea, and Populus tremula, which colonise disturbed areas and are generally considered more drought-tolerant than alder ([37]). In dry locations near the permanent wilting point, alder seedlings can successfully establish and grow in open soil. Shading can significantly enhance germination success on dry soils. If Alnus glutinosa could reduce its water consumption under drought stress, a potential for reforestation of disturbed terrestrial forest areas would be possible ([41]). However, covering the soil with litter and moss presents challenges. Alder requires mineral soil for successful germination. Seeds can only germinate on litter and moss when sufficient moisture is persistently available, which is unlikely in riparian ecosystems due to climate change. Droughts hamper the germination and development of Alnus glutinosa seedlings by delaying and prolonging these processes. However, drought does not entirely prevent germination and the establishment of alder seedlings in dry locations.
Acknowledgments
KT conceived the study, conducted the greenhouse measurements, performed the statistical analyses, and wrote the manuscript; AK conducted the greenhouse measurements and helped draft the manuscript.
The study is part of the research project “Study on the natural regeneration potential of pioneer tree species on windthrown sites under changing environmental conditions due to climate change (WISTUPIO)” and was funded by the German Federal Ministry of Food and Agriculture (BMEL) through the Agency of Renewable Resources (FNR) - Forest Climate Fund (WKF) (grant no. 2218WK45X4). We would like to thank Dániel Oroszlán, Angelika Mann, and Jörg Wollmerstädt for their help during the experiments and Robert Schlicht for his statistical advice.
References
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Supplementary Material
Authors’ Info
Authors’ Affiliation
Antje Karge
Chair of Silviculture, Institute of Silviculture and Forest Protection, TUD Dresden University of Technology, Pienner Str. 8, 01737 Tharandt (Germany)
Corresponding author
Paper Info
Citation
Tiebel K, Karge A (2026). Can we expect more from Alnus glutinosa (L.) Gaertn. in a changing climate?. iForest 19: 69-76. - doi: 10.3832/ifor4842-019
Academic Editor
Michele Carbognani
Paper history
Received: Feb 27, 2025
Accepted: Jan 09, 2026
First online: Mar 08, 2026
Publication Date: Apr 30, 2026
Publication Time: 1.93 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2026
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 1732
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 321
Abstract Page Views: 940
PDF Downloads: 405
Citation/Reference Downloads: 0
XML Downloads: 66
Web Metrics
Days since publication: 92
Overall contacts: 1732
Avg. contacts per week: 131.78
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
(No citations were found up to date. Please come back later)
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Effects of brassinosteroid application on seed germination of Norway spruce, Scots pine, Douglas fir and English oak
vol. 10, pp. 121-127 (online: 02 October 2016)
Research Articles
Influence of mother plant and scarification agents on seed germination rate and vigor in Retama sphaerocarpa L. (Boissier)
vol. 7, pp. 306-312 (online: 08 April 2014)
Research Articles
Conservation of Betula oycoviensis, an endangered rare taxon, using vegetative propagation methods
vol. 13, pp. 107-113 (online: 23 March 2020)
Research Articles
Germination and seedling growth of holm oak (Quercus ilex L.): effects of provenance, temperature, and radicle pruning
vol. 7, pp. 103-109 (online: 18 December 2013)
Technical Reports
Effects of different mechanical treatments on Quercus variabilis, Q. wutaishanica and Q. robur acorn germination
vol. 8, pp. 728-734 (online: 05 May 2015)
Research Articles
The effectiveness of short-term microwave irradiation on the process of seed extraction from Scots pine cones (Pinus sylvestris L.)
vol. 13, pp. 73-79 (online: 13 February 2020)
Research Articles
Effects of different nut pretreatments and substrates on germination and seedlings growth of Neocarya macrophylla Sabine in Basse Casamance, Senegal
vol. 17, pp. 346-352 (online: 03 November 2024)
Research Articles
Optimum light transmittance for seed germination and early seedling recruitment of Pinus koraiensis: implications for natural regeneration
vol. 8, pp. 853-859 (online: 22 May 2015)
Research Articles
Size and age: intrinsic confounding factors affecting the responses to a water deficit in black spruce seedlings
vol. 8, pp. 401-409 (online: 09 December 2014)
Technical Reports
Nursery practices increase seedling performance on nutrient-poor soils in Swietenia humilis
vol. 8, pp. 552-557 (online: 09 December 2014)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword