The impact of seed predation and browsing on natural sessile oak regeneration under different light conditions in an over-aged coppice stand
iForest - Biogeosciences and Forestry, Volume 9, Issue 4, Pages 569-576 (2016)
doi: https://doi.org/10.3832/ifor1835-009
Published: Apr 04, 2016 - Copyright © 2016 SISEF
Research Articles
Collection/Special Issue: IUFRO division 8.02 - Mendel University Brno (Czech Republic) 2015
Coppice forests: past, present and future
Guest Editors: Tomas Vrska, Renzo Motta, Alex Mosseler
Abstract
Sessile oak (Quercus petraea (Matt.) Liebl.) is one of the most important commercial species cultivated at low altitudes in the Czech Republic, and over-aged coppices are a significant part of oak stands in the region. In order to secure a high-valuable timber production (e.g., through conversion of such stands into coppices-with-standards), knowledge of the potential and limits of generative regeneration is essential. This study was conducted in three oak-dominated over-aged coppice stands in different stages of conversion into coppices-with-standards and characterized by different basal area (BA, from 9.3 to 14.1 m2 ha-1) and relative diffuse radiation (ISF, from 12.1 to 35.5%). The study stands were compared with respect to seed predation following acorn fall and oak regeneration parameters. At the time of their fall the acorns represented a sought-after source of food for large mammals (particularly wild boar). At the end of acorn fall, 13-67% acorns were lost due to animal predation. A control evaluation conducted the following spring revealed a decrease of 92-97% in fallen acorns. Despite the major animal impact, a high reserve of acorns and saplings remained in the stands (4 600-29 000 acorns and 66 000-310 000 saplings per ha). With increasing light intensity the oak regeneration density decreased, while the height and age variability of oak regeneration increased. Although saplings were capable of surviving several years under unfavorable light conditions (even below 12% ISF), they require a minimum of 20% ISF (i.e., BA < 16 m2 ha-1) to achieve sustainable height increment. Based on our results, for conversion of such stands into coppices-with-standards we recommend a maximum of 200 reserved trees (BA = 16 m2 ha-1) to achieve successful height growth of the understorey.
Keywords
Over-aged Coppice, Quercus petraea, Natural Regeneration, Herbivore Impact, Acorn, Light Intensity, Wild Boar
Introduction
Modern techniques of forest regeneration in Central Europe aim at an optimal utilization of natural regeneration, both for economic reasons and for enhancing forest stand stability and species diversity. In the lower elevation areas of the Czech Republic (hereafter CR), sessile oak (Quercus petraea (Matt.) Liebl.) is one of the most important commercial species. The natural proportion of oak in the CR is estimated to be about 19% of the forest area, with a total stand area consisting of 96% high forest, 1.9% coppice-with-standards and 0.7% low forests. The relatively significant proportion of oak high forests is actually represented by over-aged coppices (i.e., stands of vegetative origin with a longer rotation period) with age similar to that of high forests (i.e., stands of generative origin).
There are various silvicultural systems prescribing how to manage stands using vegetative or generative regeneration or both ([29]). In this context, a proportion of generative oak regeneration in vegetative-regenerated coppices is often kept for a high-valued timber production, as in the coppice-with-standards management. Such silvicultural system consists in a two-storied stand with an upper story of trees of different ages (standards) and a lower storey dominated by stems originated from sprouts. The harvest of standards occurs periodically (usually 30 years - [13]) and simultaneously with the lower story harvesting. Approximately 50-100 trees of new generation per ha are usually left in the lower story to replace older standards which are selectively harvested. Standards are ordinarily divided into four age cohorts (31-60, 61-90, 91-120 and 121+ years), and selected for harvesting based on criteria such as their economic value, health, age and the surrounding stand density ([3]).
Knowledge of the potential and limits of oak generative regeneration in coppice-with-standards stands is essential for their maintenance and stability. However, the success of natural regeneration is conditioned by a number of biotic and abiotic factors ([39]), in particular the climatic conditions ([26]), stand structure ([25]), seed predation ([9]) or annual shoot browsing ([1], [15]).
Oak reaches fructification at the age of 40-50 years in open stands and 70-80 years in closed stands. Fruit production in open stands occurs almost every year, while in closed stands every 4-8 years (mast years). The mature stand production is 0.7-2.0 t acorns ha-1, a well-developed free-growing oak produces 40-100 kg acorns year-1. Mast years with production exceeding 50 acorns m-2 are rare, mainly due to late frost and insect pests (predominantly Tortrix viridana and Thaumetopoea processionea) destroying the first blossoms. Further, part of the mast is consumed by birds and squirrels and attacked by insect pests while still on the tree. Losses occurring after seed fall are even more substantial. Seeds on the ground are subjected to a complex of negative abiotic and biotic factors, mainly predation (insects, birds, mice, squirrels, ungulates), water availability, late frost, fungal attack and diseases ([30]). On the other hand, animals (particularly rodents and birds) play a positive role in acorn dispersal, with jays being able to spread acorns up to several kilometers ([18], [11]). However, in autumn acorns represent a source of a highly attractive food for all ungulates ([14]) and rodents ([28]). The cumulative impact of all consumers may result in the destruction of all oak seeds and sapling production ([8], [39]). Cutini et al. ([8]) described the high yearly variability of seed production as a defense strategy for oaks, resulting in the satiation of consumers in mast years and their starvation in the others (the “predator satiation” hypothesis). Indeed, a direct correlation between the occurrence of mast years and the density of consumers has been confirmed for wild boar ([12]) or rodents and birds ([43], [7]).
Successful natural regeneration of oak stands is suppressed by ruminant ungulates, such as roe deer (Capreolus capreolus) and red deer (Cervus elaphus), that can cause significant damage to stand regeneration by retarding tree growth ([19], [2]) or preventing seed emergence. However, the population of free living ruminants in the Czech Republic is relatively stable over time, while the abundance of wild boar (Sus scrofa) has been recently increasing ([24]), leading to an extended consumption of acorns and a consequent reduction of the natural regeneration of oak stands ([9]). Wild boar is characterized by a high ecological adaptability and reproductive potential, and feeds on nourishment of high nutritional value ([40]), such as seeds and field crops in spring/summer and forest food from autumn to spring ([34]). In relation to forests management, wild boar had not been perceived as a harmful species for a long time, as its disturbance of the topsoil is considered beneficial for stand regeneration ([31]). However, its current high abundance and feeding behavior can dramatically impact on forest ecosystems and thus requires a closer attention.
