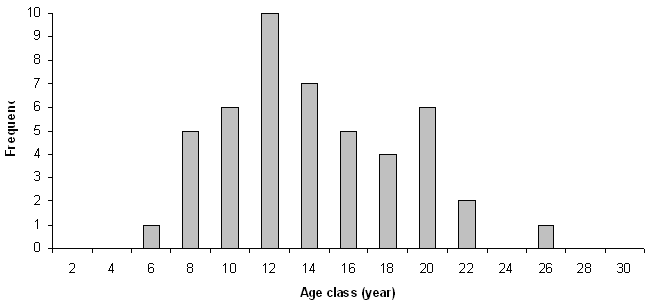
Shrub facilitation of Quercus ilex and Quercus pubescens regeneration in a wooded pasture in central Sardinia (Italy)
iForest - Biogeosciences and Forestry, Volume 3, Issue 1, Pages 16-22 (2010)
doi: https://doi.org/10.3832/ifor0517-003
Published: Jan 22, 2010 - Copyright © 2010 SISEF
Research Articles
Abstract
In the woodlands of Sardinia, as in many other areas of the Mediterranean region, grazing of domestic animals is still very common, though often in the absence of any sustainable management logic or technique. The present work analyzes the effects of excessive grazing pressure on a wooded pasture in the municipality of Orgosolo (Nuoro province), emphasizing the effects on the natural regeneration of the oak species (Quercus ilex and Quercus pubescens). This study has revealed the positive effects of the interaction between shrubs and seedlings of tree species. Crataegus monogyna proved to be the most efficient shrub species in favouring the establishment and growth of saplings; Rubus ulmifolius is efficient in the establishing phase but somewhat less so in the following stages. The other shrub species (Prunus spinosa, Genista pichi-sermolliana, Stachys glutinosa) play a lesser facilitating role. Despite the fact that seedlings are found in more than half (56%) of the shrub patches, the average age of the seedlings (14±0.6 yrs) and their small average size (height 24±1.4cm) lead to think that the present grazing pressure is incompatible with any concrete chance of success for their natural regeneration.
Keywords
Shrub facilitation, Tree regeneration, Wood pasture management, Overgrazing
Introduction
Pastures with consistent tree cover, composed mostly of evergreen oaks like Quercus ilex, Quercus rotundifolia and Q. suber, are quite widespread in Western Mediterranean countries. These landscapes assume a peculiar physiognomy and are called “dehesa” in Spain and “montado” in Portugal. The same stands are also present in Italy, especially on the island of Sardinia.
The features and functions of these formations are the result of specific climatic conditions, from somewhat mild summer droughts, combined with grazing and related human activities over the centuries to exploit natural resources ([43], [42], [38]). What we are looking at today must therefore be considered a peculiar agro-forestry system ([33], [34]), rather than the remains of a previously closed forest ecosystem.
Today, a general tendency for overgrazing with progressive symptoms of degradation can be observed ([47], [45], [44], [41], [25]). In particular, the open woods of the meso-Mediterranean belt dominated by the Holm oak, maintain their functionality only with low grazing pressure. High livestock densities produce negative effects, especially on the natural regeneration of the tree component ([21]), which is often confined under the protective shadow of shrubs which are not palatable to the animals. These patches then become potential safe sites for seedlings and saplings as recognized by many authors ([12], [11], [15], [49], [31], [35], [3], [52], [54]).
In the majority of the hilly and mountain areas of Sardinia, continuous stocking is the main grazing method ([16]). This is true also for the Pradu woodland, which is the subject of the present study. In recent decades, the pastoral activity in Mediterranean areas has been subjected to important modifications, with increasing livestock density and the shift from transhumance to more stable and intensive forms of breeding. The Pradu woodland has also experienced these transformations, together with a considerable increase in the number of animal species feeding at the same time (cattle and horses have joined the more traditional herds of sheep, goats and swine). As a result, the natural regeneration of the tree component has sharply decreased in density and spatial distribution, failing to overcome the progressive senescence of the existing stand under natural or human-induced stresses ([47], [24]). The conservation of the Pradu woodland is further compromised by the increasing, uncontrolled harvest of large branches, and even entire trees, for fuel ([1]).
This paper aims to quantify the consistence and potential of natural tree regeneration at the Pradu site, by analyzing in detail the characteristics of the shrub components, with respect to their ability to become nurse plants for tree saplings.
