Recovery of water potential and leaf gas exchange performance following drought stress in Quercus cerris populations
iForest - Biogeosciences and Forestry, Volume 19, Issue 2, Pages 94-101 (2026)
doi: https://doi.org/10.3832/ifor4743-018
Published: Mar 17, 2026 - Copyright © 2026 SISEF
Research Articles
Abstract
Drought and global warming are major environmental stressors that significantly affect forest ecosystems and the survival of tree species. This study investigates the physiological and biochemical responses of Turkey oak (Quercus cerris L.) seedlings from five populations across three bioclimatic zones in Turkey - semi-arid (Yozgat), semi-humid (Isparta, Balikesir), and humid (Çanakkale and Sinop) - to drought stress and subsequent re-watering. Seedlings were subjected to control (regular irrigation) and drought stress (no irrigation for 30 days), with ecophysiological (soil water content, midday water potential, gas exchange) and biochemical (total soluble sugars and chlorophyll content) traits measured on days 10, 20, 30, and after re-watering (days R3 and R10). Drought stress reduced midday water potential and gas exchange parameters across all populations. Total soluble sugar content increased under drought, except in the Sinop population, which showed a decline in photosynthetic rate and failed to accumulate soluble sugars. In contrast, the Yozgat population maintained higher midday water potential, suggesting effective osmotic regulation. Other populations (Isparta, Balikesir, and Çanakkale) coped with drought by increasing total soluble sugar content despite lower water potential. Following re-watering, water potential and sugar content recovered in all populations, with a gradual improvement in photosynthetic performance. These findings highlight significant intraspecific variation in drought response among Q. cerris populations and underscore the importance of considering population-level differences in forest management and afforestation strategies under changing climate conditions.
Keywords
Drought, Gas Exchange, Recovery, Soluble Sugar, Quercus Populations
Introduction
Climate is one of the most critical factors shaping life on Earth and determining the distribution of organisms across ecosystems ([7]). Global climate variability has a major impact on forests ([28], [33]). Among these factors, global warming is a key driver of environmental change, directly impacting plant species’ survival, growth, and overall performance. As climate change intensifies, a better understanding of the functional traits of tree species, particularly those related to drought tolerance, is crucial for predicting ecosystem responses ([10]).
Woody species show considerable variation in their ability to cope with drought, particularly in regions facing prolonged aridity ([2]). Extensive research has been conducted on plant responses to drought stress, which occur at morphological, physiological, and molecular levels across various forest species ([53], [11], [24]). Trees adapt to water stress through changes in growth, gas exchange, and water relations, with stomatal closure being a key indicator of plant water stress ([22], [40]). Drought leads to reduced growth and photosynthesis ([16], [14]), lower stomatal conductance ([11]), reduced hydraulic conductance ([39]), and altered nonstructural carbohydrate dynamics ([31]).
A comprehensive understanding of geographic patterns and genetic variation in functional and structural traits of key tree species is critical for informing adaptive forest management strategies to mitigate the projected impacts of climate change on plant growth and drought tolerance ([9]). In this context, provenance trials are valuable tools for assessing individual trees’ capacity to adapt to varying environmental conditions ([46]).
The genus Quercus, comprising nearly 600 species worldwide, includes both trees and shrubs that occupy a wide range of habitats, from temperate to subtropical forests and from humid to extremely dry environments ([32]) and form forests of high cultural, ecological, and commercial value ([43]). In Turkey, oak species cover a significant area, with approximately 6.92 million hectares of forest, representing a substantial part of the country’s forest ecosystem ([20]). Genetic variation plays a key role in the observed differences in drought tolerance within and among Quercus species ([12], [4], [17], [5]). Among these species, Quercus cerris, commonly known as the Turkey oak, has shown particular promise in the context of climate change, especially in Central and Western Europe, due to its notable drought tolerance. This species offers substantial potential for adaptive forestry practices owing to its distinct evolutionary history, drought-resistant strategies, and high phenotypic and genetic variability ([27]).
In Mediterranean regions, where oaks experience prolonged summer droughts, evergreen and deciduous species exhibit distinct drought-response strategies. Evergreen oaks, such as Quercus ilex L., often recover more quickly from dry periods by resuming growth after drought stress ([8]). Manes et al. ([30]) found that during drought, Q. ilex maintains root growth and keeps its stomata open, while the deciduous Q. cerris exhibits higher photosynthetic capacity under water-abundant conditions. Moreover, even among deciduous oaks, drought tolerance varies; for instance, Q. cerris is more drought-tolerant than Q. pubescens ([50]). The ability of species such as Q. cerris to maintain water uptake as soil moisture declines is vital to their success in increasingly arid climates ([26]).
A thorough understanding of how tree populations respond to drought is particularly important, as it is essential for predicting species adaptability to climate change and for guiding afforestation efforts in vulnerable Mediterranean ecosystems. Although interest in Q. cerris and other Mediterranean oaks is growing, studies comparing early drought responses across populations from different bioclimatic zones remain limited. Despite Quercus cerris is recognized for its drought tolerance, population-level variation in physiological and biochemical responses to water deficit remains insufficiently characterized.
By examining multiple populations from contrasting bioclimatic zones, we explore intraspecific variation in drought tolerance, a topic for which limited data exist for this species. This study investigates drought-induced changes in midday water potential, gas exchange, and osmotic regulation in three-month-old seedlings from five Q. cerris populations originating from contrasting bioclimatic zones in Turkey. We hypothesize that seedlings from semi-arid regions exhibit greater drought tolerance than those from more humid climates, as reflected in their ecophysiological and biochemical traits. By integrating climate-origin information with drought-response metrics across multiple populations, this study provides a population-level framework for identifying adaptive traits relevant to future seed transfer, provenance selection, and breeding programs under increased drought frequency. Identifying populations better suited to cope with future droughts will enhance the sustainability and productivity of Mediterranean forest ecosystems under climate change.
Material and methods
Plant material and climate data
Seed material was obtained from five populations of Turkey oak trees (Yozgat, Balikesir, Isparta, Çanakkale, Sinop - see Fig. S1 in Supplementary material) representing three different bioclimate zones (semi-arid, semi-humid, and humid) based on Emberger’s bioclimate categories ([1]). In October 2021, seeds were collected from at least 10 trees per population and stored in polyethylene bags at +4 °C. At the beginning of March 2022, the seeds were planted in polyethylene tubes (11.0 cm in diameter × 22.0 cm in height) with a humus-perlite mixture (3:1, v/v). Climate data were gathered from the WorldClim database, which provides worldwide climate data as spatial data layers ([15]). Based on the Emberger’s bioclimatic classification, the five populations belong to three bioclimate classes: semi-arid, low-humid, and humid (Tab. 1).
Tab. 1 - Coordinate and climate data and bioclimatic types of Q. cerris populations. (S): summer drought index value; (Q): precipitation-temperature coefficient; (Med.): Mediterranean.
| Population | Latitude N |
Longitude E |
Altitude (m) |
S | Q | Bioclimatic type | |
|---|---|---|---|---|---|---|---|
| S | Q | ||||||
| Yozgat | 39° 59′ 05″ | 35° 13′ 42″ | 1304 | 2.5 | 53.4 | Med. | Semi-arid |
| Balikesir | 39° 27′ 31″ | 28° 29′ 52″ | 1010 | 2.7 | 94.3 | Med. | Semi-humid |
| Isparta | 37° 39′ 31″ | 31° 12′ 14″ | 1400 | 2.6 | 69.3 | Med. | Semi-humid |
| Çanakkale | 39° 42′ 33″ | 26° 44′ 52″ | 988 | 2.5 | 104.1 | Med. | Humid |
| Sinop | 41° 52′ 34″ | 34° 54′ 15″ | 305 | 4.8 | 103.5 | Med. | Humid |
Experimental design and stress application
All seeds had germinated by the start of the experiment, and seedlings with uniform height and diameter were used. Seedlings were regularly irrigated in the greenhouse, and routine maintenance (e.g., cleaning and weeding) was performed. Three-month-old oak seedlings were then subjected to two water treatments (control and drought), with three replicates each, according to Emberger’s bioclimate classification. We used a randomized trial design, with 2 water treatments [irrigation (control) and no irrigation (drought stress)] × 5 populations × 3 replicates × 30 seedlings per replicate, totaling 900 seedlings. As for the first treatment (control), seedlings were kept under well-watered conditions throughout the experimental period, i.e., they were watered to field capacity every 2 or 3 days. In the second treatment, seedlings were subjected to water-deficit stress by withholding water for 30 days. On the 10th, 20th, and 30th days, measurements were taken in the greenhouse. The seedlings were irrigated to field capacity (recovery) on the 30th day, and physiological and biochemical properties were measured on the 3rd and 10th days after rewatering (see also Fig. S2 in Supplementary material). To facilitate the presentation of the results, measurements taken 3 days after rewatering are shown as “R3”, and those taken on the 10th day after rewatering are shown as “R10”. Accordingly, results are displayed in diagrams using the labels 10, 20, 30, R3, and R10 (Fig. S2).
The temperature and relative humidity in the greenhouse were measured using a datalogger since the beginning of the experiment. During the experiment, the average air temperature ranged from 15.8 to 28.9 °C, and relative humidity from 23.3% to 65.3%. On the measurement days (10, 20, 30, R3 and R10), the average air temperature ranged from 21.4 to 25.5 °C, while the average air humidity ranged from 35.7% to 58.65%.
Ecophysiological traits
Physiological traits (midday water potential, net photosynthetic rate, stomatal conductance, and transpiration rate) and biochemical traits (total soluble sugar and chlorophyll content) of seedlings from the five populations of Q. cerris were measured in a greenhouse during July and August 2022, and subjected to drought stress followed by rewatering.
Midday water potential (Ψmd) and soil water content (SWC)
A Scholander pressure chamber (Model 600, PMS Instruments, Corvallis, OR, USA) was used to measure the leaf water potential (Ψmd, Mpa - [42]). Shoot samples were randomly collected from the six seedlings in each group (control and drought stress) and measured between 12:00 and 13:00 pm. At the same time, soil volumetric content was measured with 5 replications on the same seedlings used for physiological measurements by the probe TDR 300 (Fieldscout, Portland, OR, USA) in the top 12.5 cm of soil.
Gas exchange measurements
Gas exchange parameters were determined on mature leaves in the greenhouse using a portable photosynthetic equipment model LI-6400XT (LiCOR, Lincoln, NE, USA), in three replicates. Between 08:30 and 11:30 and 13:30 and 15:00, all measurements were carried out using a 6 cm2 chamber and a linked light source (6400-02B-red/blue/light). The manufacturer’s recommended calibration procedure was followed. A reference CO2 level of 400 ppm, an air flow rate of 500 μmol s-1, and photosynthetic active radiation (PPFD) of 1500 μmol m-2 s-1 were maintained. During the measurement, the leaves’ temperature was adjusted to match the ambient temperature. The net photosynthetic rate (μmol CO2 m-2 s-1), stomatal conductance (mmol H2O m-2 s-1), transpiration rate (mmol H2O m-2 s-1), and intercellular CO2 concentration (μmol mol-1) were determined.
Total soluble sugar (TSC)
For total soluble sugar analysis, leaf samples were collected from seedlings on the 10th, 20th, and 30th day of treatment, as well as on the 3rd and 10th days after rewatering. Leaf samples were taken from the same seedlings used for physiological measurements. An average of 6 seedlings (all leaves) for each sampling day in each treatment were analyzed. After cleaning in distilled water, leaves were dried at 65 °C for 48 hours and then ground using a coffee grinder. For each sample, 100 mg of ground sample were incubated, and total soluble sugar content (mg g-1 DW) was determined according to Dubois et al. ([13]).
Leaf chlorophyll content (Chl)
Chlorophyll concentration was determined using four replicates of leaves from each treatment using the portable device SPAD-502 Plus (Konica Minolta, Inc., Osaka, Japan). It was measured three times by cleaning the leaf surface on one leaf (tip, middle, and near the petiole), and the average value was calculated.
Data analysis
A two-way analysis of variance (ANOVA) was used to examine the biochemical and physiological data, with “population” and “drought stress” acting as the constant factors. Sampling time served as the within-subjects factor, and “population” and “drought stress” as the main between-subjects factors. Changes in leaf gas exchange metrics during the investigation were tested. The relationship between factors, physiological and biochemical characteristics of seedlings was also assessed using principal component analysis (PCA).
Results
Water potential and soil water content
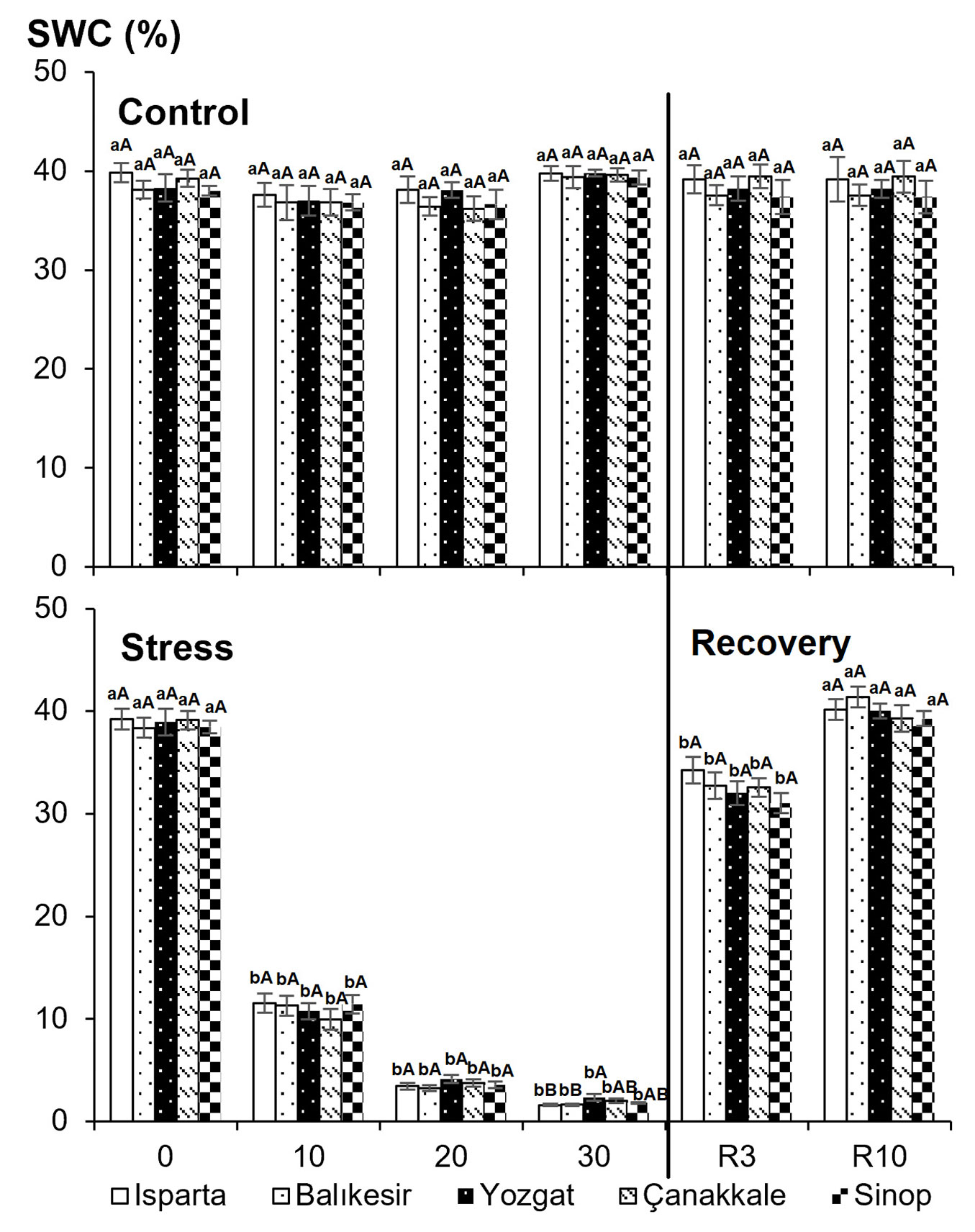
On day 30, the volumetric soil water content of drought-stressed seedlings ranged from 1.6% to 2.3%, while the populations’ midday water potential ranged from -3.5 to -4.5 MPa. Quercus cerris seedlings subjected to drought treatment on days 10, 20, and 30, followed by re-watering, showed significant changes in midday water potential and volumetric soil water content between treatments (Fig. 1, Fig. 2). Three days after re-irrigation, a significant difference in volumetric soil water content was observed between control and drought-stressed seedlings. Additionally, there was a significant positive correlation (R2 = 0.783, P < 0.01) between soil water content and midday water potential. As soil water content decreased, the midday water potential also decreased significantly. Volumetric soil water content was restored by day 10 after re-watering (Fig. 1).
Fig. 1 - Effects of drought stress on volumetric soil water content in five Q. cerris populations subjected to different drought treatments. Data are presented as means ± standard error (SE). Differences between treatments are shown with lower-case letters, while differences between populations are indicated with upper-case letters.
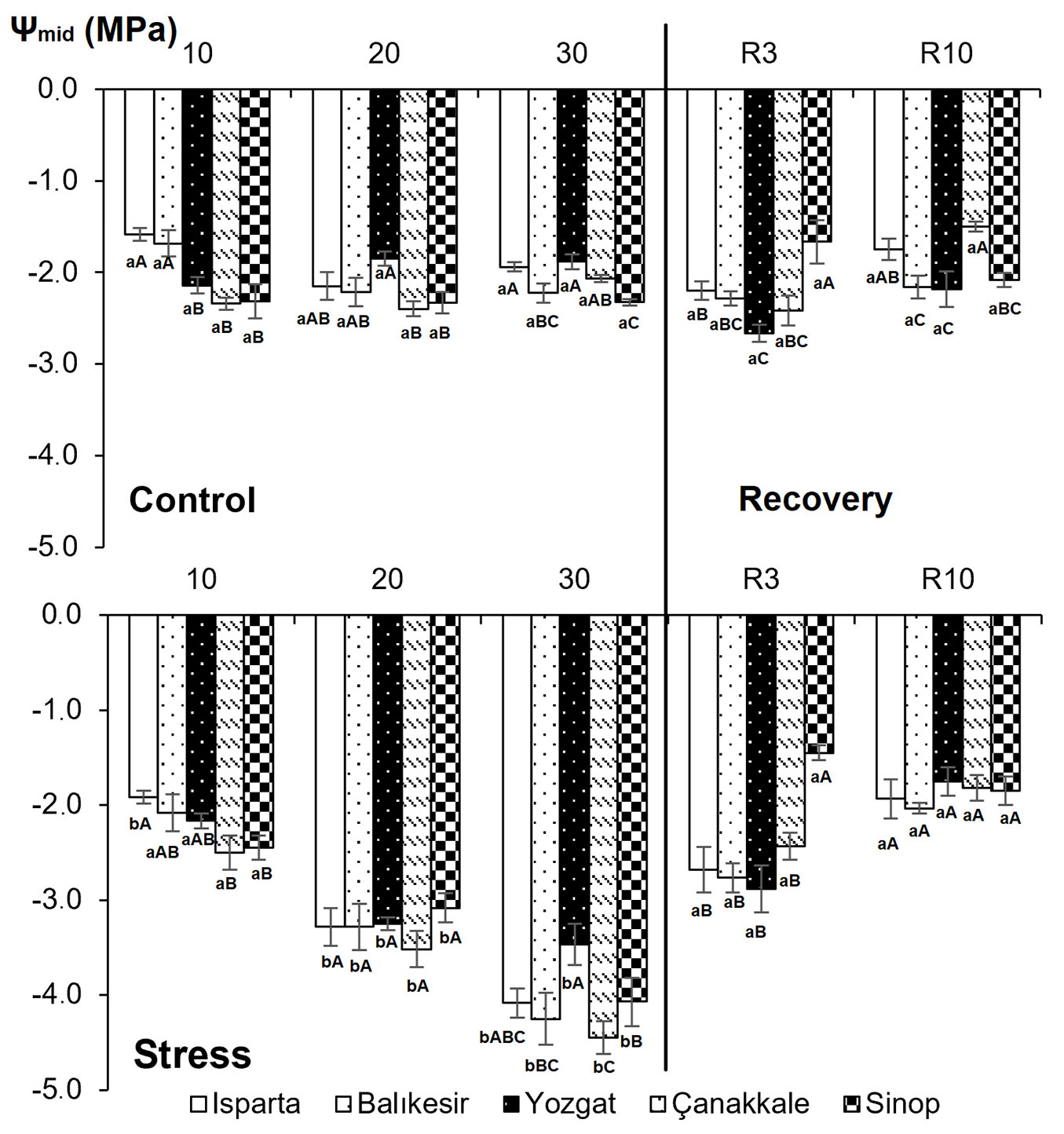
Fig. 2 - Effects of drought stress on midday water potential in five Q. cerris populations subjected to different drought treatments. Data are presented as means ± standard error (SE). Differences between treatments are shown with lower-case letters, while differences between populations are indicated with upper-case letters.
