Effect of plant species on P cycle-related microorganisms associated with litter decomposition and P soil availability: implications for agroforestry management
Eduardo Correa (1), Lilia Carvalhais (1), Mirian Utida (2), Christiane Abreu Oliveira (2), Maria Rita Scotti (1)
iForest - Biogeosciences and Forestry, Volume 9, Issue 2, Pages 294-302 (2015)
doi: https://doi.org/10.3832/ifor1459-008
Published: Oct 05, 2015 - Copyright © 2015 SISEF
Research Articles
Abstract
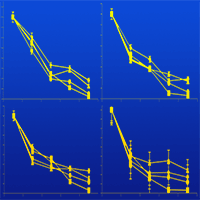
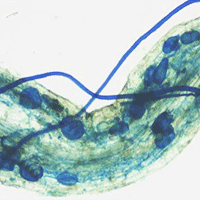
Cutting dry deciduous forest (preserved site) for wood supply in semi-arid Brazil has led to invasion of a pioneer shrub vegetation called “Carrasco” (disturbed site), which inhibits the sprouting of native species. A land restoration project was undertaken in a cleared Carrasco area where a mixed plantation of native species and Eucalyptus spp. (experimental site) was established to preserve the forest and ensure wood supply for the local population. We considered phosphorus as a limiting soil nutrient to plant growth, and we addressed the roles of litter decomposition and microbial activity on phosphorus release in the disturbed, preserved and experimental sites. The phosphorus released from leaf litter was affected by the vegetation type, which favored specific soil microbial populations during decomposition. The Carrasco vegetation predominantly favored arbuscular mycorrhizal fungi (AMF), as shown by root colonization in the litter bags; the Eucalyptus plants favored AMF and ectomycorrhizal fungi (EM), as well as phosphate solubilizing microorganisms (PSM), and the intercropping system favored AMF and PSM groups. In contrast, the preserved site favored the PSM population. High phosphatase activity was found in the preserved and experimental sites in contrast to the Carrasco soil. Principal component analysis showed that AMF root colonization and phosphatase activity were the main parameters influencing the increase in soil phosphorus. Based on the above results, rehabilitation appeared to be underway in the experimental site, since the samples were more similar to the preserved site than to the disturbed site. This effect was attributed to Eucalyptus camaldulensis that promote the establishment of all phosphorus cycle-related microorganisms (AMF, EM and PSF). E. camaldulensis associated with mycorrhizal fungi and PSM are recommended for inclusion in agroforestry systems.
Keywords
Agroforestry System, Arbuscular Mycorrhizal Fungi, Land Restoration, Litter Decomposition, Phosphate Solubilizing Microorganisms, Soil Phosphorus
Authors’ Info
Authors’ address
Lilia Carvalhais
Maria Rita Scotti
Department of Botany, Institute of Biological Science / Federal University of Minas Gerais . Avenida Antonio Carlos, 6627, Pampulha, Cep: 31.270-901 Belo Horizonte Minas Gerais (Brazil)
Christiane Abreu Oliveira
EMBRAPA- National Center of Maize and Sorgum Rod. MG. 424 Km 45 CEP: 35701970 Sete Lagoas- Minas Gerais (Brazil)
Corresponding author
Paper Info
Citation
Correa E, Carvalhais L, Utida M, Oliveira CA, Scotti MR (2015). Effect of plant species on P cycle-related microorganisms associated with litter decomposition and P soil availability: implications for agroforestry management. iForest 9: 294-302. - doi: 10.3832/ifor1459-008
Academic Editor
Gianfranco Minotta
Paper history
Received: Sep 29, 2014
Accepted: Jun 22, 2015
First online: Oct 05, 2015
Publication Date: Apr 26, 2016
Publication Time: 3.50 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2015
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 55909
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 45778
Abstract Page Views: 4173
PDF Downloads: 4529
Citation/Reference Downloads: 27
XML Downloads: 1402
Web Metrics
Days since publication: 3872
Overall contacts: 55909
Avg. contacts per week: 101.08
Citation Metrics
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2016): 8
Average cites per year: 0.80
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
References
Vegetação e manejo de solos - projeto Jaiba [Vegetation and soil management - Jaiba Project]. Relatório n°5, Distrito de Irrigação do Jaiba, Belo Horizonte, Brazil, pp. 25-60. [in Portuguese]
Gscholar
Manual de métodos de análises do solo [Manual of soil analysis methods] (2nd edn). Centro Nacional de Pesquisas de Solos, Empresa Brasileira de Pesquisas Agropecuária - Embrapa solos, Rio de Janeiro, Brazil, pp. 13-196. [in Portuguese]
Gscholar
Forage fiber analyses: apparatus, reagents, procedures, and some applications. Agricultural Handbook No. 379, USDA, Washington, DC, USA, pp. 20.
Gscholar
Isolation of DNA from fungal mycelia and single spores. In: “PCR Protocols. A Guide to Methods and Applications” (Innis MA, Gelfand DH, Sninsky JJ, White TJ eds). Academic Press, San Diego, USA, pp. 282-287.
Gscholar
Effect of rhizobia, mycorrhizal fungi and phosphate-solubilizing microorganisms in the rhizosphere of native plants used to recover an iron ore area in Brazil. European Journal of Soil Biology 45: 259-266.
CrossRef | Gscholar
Método simplificado para determinação colorimétrica de nitrogênio em plantas. [Simplified method for colorimetric determination of nitrogen in plants]. Ciência y Cultura 38: 178-180. [in Portuguese]
Gscholar
Mobilization of phosphorus in soil in connection with vital activities by some microbial species. Mikrobiologia 17: 362-370.
Gscholar
Tratado de fitogeografia do Brasil: aspectos ecológicos, sociológicos e florísticos [Treaty of plant geography of Brazil: ecological, sociological and floristic aspects]. Ambito Cultural Edições Ltda, Rio de Janeiro, Brazil, pp. 747 [in Portuguese]
Gscholar
Estatística aplicada à experimentação animal [Statistics applied to animal experimentation]. Fundação de Estudo e Pesquisa em Medicina Veterinária, Belo Horizonte, Brazil, pp. 221. [in Portuguese]
Gscholar
Análises químicas em plantas [Chemical analysis in plants]. Escola Superior de Agricultura Luiz de Queiroz (ESALQ). Piracicaba, São Paulo, Brazil, pp. 56 . [in Portuguese]
Gscholar
Methods in legume - Rhizobium technology. NifTAL, MIRCEN University of Hawaii Press, Maui, Hawaii, USA, pp. 361.
Gscholar
Soil enzymes. In: “Methods of Soil Analysis - Part 2” (Page AL, Miller RH, Keeney DR eds). American Society Agronomy, Madison, WI, USA, pp. 943-947.
Gscholar
Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: “PCR Protocols: A Guide to Methods and Applications” (Innis MA, Gelfand DH, Sninsky JJ, White TJ eds). Academic Press, San Diego, CA, USA, pp. 315-322.
Gscholar