Material and methods
Study area
The study was conducted in mid-eastern Sardinia in the municipal territory of Orgosolo (Nuoro province) in a part of the Pradu woodland (N 40°10’; E 9°20’) with an area of about 280 ha at an altitude of about 1000m above sea level, with a mostly western exposure.
The climate, according to the bioclimatic classification of Rivas-Martínez & Rivas-Sáenz ([48]), is mid-Mediterranean with an oceanic footprint and an average yearly temperature and rainfall of 12 °C and 1100 mm respectively ([8]).
Paleozoic intrusive rocks dominate the landscape, with outcrops of granite and filladic schists ([39]). Dystric, Typic and Lithic Xerorthens ([55]) are the more common soil types over both intrusive and metamorphic substrates; they are usually shallow, coarse-textured, rich in rock fragments and with quite a low content of organic matter. Typic and Lithic Dystroxerepts have limited diffusion over more gentle slopes and in concavities. Despite the generally good soil permeability, recent gully phenomena may be observed locally.
Vegetation type refers to the climax of Sardinian mountain forests dominated by Holm oak and Downy oak stands ([2]). The tree component of the vegetation consists of a high forest with an unevenly aged structure (the diameter ranges from 40-160 cm) where Holm oak (Quercus ilex) is the dominant species (96%), and Downy oak (Quercus pubescens) (3%) and Montpellier maple (Acer monspessolanum - 1%) the associated species. The average density is 65 trees per hectare, but the spatial distribution is very irregular with scarcely covered (minimum of 11 plants ha-1) and densely covered (121 plants ha-1) areas. Historical record refer that until 150 years ago the forest physiognomy was like overaged coppice with standards of oak. At those time the forest was irregularly and occasionally logged and the grazing was transhumant and periodically suspended. Actually, the woodland is grazed by several domestic animals which, at various trophic levels, play an important role in favouring and shaping the floristic composition of the herbaceous vegetation. The authorizations for grazing was granted by municipality board to farmers of neighbourhood without any control on the number of livestock and grazing system. The entire study area is, in fact, subject to grazing year round; according to official data from the Orgosolo municipality, the average livestock pressure in 2003 was 1.7 LU (adult livestock unit) per hectare, of which more than 70% are sheep, 14% swine, and the rest divided among cattle, goats and equines ([17]). Shading, nitrification, and selection of species less palatable to animals have favoured the presence of thorny species (Genista pichi-sermolliana, G. corsica, Carlina corymbosa, Centaurea calcitrapa, Ononis spinosa, Ptilostemon casabonae) or toxic species (Euphorbia cupani, Paeonia morisii, Digitalis purpurea, Ranunculus spp.). Prominent among the ruderal and the nitrophilus species are Onopordon illyricum, Urtica atrovirens and U. pilulifera and the endemic Rumex suffocatus. The presence of Rubus ulmifolius is quite remarkable, because although browsed by sheep and especially goats, it partially characterises an anomalous undergrowth, together with dogrose (Rosa canina) which rises in big tufts. Other woody species are wild pear (Pyrus spinosa) and hawthorn (Crataegus monogyna), found sporadically throughout the area. Exploitation, practiced regularly for centuries, as testified by the important archaeological remains of the nuraghic era, has selected a number of species of scarce foraging and grazing interest. The species of the genus Aira, Vulpia, Bromus are the most common among the gramineae, whereas among the compositae the genus Crepis, Reichardia and Carlina are those which give a significant contribution to the grass turf. Among the leguminosae, in addition to the above-mentioned Ononis spinosa, two species are of remarkable pabular interest: Lotus alpinus, exclusively in the high mountain areas of the Gennargentu and the Marghine-Goceano region of Sardinia, but most of all Trifolium subterraneum, which protects itself from overgrazing thanks to its self-reseeding character. Present among the labiatae are thyme (Thymus catharinae) and helichrysum (Helichrysum microphyllum), small fragrant suffrutexes usually associated with gorses. Asphodel (Asphodelus microcarpus) is sporadically browsed in its vegetative stage, but completely rejected during flowering; this facilitates its diffusion, which is also helped by its root system, very rich in nourishing supplies. Similar to asphodel, geophytes provided with tubers, bulbs and rhizomes have an advantage, particularly those of the genus Gagea, Scilla, Ornithogalum, and Leucojum, which also produce showy and characteristic flowerings.