Regarding midday water potential, interactions across populations, treatments, and sampling times were highly significant (P < 0.01 - Tab. 2). Midday water potential decreased gradually with increasing drought stress. In the semi-arid Yozgat population, the highest midday water potential (-3.5 MPa) was found on the 30th day of drought stress. The lowest midday water potential (-4.5 MPa) was recorded in the Çanakkale population, originating from a humid climate. Midday water potential of previously stressed plants reached levels comparable to those of control plants by the end of the drought-recovery phase (R3 - Fig. 2). On the third day of recovery following drought stress, midday water potential was similar across all treatments.
Tab. 2 - Effects of drought stress on physiological and biochemical characteristics. (T): sampling time; (P): populations; (D): drought stress; (TXP): interactive effect of sampling time and population; (PXD): interactive effect of populations and drought stress; (DXPXT): interactive effect of sampling time, population, and drought stress; (*): P < 0.05; (**): P < 0.01; (ns): non-significant.
| Variables | F value | ||||||
|---|---|---|---|---|---|---|---|
| T | P | D | TXP | PXD | TXD | TXPXD | |
| SWC | 134.3** | 1.9ns | 948.2** | 0.8 ns | 0.3 ns | 94.8** | 1.4 ns |
| Ψmd | 55.7** | 5.2** | 156.9** | 6.8** | 3.6** | 42.4** | 2.0* |
| A net | 175.4** | 45.3** | 906.6** | 26.6** | 14.8** | 130.2** | 36.1** |
| gs | 163.7** | 187.6** | 2509.4** | 37.3** | 117.7** | 226.4** | 47.8** |
| E | 114.9** | 13.8** | 1909.3** | 16.7** | 11.7** | 161.4** | 32.1** |
| Ci | 85.1** | 21.0** | 1.4ns | 10.8** | 4.0** | 23.9** | 4.2** |
| TSC | 4.8** | 13.1ns | 6.0* | 18.4** | 1.2 ns | 7.0** | 1.7 ns |
| Chl | 1.2ns | 3.8* | 8.1* | 1.3 ns | 2.9 * | 4.3* | 0.5 ns |
Photosynthetic traits at the leaf level
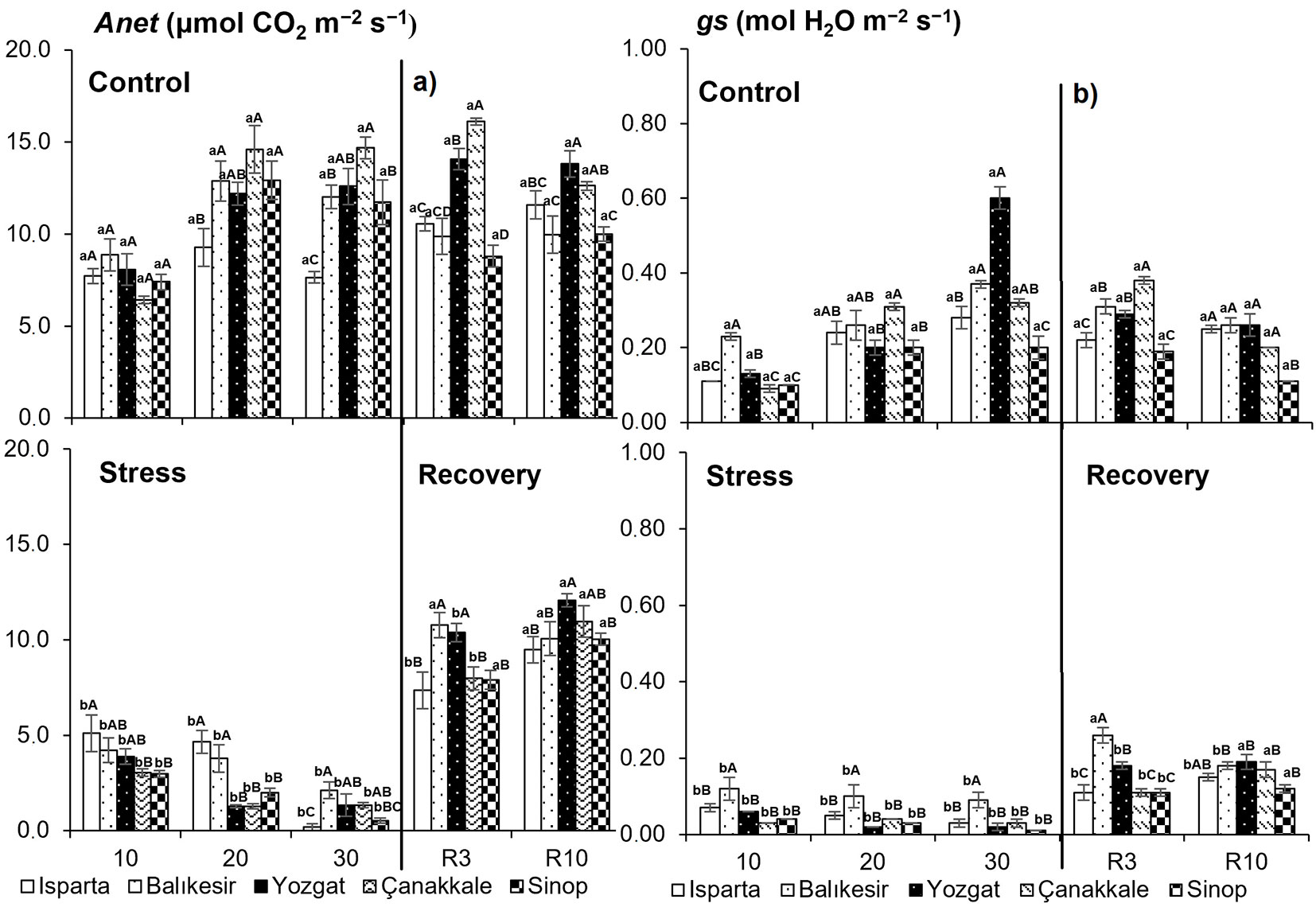
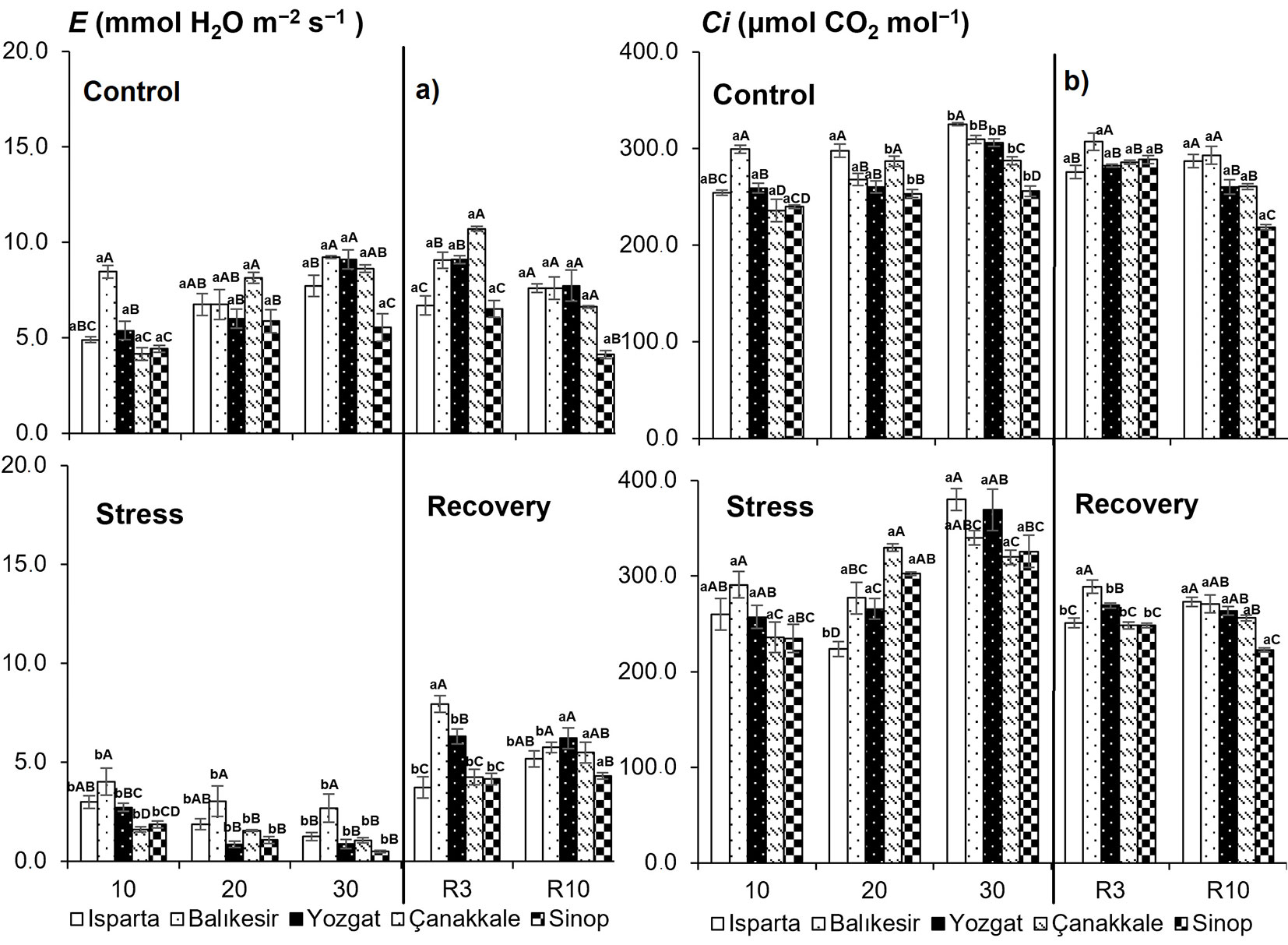
Drought stress was followed by a progressive decrease in net photosynthetic rate (Anet), stomatal conductance (gs), and transpiration (E) in all populations (Fig. 3a-b, Fig. 4a). The rate of decline in gas exchange parameters varied among populations. The interactions between the sampling time, populations and treatments for these variables (sampling time × population, population × drought stress and sampling time × drought stress interaction, P < 0.01) were also highly significant (Tab. 2). During the drought-recovery period (days 3-10), previously stressed plants began to increase Anet, gs, and E in some populations by day 3 and in others by day 10 after re-watering. By the end of the recovery period, Anet generally reached levels comparable to those of control seedlings. Intercellular CO2 concentrations differed significantly between treatments in all populations on the 30th day of drought stress. Accordingly, drought-stressed seedlings had higher intercellular CO2 concentrations (Fig. 4b). No significant differences in intercellular CO2 concentration were observed between treatments on the 10th day after re-watering.
Fig. 3 - Photosynthetic characteristics (a: net photosynthetic rate, Anet; b: stomatal conductance, gs) of Q. cerris seedlings grown under the different drought treatments. Data are presented as means ± standard error (SE). Differences between treatments are shown with lower-case letters, while differences between populations are indicated with upper-case letters.
Fig. 4 - Impact of drought stress on (a) transpiration rate (E) and (b) intercellular CO2 concentration (Ci) under different drought treatments. Data are presented as means ± standard error (SE). Differences between treatments are shown with lower-case letters, while differences between populations are indicated with upper-case letters.
The results of the principal component analysis are summarized in Fig. S3 (Supplementary material). According to the PCA, the highest Pearson’s correlation coefficient (r) with PC1 (variance accounted for: 67.4%) was recorded for ψmid (r=0.864), SWC (r=0.920), Anet (r=0.915), gs (r=0.862), E (r=0.925), TSC (r=0.621), and Chl (r=0.843). Midday water potential was positively correlated with volumetric water content, net photosynthetic rate, stomatal conductance, transpiration rate, and chlorophyll pigment content, and negatively correlated with intercellular CO2 concentration and total soluble sugar content.
Biochemical traits in response to drought
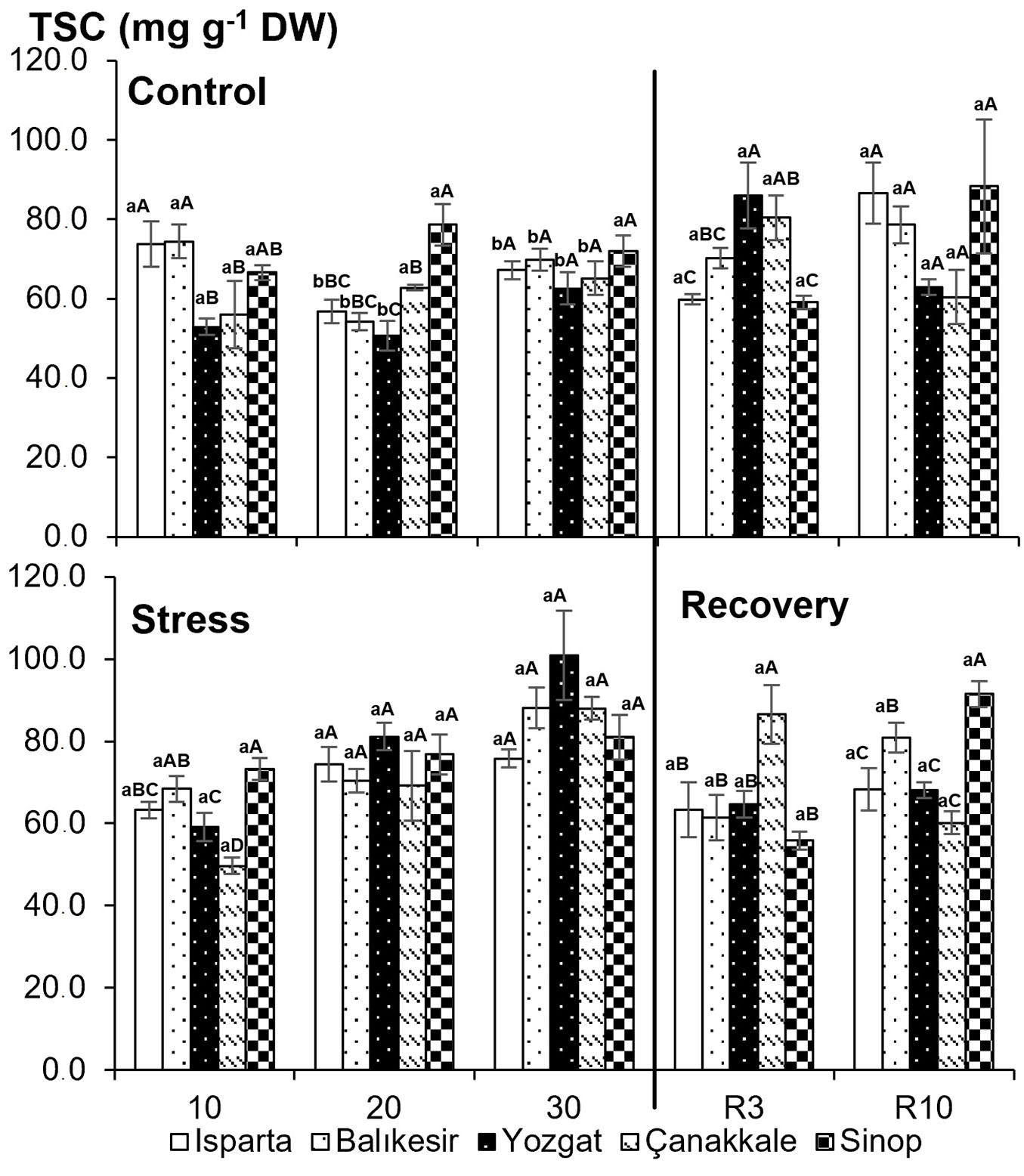
Examining the effects of sampling time and drought stress on total soluble sugar by ANOVA revealed significant main effects of time and drought stress, as well as the time × drought stress interaction (P<0.01 - Tab. 2). Total soluble sugar content increased with drought stress. Differences were significant among treatments, except for the Sinop population on day 30. On day 3 of re-watering, the total soluble sugar content in the drought-stressed treatments was close to that of the control treatment across all populations (Fig. 5). The total soluble sugar content recovered on day 3 after drought stress.
Fig. 5 - Total soluble sugar (TSC) content in five Quercus cerris populations under control and drought stress conditions. Data are presented as means ± standard error (SE). Differences between treatments are shown with lower-case letters, while differences between populations are indicated with upper-case letters.
