Windstorm disturbance triggers multiple species invasion in an urban Mediterranean forest
iForest - Biogeosciences and Forestry, Volume 11, Issue 1, Pages 64-71 (2018)
doi: https://doi.org/10.3832/ifor2374-010
Published: Jan 25, 2018 - Copyright © 2018 SISEF
Research Articles
Abstract
Plant invasion in forest ecosystems is a serious ecological and economic issue, deserving attention by researchers, managers and policy-makers worldwide. Many invasive plants have been reported as early successional species able to colonize disturbed areas following abrupt changes in microhabitat and resource availability. We investigated disturbance effects of a severe windstorm generated by a wet microburst (hail and rain at 160 mm h-1) remarkably affecting the canopy cover of an old-growth Quercus ilex urban forest in Southern Italy. This stand-replacing disturbance produced a mosaic of 103 gaps, 5.6 to 1632 m2 in size, over an area of 1.53 ha, uprooting 76% of the trees and decreasing thereby 85% of the standing above-ground dry biomass into the gaps. By intensive monitoring we compared above- and below-ground microclimate, soil moisture and mineral N availability in paired disturbed and control areas of the study forest. Within newly formed gaps we observed a seasonally consistent 70% higher content of nitrate nitrogen, 29% and 47% decreases of ammonia nitrogen in summer and autumn, respectively, and a higher moisture in topsoil, in addition to different above- and below-ground microclimatic conditions, with canopy cover mitigating extreme temperatures. One year after the windstorm, the microhabitat shift promoted the establishment in gaps of 15 native and 10 alien taxa previously absent in both disturbed and control plots. In such conditions, the rarefaction of the dominant Q. ilex canopy cover and the occurrence of empty niches prone to invasion could dramatically affect the local community structure and diversity. Our data indicate that stand-replacing windstorm can transiently transform the studied urban evergreen forest to an early allogenic successional community dominated, in the medium and large gaps, by annual and perennial non-native species. This is particularly relevant under a perspective of possible increasing frequency of windstorm events in the Mediterranean region in the near future.
Keywords
Invasive Plants, Microburst, Mediterranean Evergreen Woodland, Quercus ilex, Resources Fluctuation, Empty Niche
Introduction
Invasive, non-native species are regarded as one of the most serious issues from both an ecological and economic perspectives ([31]). Invaders are known to alter both terrestrial and aquatic ecosystem structure and function ([20]), changing hydrology, nutrient cycling and energy budgets, thereby affecting native flora, fauna and ecosystem services ([18]).
In forest ecosystems, plant invasion has drawn the attention of researchers, managers and policy-makers worldwide ([41]). The successful establishment and spread of invasive plants has been traditionally related to their capability to efficiently intercept and use water, light and nutrients. Accordingly, native plant communities can become susceptible to invasion when available resources are not fully used, leaving empty niches potentially exploitable by invaders ([24]).
The fluctuating resource hypothesis states that an increase in unused resources enhances plant community susceptibility to biological invasion ([9]). On the other hand, invasive plants are often characterized by a more rapid growth and reproduction compared to native plants ([16]), a competitive ability to modify the invaded environment (e.g., by allelochemical production - [4]), and a phenotypic plasticity allowing adaptation to a wide range of environmental conditions ([33]). Established invaders, in addition, can actively modify resource dynamics by altering the soil nutrient cycle, thus leading to plant-soil feedback fostering invader success ([13]).
The availability of unused resources, such as soil nutrients, may be ascribed to either a decreased uptake by native species after disturbance events (e.g., fire, windstorm, overgrazing etc.) or to an increased input by external processes (such as anthropogenic N deposition, increased precipitation and solar radiation) that, in turn, may accelerate nutrient release from the soil ([46]). Many invasive plants have been classified as “disturbance-dependent” or “early successional” species, able to colonize disturbed areas after shifts in microhabitat and resource availability ([17]). As an example, Arellano-Cataldo & Smith-Ramírez ([3]) recently reported that canopy gaps in the Robinson Crusoe Island forest showed higher density of the invasive species Aristotelia chilensis and Rubus ulmifolius compared to the understory of intact forest canopy.
Natural disturbances such as windstorms, fires, floods and landslides play an important role in the structure and function of forest ecosystems ([42]). Windstorms, in particular, are considered a major disturbance process at global scale ([27]). Their impacts to forests occur across different temporal and spatial scales (reviewed by [23]). Visible damages can range from the uprooting or breakage of a single tree to massive catastrophic losses up to thousands of hectares ([30]). The main effect on forest canopy is the formation of tree-fall gaps, which leave space for the development of new plants ([47]). Canopy gaps, enhancing direct solar radiation to understory vegetation, can produce a community shift towards heliophytes by promoting seeds germination and growth of light-requiring species, previously suppressed by canopy shading ([45]). Among these, many invasive plants are highly competitive in bright sunlight conditions ([32]). On the other hand, uprooting locally produces an inversion of forest soil profile ([36]), which may facilitate colonization by early successional species, including invasive plants ([17]). Gap formation can alter additional resources and properties of the forest floor, such as soil moisture, temperature, and micro-topography, which can favor invasive plants development ([6]).
Evidence of windstorm damages to forest ecosystems is widely reported for North America ([14], [30]) and Tropics ([44], [29]), while reports for Europe ([19], [39]) and the Mediterranean region are more rare, as related to the lower occurrence and intensity of extreme events. However, windstorm frequency and impact in Mediterranean forests have been increasing since the mid-twentieth century, putatively due to climate change and forest management activities ([11], [12]). Such trend is expected to increase in the near future, possibly leading to a severe loss in forest structure, functioning and economic value ([37]). In addition, the impact of windstorm on forest ecosystems can interact with anthropogenic activities. In the Mediterranean basin, urbanization dramatically increased in the last decades ([35]). Urban and suburban forests are characterized by a specific ecology, being negatively affected by the surrounding urban landscape that create habitat fragmentation, induce community homogenization and so promote the spread of non-native species ([26]).
In this study, we investigated the disturbance effects of a stand-replacing windstorm generated by an extreme wet microburst in an old-growth, urban Quercus ilex forest in Southern Italy. By monitoring above- and below-ground microclimate, soil moisture and mineral nitrogen content in both disturbed and non-disturbed areas, we tested the hypothesis that altered environmental conditions promote changes in plant community composition. Specific hypothesis addressed in this study are: (i) above-ground microclimate changes in disturbed compared to non-disturbed area; (ii) windstorm disturbance drives an increase of soil resources in the forest gaps (i.e., nitrogen and soil moisture); (iii) abrupt shifts of microclimate and resources availability promote the spread of invasive plant species.
Materials and methods
Study site description
The Gussone Park of the Royal Palace of Portici (40° 48′ 40.3″ N, 14° 20′ 33.8″ E, elevation 75 m a.s.l.; about 1 km distant from the coast line) hosts a 16.4 ha, even-aged old-growth evergreen holm oak Mediterranean forest, surrounded by a densely urbanized metropolitan area. Small preexisting canopy gaps and newly storm-generated gaps suggest that the dynamic of stand structure will shift from a monocohort to a more complex multi-cohort stand. The study site is located at the base of the south-western slopes of Mt. Vesuvius volcano at an elevation of 84 m a.s.l., facing the sea of the Gulf of Naples.
The holm oak (Quercus ilex) is the dominant tree in the forest with scattered occurrence of some deciduous species such as Fraxinus ornus, Quercus pubescens, Ulmus campestris, and Celtis australis. In addition, it includes some creeper plants (Hedera helix, Rubus ulmifolius, Clematis vitalba, Tamus communis) and shrubs such as Viburnum tinus. Overall, the vascular flora includes 454 species ([38]).
A shallow soil with andic properties lays over pyroclastic deposits from eruptions of the Mount Vesuvius dated 1631 CE. The climate is Mediterranean, with humid winter and dry summer, total yearly rainfall of 929 mm (290, 200, 89 and 348 mm in winter, spring, summer, and fall, respectively), and mean monthly temperatures ranging between 11 °C (January) and 26 °C (August).
On 16 June 2014 at ~13:30 (CEST), a wet microburst, part of a large front of severe thunderstorms hitting the Tyrrhenian coastline of Southern Italy, moved from the Gulf of Naples towards the study site. The microburst generated irregular wind patterns with local speeds exceeding 100 km h-1 and hail and rain precipitations with intensity above 160 mm h-1.
Forest structure and vegetation analysis
Few days after the event, a field survey was carried out in the whole Gussone Park to assess the tree damage occurred in the forest. For all damaged trees (i.e., standing, uprooted or snapped), we measured diameter at breast height with a minimum threshold diameter of 7.5 cm; above-ground dry biomass was estimated using ad hoc forest height curve and allometric equation for total above-ground dry biomass of Q. ilex reported in the National Inventory of Forest and Carbon of Italy (equation dw4, p. 345 - [40]). Number and extension of canopy gaps were estimated after the windstorm in Google Earth Pro® using satellite images dated 11th of August 2014. In a selected canopy gap of 1318 m2, one month after the windstorm, stem diameter of standing and damaged trees (i.e., uprooted and snapped) were also measured in order to compare stand attributes and above-ground dry biomass before and after the event using the above mentioned equation.
In June 2015, one year after the windstorm event, we randomly selected 5 locations where disturbance occurred. A location consisted in a disturbed area with canopy gap and a control area under closed canopy. In each location, we estimated vascular species abundance within two paired adjacent plots of 10 × 10 m, one in the control area and the other in the disturbed one. Then, in each selected location two paired adjacent plots, the first under closed canopy and the other within the canopy gap (hereafter referred to as “control” and “disturbed area”, respectively). Plant species cover was visually assessed based on the following abundance scores: (1) rare; (2) <1%; (3) 1-5%; (4) 6-15%; (5) 16 -25%; (6) 26-50%; (7) 51-75%; (8) 76-100%. Plant specimens were identified according to Tutin ([43]). For alien exotic species and their naturalization status, reference was made to Celesti-Grapow et al. ([8]).
Litter and soil nitrogen availability
In mid-June 2015, we collected from the forest floor leaf litter using randomly positioned squared frames (10 × 10 cm). A total of 20 litter samples were collected, 2 in each of the previously selected area (5 disturbed and 5 control). Litter samples were placed in plastic bags, transported to the laboratory, and dried at 80 °C until reaching constant weight.
In the same areas, to assess the effects of disturbance on mineral nitrogen (N) availability, a total of 40 soil samples were randomly collected, away from uprooted trees, in summer (July) and autumn (October) 2015 (10 areas × 2 seasons × 2 samples). All samples (~1 kg each) were collected from the topsoil layer (0-20 cm), after removing the litter layer. Samples were packed in polyethylene bags, transferred to the laboratory within the sampling day and sieved at 2 mm, with subsequent quantification of the coarse (diameter >2 mm) and fine (diameter <2 mm) particle fractions. Soil samples for determination of nitrate (NO3-) and ammonium (NH4+) content were prepared by mixing 1 g of dry pulverized soil with 1 ml of distilled water in 2 ml Eppendorf tube. This mixture was shaken for 20 min and subsequently centrifuged for 5 min at 13.000 rpm. The samples were analyzed with a DR 3900 Spectrophotometer (Hach, Loveland, CO, USA) by using the manufacturer kits LCK 340 for NO3- and LCK 303 for NH4+.
Above- and below-ground microclimate and soil moisture
Microclimate and soil moisture were monitored for one year (from December 9th, 2014 to December 9th, 2015) using two monitoring stations in either disturbed and control areas. Each station was equipped with an Em 50 data logger, two 5TM sensors for soil moisture content and temperature and one VP3 sensor for air temperature and relative humidity (all instruments from Decagon, Pullman, WA, USA). Air temperature/humidity sensors, protected by a radiation shield, were placed 10 cm above the ground. The two soil moisture and temperature probes were diagonally buried into the soil at -5 cm and -20 cm depths, respectively. No specific direct gravimetric measurements were performed for calibrating the relation between 5TM sensor responses and actual moisture content, because the sensors were installed for comparing relative changes over time by meteorological forcing in disturbed and control areas, rather than for retrieving absolute soil moisture values. Volumetric water content data were estimated according to the standard relation proposed by the manufacturer, certifying an accuracy of ± 3% for soils. The data loggers were programmed to store sensor data every 30 minutes over the whole monitoring period.
Photosynthetic photon flux density (PPFD, λ = 400-700 nm) was measured using a LI-250A light meter as quantum sensor (Li-Cor Inc. Environmental, Lincoln, Nebraska, USA), located at 0, 0.5, 1.0, and 2.0 m above the ground of 10 locations randomly selected within disturbed and control areas. Instantaneous measurements averaged every 15 seconds were taken during bright, sunny days in July 2014.
Data analysis
Tree data from disturbed and control areas were used to estimate tree density, above-ground biomass, basal area and mean height of standing and damaged trees, the latter separately assessed for uprooted and snapped trees. Gap data from aerial imagery were used to calculate gap-size frequency distribution. Species abundance scores were transformed in percent cover data, using the mean percent value for each ordinal class, and submitted to two-way factorial ANOVA including species, area (either disturbed or control) and their interaction as fixed factors. Species-specific differences between disturbed and control area were assessed by means of Duncan’s post-hoc test. The matrix plots × species of transformed cover data was also submitted to unconstrained ordination by Principal Component Analysis (PCA).
Litter data were tested for significant differences between disturbed and control areas fitting a Generalized Linear Mixed Model (GLMM) with gaussian error distribution and identity link function, considering area and location as fixed and random factors, respectively. Further GLMMs were fitted to test differences of soil mineral N content, either NO3- or NH4+, between disturbed and control areas in summer and autumn, including fixed effects of area and season, and random effect of location where soil samples were collected. In all GLMMs, pairwise differences were tested by Duncan’s post-hoc test. Statistical significance was tested in all cases at P < 0.05.
Results
Effect of windstorm on stand structure, vegetation and plant invasion
Damages produced by the windstorm on the forest canopy were highly variable in space, as related to high spatial variability of the wind pattern generated by the microburst. Windstorm was stand-replacing on 1.53 ha (i.e., 9.3% of the total forest area), which correspond to ~8% of the total above-ground dry biomass (estimated in ~5000 Mg in a survey before the storm event), with a total of 103 canopy gaps. Gap size ranged from 5.6 m2 up to 1632 m2, although 96.5% of the gaps had a size of less than 500 m2 (see Tab. S1 in Supplementary material). We counted 404 trees (i.e., 352 Q. ilex and 52 other broadleaved trees and conifers) uprooted or with stem breakage, mostly inside the stands (367 trees) and to a lesser extent along internal roads (37 trees).
Tab. S2 (Supplementary material) reports the number, dry biomass, basal area and quadratic mean diameter of the trees growing in the disturbed area selected for the vegetation analysis, before and after the windstorm. After the windstorm, about 65% of trees were uprooted in disturbed areas (Tab. S2). Above-ground dry biomass declined by ~87%. The quadratic mean diameter of the trees before the storm was 33.1 cm, while after windstorm it became 34.3 cm and 30.8 cm for uprooted and snapped trees, respectively.
Plant species cover was significantly affected by disturbance, with effects differing for different species (Tab. S3). Unconstrained ordination by PCA showed a distinct segregation of the plots according to disturbance (Fig. S1 in Supplementary material), with disturbed and control plots being positively and negatively correlated to the first ordination axis, respectively, and all invasive plant species showing positive scores on the same axis. Thirty-nine plant species were recorded in the plots surveyed, with 14 life forms overall representing 32 families (Tab. S4). Among these, 10 alien invasive plant species were recorded in the disturbed area, while the control area was not subject to plant invasion. Phytolacca americana and Solanum chenopodioides were the most abundant invasive herbs (Fig. 1, Fig. S1), with other invasive species that include 3 deciduous broadleaved trees (Ailanthus altissima, Diospyros kaki and Morus alba), 1 palm (Phoenix canariensis), 2 vines (Araujia sericifera and Parthenocissus quinquefolia) and 2 annual herbs (Chamaesyce maculata and Erigeron sumatrensis). Concerning native plants, with the exclusion of Q. ilex, 15 species were exclusively recorded and 3 showed highest cover in the disturbed area, while 6 species were found in both areas with similar coverage, and 4 species were exclusively recorded in the control areas (Fig. 1, Tab. S4).