The whole area is crossed by paths open to lorry traffic which favours channelled erosion, as they interrupt the already poor grass turf, compact the soil and constitute preferential path for run off concentration. This process has considerably increased in recent years, adding to the actions of the swine, which are present in far greater number with respect to the sustainability of the area, and dig looking for tubers, bulbs and larvae, thus worsening the deterioration of the entire area.
The flora of the Pradu woodland, despite being poor in species suitable for grazing, has an endemic component common to the other areas of the Gennargentu which increases its environmental value.
Plot sampling
The research area was delimited and georeferenced using regional orthophotographs (1998) on a scale of 1:10 000 in a GIS environment. The total area was divided into a squared 100 m wide grid following the Gauss-Boaga geographic projection; the area of each square was further divided into 25 smaller 20 m wide squares (400 m2), also referred to as the reticule. Next were the stratification of the big squares according to the degree of canopy cover in the GIS environment (3 classes: A <30 %, 31< B <60 % and C >60%), and the random selection of 45 small squares (specimen areas - SA) in proportion to the frequency observed in each cover class.
Field observations
For each SA the general characteristics such as slope, exposure, and degree of vegetation cover were recorded. The crown height (H), as well as the bigger measurement (L1) and the one perpendicular to it (L2), were taken for each shrub present.
Every seedling of tree species (height < 5 m) was surveyed distinguishing between those growing in the open and those growing among shrubs, and its height (h) was also measured; in the presence of groups of seedlings, the total number (ns) and the height of the tallest saplings were taken.
Finally, in a random sample of shrubs with seedlings, 50 of these were taken, and their height (h), and the collar diameter (d) were measured. Seedling age was estimated by counting the annual rings using a stereomicroscope.
Data processing
The area occupied by the crown of each shrub (S) was estimated through the ellipse formula (eqn. 1):
As a whole and for each shrub species, we determined the number of shrubs per hectare (N ha-1), the average occupied area and the average height. For the seedling sample we also calculated the average yearly increase in height (Ih) and diameter (Id).
All data were elaborated using common tests of inferential statistics:
- χ2 test to assess the differences between the shrub species for the presence or absence of seedlings within (in cases of just 1 degree of freedom Yates’s correction was utilized);
- two-way ANOVA (factorial scheme with interaction) for the variables S, H, h (variation sources: shrub species; presence or absence of seedlings);
- one-way ANOVA for the variables S (variation source: tree cover degrees) and h (variation source: shrub species);
- Duncan’s test in all cases where P <0.05;
- analysis of the regression of h, d, Ih, Id versus the age of the seedlings;
- linear correlation between S, H and h;
- Spearman’s correlation between S, H and the number of seedlings (ns).
Results
Shrubs
An overall census of 502 shrubs with an average density of 279 (s.e. ± 73.5) per hectare resulted in a field survey of the 45 sample plots; 8 of them (18%) were lacking in both tree seedlings and shrubs. Of these, 6 are concentrated in the upper part of the research area, near some enclosures used as temporary shelters for livestock, and 2 are located in the lower part, in an area periodically subject to waterlogging and presumably unsuitable for woody plants.
In the remaining 37 specimen areas the following shrub species were registered (Tab. 1): Stachys glutinosa (S.g.), Crataegus monogyna (C.m.); Genista pichi-sermolliana (G.s.); Prunus spinosa (P.s.); Rubus ulmifolius (R.u.). The most abundant species are G.s. (56%) and R.u. (29%) followed by C.m. (7%), S.g. (5%) and P.s. (3%). G.s. is exclusive to the cover class A, where the average density of shrubs is higher (443 plants ha-1; s.e. ± 3.8). This density generally decreases with the increase of the degree of tree cover.
Tab. 1 - Mean density of shrubs (N ha-1 ± standard error) for the 45 sample squares.
| Species | Tree canopy cover classes | Overall | ||
|---|---|---|---|---|
| <30% | 30-60% | >60% | ||
| Stachys glutinosa | 81 (±0.7) | 100 (±15.8) | 75 (±0.5) | 100 (±14.1) |
| Crataegus monogyna | 56 (±0.2) | 25 (±0.5) | 50 (±2.4) | 47 (±4.1) |
| Genista pichi-sermolliana | 996 (±387.2) | - | - | - |
| Prunus spinosa | 46 (±0.2) | 38 (±2.6) | 25 (±0.5) | 42 (±4.2) |
| Rubus ulmufolius | 152 (±1.8) | 175 (±19.3) | 68 (±12.1) | 135 (±34.6) |
The average area of the shrub crown, with the exclusion of G.s. which has an area of 2.6 m2, is not significantly differentiated in the various classes of tree cover (F2;218= 1.48) and is on average 1.3 m2 (s.e. ± 0.12).
Saplings
The total number of registered seedlings comes to 1079, with an average density of 599 (s.e. ± 122) per ha; Holm oak is the most abundant species (92%) with 553 (s.e.±117) seedlings per hectare, whereas Downy oak (8%) is much less present (46 ± 18 seedlings per hectare).
The average age of the seedlings is 14 years (s.e. ± 0.6) ranging from 6 to 25 years; the subdivision of the sampled individuals into age classes (Fig. 1) reveals a normal distribution with a slight asymmetry towards the right side.
The height of the seedlings (48 cm; s.e.± 18) is not significantly correlated to their age (r=0.2); on the contrary d (1 cm; s.e. ± 0.4) is positively correlated to this feature (r = 0.7, p < 0.01). The mean Ih is 3.9 cm/year (s.e. ± 0.4) and tends to decrease considerably with age (r = -0.68, p < 0.01), the average Id is 0.7 cm/year (s.e. ± 0.04), with no significant correlation to age.
Interaction between shrubs and saplings (shrub facilitation)
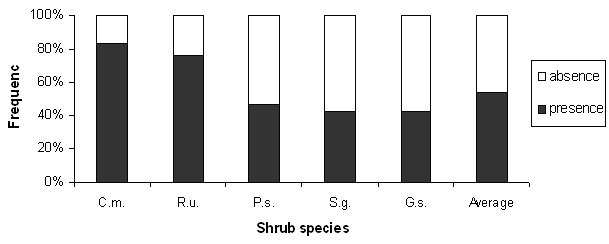
Fifty-five percent of the shrubs were shown to host saplings (Fig. 2, Tab. 2) with significant differences among species (χ2[4] = 59.1, p<0.01).
Tab. 2 - Comparison among sapling densities inside the different shrub species (n.s.= not significant).
| Comparisons | d.f. | χ2 | P |
|---|---|---|---|
| 1. All species | 4 | 59.1 | <0.01 |
| 2. C.m. vs. R.u. | 1 | 1.1 | n.s. |
| 3. P.s. vs. G.s. vs. S.g. | 2 | 0.13 | n.s. |
| 4. 2 vs. 3 | 1 | 58.4 | <0.01 |
C.m. is the species with the highest frequency of patches containing saplings (83%) with values comparable to those of R.u. (76 %; χ2[1] = 1.1); all in all, the other kinds of shrubs have significantly lower values with respect to the previous ones (χ2[1] = 58.4, p < 0.01), with comparable frequencies (χ2[2] = 0.13) though consistently below 45%.
The ANOVA test (Tab. 3) has highlighted significant differences with respect to the average area (S), both among the examined species and regarding the presence or absence of saplings inside.
Tab. 3 - ANOVA test, variable S, variation sources are shrub species (1), the presence or absence of seedlings (2) and the interaction 1x2 (n.s.= not significant).
| Effect | d.f. | F | P |
|---|---|---|---|
| 1. Shrub species | 4 | 17.35 | <0.001 |
| 2. Sapling occurrence | 1 | 7.15 | <0.01 |
| 3. Interaction 1x2 | 4 | 1.55 | n.s. |
| Error | 492 | - | - |
On the basis of the results of the Duncan test, G.s. has an average area (2.6 m2) considerably larger than S.g. (0.2 m2) and P.s. (0.8 m2), whereas R.u. and C.m. have intermediate values (1.4 and 1.9 m2 respectively); moreover, the shrubs giving shelter to saplings have a larger average area than the others (2.5 and 1.5 m2 respectively).
The ANOVA test (Tab. 4) showed significant differences with respect to the average H among species.
Tab. 4 - ANOVA test, variable H, variation sources are shrub species (1), the presence or absence of seedlings (2) and the interaction 1x2 (n.s.= not significant).
| Effect | d.f. | F | P |
|---|---|---|---|
| 1. Shrub species | 4 | 15.8 | <0.001 |
| 2. Sapling occurrence | 1 | 1.45 | n.s. |
| 3. Interaction 1x2 | 4 | 0.13 | n.s. |
| Error | 492 | - | - |
Based on the results of the Duncan test, the average height of C.m. shrubs (1.34 m) is higher than that of all other shrubs, those of P.s. and S.g. are the lowest (0.41 and 0.29 m respectively), whereas R.u. and G.s. have intermediate heights (0.82 and 0.83 m respectively).
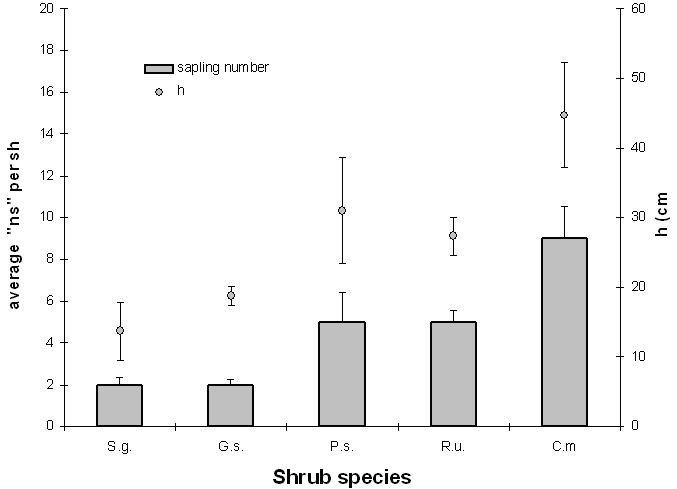
Considering all shrubs with saplings inside, the average ns (Fig. 3) varies considerably among the species (F4;270=11.67, p<0.01); from the Duncan test it appears that C.m. has a higher average ns (9), whereas G.s. and S.g. have the lowest (2), and P.s. and R.u. have intermediate values (5).
Fig. 3 - Means of sapling number (ns) and shrub height (h) by species (vertical bars indicate 1 standard error).
The average height of the saplings (24 ± 1.4 cm) varies quite considerably among the shrub species (F4;270= 5.31, p<0.01). In particular, the Duncan test shows that the average h is higher for C.m. (44.8 cm), lower for S.g. and G.s. (13.7 and 18.8 cm respectively), whereas R.u. and P.s. have intermediate values (27.3 and 31 cm).
The number of saplings per shrubs is positively correlated (Tab. 5) to the S and H of the shrubs (r = 0.23 and 0.32 respectively, p < 0.05), but the correlation is very high and significant for R.u. (r = 0.46 and 0.48 respectively, p < 0.01) and for C.m. (r= 0.81 and 0.66 respectively, p < 0.01).
Tab. 5 - Values of the Spearman correlation between H, S and ns. (*) = P < 0.05; (**) = P < 0.01).
| Species | H | S | N. of cases |
|---|---|---|---|
| S.g. | 0.40 | -0.12 | 10 |
| P.s. | -0.04 | 0.15 | 29 |
| G.s. | 0.06 | 0.19 | 119 |
| Ru. | 0.48** | 0.46** | 7 |
| C.m. | 0.66** | 0.81** | 110 |
| Total | 0.32* | 0.23* | 275 |
The height of the saplings is positively correlated to S and most of all to H (r = 0.28 and 0.6 respectively, p < 0.01). The highest correlation values are those of C.m. and R.u. (Tab. 6).
Tab. 6 - Values of the linear correlation between h, H and S.(*) = P < 0.05; (**) = P < 0.01).
| Species | h-H | h-S | N. of cases |
|---|---|---|---|
| S.g. | 0.66* | 0.47 | 10 |
| P.s. | 0.72 | 0.04 | 29 |
| G.s. | 0.33* | 0.35** | 119 |
| R.u. | 0.57** | 0.58** | 7 |
| C.m. | 0.81** | 0.68** | 110 |
| Total | 0.60** | 0.28** | 275 |
Discussion
Shrubs
At present the shrub cover consists of five main species and represents a minor part (12%) of the total ground cover, compared to grass (31%) and bare soil (57%). Over half of the registered shrubs are Genista (56%); they form very dense patches located in areas with large granitic outcrops, thus indicating a preference for relatively scant pedological conditions (more dryness). Rubus ulmifolius is the second most common shrub species (29%), followed by Crataegus monogyna (7%) Prunus spinosa and Stachys glutinosa (3% and 5% respectively), both were distributed quite homogeneously over the examined area. All these shrub species have thorns basically as a result of the selecting action of animal grazing ([57], [58]). It is a known fact that in environments with a Mediterranean climate, excessive grazing pressure initially causes a simplification of biodiversity with the disappearance of the most palatable species and/or the substitution of perennial herbaceous species. The last regression stage is the complete disappearance of vegetation and the beginning of erosive phenomena ([42]).
The density and composition of the shrub cover in the Pradu woodland are clearly connected with tree cover: where the latter is higher, the shrub cover is minimal. This condition is largely ascribable to the ecological needs of the shrub species, all of which are basically heliophilus; the action of animal grazing, however, also influences their distribution, which tends to thicken in the less browsed areas and in the presence of large rock outcrops.
Seedlings
The specific composition of regeneration is directly related to the species proportion of the tree layer: in fact, Holm oak seedlings are the most common (80%) and are occasionally associated with those of the Downy oak (17%) which is sometimes the only species present inside the shrubs. Under no circumstances has the regeneration of Montpellier maple been observed.
The light microclimate inside small shrub patches is known as a potential selecting factor for the saplings of tree species ([26], [36]). In our case, the higher shade tolerance of Holm oak compared to Downy oak and Montpellier maple could facilitate its survival inside shrubs. Moreover, the morphology of seedling leaves should also add competitive advantages to Holm oak: under conditions of repeated/continuous grazing the leaf tends to become more coriaceous and thorny, expressing a substantial adaptability to grazing ([6]).
The age of the saplings is undoubtedly a very important factor within the Pradu woodland. As in other studies carried out in the Mediterranean region ([15], [47]), our observations have demonstrated that a general, uneven age distribution exists; a shortage of young and very young subjects is reported, however. More specifically, the sampled individuals are rather old and their age class distribution shows some peaks that might be the consequence of years of larger seed production. In such years, in fact, the population of predators leaves a higher number of seeds available for germination ([56], [22], Murphy & Janzen in [5]). The absence of seedlings younger than 6 years of age could be symptomatic of a tendency towards a worsening of the seed quality produced by the plants: according to Cierjacks & Hensen ([21]), increased grazing pressure results in a higher percentage of aborted Holm oak acorns. However, worsening climatic conditions (dryness) should not be ruled out.
According to Smit et al. ([54]) the facilitating action exerted by shrubs on tree regeneration decreases as biotic stress increases: intensive grazing causes severe damage to shrubs, thus leading to heavier grazing and higher seedling mortality. The yearly average height growth of the seedlings (3.9 cm) decreases considerably with the increase in age (r = -0.7, p < 0.01) as a result of livestock browsing; nonetheless, the diameter growth remains constant (0.7 cm/year), thus confirming that oak species tolerate grazing well (Shaw 1974 and Hilton et al. 1987, in [40], [59]).
Shrubs facilitation
In the Pradu woodland, shrub patches represent the only habitats where processes of natural regeneration of the tree components have been observed: no seedlings have been found in the open. This result confirms that facilitation is especially important in cases of heavy grazing pressure ([7]). However, the fact that no regeneration has been observed in many shrubs could indicate that intensive grazing causes severe damage even to shrubs and reduces their defensive action ([54]).
The interaction between shrubs and tree regeneration occurs in different ways during the various stages of the regeneration process ([26], [53]). Due to their morphological characteristics and to the richness of nourishing supplies, the seeds of oak species are more likely to be consumed ([51], [4]). The same authors highlight the role played by rodents and birds in the dispersal of these seeds: among them, the jay (Garrulus glandarius) appears abundant in the study area (Shenk & Torre in [16]). The dispersal of seeds from the parent plants and their establishment in small repositories thanks to these “secondary dispersal agents” become more important when the number of safe sites is small ([46]). It has been widely confirmed that shrub patches are very important sites both in the dispersal phase ([9], [23], [26], [15], [49]) and in the establishment phase, since seeds and seedlings are protected from predation and trampling ([32], [49], [35], [52], [54]). This role remains consistent even if seed consumption by insects, rodents and birds reaches significant levels ([37], [53], [59]). Moreover, the shrub patches create the most favourable micro environmental conditions for the germination of seedlings ([29], [30]): less soil compaction, higher humidity, less radiation, greater availability of nutrients, and less competition with herbaceous species ([11], [26], [49], [20], [3], [28], [36]).