Light availability is another major limiting factor affecting the survival, growth and competitive ability of oak regeneration (particularly with respect to shade-tolerant species, such as beech, hornbeam or lime). Sessile oak is a shade-tolerant species in the earlier life-cycle stages, being able to survive at only 15% of the full solar radiation. However, at least 20% of relative radiation is required for its sustainable growth, with an optimum relative light intensity of 20-40% and 25-50% for one-year-old and two-year-old saplings, respectively ([30]). At older age, oak is rather a heliophilous species. Light reduction results in higher height increment, larger leaf area and a higher content of chlorophyll, but also in an insufficient development of the root system, lower metabolism, lower assimilatory capacity, and generally lower growth performances.
The natural regeneration of oak stands is therefore affected by many factors. Tree density is related to seed production and distribution, and high density strongly limits sapling growth by reducing light intensity and water availability. Moreover, the success of regeneration is influenced by seed consumption and shoots browsing caused by mammals. The aim of the study was to evaluate the impact of wild boar, roe deer and rodents on acorn abundance and on the growth of oak regeneration under different light conditions in an oak over-aged coppice stand.
Materials and methods
Study area
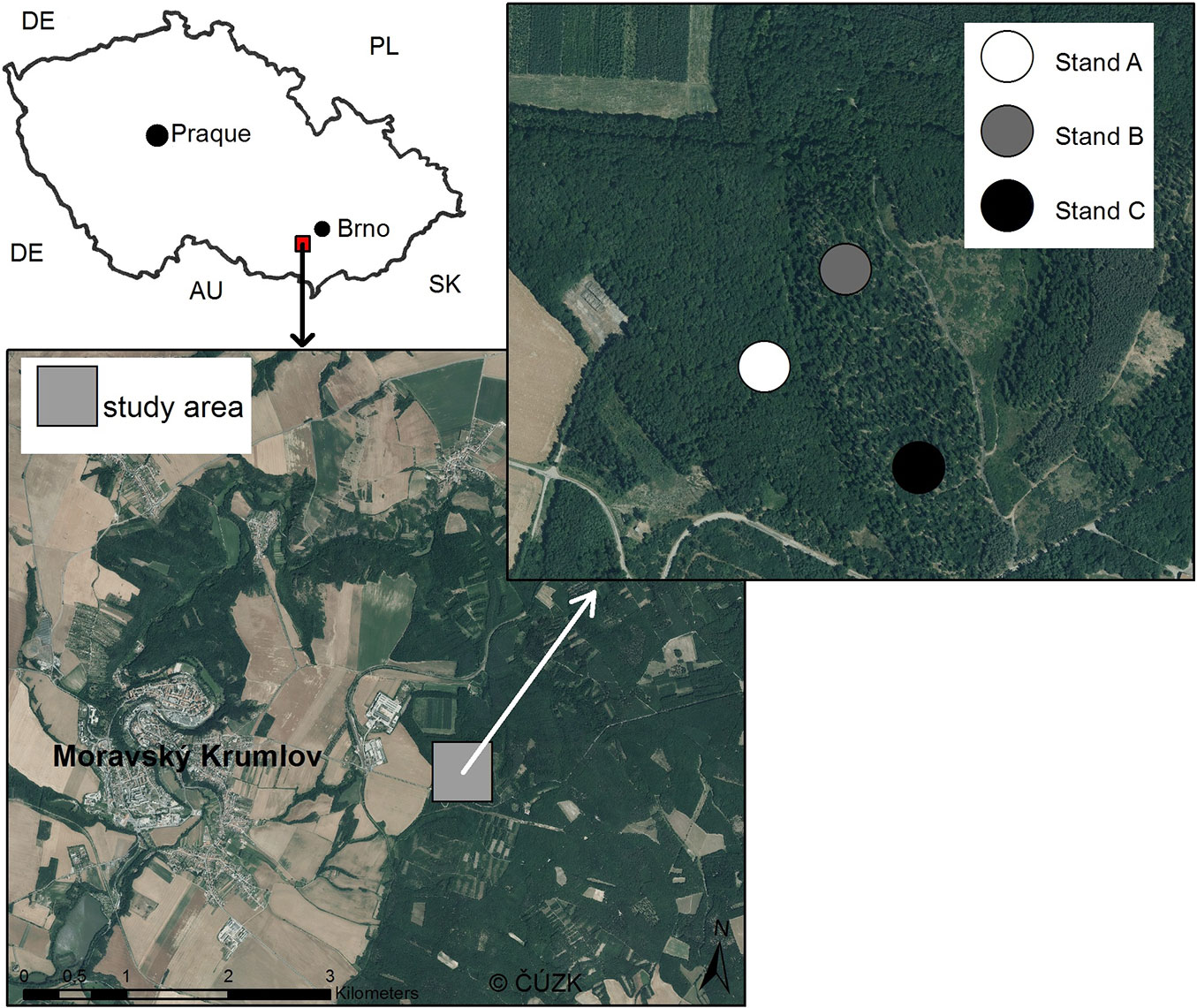
The study area was located in the southern part of the Czech Republic near the town of Moravský Krumlov (49° 2′ 42.849″ N, 16° 21′ 13.071″ E - Fig. 1). Elevation of the area ranges between 250 and 380 m a.s.l. and the mean annual temperature is 8-9 °C. The parent rock is predominantly composed of metamorphosed granodiorites of proterozoic age, while the local soils are medium-rich with modal Cambisol as the dominant soil type. The region is characterized by the occurrence of thermophilic deciduous forests dominated by oak.
From a management perspective, the investigated oak stands are over-aged coppices and coppices-with-standards located in an area where the traditional coppice-with-standards was converted in 1955 to high forests with long rotation. Sessile oak was the dominant tree species. Afterward, a reverse conversion to coppice-with-standards was initiated 20 years ago, with the goal of enhancing biodiversity and the economic value of harvested standards.
Three oak-dominated forest stands with similar site conditions were chosen, representing loamy and fresh beech-oak forests ([41]). The selected stands differed in the intensity of canopy closure: (A) closed-canopy stand; (B) medium-canopy stand; (C): open-canopy stand (Tab. 1). All the study stands have been converted to coppice-with-standards. The process of conversion is generally divided into three steps: (1) regeneration with the preservation of standards; (2) formation of a fully established coppice-with-standards; (3) management of already established coppice-with-standards ([38]). The time range of both first and second steps should equal to the rotation of previous coppice (approx. 40 years), while the third step has unlimited length. Stands B and C are currently in the half of the first step of conversion, while the stand A is at the very beginning (Tab. 1). From the beginning of the reverse conversion, sanitary thinnings (stands A-C) and the third-phase shelterwood cutting (stands B and C) were performed according to Utinek ([38]). All standards were pruned.
Tab. 1 - Characteristics of studied forest stands taken from the management plan. (QP): Sessile oak, Quercus petraea (Matt.) Liebl.; (CB): European hornbeam, Carpinus betulus L.; (TC): small-leaved lime, Tilia cordata Mill.; (PS): Scots pine, Pinus sylvestris L.; (LD): European larch, Larix decidua Mill.
| Stand | Area (ha) |
Age (years) |
Species composition | Standing volume (m3 ha-1) | Stand density (%) |
|---|---|---|---|---|---|
| A | 14.05 | 85 | QP 97%; CB 2%; TC 1% | 270 | 100 |
| B | 9.91 | 101 | QP 95%; PS 3%; LD 1%; CB 1% | 145 | 50 |
| C | 9.91 | 101 | QP 95%; PS 3%; LD 1%; CB 1% | 93 | 30 |
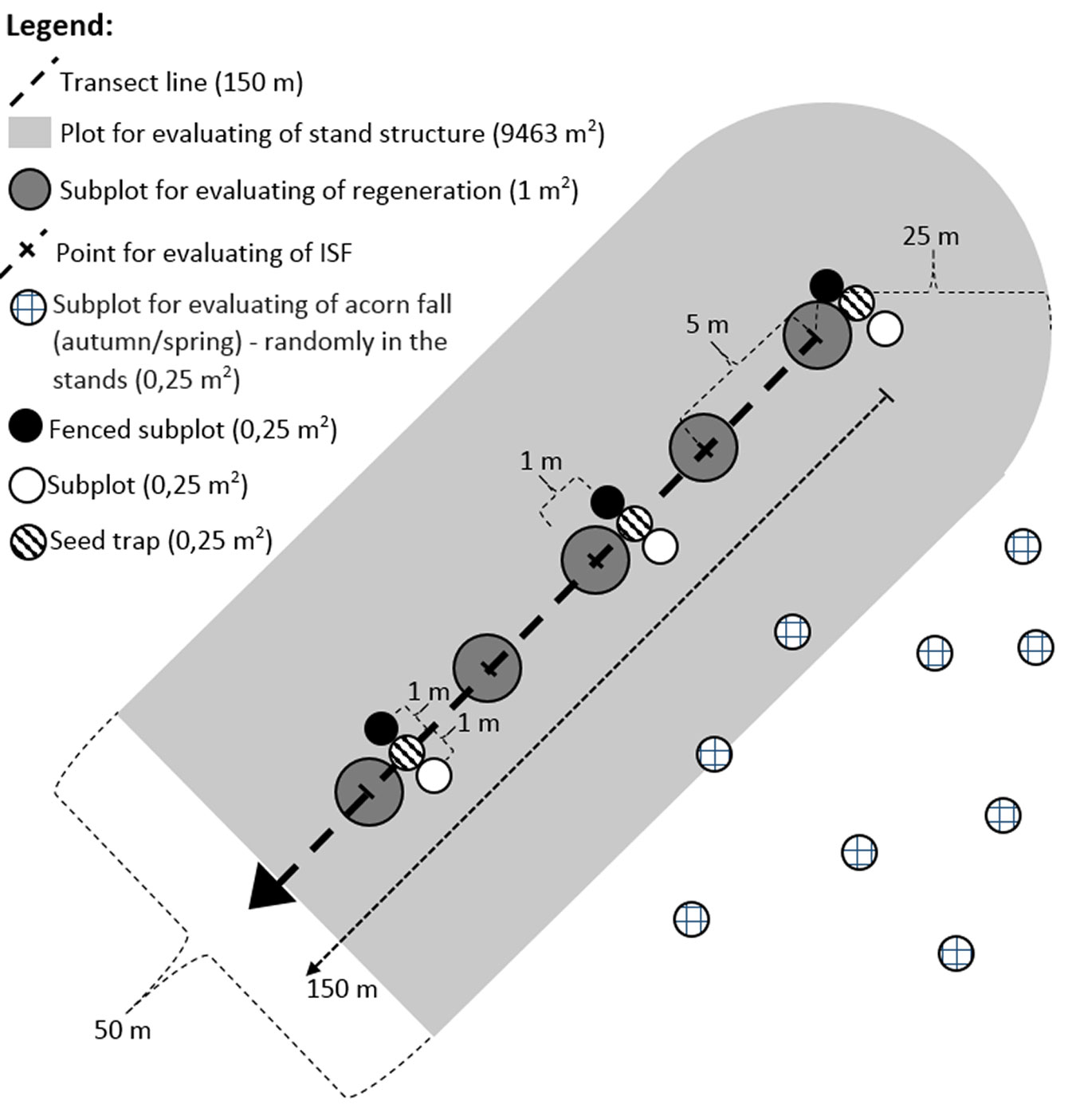
Using the Field-Map system, a linear transect of length 150 m was established in the central, homogeneous part of each stand, far from neighboring stands, roads, etc. Each transect was then divided into 5-meter sections using pegs, obtaining 31 sampling points per transect (overall, 93 sampling points). The following stand characteristics were evaluated within each transect (see below): (i) stand structure and light conditions; (ii) acorn production and consumption; (iii) inventory of a natural regeneration. The experimental design adopted is skecthed in Fig. 2.
Mammals which may potentially affect natural oak regeneration in the study area include wild boar (Sus scrofa) and roe deer (Capreolus capreolus). Small mammals encompass rodents: yellow-necked mouse (Apodemus flavicollis), wood mouse (Apodemus sylvaticus) and the bank vole (Myodes glareolus). The study area was situated in the vicinity of a game preserve surrounded by fences, which however did not hampered wild boars from moving inside and outside the preserve. Farmland did not provide any shelter or sufficient food supply for the wild boars during the experiment. Throughout the monitored period wild boar spent most of the daytime hidden in the thick brushwood and ventured out at night to forage in the open parts of the stands and in the fields surrounding the studied forest complex.
Stand structure and light conditions
All trees exceeding 50 mm diameter at breast height (1.3 m above the ground) were localized within 25 m at each side of the transect (including the endpoints), covering a total area 9463 m2 in each stand. We determined the species of each tree and measured its height and diameter at breast height. Hemispherical photographs were also taken (1.5 m above the ground, weather: overcast) at each sampling point along the transect (93 photographs in total) using a digital Nikon Coolpix 4500 camera with FC-E8 fisheye convertor (camera settings: Shooting-Mode: P; Programmed: Auto; Focus Mode: infinity; image format: TIFF). The indirect site factor ISF (i.e., the intensity of relative diffuse radiation) was evaluated using the WinsCanopy® 2008a software package (Regent Instruments Inc., Canada) using the following settings: automatic thresholding, no other corrections. The ISF values differed significantly in the individual stands A, B, C (F[2, 90] = 37.367, p < 0.001 - Tab. 2).
Tab. 2 - Altitude, stand composition, diameter at breast height (DBH), basal area and indirect site factor (ISF) of the transects. (QP): Quercus petraea (Matt.) Liebl.; (CB): Carpinus betulus L.; (TC): Tilia cordata Mill.; (PS): Pinus sylvestris L.; (SD): standard deviation.
| Stand | Altitude (m a.s.l.) |
Species composition (%) |
Trees (n ha-1) |
DBH (cm) mean ± SD |
Basal area (m2 ha-1) |
ISF (%) mean ± SD (median) |
|---|---|---|---|---|---|---|
| A | 340-355 | QP 100 (PS+) | 401 | 22.4 ± 7.5 | 19.1 | 12.1 ± 2.0 (11.7) |
| B | 345-365 | QP 100 | 243 | 26.2 ± 6.0 | 14.1 | 25.5 ± 10.9 (22.7) |
| C | 370-380 | QP 100 (CB, TC+) | 120 | 30.8 ± 10.1 | 9.3 | 35.3 ± 14.3 (31.8) |
Natural regeneration inventory
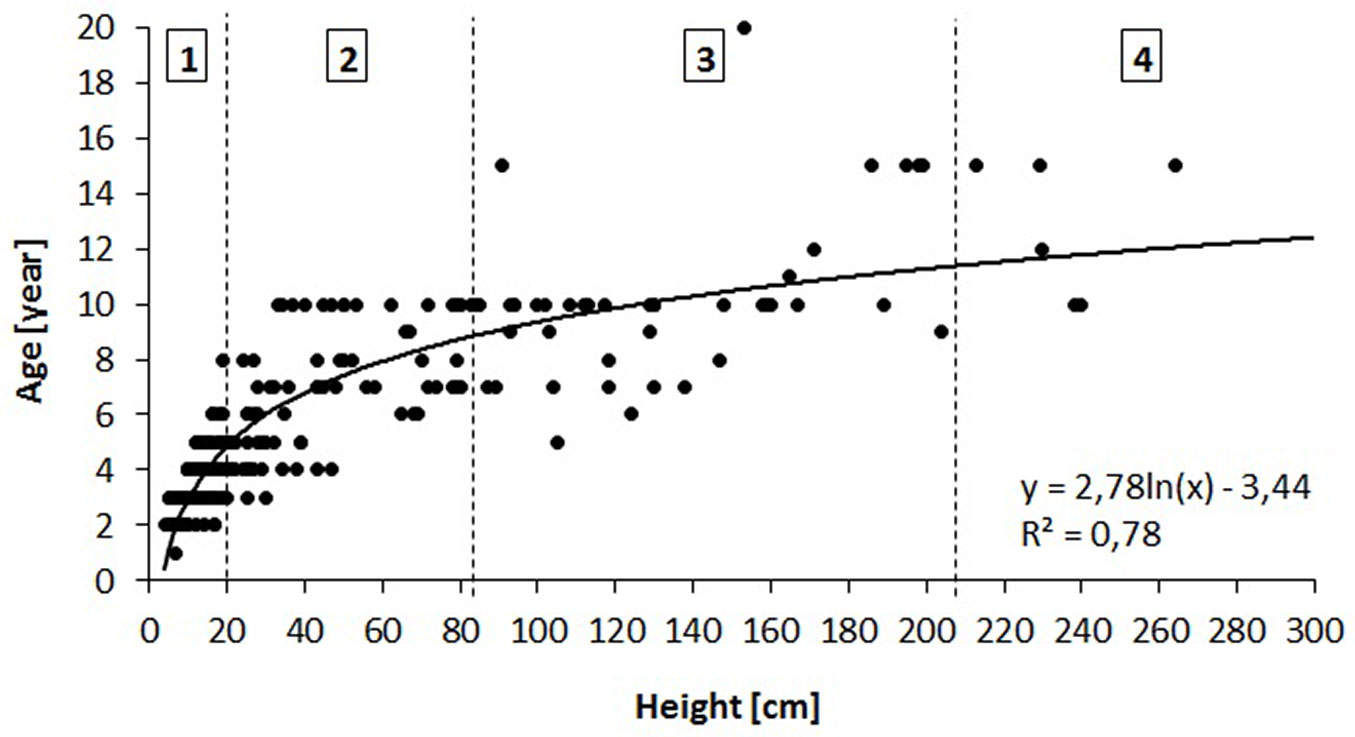
A circular plot of 1 m2 was established centered at each sampling point along the transects, and the species, density and height class of each sapling within the plot were determined. Sample oak saplings were also characterized by the height increments (cm) of the two preceding years (mean of both values), height (cm) and age (number of internodes). Based on the relationship between height and age (Fig. 3), we divided all saplings into four height/age classes (1-4), each characterized by a given height and age (Tab. 3, Fig. 3).
Fig. 3 - Relationship between height and age of oak saplings in the three studied stands and the derived height/age classes (1-4) for saplings.
Tab. 3 - Characteristics of the height/age classes (1-4) of oak saplings. The age of saplings in each height class (Expected Age) was derived from the relationship shown in Fig. 3.
| Height class (cm) |
Expected Age (year) |
Height/ age class |
|---|---|---|
| ≤ 20 | 1-5 | 1 |
| 20.1-50 | 6-9 | 2 |
| 50.1-80 | 6-9 | 2 |
| 80.1-130 | 10-11 | 3 |
| 130.1-200 | 10-11 | 3 |
| 200.1-300 | 12-13 | 4 |
| 300.1-400 | 12-13 | 4 |
Acorn production and consumption
To monitor seed fall and its consumption, three plots were placed in close proximity at each sampling point along the transect, each covering 0.25 m2 (15 sets in each stand, 45 in total). The first plot (seed trap) was used to determine the total seed fall, and consisted of a metal stand and a cloth-made seed trap which prevented animals from consuming the acorns. The second plot (fenced) was surrounded by a 1 m-high wire mesh with 10×10 cm openings which enabled unlimited access to small mammals, but prevented ungulates from consuming the acorns. The third plot (control) was only marked out and the acorns within it were freely accessible to animals. The consumption by small mammals was calculated as the difference between the total seed fall in the seed trap and the number of acorns left in the fenced plot. The consuption by ungulates was assessed based on the acorns left in the open plot. We checked the plots at one week intervals throughout the seed fall period (a total of 8 samplings from 10th September 2013 to 9th November 2013).
The intensity of acorn consumption during the seed fall determined from 15 sets of plots was used to estimate the total stand production. The counting of acorns was conducted after the seed fall (at the time of the last check of the sample plots) at 100 randomly selected plots covering 0.25 m2 in each stand, i.e., a total of 300 plots. The characteristics of trees in the vicinity of each plot were also taken. The determined quantity of acorns sampled during this one-off monitoring was adjusted to the real number of fallen acorns by including the percentage of acorns which had been consumed by all animals by the sampling date (data from subplots on the lines). The survey was repeated in the spring, when acorns began to germinate, and the number of acorns preserved at the plots until the beginning of the vegetative stage was counted.
Data analysis
To compare acorn production, acorn consumption and the density of oak saplings in the individual stands, one-way ANOVA (analysis of variance) and Tukey’s post-hoc HSD test were used. The same analysis was also applied for the comparison of indirect site factor (ISF) between the monitored stands. For stands B and C, two-sample t-test was used to compare the density of oak saplings in height/age classes 3 and 4. All data were tested for normality by Shapiro-Wilk’s test. The data were normally distributed (p > 0.05 in all cases). All analyses were performed at a significance level α = 0.05. Linear regression was used to estimate the relationships between measured characteristics of stand/light and oak saplings.
Results
Stand characteristics and natural regeneration
A total of 722 trees were surveyed in the studied stands. At the time of establishing the study areas, the monitored stands were almost exclusively composed of oaks, except 1 individual of hornbeam and 1 individual of lime (Tab. 2). Other tree species were found only in the shrub layer (hornbeam in dense stand).
Oak dominated the composition of natural regeneration in all studied stands (Tab. 4). While in the dense stand A (ISF 12.1% - Tab. 2) only saplings in the height/age class 1 or 2 were detected (age: 1-9 years; height: up to 80 cm), in the more opened stands B and C (ISF; 25.5% and 35.3%, respectively - Tab. 2) all four classes were present (age: 1-20 years; height: up to 400 cm - Tab. 4). The highest density of oak saplings was found in the stand A. The comparison of the average density of individuals in the height/age class 1 revealed statistically significant differences among the three stands (F[2, 90] = 26.177, p < 0.001). Contrastingly, the comparison of the average density of saplings in classes 2, 3 and 4 revealed no statistically significant differences among stands (p > 0.05).
Tab. 4 - Average density of the natural regeneration in the studied stands, in total and partitioned into height/age classes (1-4). Values in brackets are 95% confidence intervals. (QP): Quercus petraea (Matt.) Liebl.; (CB): Carpinus betulus L.
| Stand | Density - total (n ha-1) |
Species Composition (%) |
Density - total oak (n ha-1) |
Decrease of density oak % |
Oak Density (n ha-1) | |||
|---|---|---|---|---|---|---|---|---|
| Class 1 | Class 2 | Class 3 | Class 4 | |||||
| A | 310000.0 (101136.3) |
QP 100% | 310000.0 (101136.3) |
0 | 298333.3 (97688.3) |
11666.7 (8091.3) |
0.0 | 0.0 |
| B | 112758.6 (29880.2) |
QP 99%, CB 1% | 111379.3 (30118.4) |
64 | 86551.7 (31926.6) |
16206.9 (7360.0) |
8620.7 (6326.2) |
344.8 (706.4) |
| C | 66129.0 (29959.5) |
QP 100% | 66129.0 (29959.5) |
79 | 14516.1 (5902.3) |
27096.8 (14462.6) |
24516.1 (17612.6) |
1935.5 (1473.1) |
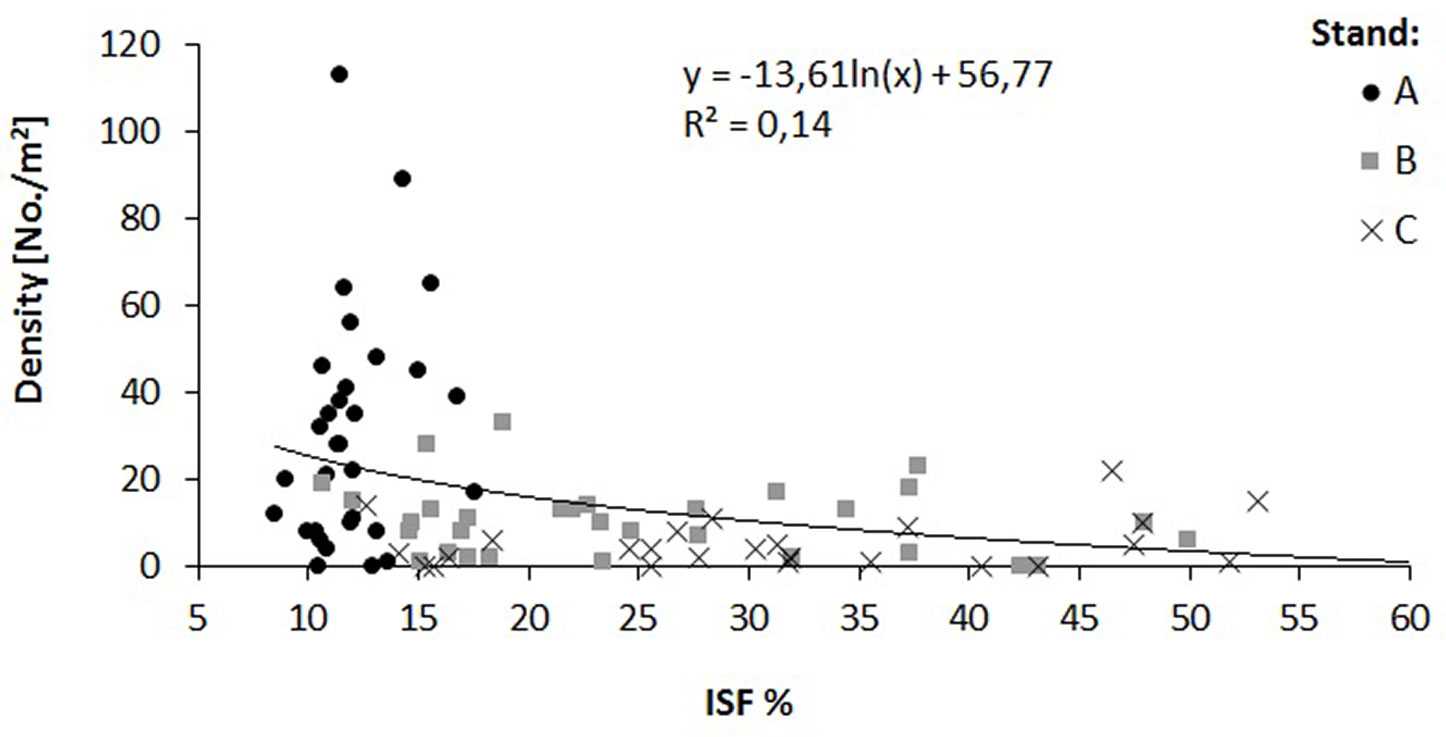
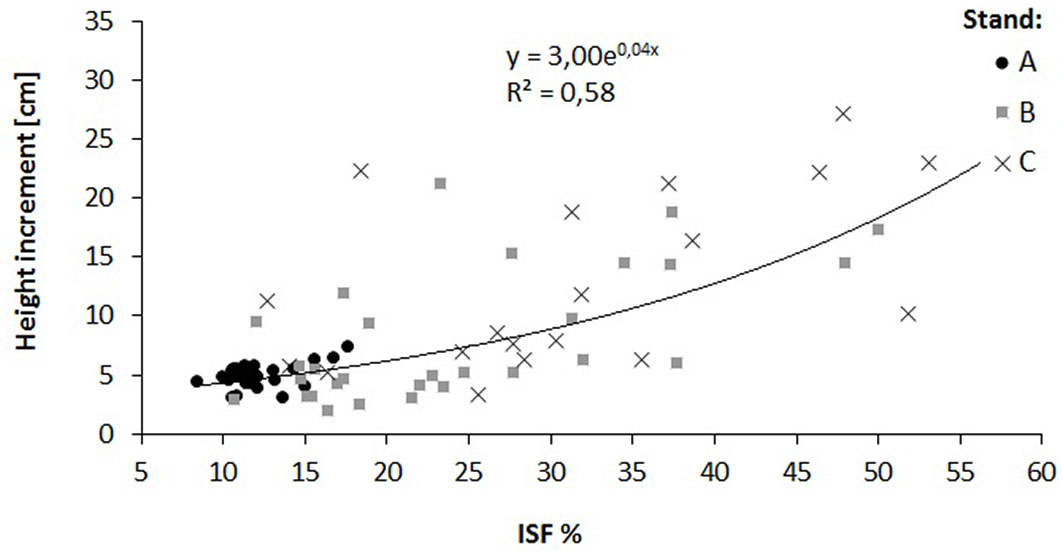
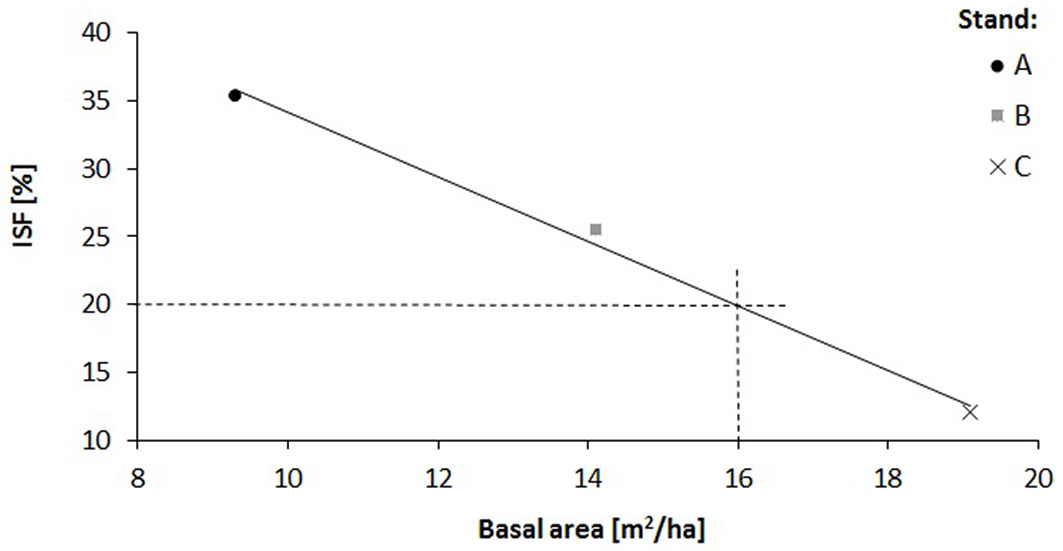
The relationship of the relative diffuse radiation with density (R2 = 0.14) and height increment (R2 = 0.58) of oak saplings was verified by a regression analysis (Fig. 4, Fig. 5). A decrease in sapling density was recorded when ISF values exceeded 15%, while a distinct increase in the height increment was recorded when ISF values exceeded 20%. In this context, a reduction of the stand basal area below 16 m2 ha-1 is necessary to enhance height increment of oak saplings (Fig. 6).
Fig. 4 - Relation between relative diffuse radiation (ISF) and oak regeneration density in the three investigated stands (A, B and C).
Fig. 5 - Relationship between relative diffuse radiation (ISF) and oak height increment in the three investigated stands (A, B and C).
Fig. 6 - Relationship between the stand basal area and the relative diffuse radiation (ISF) in the investigated oak stands (A. B. and C).
Acorn production
Taking into consideration the acorn consumption in individual stands, we assessed a production of 731 000 (stand A), 350 000 (stand B) and 108 000 (stand C) acorns per hectare, with significant differences among stands (F[51, 180] = 2.1712, p < 0.001 - Tab. 3). While the overall acorn production differed among stands as a consequence of the different number of trees, acorn production per tree was similar in all surveyed stands.
Acorn consumption
Acorns represented a sought-after source of food for animals in the study area. By the end of the seed fall, approximately half of the fallen acorns had been consumed, on average. The highest proportion of consumed acorns was observed in stand C (67%), where the lowest seed fall was also registered. As for stand A, 44% acorns were consumed, while the lowest acorn consumption intensity was recorded in stand C (only 13% - Tab. 5). The differences in the intensity of acorn predation among stands were not significant in all cases (F[1, 293] = 1.171, p = 0.28). The quantity of acorns in spring corresponded to their production and predation. The significantly smallest amount was found in the stand C (χ2 = 25.028, p < 0.001), where acorn production was the lowest and only 4600 acorns per ha remained in spring. Over 20 000 acorns were found in the remaining stands. Both large ungulates and small mammals participated in acorn consumption, albeit their proportional ratio differed significantly in individual monitored stands.
Tab. 5 - Acorn production in the monitored stands and intensity of acorn consumption by small mammals and ungulates during the seed fall.
| Stand | A | B | C |
|---|---|---|---|
| Production (thousands of acorns ha-1) | 731 | 350 | 108 |
| Consumption during the seed fall (%) | 44 | 13 | 67 |
| Small mammals (%) | 40 | 7 | 18 |
| Ungulates (%) | 4 | 6 | 49 |
| Remnants in spring (thousands of acorns ha-1) | 22 | 29 | 4.6 |
Discussion
Acorn production and predation
Acorn production and predation is the key factor influencing the process of oak natural regeneration. Our study demonstrated that acorn production in over-aged coppice stands (stand A: 73 seeds m-2; B: 35 seeds m-2; C: 11 seeds m-2) can be comparable to that of generative forests, where mast years with acorn production exceeding 50 seeds m-2 are rare ([21]). Acorn production is influenced by a number of biotic and abiotic factors ([30], [39]). Animal predation can strongly lower the density of acorns ([17], [11], [1]), as corroborated by the results of our study. Acorns are very attractive food preferred by many species (e.g. birds, especially wood-pigeons and jays, voles, rabbits, badgers and deer - [32], [17]).
Wild boar impact
Wild boar is the main ungulate species in our study area. The density of roe deer was low, as reported by local forest managers. Wild boar, which is considered the major acorn consumer in other parts of the world, is not present in Great Britain ([11]). However, wild boar populations are globally growing ([36], [23]) and expanding their range to new regions ([6]). This further aggravates the negative impact of the wild boar to forest stands, and represents a threat to the natural regeneration of forest tree species ([14]).
Consumption of acorns by large and small mammals differed according to the canopy openness. The dense stand A with a low shrub layer showed the highest impact of small mammals, while the stand C with a low density of mature trees and a rich undergrowth in the higher height/age classes (exceeding 50 cm) showed the highest impact by large animals. We assume that a shelter extends the duration of animal activities ([27], [37]). Therefore, the proportion of acorns consumed by ungulates in a safe place was significantly higher. The importance of a shelter also underlies the typical seasonal migration of wild boar between fields and forests ([5]). During the vegetation period, wild boar takes advantage of rape, cereal and maize cultures until their harvest in September or October ([33]). Subsequently, they move to forests where sufficient food sources and shelter are available ([10]). Indeed, animals with adequate food supplies do not migrate far from their resting places in search of uncertain sources ([22], [20]), and they move to more distant places only after the local food sources have been completely depleted ([37]). Therefore, their feeding behavior is conditioned by the distance from food sources, escape possibilities and cover opportunities ([35]). Such tendency of wild boar to linger within the forest in autumn and winter further increases acorn consumption and decreases the recruitment of the forest stands. In our study, 91-97% of the total acorn production was consumed from autumn to spring, similarly to previous studies ([11]). The acorn consumption by wild boar was the lowest in the stand A, devoid of cover possibilities. However, wild boar impact on the natural regeneration of trees is conditioned by other factors, such as the distance from water and other food sources, hunting activities in the stand or the length of the period spent in cropfields). Therefore, the extent of its impact on acorn survival cannot be generalized ([16]), and may differ from stand to stand.
Rodent impact
With regards to rodents, no increasing or decreasing trend with respect to the regeneration density or the stand characteristics was found in this study. However, acorn consumption was the highest in the stand A, characterized by a dense regeneration up to 20 cm. Rodents were responsible for a decrease of 292 000 acorns per hectare in the studied stand (40%). Nonetheless, the absence of acorns due to rodent activities can not be considered a direct consumption, because rodents build underground food storage ([42], [11]), which favor natural regeneration through seed dispersal ([18]). Experiments conducted by Wang et al. ([42]) showed that acorns disappeared very quickly after artificial exposure due to rodent activities (all acorns disappeared within 5-6 hours since exposure during the day, or within 10 hours during the night). The study also concluded that rodents moved more than 82% of acorns to underground food storages. Focardi et al. ([9]) notes that these supplies may become a source of food for wild boar in early spring, when acorns lying on the ground have already been consumed. Due to the scarcity of sufficient food source at this time, these rodent-amassed deposits of acorns may become an important factor affecting the wild boar population dynamics.
Other factors affecting oak regeneration
With respect to the estimated total regeneration density (66 000-310 000 saplings ha-1) the observed quantity of acorns in spring (4600-22 000 acorns ha-1) is more than sufficient for the purposes of forest management. The high density of natural regeneration found in this study was probably due to the accumulation of saplings from previous mast years. The density of oak regeneration decreased with increasing canopy openness, while both height and age variability of saplings increased. This is probably due to intraspecific competition (autoreduction) of oak saplings and to different growth dynamics at different age. Nevertheless, oak survived (in our case up to 9 years with a height up to 50- 80 cm) even under full canopy (about 12% ISF). Generally, oak is able to survive several years at 15% relative radiation and oak sapling decrease in stocking of 0.8-0.9 after 6-7 years. We even recorded the occasional occurrence of oak saplings under extremely low radiation (as low as 5% ISF), according to several studies which describe oak as a shade-tolerant species in its early years ([4]). However, based on our results oak needs at least 20% ISF for its sustainable growth. This finding is corroborated by other studies which consider relative radiation values of 30-60% as optimal for sustainable height growth ([30]).
We estimated also the impact of shoot browsing by roe deer. In the studied area the intensity of damaged shoots was less than 1% and had no effect on oak regeneration. Indeed, in this area characterized by mild winter and rich food supply, the roe deer prefer other food. Other biotic and abiotic factors, not only light and stand structure, influence oak saplings emergence and growth, e.g. interspecific competition, ground vegetation, drought, richness of nutrients, browsing, etc. ([30]). For example, at a rich site with a similar stand structure and light conditions, other species like hornbeam, field maple, ash, lime and shrubs were dominant in the natural regeneration. Consequently, in the young stands without targeted silvicultural measures the proportion of oak was less than 1 %. The remaining living oaks in the upper layer (about 55 indiv. ha-1) were distributed irregularly (cluster pattern) and their basal area reached about 5 m2. In our case, oak regeneration at the acidic site was abundant. However, to increase height growth of oak saplings a reduction of the basal area of adult oak stands under 16 m2 is recommended. For the proper mechanical stability and optimal diameter increment of the young oak stand, the density of oaks should be reduced to 10 000 indiv. ha-1 through the first juvenile thinning.
Conclusions
Our results proved that over-aged coppice stands are characterized by abundant fructification and successful generative regeneration under different light conditions and despite the strong animal impact. Indeed, more than half of acorns are consumed by the end of acorn fall and less than 10% of the total remain until spring. Nevertheless, a high stocks of acorns and saplings remained in the stands. Based on the number of different height/age classes of saplings observed in this study, the regeneration capacity of the studied stands was not threatened in the past years. Although the saplings may survive under unfavorable light conditions for several years, a basal areas of less than 16 m2 ha-1 of the parent stands is required to achieve a sustainable height growth. Therefore, generative natural oak regeneration in over-aged coppice stands can be relied upon in the conversion into a coppice-with-standards, which was carried out 20 years in the studied forest stands. The results obtained provide complementary information on the widely discussed issue of the optimal number of reserved trees and optimal basal area in a stand (in our case, max. 200 reserved trees ha-1 and 16 m2 ha-1, respectively) to secure a successful natural generative regeneration under the optimum relative diffuse radiation (min. 20% ISF).
Acknowledgements
This study was supported by the Ministry of Education, Youth and Sports of the Czech Republic, project No. CZ.1.07/2.3.00/ 20.0267 and by the Grant Agency of MENDELU, project No. 80/2013.
References
Gscholar
Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Jakub Drimaj
Radim Plhal
Jaroslav Zeman
Jan Hrbek
Department of Forest Protection and Wildlife Management, Faculty of Forestry and Wood Technology, Mendel University in Brno, Zemedelská 3, 613 00 Brno (Czech Republic)
Robert Knott
Antonín Martiník
Department of Silviculture, Faculty of Forestry and Wood Technology, Mendel University in Brno, Zemedelská 3, 613 00 Brno (Czech Republic)
Michal Kneifl
Zdenek Adamec
Department of Forest Management and Applied Geoinformatics, Faculty of Forestry and Wood Technology, Mendel University in Brno, Zemedelská 3, 613 00 Brno (Czech Republic)
Corresponding author
Paper Info
Citation
Kamler J, Dobrovolný L, Drimaj J, Kadavý J, Kneifl M, Adamec Z, Knott R, Martiník A, Plhal R, Zeman J, Hrbek J (2016). The impact of seed predation and browsing on natural sessile oak regeneration under different light conditions in an over-aged coppice stand. iForest 9: 569-576. - doi: 10.3832/ifor1835-009
Academic Editor
Tomas Vrska
Paper history
Received: Aug 31, 2015
Accepted: Feb 24, 2016
First online: Apr 04, 2016
Publication Date: Aug 09, 2016
Publication Time: 1.33 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2016
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 53251
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 43959
Abstract Page Views: 3864
PDF Downloads: 3938
Citation/Reference Downloads: 35
XML Downloads: 1455
Web Metrics
Days since publication: 3514
Overall contacts: 53251
Avg. contacts per week: 106.08
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2016): 24
Average cites per year: 2.40
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Dispersal and hoarding of sympatric forest seeds by rodents in a temperate forest from northern China
vol. 7, pp. 70-74 (online: 18 November 2013)
Review Papers
Problems and solutions to cork oak (Quercus suber L.) regeneration: a review
vol. 16, pp. 10-22 (online: 09 January 2023)
Technical Reports
Effects of different mechanical treatments on Quercus variabilis, Q. wutaishanica and Q. robur acorn germination
vol. 8, pp. 728-734 (online: 05 May 2015)
Research Articles
Optimum light transmittance for seed germination and early seedling recruitment of Pinus koraiensis: implications for natural regeneration
vol. 8, pp. 853-859 (online: 22 May 2015)
Research Articles
Shrub facilitation of Quercus ilex and Quercus pubescens regeneration in a wooded pasture in central Sardinia (Italy)
vol. 3, pp. 16-22 (online: 22 January 2010)
Research Articles
Understory vegetation dynamics and tree regeneration as affected by deer herbivory in temperate hardwood forests
vol. 10, pp. 837-844 (online: 26 October 2017)
Research Articles
Richness and abundance of granivorous vertebrates determine acorn removal patterns in a human modified oak forest
vol. 11, pp. 329-337 (online: 18 April 2018)
Research Articles
Germination and seedling growth of holm oak (Quercus ilex L.): effects of provenance, temperature, and radicle pruning
vol. 7, pp. 103-109 (online: 18 December 2013)
Research Articles
Soil CO2 efflux in uneven-aged and even-aged Norway spruce stands in southern Finland
vol. 11, pp. 705-712 (online: 06 November 2018)
Research Articles
What if Eurasian jay Garrulus glandarius would larder acorns instead of scatter them?
vol. 11, pp. 685-689 (online: 23 October 2018)