Fig. 1 - Plant cover percentage of native and invasive plant species within disturbed and control areas of the Gussone Park forest in June 2015, one year after the windstorm. Asterisks indicate significant difference for each species between disturbed and control areas (P < 0.05, Duncan’s post-hoc test for the interactive effect of area and species from two-way factorial ANOVA is shown in Tab. S4, Supplementary material).
Litter and mineral N availability
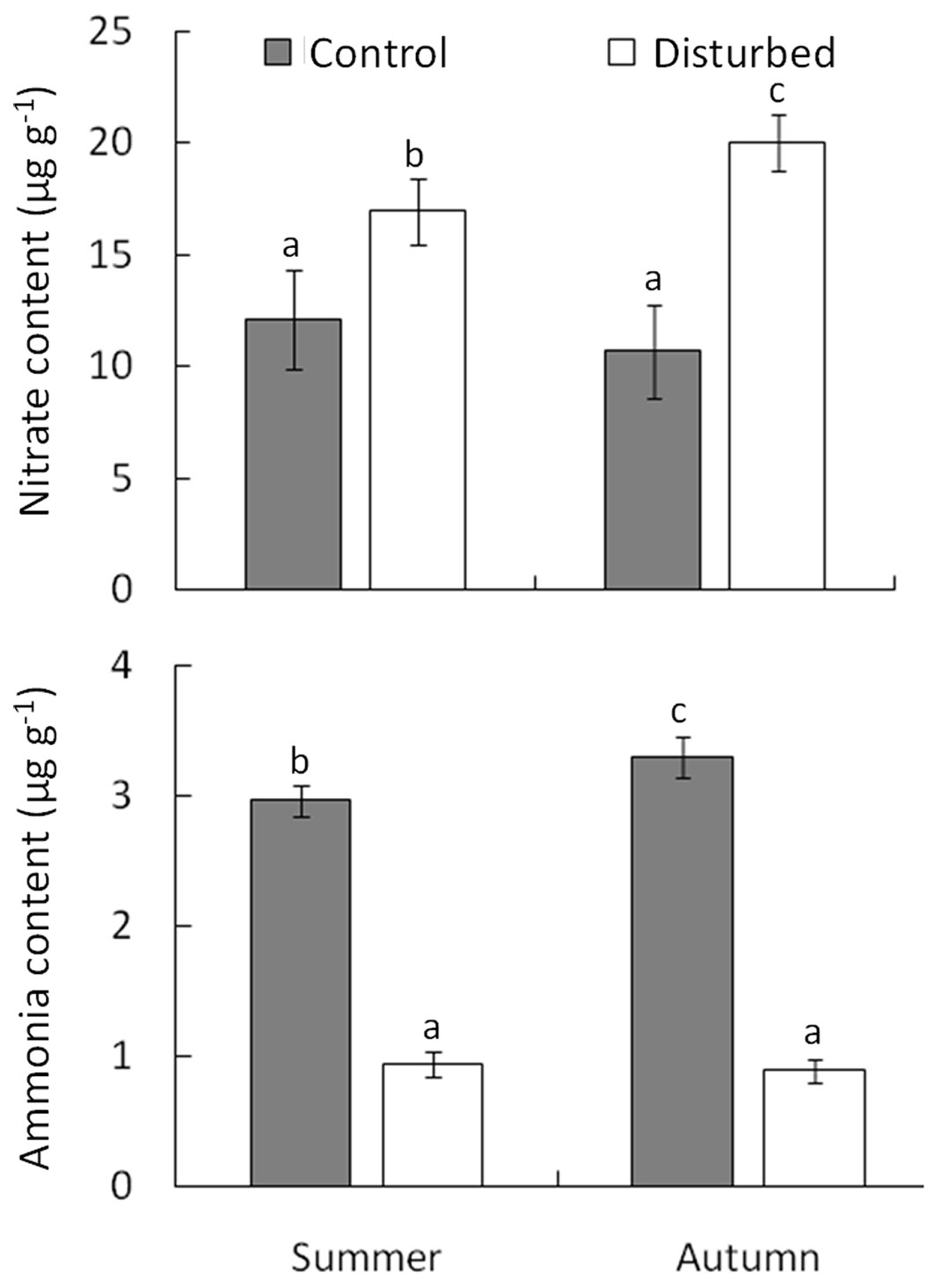
The amount of litter was significantly lower in disturbed areas compared to control ones (1745.31 ± 17.28 and 2496.88 ± 47.05 g m-2, respectively; F1.20 = 10.39, P = 0.0104, mixed effect model - Tab. S5). On average, the content of nitrate nitrogen (NO3-N) was significantly higher in the disturbed area (Fig. 2, Tab. S6). In addition, seasonal changes in NO3-N were not consistent between disturbed and control areas (significant interaction term in Tab. S6), with differences of 28.5% and 46.6% in summer and autumn, respectively, compared to the control (Fig. 2). Differently, ammonium nitrogen (NH4-N) content in the disturbed area was lower than the control (Tab. S6), with also significant seasonal differences between disturbed and control areas (interaction term in Tab. S6) of 68.2% and 72.9% in summer and autumn, respectively (Fig. 2).
Fig. 2 - Mineral nitrogen NO3- (upper panel) and NH4+ (lower panel) content in disturbed and control areas in summer (June) and autumn (October) 2015, following the windstorm of 16 June 2014. Values are average ± standard error of five replicates. Within each panel, different letters indicate significantly different groups (Duncan’s post-hoc test for the interactive effect of disturbance occurrence and season from GLMMs is shown in Tab. S6, Supplementary material).
Microclimate and soil moisture content
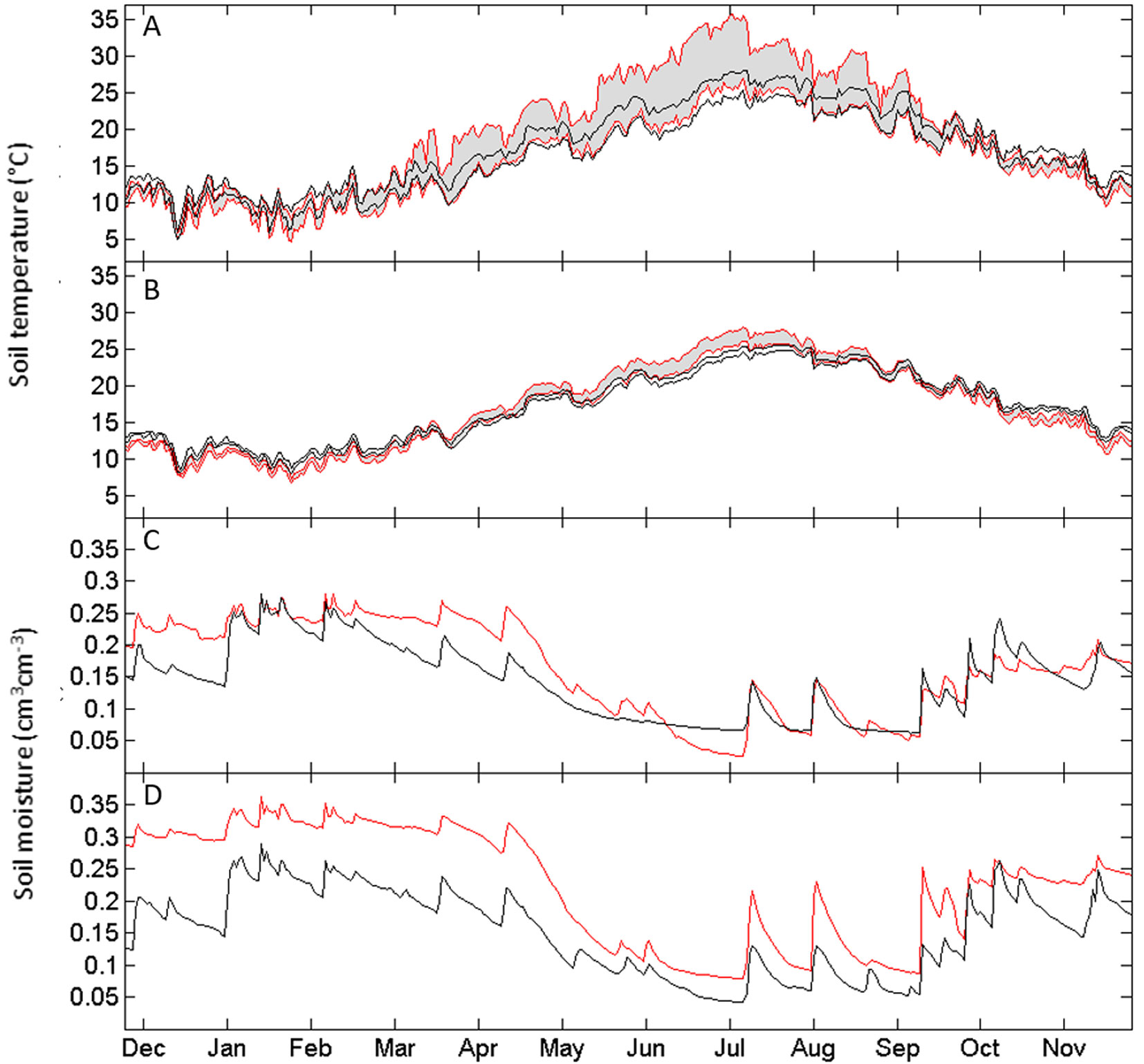
Microclimate, in terms of temperatures and moisture, exhibited a strong seasonality. Soil temperature was slightly warmer in the control area during winter months. Differently, from April to September mean daily temperature in the soil under the intact forest canopy was largely lower than that observed in the disturbed area (Fig. 3A, Fig. 3B; Fig. S2). In the disturbed areas, soil temperature at -5 cm depth often reached diurnal values close to 35 °C and remained above 30 °C for several summer weeks, while in the control area the diurnal soil surface temperature never exceeded 25 °C (the recorded maximum was 28.4 °C). Air temperature in disturbed areas, compared to the control, was lower in winter but higher in spring and especially in summer (Fig. S2 in Supplementary material), with a maximum of 48.4 °C, exceeding by 14.8 °C that of the intact forest canopy.
Fig. 3 - Canopy effects on below-ground microclimate assessed by monitoring soil temperature and moisture from December 9, 2014 to December 9, 2015 in disturbed (red line) and control (black line) areas. Panels show daily soil temperature at -5 cm (A) and -20 cm (B) depths and soil moisture at the same depths (C and D, respectively). Grey shaded areas (bounded by red lines) highlight daily temperature ranges in the disturbed areas as compared to observations under the canopy in control areas (bounded by black lines).
Soil moisture exhibited a wet stage from October to May, followed by a relatively dry stage from June to September (Fig. 3C, Fig. 3D). During the wet stage, the time series showed a sequence of small pulses corresponding to rainfall events in both areas, which however resulted in higher soil moisture in the disturbed area, especially at -20 cm depth (Fig. 3C, Fig. 3D).
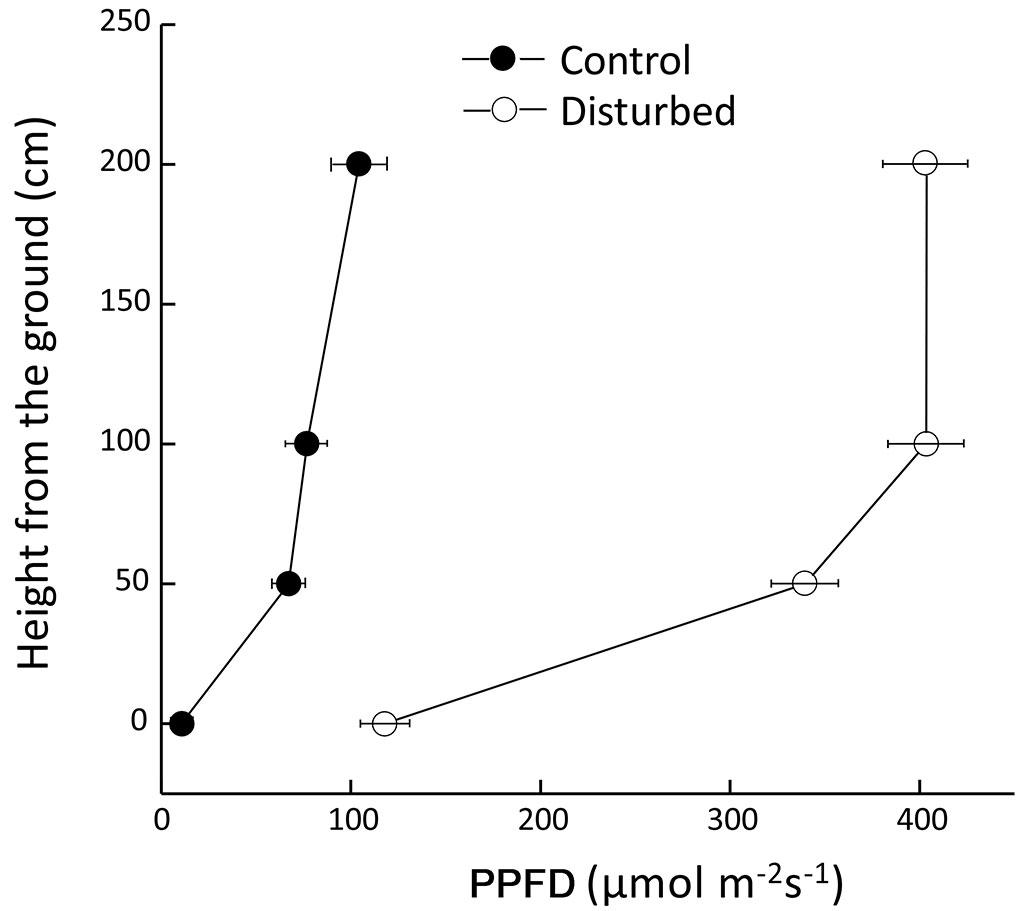
Expectedly, PPFD within the disturbed area was significantly higher than the control, with maximum relative difference recorded at ground level, where the PPFD exceeded that of the control area by one order of magnitude (Fig. 4).
Fig. 4 - Photosynthetic photon flux density (PPFD) in disturbed and control areas. Values are average ± standard error of ten replicates.
Discussion
Effects of windstorm on plant invasion
Abrupt environmental shifts produced by uprooted trees affected plant community recovery from the disturbance and recolonization within the gaps. In medium and large gaps, early colonizer species prevailed over pre-existing established herbs, lianas and tree seedlings. Noteworthy, one of the most obvious windstorm effects was the invasion of non-native species that occurred only in the disturbed plots. At the community level, the windstorm acted as a disturbance removing previously established vegetation and releasing space for new pioneer species. Such alien species spreading can be explained by two complementary factors: the increase of community invasibility and the high propagule pressure at the study site. With regard to the first factor, one year after the disturbance, micro-environmental conditions within the gaps showed higher temperature with larger diurnal and seasonal fluctuations, higher light flux-density at ground level, and higher soil moisture and nitrate nitrogen content compared to undisturbed understory. As a consequence, fast-growing, early colonizer species rooted in the gaps could benefit from favorable microclimatic conditions, at least during spring and early summer seasons, as well as from improved resource availability, compared to pre-disturbance conditions. Our data are consistent with the fluctuating resource hypothesis, which states that an increase in quantity of unused resources will enhance the susceptibility of a plant community to biological invasion ([9]). In this regards, the absence of early recruitment of shade-tolerant species (e.g., Q. ilex) in the gaps could be due to their more limited response to the increased soil resource availability even at high light radiation levels, compared to light-demanding and/or pioneer species. An alternative hypothesis for the lack of Q. ilex seedlings, in spite of the conspicuous seed fall, could be the build-up of negative plant soil feedback due to litter autotoxicity ([22], [25]) that may limit recruitment under conspecifics. On the other hand, our data suggest that invasive plants grow more rapidly under post-disturbance conditions compared to early colonizer native perennial species. For instance, it is surprisingly that recruitments of early colonizer, light-demanding (U. campestris, C. australis) and juvenile shade-tolerant (F. ornus) native tree species, were absent within the gaps, despite being relatively abundant in the adjacent undisturbed forest stands. Differently, seedlings of the invasive Ailanthus altissima were the most abundant in the gaps formed by the windstorm, while being absent in the control area. The impact of the light-requiring pioneer species A. altissima on Mediterranean ecosystems has been previously reported ([8]), and its invasion capability has been related to the sprouting of its sub-surficial roots, which spread rapidly in space ([21]). Disturbance can also favor clonal regeneration that leads to an increase of ramets from the edge of the wood or from the ramet bank, which may persist under shady condition in undisturbed stands. Our data indicate that A. altissima rapidly and massively colonizes post-disturbance forest gaps mainly by seed regeneration. However, further experimental work is needed to clarify the relative importance of within-gap nutrient, water, and microclimatic conditions among the factors that favour the colonization of this species after windstorm disturbance.
The increased community invasibility, alone, cannot explain the increased abundance and diversity of alien species recorded in forest gaps after the windstorm. The burst of several herbaceous species (e.g., P. americana and S. chenopodioides) few months after the disturbance indicates the presence of a persistent soil seed bank of non-native plants in the study site. In the case of P. americana, its longevity in the soil is well documented ([28]). Moreover, the occurrence of alien species related to agricultural (e.g., Diospyros kaki and Morus alba) and gardening (e.g., Araujia sericifera and Phoenix canariensis) systems suggests that the study site is subject to a considerable propagule pressure from the adjacent open areas. In fact, the study site is an old growth forest embedded in an urban landscape, with high frequency of cultivated fields and gardens. In this context, our findings suggest that natural forests at the urban interface are particularly vulnerable to invasion by non-native plants, because suburban gardens and agricultural activity can act as significant sources of plant propagules ([2]).
Effects of windstorm on microclimatic conditions and resource availability
The catastrophic windstorm of 16 June 2014 produced many gaps in the forest canopy caused by individual or neighborhood groups of tree falls, with gap size ranging from few square meters to more than 1600 m2. According to previous studies, gaps formed by the windstorm are characterized by greater light availability at ground level compared to closed canopy ([32]). Besides light availability, other major environmental shifts occurred within the gaps compared to undisturbed forest areas. Among these, air and surface soil temperatures within the gaps were higher than in adjacent control areas during spring and summer, in agreement with previous studies ([15], [34]), but lower during winter, especially during the night. As a consequence, plants established within the gaps experienced a much wider temperature fluctuation at both daily (day vs. night) and seasonal (summer vs. winter) scale.
Among the possible environmental modifications induced by the windstorm, we showed a systematic modification of soil moisture regime within the gaps. The study area is characterized by a Mediterranean climate with alternation of cold-wet (autumn-winter) and hot-dry (spring-summer) seasons, with very short wet-to-dry and dry-to-wet transition stages. Several studies highlighted that Mediterranean summer drought establishes microclimatic conditions particularly severe for the regeneration of shrub and tree species ([1]). In such conditions, the forest canopy buffers the local microclimatic severity by favoring a decrease of air temperature and a simultaneous increase of air relative humidity during the hottest hours of the summer days, thus contributing to mitigate vapor pressure deficit, leaf temperature and transpiration losses. Canopy light interception also reduces the amount and modifies the quality of incident solar radiation transmitted to the ground, reducing soil heating and soil evaporation losses. In spite of higher soil temperature and lower air relative humidity, soil moisture was higher within the gaps during winter and spring. A higher soil water content in forest gaps has been previously reported ([15], [34]), but not in the Mediterranean region. Higher soil moisture in the gap is probably due to lower canopy interception, which can reach ~13% of total rainfall in olm oak forests ([5]), and lower transpiration losses. Some previous studies ([10]) reported a reduction in fine root biomass following treefall, suggesting the hypothesis of reduced transpiration losses in gaps soils. Our data are consistent with such hypothesis, because soil water content differences between intact canopy and gaps are highest in winter and spring but not in the driest summer days, when soil water reached the minimum content values.
The soil in gaps, in addition to higher moisture content, had higher nitrate nitrogen content compared to undisturbed areas. Our results are apparently contrasting with previous findings by Zimmerman et al. ([48]), reporting that, after a hurricane in tropical forest, decomposition of fresh leaf litter and wood debris from tree falls induced long-term N immobilization, with depressive effects on net primary productivity in the first 13 years following the disturbance. In addition, our previous studies carried out in the study site showed that decomposition of Q. ilex leaf litter determines a net N immobilization lasting for several months ([7]), as related to its relatively high C/N ratio. This apparent contradiction could be explained by a fast mineralization of soil organic matter within the gaps that may overbalance the N immobilization by degradation of Q. ilex leaves and especially wood debris with high C/N ratio (>100). Indeed, N transfer and immobilization by microbes becomes ecologically relevant during the early phases of litter decomposition, when C/N ratio of the decomposing substrate is above a critical threshold of ~30-35, which leads to microbial N starvation. In this context, our hypothesis is that the accumulation of nitrate nitrogen could be related to higher soil temperature and water content enhancing organic matter mineralization rates during spring and early summer. On the other hands, we expect that N immobilization will become quantitatively more relevant in the long term (i.e., some years after the windstorm), as a consequence of the progressive degradation of wood debris and the litter migration to ground level. Quantification of accumulated wood debris and of the subsequent decomposition process is important to understand the impact of necromass on soil processes as well as on food-web structure and functionality. Our on-going studies are addressing the impact of dead wood and leaf litter on microbiome structure and diversity by using high-throughput sequencing of bacterial and eukaryotic rRNA gene markers.
Conclusions
We showed that extreme windstorm events affect forest structure driving to sudden environmental shifts, such as modifications of micro-climatic conditions and soil resources. At the community level, windstorms can foster the spread of several invasive species, likely by removing the dominant evergreen tree Q. ilex, thus providing empty niches for many short-lived herbs, as well as some early colonizer and non-native trees. The spread of alien plants could eventually drive to alternative successional trajectories that may dramatically affect community structure and diversity. These data are the first to show a multiple plant invasion after a windstorm disturbance in the Mediterranean region that can potentially transform an urban evergreen forest to an early successional community dominated by non-native deciduous species. This finding is relevant to appreciate the potential ecological implications of infrequent disturbance in Mediterranean forest ecosystems in the context of climatic change. Under global warming, the frequency and intensity of windstorm is expected to increase ([37]), potentially enhancing plant community invasion by non-native plants. Future studies are urgently required to assess the interactive effects of extreme weather events, community invasibility, and propagule pressure in order to understand their relative importance for the spreading of invasive plants, and evaluate their ecological and economic impacts.
References
CrossRef | Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Tushar C Sarker
Riccardo Motti
Gaspare Cesarano
Maurizio Teobaldelli
Luigi Saulino
Francesco Cona
Giovanni Battista Chirico
Stefano Mazzoleni
Antonio Saracino
Department of Agricultural Sciences, University of Naples Federico II, v. Università 100, I-80055 Portici, Naples (Italy)
Department of Agri-Food, Animal and Environmental Sciences, University of Udine, v. delle Scienze 206, I-33100 Udine (Italy)
Botany Department, Faculty of Science, Mansoura University, Mansoura 35516 (Egypt)
Department of Environmental, Biological and Pharmaceutical Sciences and Technologies, University of Campania Luigi Vanvitelli, v. Vivaldi 43, I-81100 Caserta (Italy)
Corresponding author
Paper Info
Citation
Bonanomi G, Incerti G, Abd El-Gawad AM, Sarker TC, Stinca A, Motti R, Cesarano G, Teobaldelli M, Saulino L, Cona F, Chirico GB, Mazzoleni S, Saracino A (2018). Windstorm disturbance triggers multiple species invasion in an urban Mediterranean forest. iForest 11: 64-71. - doi: 10.3832/ifor2374-010
Academic Editor
Michele Carbognani
Paper history
Received: Jan 23, 2017
Accepted: Nov 18, 2017
First online: Jan 25, 2018
Publication Date: Feb 28, 2018
Publication Time: 2.27 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2018
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 51672
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 42616
Abstract Page Views: 4069
PDF Downloads: 3850
Citation/Reference Downloads: 19
XML Downloads: 1118
Web Metrics
Days since publication: 3017
Overall contacts: 51672
Avg. contacts per week: 119.89
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2018): 21
Average cites per year: 2.63
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Shrub facilitation of Quercus ilex and Quercus pubescens regeneration in a wooded pasture in central Sardinia (Italy)
vol. 3, pp. 16-22 (online: 22 January 2010)
Research Articles
Germination and seedling growth of holm oak (Quercus ilex L.): effects of provenance, temperature, and radicle pruning
vol. 7, pp. 103-109 (online: 18 December 2013)
Research Articles
Assessing escapes from short rotation plantations of the invasive tree species Robinia pseudoacacia L. in Mediterranean ecosystems: a study in central Italy
vol. 9, pp. 822-828 (online: 25 May 2016)
Research Articles
Is cork oak (Quercus suber L.) woodland loss driven by eucalyptus plantation? A case-study in southwestern Portugal
vol. 7, pp. 193-203 (online: 17 February 2014)
Research Articles
Hemlock woolly adelgid niche models from the invasive eastern North American range with projections to native ranges and future climates
vol. 12, pp. 149-159 (online: 04 March 2019)
Research Articles
Evergreen species response to Mediterranean climate stress factors
vol. 9, pp. 946-953 (online: 07 July 2016)
Research Articles
SimHyb: a simulation software for the study of the evolution of hybridizing populations. Application to Quercus ilex and Q. suber suggests hybridization could be underestimated
vol. 11, pp. 99-103 (online: 31 January 2018)
Research Articles
Fluctuation of the ecological niche of Moringa peregrina (Forssk.) Fiori with topoclimatic heterogeneity in southern Iran
vol. 16, pp. 53-61 (online: 16 February 2023)
Research Articles
Are Mediterranean forest ecosystems under the threat of invasive species Solanum elaeagnifolium?
vol. 14, pp. 236-241 (online: 10 May 2021)
Research Articles
Post-fire effects and short-term regeneration dynamics following high-severity crown fires in a Mediterranean forest
vol. 5, pp. 93-100 (online: 30 May 2012)
iForest Database Search
Google Scholar Search
Citing Articles
Search By Author
- G Bonanomi
- G Incerti
- AM Abd El-Gawad
- TC Sarker
- A Stinca
- R Motti
- G Cesarano
- M Teobaldelli
- L Saulino
- F Cona
- GB Chirico
- S Mazzoleni
- A Saracino
Search By Keywords