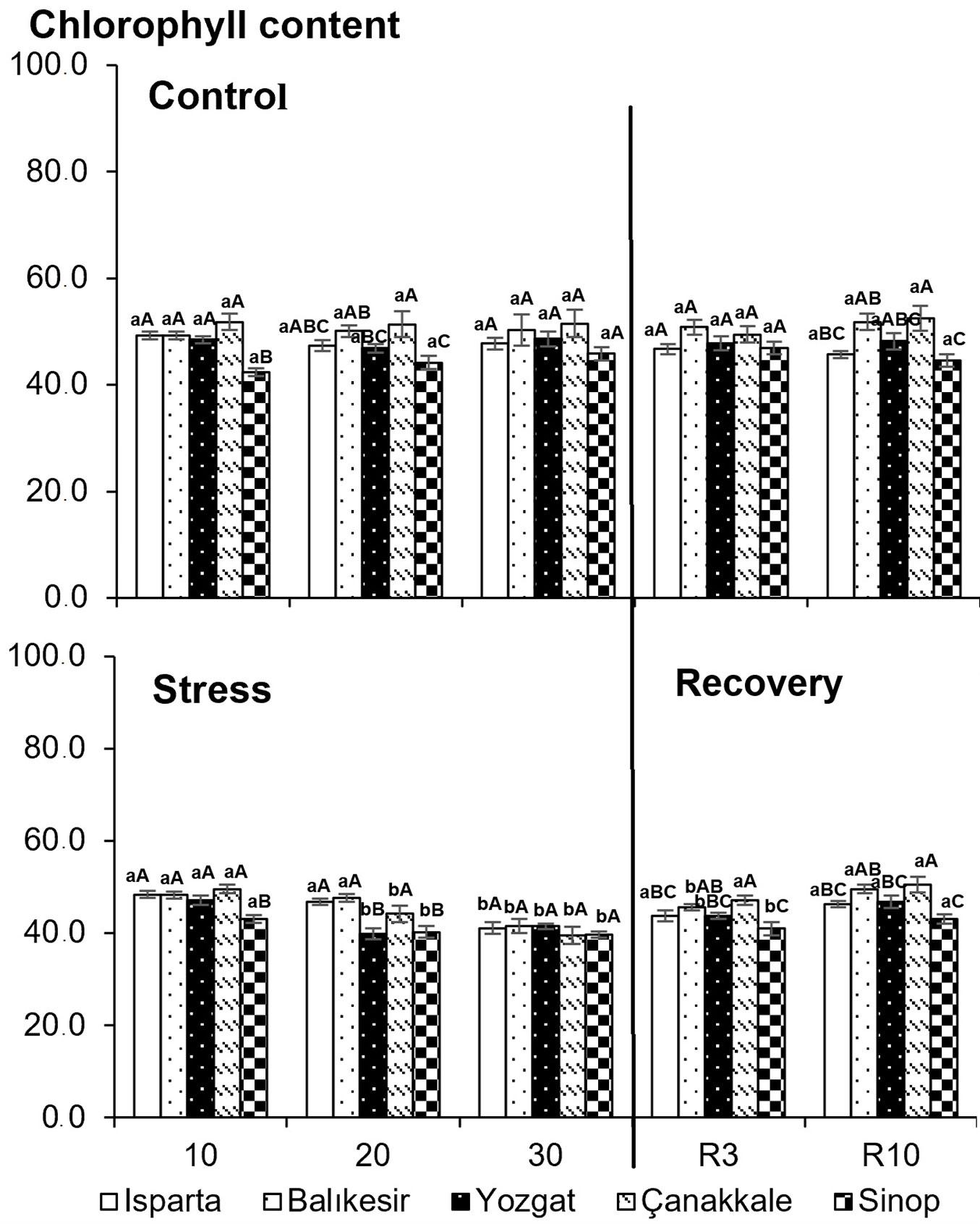
Chlorophyll content decreased significantly in all populations on the 30th day of drought stress (Fig. 6). After rewatering, chlorophyll content was similar to that of the control treatments in Isparta and Çanakkale populations on the 3rd day and in other populations on the 10th day.
Fig. 6 - Effects of drought stress on chlorophyll content in five Q. cerris populations grown under different drought treatments. Data are presented as means ± standard error (SE). Differences between treatments are shown with lower-case letters, while differences between populations are indicated with upper-case letters.
Discussion
Applying drought stress to different populations of Q. cerris across semi-arid, semi-humid, and humid climates revealed significant insights into their physiological responses, in terms of midday water potential, gas exchange parameters, total soluble sugar, and chlorophyll content. Drought stress significantly decreased midday water potential in seedlings from all five populations. The interactions among populations, treatments, and sampling time were also significant for midday water potential. By the 30th day of drought stress, seedlings from different populations had varying reductions in midday water potential. Those of the Yozgat population, originated from a semi-arid climate, maintained a relatively higher midday water potential (-3.5 MPa) than those from other populations, whereas the Çanakkale seedlings, originated from a humid climate, exhibited a lower value (-4.5 MPa). A mean midday water potential of -4.7 MPa was reported after 58 days of water deficit ([41]). Each species can adjust its physiological responses to environmental conditions under water deficit, and the degree of drought adaptation can vary significantly among genera and species ([49]). By the end of the drought-recovery phase (R3), the midday water potential of previously stressed plants reached values similar to those of control seedlings across all populations. Following re-watering, soil water content remained low, but leaf water status recovered.
Water shortage affects a wide range of plant characteristics and processes, including gas exchange ([36], [25], [45]), predawn and midday water potential ([11], [5]). Stomatal management controls how much water plants take up and how long they can withstand drought stress ([49]). Anet, gs, and E differed significantly in sampling time, population, drought stress, and their interactions. On the 30th day of drought stress, stomatal conductance approached zero. With increasing drought stress, a decrease in net photosynthetic rate and stomatal conductance has been reported in Q. ilex ([38]), Q. cerris ([11]), and Q. brantii Lindl ([24]). During drought stress, early stomatal closure is typical of isohydric plants. In contrast, when water is scarce, anisohydric plants delay stomatal closure, allowing the leaf water potential to drop as soil water content declines ([48]). Nine species showed this typical response, with a significant reduction in Ci during the early stomatal control phase, when stomatal conductance decreased. A substantial nonstomatal limiting phase was observed as gs dropped to low values, leading to a rise in Ci as gs approached a minimum ([6]). In Q. cerris seedlings, Ci increased with drought stress. Since closed stomata keep CO2 out of plant leaves, this may be the cause of the intercellular build-up of CO2. However, because photosynthesis cannot proceed effectively without sufficient CO2, the rate of photosynthesis can be low. Diffusion constraints on CO2 uptake mostly limit photosynthesis until water stress reaches an extreme level at which practically no water remains for transpiration ([47]). Ying et al. ([52]) found that the reductions in gs, E, and Ci under severe-stress conditions in the Kunming provenance were higher than those in the other populations, suggesting a better adaptation of that provenance to dry conditions. In this study, the decrease in net photosynthetic rate under stress in Q. cerris seedlings from Yozgat, Çanakkale, and Balikesir populations was relatively greater than in Isparta and Sinop provenances. In seedlings from both these populations, gs and Anet declined dramatically following drought stress exposure, and there was a clear positive correlation between Anet and gs ([29]). The rates of transpiration, stomatal conductance, and Anet were found to be strongly positively correlated in Q. cerris seedlings. In drought-stressed oak seedlings, the net photosynthetic rate of all populations increased on the 3rd day after re-watering, but Anet, gs, and E fully recovered on the 3rd day after rewatering. This suggests that incomplete recovery of the net photosynthetic rate can be due to the persistence of stomatal limitations ([21]). Across all populations, Anet reached values comparable to those of the control seedlings by the 10th day, at the end of the recovery period. Photosynthetic performance improved progressively during the re-watering period ([19]). Previous studies demonstrated that drought-tolerant angiosperms have a rapid recovery of net photosynthetic rate after a few days of rewatering ([51]). Anet and leaf have been reported to recover to control levels 16 days after rehydration in Bertholletia excelsa ([41]).
Drought stress increased the total soluble sugar content and decreased chlorophyll content in Q. cerris seedlings. Total sugar accumulation in response to drought stress was observed in seedlings from all populations, except Sinop. Following rewatering, total sugar levels decreased rapidly. The concentration of soluble sugar increased as a result of the drought in Pinus tabulaeformis Carr. Re-watering dramatically reduced the content of leaf-soluble sugars under severe and moderate drought treatments ([23]). Sugar accumulation has been observed in Quercus seedlings grown under drought stress ([24], [5]). In this study, after three days of re-watering, the total soluble sugar content in all populations in the drought-stress treatments was nearly identical to that of the control. Chlorophyll content was affected by populations, drought stress, the populations × drought stress interaction, and the sampling time × drought stress interaction. Drought stress also affected leaf chlorophyll concentration. Chlorophyll content has been reported to decrease significantly in the leaves of two Quercus species as the duration of drought stress increased ([35]). A considerable drop in chlorophyll content may indicate chlorophyll degradation caused by water deficit. In stressed Q. cerris plants, chlorophyll content steadily improved throughout the rewatering phase. Low water availability could reduce CO2 concentrations inside the chloroplast due to stomatal closure, therefore the drop in photosynthetic pigment concentrations could help prevent serious damage to the photosynthetic apparatus. After a period of rewatering, chlorophyll levels were fully restored, accelerating the recovery of photosynthesis ([34]).
Provenances from higher altitudes have been reported to survive better to drought-stress than those from lower altitudes ([44]). The altitude of the Sinop population is the lowest among other populations of Q. cerris analyzed in this study. Compared with seedlings from drier climatic provenances, those from wetter provenances showed higher tolerance to drought stress ([24]). In this study, the Yozgat population withstood drought by maintaining higher water potential, whereas seedlings from the Çanakkale population tolerated low water potential by increasing total soluble sugar content. This could be hypothesized to be due to the severe summer drought occurring in Çanakkale, which was classified as a humid climate site. Indeed, Q. cerris populations with a low summer drought index may be well adapted to dry conditions. Regarding leaf gas exchange, provenances from humid climate demostrated the lowest resistance to water deficiency and the lowest capacity of recovery following rewatering ([37]). Alonso-Forn et al. ([3]) noted that water stress applied gradually to Mediterranean oak species (Q. faginea Lam. and Q. ilex subsp. rotundifolia Lam.) exhibited different functional strategies to cope with water deficit. In addition, the evergreen species Q. ilex, with a less developed root system, likely maintains a lower net photosynthetic rate and leaf area than deciduous species. In contrast, deciduous European oaks such as Q. robur and Q. petraea, tend to adopt an avoidance strategy by establishing deep root systems to cope with drought stress ([18]).
Conclusions
This study highlights the significance of population-level variation in drought tolerance in Q. cerris, demonstrating how local climatic conditions can shape physiological and biochemical responses to drought stress. Q. cerris populations in Isparta, Balikesir, Çanakkale, and Yozgat exhibited adaptive responses to drought, in terms of decreased midday water potential and gas exchange, along with increased total soluble sugar accumulation. While these findings support our initial hypothesis, they also suggest that drought tolerance may be more strongly associated with the severity of summer drought experienced by populations than with their classification as originating from semi-humid or humid regions. Our results underline the importance of identifying well-adapted populations for future afforestation and seed transfer programs, particularly in regions anticipated to experience drought as a consequence of climate change.
Acknowledgments
This work was supported by the Research Fund of the Isparta University of Applied Sciences. Project Number: 2021-ILK1-0158.
References
Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Isparta University of Applied Sciences, Faculty of Forestry, Isparta (Turkey)
Corresponding author
Paper Info
Citation
Bayar E (2026). Recovery of water potential and leaf gas exchange performance following drought stress in Quercus cerris populations. iForest 19: 94-101. - doi: 10.3832/ifor4743-018
Academic Editor
Claudia Cocozza
Paper history
Received: Oct 10, 2024
Accepted: Aug 27, 2025
First online: Mar 17, 2026
Publication Date: Apr 30, 2026
Publication Time: 6.73 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2026
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 736
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 185
Abstract Page Views: 328
PDF Downloads: 196
Citation/Reference Downloads: 0
XML Downloads: 27
Web Metrics
Days since publication: 46
Overall contacts: 736
Avg. contacts per week: 112.00
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
(No citations were found up to date. Please come back later)
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Effect of drought stress on some growth, morphological, physiological, and biochemical parameters of two different populations of Quercus brantii
vol. 11, pp. 212-220 (online: 01 March 2018)
Research Articles
Light acclimation of leaf gas exchange in two Tunisian cork oak populations from contrasting environmental conditions
vol. 8, pp. 700-706 (online: 08 January 2015)
Research Articles
Links between phenology and ecophysiology in a European beech forest
vol. 8, pp. 438-447 (online: 15 December 2014)
Research Articles
Carbohydrate metabolism during new root growth in transplanted Larix olgensis seedlings: post-transplant response to nursery-applied inorganic fertilizer and organic amendment
vol. 10, pp. 15-22 (online: 22 September 2016)
Research Articles
Multi-temporal influence of vegetation on soil respiration in a drought-affected forest
vol. 11, pp. 189-198 (online: 01 March 2018)
Short Communications
Variation in growth, photosynthesis and water-soluble polysaccharide of Cyclocarya paliurus under different light regimes
vol. 10, pp. 468-474 (online: 04 April 2017)
Research Articles
Magnolia grandiflora L. shows better responses to drought than Magnolia × soulangeana in urban environment
vol. 13, pp. 575-583 (online: 07 December 2020)
Research Articles
Wintertime photosynthesis and spring recovery of Ilex aquifolium L.
vol. 12, pp. 389-396 (online: 31 July 2019)
Research Articles
Auxin (IAA) and soluble carbohydrate seasonal dynamics monitored during xylogenesis and phloemogenesis in Scots pine
vol. 11, pp. 553-562 (online: 01 September 2018)
Research Articles
Response of juvenile progeny of seven forest tree species and their populations to simulated climate change-related stressors, heat, elevated humidity and drought
vol. 11, pp. 374-388 (online: 15 May 2018)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword