The use of branch enclosures to assess direct and indirect effects of elevated CO2 on photosynthesis, respiration and isoprene emission of Populus alba leaves
Federico Brilli (1) , Domenico Tricoli (1), Silvano Fares (1), Mauro Centritto (2), Francesco Loreto (1)
iForest - Biogeosciences and Forestry, Volume 1, Issue 1, Pages 49-54 (2008)
doi: https://doi.org/10.3832/ifor0429-0010049
Published: Feb 28, 2008 - Copyright © 2008 SISEF
Research Articles
Abstract
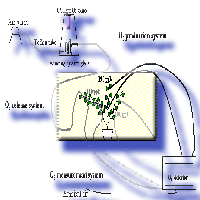
We used a novel system of branch enclosures to study the impact of elevated CO2 (900 ppm) on the gas-exchange characteristics of developed and developing leaves of white poplar (Populus alba L.), as well as of leaves subsequently developing at ambient CO2, outside the enclosures in which the CO2 concentration was raised. We found no significant effect of elevated CO2 on photosynthesis, respiration and isoprene emission, as the rates of developed and developing leaves inside the enclosures, and of leaves developing outside the enclosures, were similar to those recorded using enclosures maintained at ambient CO2. The enclosure system, however, largely influenced the rates of gas-exchange. In fact, leaves already developed inside the enclosures showed rates of photosynthesis, stomatal conductance, and isoprene emission higher than leaves developing inside the enclosures, and also higher than leaves developing outside the enclosure. These differences were caused by a higher efficiency in the light use and by a higher Ribulose 1.5 bisphosphate carboxylase (Rubisco) activity in leaves fully developed inside enclosures than in the other leaf classes. The experiment overall suggests that branch enclosures may alter the physiology of the plants, reducing or counteracting the impact of elevated CO2, which we predicted to stimulate photosynthesis and uncouple isoprene emission from photosynthesis. This may be an important bias against the use of enclosure systems for studies of the impact of environmental constraints and global change factors on physiological features.
Keywords
Populus alba, Elevated CO2, Branch Enclosure, Photosynthesis, Isoprene Emission
Authors’ Info
Authors’ address
Domenico Tricoli
Silvano Fares
Francesco Loreto
CNR - Istituto di Biologia Agroambientale e Forestale, v. Salaria Km. 29.300, I-00016 Monterotondo Scalo, RM (Italy)
CNR - Istituto sull’Inquinamento Atmosferico, v. Salaria Km. 29.300, I-00016 Monterotondo Scalo, RM (Italy)
Corresponding author
Paper Info
Citation
Brilli F, Tricoli D, Fares S, Centritto M, Loreto F (2008). The use of branch enclosures to assess direct and indirect effects of elevated CO2 on photosynthesis, respiration and isoprene emission of Populus alba leaves. iForest 1: 49-54. - doi: 10.3832/ifor0429-0010049
Paper history
Received: Mar 09, 2006
Accepted: Jan 01, 2007
First online: Feb 28, 2008
Publication Date: Feb 28, 2008
Publication Time: 14.10 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2008
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 57901
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 46254
Abstract Page Views: 3968
PDF Downloads: 6417
Citation/Reference Downloads: 52
XML Downloads: 1210
Web Metrics
Days since publication: 6660
Overall contacts: 57901
Avg. contacts per week: 60.86
Citation Metrics
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2008): 2
Average cites per year: 0.11
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
References
Monoterpene emission and monoterpene synthase activities in the Mediterranean evergreen oak <i>Quercus ilex</i> L. grown at elevated CO<sub>2</sub> concentrations. Global Change Biology 7: 709-717.
CrossRef | Gscholar
Respiration in the light measured by <sup>12</sup>CO<sub>2</sub> emission in <sup>13</sup>CO<sub>2</sub> atmosphere in maize leaves. Australian Journal of Plant Physiology 28: 1103-1108.
Gscholar
Impact of rising CO<sub>2</sub> on emission of volatile organic compounds: isoprene emission from <i>Phragmites australis</i> growing at elevated CO<sub>2</sub> on a natural carbon dioxide spring. Plant Cell & Environment 27: 393-401.
CrossRef | Gscholar