As observed in research conducted in Holland ([3]), the size of the shrubs has an influence on the seedlings’ chance of survival. Our results indicate that at this stage the height of the shrubs does not affect the establishment of seedlings. On the contrary, the crown area is a decisive factor; the wider the shrubs, the greater the difficulty for animals (especially swine) to feed on acorns and browse seedlings. At Pradu the minimum effective area has proven to be somewhat larger than 2 m2; there are, however, species-related differences, with the cover of Crataegus monogyna and Rubus ulmifolius representing the safest site for the establishment of seedlings. A minor portion of Genista pichi-sermolliana shrubs has seedlings inside, but the seedling density is nevertheless low. This may be due to the scarce massiveness of the crown of this species, with a greater accessibility to animals compared to previous ones; however, it must also be considered that Genista patches concentrate on rock outcrops, which have more adverse edaphic conditions for seed development and are generally located away from the most vigorous seed-bearer trees.
Prunus spinosa and Stachys glutinosa are not very common and generally form small-sized shrubs less effective in facilitating the establishment of seedlings.
In the consolidation stage the survival and growth of the seedlings are mainly influenced by animal browsing ([40], [12], [11], [27], [10], [49], [35], [3], [19], [54]). Competition between shrubs and saplings for light, water and nutrients is also important at this stage, and, according to some authors, results in reduced growth of tree species ([10], [50]).
The average height of saplings at Pradu is considerably greater in shrubs of Crataegus monogyna, Prunus spinosa and Rubus ulmifolius compared to the other species; nevertheless no sampling has reached such a size as to definitely escape browsing action by livestock (roughly estimated within 2 m).
Seedlings have shown the tendency to parallel the height growth of the hosting shrubs; this correlation is particularly marked in the case of Crataegus monogyna. This phenomenon is probably due to the morphological characteristics of hawthorn which, in optimum conditions and unlike Prunus spinosa, is able to grow in height at the same rate of the saplings. Shrubs of Rubus ulmifolius tend to grow enveloping seedlings and follow them in their growth; since young shoots of this plant are susceptible to grazing, the height growth is, nonetheless, conditioned. Studies conducted in California ([59]) have also shown that shrubs of Rubus discolor facilitate the establishment of seedlings of oak species; however, they interfere with their subsequent development by confining their growth.
At Pradu, the seedlings’ growth inside Genista pichi-sermolliana patches, though proportional to the shrubs’ size, is less marked: this could be due both to the poor conditions of the site where these shrubs grow and to the poor ability of this species to protect itself from grazing. Clefts between rocks occasionally proved to be safe sites for the establishment and survival of seedlings of tree species; this phenomenon has already been described for Picea abies seedlings in the timbered grasslands of Switzerland ([52]). Still our observations indicate that saplings remain confined inside the rock clefts with no chance for further growth, both because of punctual animal browsing as soon as the seedlings emerge, and because of the poor soil condition of such microenvironments.
Conclusions
The effectiveness of the interaction between shrubs and seedlings with respect to tree species regeneration in wooded pastures has been demonstrated at the Pradu site as well. In particular, shrub facilitation has proved to be species-specific ([13], [14], [28]). Crataegus monogyna is the most efficient species in facilitating the establishment and development of tree species seedlings; Rubusulmifolius is efficient in the establishment phase, but somewhat less so in the following stages. The other shrubs species have a weaker facilitating role.
Despite the fact that the density of shrubs hosting Holm oak or Downy oak seedlings is quite considerable, the high average age and the small size of the seedlings lead us to think that the present grazing pressure is incompatible with any concrete chance of consolidation for these saplings, and thus for the natural regeneration of the tree component of this peculiar ecosystem. At present, most of the shrubs and the hosted saplings have not attained a height greater than the browsing line of cattle and equines; this means that their future is still dubious.
At the same time, the local social and economic situation does not allow us to advance any hypothesis about even a temporary suspension of grazing activities; as a result, the survival of these ecosystems seems unlikely, and there are important economic, hydrologic and aesthetic implications. A great cultural heritage, based on the local population’s management of natural resources over the centuries, risks extinction as well. The conservation of these stands must therefore rely on the rationalisation of human activities through the exploitation of a whole range of resources (not least tourist appeal). This approach appears to be the only way to face this threat and effectively solve the problem of tree regeneration at Pradu.
The adjustment of the grazing pressure to sustainable levels is certainly a critical measure. According to recent studies conducted in Q. ithaburensis forests in Israel ([25]), a cattle density of 0.7 head per hectare is already able to cause considerable damage to regeneration. It is obvious that an agreement must be reached with local breeders, perhaps by compensating the reduced yield with a better profitability of dairy and meat products. Another possibility could be the introduction of systems that provide for the integration of natural regeneration by means of planting seedlings either inside natural shrub patches, as positively experimented in Spain for Q. pirenaica ([18], [19]), or inside special enclosures.
Acknowledgements
This work was conducted with the cooperation of the Orgosolo municipality (Nuoro province). The authors would like to thank Prof. Ignazio Camarda of the University of Sassari, for his assistance with the floristic classification. Special thanks also go to Dr. Tonina Muggianu and Dr. Nicola Sanna for their contribution to field surveys. The experiments comply with the current laws of the country in which they were performed.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
University of Sassari, v. Sa Terra Mala, I-08100 Nuoro (Italy)
G Calamini
S Sioni
Department of Environmental Forestry Science and Technology, University of Florence, v. San Bonaventura 16, I-50145 Florence (Italy)
CRA - ABP Florence, p.zza M. D’Azeglio 30, I-50121 Florence (Italy)
Corresponding author
Paper Info
Citation
Alias S, Bianchi L, Calamini G, Gregori E, Sioni S (2010). Shrub facilitation of Quercus ilex and Quercus pubescens regeneration in a wooded pasture in central Sardinia (Italy). iForest 3: 16-22. - doi: 10.3832/ifor0517-003
Academic Editor
Renzo Motta
Paper history
Received: Mar 02, 2009
Accepted: Sep 24, 2009
First online: Jan 22, 2010
Publication Date: Jan 22, 2010
Publication Time: 4.00 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2010
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 51359
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 44248
Abstract Page Views: 2530
PDF Downloads: 3814
Citation/Reference Downloads: 26
XML Downloads: 741
Web Metrics
Days since publication: 5207
Overall contacts: 51359
Avg. contacts per week: 69.04
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Feb 2023)
Total number of cites (since 2010): 7
Average cites per year: 0.50
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Germination and seedling growth of holm oak (Quercus ilex L.): effects of provenance, temperature, and radicle pruning
vol. 7, pp. 103-109 (online: 18 December 2013)
Research Articles
SimHyb: a simulation software for the study of the evolution of hybridizing populations. Application to Quercus ilex and Q. suber suggests hybridization could be underestimated
vol. 11, pp. 99-103 (online: 31 January 2018)
Review Papers
Problems and solutions to cork oak (Quercus suber L.) regeneration: a review
vol. 16, pp. 10-22 (online: 09 January 2023)
Research Articles
Earlywood vessel features in Quercus faginea: relationship between ring width and wood density at two sites in Portugal
vol. 8, pp. 866-873 (online: 27 April 2015)
Research Articles
Behavior of pubescent oak (Quercus pubescens Willd.) wood to different thermal treatments
vol. 8, pp. 748-755 (online: 16 February 2015)
Research Articles
Regeneration of Abies pinsapo within gaps created by Heterobasidion annosum-induced tree mortality in southern Spain
vol. 7, pp. 209-215 (online: 27 February 2014)
Review Papers
Should the silviculture of Aleppo pine (Pinus halepensis Mill.) stands in northern Africa be oriented towards wood or seed and cone production? Diagnosis and current potentiality
vol. 12, pp. 297-305 (online: 27 May 2019)
Research Articles
Tree-oriented silviculture: a new approach for coppice stands
vol. 9, pp. 791-800 (online: 04 August 2016)
Research Articles
A model of shrub biomass accumulation as a tool to support management of Portuguese forests
vol. 8, pp. 114-125 (online: 27 July 2014)
Research Articles
Natural regeneration and species diversification after seed-tree method cutting in a maritime pine reforestation
vol. 15, pp. 500-508 (online: 14 December 2022)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword