Effects of gap size and within-gap position on seedlings establishment in silver fir stands
iForest - Biogeosciences and Forestry, Volume 1, Issue 1, Pages 55-59 (2008)
doi: https://doi.org/10.3832/ifor0448-0010055
Published: Feb 28, 2008 - Copyright © 2008 SISEF
Research Articles
Abstract
The conversion of pure conifer plantations into stands with heterogeneous structure and composition is a current silvicultural challenge in Italy. A silvicultural system aiming to the regeneration of natural forests by means of openings in the canopy cover is considered. The purposes of this study are: I) to evaluate if the creation of gaps is an appropriate way to transform homogeneous man-made stands of silver fir (Abies alba Mill.) into naturally regenerated stands; II) to analyse the influence of micro-environmental conditions in gaps of different size on the recruitment and establishment of natural regeneration of forest tree species. After three growing seasons silver fir seedlings recruitment was greater in small gaps and in the central-southern positions where PAR was lower and the shading influence of ground vegetation less relevant. The most suitable gap size for silver fir regeneration, in the southern Apennine, is 200 (300) m2, D/H = 0.5 and anyway <0.75. The silvicultural treatment proposed seems to foster natural regeneration of silver fir and other broadleaves, enhancing the development of a multi-layered forest structure and fulfilling timber production objectives without compromising ecosystem management principles.
Keywords
Forest gaps, light, natural regeneration, silvicultural systems, Abies alba, Italy
Introduction
Silver fir (Abies alba Mill.) was artificially spread in many areas of Apennines since XIV century. Later on, a lack of attention to the forest management, tree ageing processes, insects and fungi outbreaks lead to an unsteady biological equilibrium in many plantations. The conversion of conifer plantations into stands with heterogeneous structure and composition is a current challenge for Italian silviculture. A silvicultural system aiming to the natural regeneration by means of canopy gaps is here considered.
Watt ([42]) first recognized the importance of gaps in ecological regeneration. Treefall gap disturbances are important to the species dynamics of many forest types ([33], [35]). In addition, gaps are important in maintaining and enhancing biological diversity ([15], [13], [43], [3], [36], [5], [10], [21]).
This approach meets the requirement of a forest management aiming to low environmental impact in accordance with the principles of sustainable silviculture, biodiversity conservation and the concepts of “the silviculture of transformation” ([30]).
Coates & Burton ([10]) review the gap dynamics literature with special attention to the use of gap size or position as predictive variables for silvicultural success or maintenance of ecosystem function. The role of gap size and within-gap position on the establishment of tree species has been studied in many different managed forests ([19], [40], [8], [26], [12], [17], [44]).
The purposes of this study are: 1) to assess if the opening of gaps is an appropriate tool to transform man-made pure stands of Abies alba into naturally regenerated stands; 2) to analyse the influence of micro-environmental conditions in different gap size on the recruitment and establishment of natural regeneration of forest tree species three years after gaps opening.
Materials and methods
The study area was located in artificial silver fir (Abies alba Mill.) stands on the north aspect of Serra S. Bruno (VV - Calabrian Apennine: 38°33’N; 16°19’E) at 900-940 m a.s.l., with an inclination of 5%.
Mean annual precipitations are 1848 mm, summer precipitations are 102 mm, with a dry period of 46 days, air humidity is normally high and never falls below 60%. Mean annual temperature is 10.8 °C, mean temperature of the coldest month is 3.1 °C, mean temperature of the warmest month is 21.5 °C. Soils classified as Humic Dystrudept ([41]), are acid, deep, well drained and derived from granitic parent material. The natural forest vegetation is dominated by silver fir-beech stands (Aquifolio-Fagetum Gentile 1969 - [2]) with sparse plants of chestnut (Castanea sativa Mill.).
During the late summer 2001 two sizes of circular gaps were created by cutting and removing all trees in a 90 year old stand, considering small (185 m2) and medium (410 m2) gaps with three replicates of each size. The gap diameter (measured between stems edges) to the tree height ratios were respectively 0.50 and 0.75 (mean stand height 30.5 m).
Thirteen circular subplots of 3.14 m2 (100 cm radius) were established inside each gap. Subplots were positioned one in the central part of the gap and the others along the cardinal directions. Starting from the centre each subplot was placed at one third of the radius.
The percent of vegetation cover was estimated in each subplot and in the whole gap. We considered all the seedlings born after the canopy opening. Each of these seedlings was identified by species, total height was measured and the age was visually determined. Census of seedlings in each subplot was conducted at the end of each growing season. Seedlings mortality was calculated as a difference between the inventories taken in the years 2002, 2003 and 2004.
To follow the seedlings growth in different gap micro-environments, 10 seedlings were marked with small pin flags in each sub-plot and total height of each seedling was recorded at the beginning (May) and at the end (October) of growing season 2003 and 2004. Seedling height increments were calculated over the course of growing season.
Four soil cores (8 cm diameter x 15 cm depth) were taken, every month, in the central part and four in the edge of each gap to assess the soil moisture content with gravimetric method. Four core samplings were collected also below the stand adjacent to the gaps to assess the microclimatic conditions under closed forest (control).
Only one depth level of sampling was chosen as seedlings concentrate the greatest part of their root system in the superficial layers of the soil. The soil samples were weighed immediately after the collection (wet weight). Later, in the laboratory, soil samples were dried in a drying oven at 105 ± 2 °C to constant weight and reweighed (dry weight).
Soil moisture content was expressed as percentage of dry weight (eqn. 1):
Measurements of the Photosynthetically Active Radiation (PAR - radiation in the 0.4-0.7 µm waveband) were collected with a ceptometer (AccuPAR, Degagon Devices Inc., Pullman, WA, USA - [34]), at 1.00 m above the ground, with the instrument held horizontally. Four instantaneous readings were done at each sampling point, with reference to the cardinal directions.
These measurements were carried out monthly, during growing season (May-September), in bright sunny days, at 12.00 (local solar time). Measurements were taken:
- in the full open, in a large clearing near the experimental area
- inside the adjacent stands
- in correspondence of each subplot.
After each measurement cycle for each sampling point the PAR transmittance was calculated using the following formula (eqn. 2):
Data were grouped for analysis according to the position of the subplot inside the gap: GAP CENTRE (C,1N,1E,1S,1W) central subplot plus the four nearest subplots in the cardinal directions and GAP EDGE (2N,2E,2S,2W, 3N,3E,3S,3W) the others.
Specific leaf area (SLA, leaf area per unit of leaf dry weight) was calculated for 12 silver fir seedlings at the end of the third growing season in gap centre and gap edge. Silver fir current-year needles were sampled, bagged in plastic, refrigerated and returned to the laboratory. Total leaf area (one-sided) per seedling was measured with an area meter (LI 3100, LI-COR Inc. Lincoln, NE, USA) and needles were weighed after drying for 24 hours at 85 ± 2 °C.
Differences in soil moisture, PAR transmittance, seedling density, seedling mortality, height increment and SLA were compared by means of a paired samples t- test (p<0.05) between gap sizes and within-gap position (Statistica, Statsoft®, Inc. Tulsa, OK, USA).
Results and discussion
Soil moisture
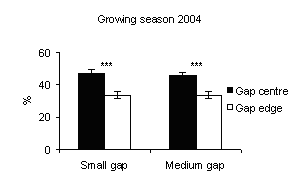
Soil moisture levels were not different between gap sizes and specifically both between gap centres and between gap edges (Fig. 1).
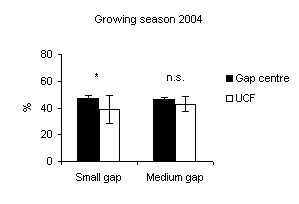
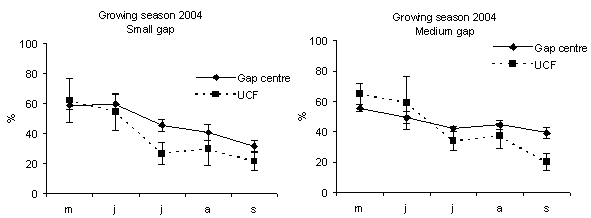
In all gaps soil moisture content was higher in the gap centre than in the gap edge with significant differences (Fig. 1). These findings are in accordance with Gray et al. ([21]) in gap sizes that ranged from 40 to 2000 m2 in mature Douglas-fir forests in the Pacific Northwest (USA). In a different way, according to the studies of McGuire et al. ([27]) and Gagnon et al. ([17]) in Pinus palustris stands in Florida and Georgia (USA), soil moisture did not vary from the gap edge to gap centre when drought conditions prevailed. Soil moisture was higher inside the gaps if compared with those recorded under closed forest (Fig. 2 and Fig. 3) with significant differences between small gaps and surrounding closed forest. This confirms the observations by Gray et al. ([21]) in gaps in mature Douglas-fir forests in the Pacific Northwest (USA); by Cutini et al. ([11]) in gaps in silver fir stands in Central Apennines (Italy) and by Ritter et al. ([38]) in gaps of beech-dominated forest in Denmark. Soil water values are higher in the gaps than in the surrounding forest probably in relation to both an increase in precipitation and a decrease in transpiration in the gaps ([45], [44]).
Fig. 2 - Means (± s.e.) of soil moisture content among gap sizes and among gap centre and under closed forest (UCF).
Fig. 3 - Monthly soil moisture content in gap centre and under closed forest (UCF). The vertical bars represent standard errors.
PAR transmittance
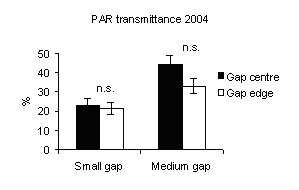
PAR transmittance was higher in medium gaps than in small ones (Fig. 4) with significant differences between gap sizes.
Fig. 4 - Means (± s.e.) of PAR transmittance (h 12.00) according to gap sizes and within-gap position.
These findings confirm that the amount of light received on the ground in a forest canopy gap is directly related to the size of the canopy opening ([6], [23], [27], [12], [21]).
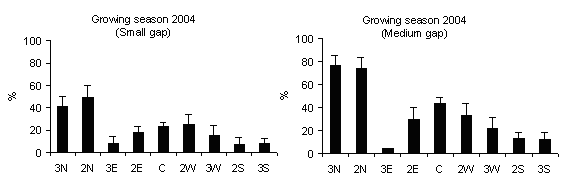
PAR transmittance varied within-gap position, as observed by Canham et al. ([6]), Brown ([4]), McGuire et al. ([27]), Diaci ([12]), Gagnon et al. ([17]). High values were recorded in gap centre rather than in gap edge (mainly in medium gap) but without significant differences (Fig. 4) and decrease from north to centre and to south positions for all gap sizes (Fig. 5). This is in accordance with Gray et al. ([21]) in experimental gaps in old-growth forests and mature forests dominated by P.menziesii and T. heterophylla in the Cascade Mountains (USA) where the gap diameter to tree high ratios were 0.2, 0.4, 0.6 and 1.0. Gagnon et al. ([17]) for a 3000 m2 experimental gap in Pinus palustris stands of Florida (USA) found that PAR transmittance was higher in the gap centre and lower near the gap edge and also between the northern and southern zones. Meanwhile, the difference in light between north and south aspects of gaps was attenuated as gap size increased ([8], [27]).
Fig. 5 - Distribution of PAR transmittance (h 12.00) according to within-gap position. The vertical bars represent standard errors.
Lower TPAR values recorded in the subplots in south zone compared to those in north zone can be explained as a consequence of the slope aspect and inclination effect and the influence of surrounding trees in gap edges.
The light levels beneath closed-canopy forests were 1 % of full sunlight, as reported (<1-2%) by Canham et al. ([6]) in many forest types. This could explain the deficiency of natural regeneration. In fact values <2% of PAR transmittance represent the threshold of silver fir seedlings survival ([25], [31], [22]).
Seedling density
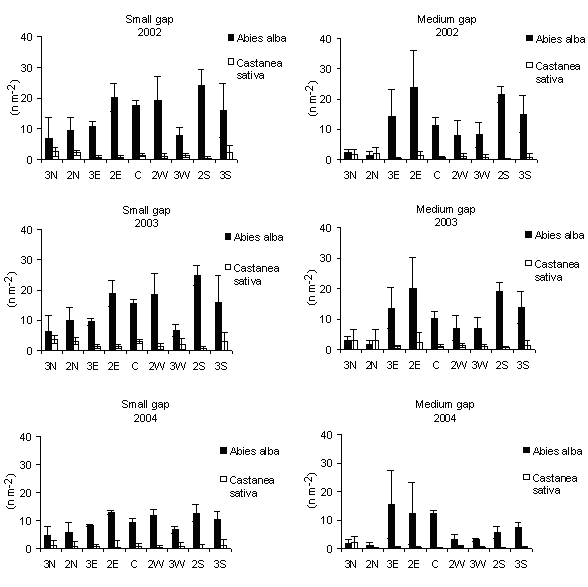
Silver fir (the most abundant species with respect to chestnut) seedling density was higher in small gaps than in medium ones (Fig. 6) with significant difference between the gap centres, but not between the gap edges. Among the possible reasons Grassi & Bagnaresi ([18]) consider that the limited ability of silver fir to exploit high-light conditions, limits its recruitment to small gaps. No significant difference of seedling density was observed between gap centres and gap edges inside the small gaps while it was significant in medium gaps (Fig. 6).
Fig. 6 - Means (± s.e.) of distribution of seedlings density according to gap sizes and within-gap position.
Silver fir seedling density tended to be higher in the central-southern positions in small gaps; in medium gaps high values were recorded also in the eastern positions (Fig. 7). This confirms that the natural regeneration of a shade-tolerant species, as silver fir, seems to occur in those sectors of the gap receiving less PAR but sufficient to allow the seedlings growth. Indeed the distribution patterns of seedlings of shade-tolerant tree species, in northern hemisphere, follow the shady southern edges of gaps, as demonstrated by Gray & Spies ([19]) in the Cascade Mountains (USA) where regeneration of shade-tolerant species was more abundant in shaded portions of gaps. In the northwestern British Columbia (Canada) Abies lasiocarpa and Abies amabilis “appeared to regenerate equally well at all gap positions but showed a clear preference for shadier small and medium sized gap” ([9]). Diaci ([12]) for a mixed forest with beech, silver fir and Norway spruce, in a Pre-alpine region of Slovenia, reported that the northern edges of the gaps can be unfavourable for germination since direct solar radiation, low precipitations and root competition occur. These results are in accordance with Ciancio et al. ([7]) reporting the occurrence of natural regeneration in the centre of natural small gaps in silver fir stands of the same area of this study. However, contrasting results were obtained by Mercurio (1994, 2000) in experimental gaps of artificial stands in the Central Apennine (Italy) in which fir regeneration was concentrated to the gap edges. This could suggest a diverse ecological behaviour of silver fir of Serra San Bruno.
Fig. 7 - Distribution of seedling density according to within-gap position. The vertical bars represent standard errors.
Seedling mortality
After three growing seasons seedling mortality was higher in the gap centres, but with no significant differences between gap sizes and within-gap positions (Fig. 8). The increase (about 40%) in seedling mortality, between 2003 and 2004, could be partially related to the detrimental effect of ground vegetation for natural regeneration ([12], [32]). In small gaps the vegetation cover (68%) was 30% lower than that of medium gaps (96%) and was negligible at the edges. This could have influenced the higher values of seedling mortality in medium gaps than in the small ones.
Fig. 8 - Seedlings mortality (2003-2004) according to gap sizes and within-gap position. The vertical bars represent standard errors.
Seedling growth
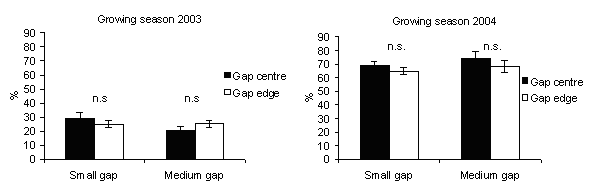
In 2003, the percentage of seedling height increments (Fig. 9) was significantly higher in gap centres of medium gaps where PAR transmittance was higher. This is in accordance with Gray & Spies ([19], [20]): seedling size increased with gap size and was greater at gap centres. In northern temperate forest of British Columbia (Canada), after five years, growth of planted seedlings of shade tolerant and intolerant species increased rapidly from small single-tree gaps to about 1000 m2 gaps thereafter showed little change in gaps up to 5000 m2 ([8]).
Fig. 9 - Seedlings height increment according to gap sizes. The vertical bars represent standard errors.
Reduced seedling growth in response to decreased light availability has been found in a variety of forest types ([4], [14], [27], [22]). Indeed, in 2004, the percentage of seedling height increment decreased with respect to 2003 in all gaps (Fig. 9) and was higher in gap edges than in gap centres. A probable reason could be that in the gap edges the shading influence of ground vegetation was less relevant than in the gap centres as observed by Mercurio ([28], [29]) in artificial gaps in silver fir stands in the Central Apennine (Italy). Also Diaci ([12]) refers about the importance of these ground vegetation-free microsites for the establishment of silver fir regeneration.
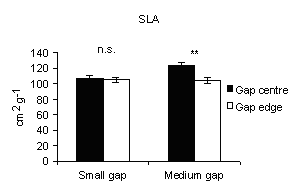
Specific leaf area
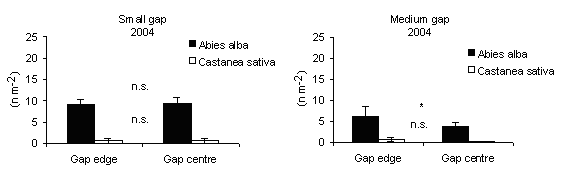
Specific leaf area (SLA) was used as an indicator of above-ground limitations on foliage growth and development arising from understory shading. In 2004, it was significantly higher at centres than at edges at medium gaps (Fig. 10), meaning that in the centres the foliage suffers from shading limitations of ground vegetation cover. On the other hand in A. alba total seedling biomass and total leaf area respond positively to increasing irradiance ([39]).
Conclusions
It is often advocated that ecologically sound silvicultural systems should be based on natural forest processes in order to obtain stable and self-sustaining forest systems ([1], [10], [16]). The gap-based approach and the gap-cutting system (sensu [10], [40]) simulate small-scale disturbances like natural gaps and are able to support the above mentioned purposes. This study shows that silver fir seedlings recruitment was greater in small gaps and in the central-southern positions. The most suitable gap size (expanded gap, sensu [37]) for silver fir regeneration, in this studied area, is 200 (300) m2, D/H = 0.5 and anyway < 0.75. As also supported by Ciancio et al. ([7]) who indicate for silver fir stands of the Calabrian Apennine a gap size of 200-400 m2 with a D/H ratios < 0.75. In order to foster seedling growth, browsing pressure by domestic cows needs to be controlled as well as, in the first years, the shrub/herb layer. The silvicultural treatment proposed may promote natural regeneration of silver fir and other broadleaves, enhance the development of a multi-layered forest structure and meet timber production objectives without compromising ecosystem management principles.
Acknowledgments
This study was supported with funds by Regione Calabria within the project “The re-naturalisation of conifer plantations”. We thank Dr. Antonio Poletto from Azienda Agricola S. Maria for logistical support.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
OI Gugliotta
I Mercurio
R Mercurio
Dipartimento di Gestione dei Sistemi, Agrari e Forestali, Università Mediterranea, loc. Feo di Vito, I-89100 Reggio Calabria (Italy)
Corresponding author
Paper Info
Citation
Albanesi E, Gugliotta OI, Mercurio I, Mercurio R (2008). Effects of gap size and within-gap position on seedlings establishment in silver fir stands. iForest 1: 55-59. - doi: 10.3832/ifor0448-0010055
Paper history
Received: Apr 27, 2005
Accepted: Nov 07, 2005
First online: Feb 28, 2008
Publication Date: Feb 28, 2008
Publication Time: 28.10 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2008
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 61131
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 49010
Abstract Page Views: 3966
PDF Downloads: 6848
Citation/Reference Downloads: 106
XML Downloads: 1201
Web Metrics
Days since publication: 6532
Overall contacts: 61131
Avg. contacts per week: 65.51
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2008): 34
Average cites per year: 1.89
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Optimizing silviculture in mixed uneven-aged forests to increase the recruitment of browse-sensitive tree species without intervening in ungulate population
vol. 11, pp. 227-236 (online: 12 March 2018)
Research Articles
Stand dynamics and natural regeneration in silver fir (Abies alba Mill.) plantations after traditional rotation age
vol. 7, pp. 313-323 (online: 08 April 2014)
Research Articles
Regeneration of Abies pinsapo within gaps created by Heterobasidion annosum-induced tree mortality in southern Spain
vol. 7, pp. 209-215 (online: 27 February 2014)
Research Articles
Optimum light transmittance for seed germination and early seedling recruitment of Pinus koraiensis: implications for natural regeneration
vol. 8, pp. 853-859 (online: 22 May 2015)
Review Papers
Structure and management of beech (Fagus sylvatica L.) forests in Italy
vol. 2, pp. 105-113 (online: 10 June 2009)
Research Articles
Short- and long-term natural regeneration after windthrow disturbances in Norway spruce forests in Bulgaria
vol. 11, pp. 675-684 (online: 23 October 2018)
Research Articles
Density and spatial distribution of beech (Fagus sylvatica L.) regeneration in Norway spruce (Picea abies (L.) Karsten) stands in the central part of the Czech Republic
vol. 9, pp. 666-672 (online: 12 March 2016)
Review Papers
Ensuring future regeneration success of Qualea grandiflora Mart. (Vochysiaceae) in neotropical savanna (cerrado) biomes by reviewing the available information and identifying research gaps
vol. 13, pp. 154-164 (online: 02 May 2020)
Research Articles
Tree-oriented silviculture: a new approach for coppice stands
vol. 9, pp. 791-800 (online: 04 August 2016)
Review Papers
Problems and solutions to cork oak (Quercus suber L.) regeneration: a review
vol. 16, pp. 10-22 (online: 09 January 2023)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword