Carbon storage in degraded cork oak (Quercus suber) forests on flat lowlands in Morocco
iForest - Biogeosciences and Forestry, Volume 9, Issue 1, Pages 125-137 (2015)
doi: https://doi.org/10.3832/ifor1364-008
Published: Aug 08, 2015 - Copyright © 2015 SISEF
Research Articles
Abstract
The present study aims to quantify the carbon stored in a degraded cork oak (Quercus suber L.) ecosystem in the north west of Morocco, in view of potential management implications. To this end, carbon stocks were evaluated in the first 100 cm of the soil, the cork oak trees, and the understorey species (both above- and belowground). Results show that the total carbon stocks in the cork oak ecosystem ranges from 65 to 237 Mg ha-1 with a mean value of 121 Mg ha-1. The first 100 cm of the soil (including the forest floor) represents the largest carbon pool (~51% of the total organic carbon) of the ecosystem. Tree biomass (above- and belowground tissues of cork oak) represents the second largest pool (47%), whereas the contribution of the understorey is less than 2%. Within the first 100 cm of the soil, over 87% of all the soil organic carbon is situated in the first 40 cm of the soil depth. The amount of carbon stored here ranges from 30 to 110 Mg ha-1and these organic carbon stocks vary considerably with the stand basal area of the cork oak (R2 = 0.82). In practice, the carbon stocks of the different pools considered are strongly correlated with the stand density of the cork oak stands. In the semi-arid forest ecosystems of our study, management prescriptions aiming at increasing the standing biomass of the cork oak should thus considerably contribute, both directly through tree biomass and indirectly through increased soil organic matter, to efficient carbon sequestration.
Keywords
Quercus suber, Mamora, Carbon Storage, Biomass, Litter and Soil
Introduction
Emissions of CO2 have largely contributed to the observed changes in global climate over the last decades ([47]) and the link with human activities is no longer debated ([46]). Terrestrial ecosystems represent a considerable part of global carbon stocks ([48]) and of all terrestrial ecosystems, forest ecosystems make up the largest part, containing around 80% of the aboveground and 40 % of the belowground terrestrial carbon ([30]). Soils represent roughly two-thirds of the terrestrial carbon in forests, but this varies across biomes with 85% in boreal forests, 60% in temperate forests and 50% in tropical rain forests ([30]). Forest soils account for 35% of the global soil C stock ([79]) and this value is 2-3 times higher than the C contained in the atmosphere ([62], [88], [33]). Clearly, whether such ecosystems are a source or a sink is thus a critical issue for the evolution of the carbon concentration in the atmosphere. At the same time, increased CO2 levels in the atmosphere have been shown to lead to increased sequestration in terrestrial ecosystems ([68]), in particular in forests. The extent to which this may occur will depend on local site conditions such as climate, nutrient and water availability, stand vitality, latitude, tree age or species ([69], [26], [38], [29]), but also on anthropogenic influences, such as management, land degradation, or overexploitation ([69], [53], [50]). Clearing vegetation or removing the forest is estimated to account for 17% of the total emissions of greenhouse gases ([47]), and recently emphasis has been laid on this in developing countries. However, to our knowledge, the effect of human (over-) exploitation on carbon storage in these forested ecosystems has not been fully addressed. Typically, carbon accounting studies or predictive models highlight differences between boreal, temperate and tropical rain forests ([70]), but do not present any values for arid or semi-arid regions. According to the FAO ([41]), the carbon stocks in forested ecosystems in North Africa (including biomass, dead organic debris, forest floors and the first 30 cm of the soil) were on average 64.9 Mg ha-1. Many forested ecosystems in these climates, such as those around the Mediterranean basin, are characterized by low stocking for the overstorey species, with variable amounts of understorey. In the present study, we tried to characterize carbon pools of stands typical of the Mediterranean basin: a cork oak forest.
Cork oak (Quercus suber L.) is a species endemic to the Mediterranean basin and has been present in the western Mediterranean for more than 60 million years. Its distribution range has greatly decreased as a result of the large variations in climatic conditions during this long period, but above all due to human activities ([27]). Cork oak forests in Morocco are representative of semi-arid forested ecosystems, often suffering from overexploitation, and currently they cover approximately 377 500 ha ([87]), equal to 15% of the world’s cork oak forests. From the ecological, economical and societal point of view, these cork oak forests are of great value. The cork oak forests of the coastal plains in the Mamora region covered more than 130 000 ha at the beginning of the 20th century ([39]), whereas the cork oak makes up less than 60 000 ha at present ([2]). The regression in surface area of the cork oak forests in Mamora is mainly due to the pressure of mankind and its livestock. Indeed, while the human population totals 300 000 inhabitants living in 300 villages, the Mamora region also has no less than 225 000 sheep and cows ([52]). Between 1913 and 1958, human activities were held responsible for the disappearance of up to 60 000 trees per year due to programmed or illegal exploitation, harvest of firewood, collection of aromatic or medicinal plants, fires, intensification of pasture grazing by livestock, cork exploitation from bark, charcoal production or transformation to crop lands for local needs ([16]). These rural pressures, combined with more frequently occurring water stress in drought periods and insect attacks ([37], [59], [45]) have led to a degradation of the cork oak ecosystem. In addition to rural pressures, the cork oak forest of Mamora is influenced by several surrounding cities, thereby exposing the forest to leisure activities as well as uncontrolled urbanization ([54]). As a result of all these factors of forest degradation and regression, the specific composition of the forest is disturbed, recruitment of new saplings has become almost impossible ([13]), and its surface area has greatly decreased over the last century ([3]).
Despite the relevance for the local economy and ecology, intensive use by mankind has resulted in severe degradation of the forest vegetation, stand structure and tree stocking. Restrictions of cutting and collection of firewood or acorns, along with many efforts to rehabilitate and replant the forest, have not been able to fully compensate for the degradation due to intensive use. A challenging issue, not addressed hitherto, is the actual and potential for carbon storage in such degraded low stocked forests. To this end, our study targets the quantification of carbon storage in this flat lowland cork oak forest. Our objectives were to: (1) quantify the carbon storage in a Mediterranean degraded forest ecosystem, such as the cork oak forest of Mamora; (2) assess the local determinants of carbon storage of degraded land; and (3) derive prescriptions for management schemes and optimization of carbon storage.
Materials and methods
Site description
The Mamora forest is located in the north west of Morocco close to the Atlantic Ocean, between 6° and 6° 45′ W and 34° and 34° 20′ N (Fig. S1 in Appendix 1). The topography is flat with altitudes ranging from 6-8 m close to the coast up to 300 m in the north easterly corner. The gentle slopes are oriented from south-west to the north-east and this flat topography enables easy access to and circulation in the forest. The Mamora geological formations are mostly marine deposits from the Pliocene to the Pleistocene ([1]) with sandy soils overlying a clayey substratum. Soils are sandy and slightly acidic throughout the region. They are classified as arenosols, based on the WRB nomenclature ([42]). The climate is Mediterranean with mean air temperatures ranging from 11 °C in January to 34 °C in August (annual mean value: 18 °C). The annual precipitation ranges from 450-600 mm, with most rain in November and December. The coastal area receives about 100 mm more rain than the inland area. Periods of summer drought easily last for more than two months, even though some alleviation may come from the humid atmosphere close to the coast. This dry season generally starts by the end of April or early May and extends until October ([1]). High summer temperatures and extreme dryness of the sandy top soils contribute to a rapid oxidation of all organic material at the surface whenever soils are bare or lose their cover, leading to very low levels of soil organic matter ([57]). As a result of these differences in microclimatic conditions, the western part of the Mamora forest is characterized by a sub humid climate with warm winters, while the eastern part is semi-arid with temperate winters ([14]).
Mamora forest has a total surface area of 133 000 ha ([51]), of which around 60 000 ha has cork oak as the main species, together with an endemic pear (Pyrus mamorensis Trab.) appearing in little thickets, but never in full stands. The mastic tree (Pistacia lentiscus L.), wild olive (Olea europaea L. subsp. oleaster [Hoffmans & Link] Negodi) and mock privet (Phillyrea latifolia L.) can be found, but remain uncommon. The understorey vegetation, when it is dense, is composed of flax-leaf broom (Genista linifolia L.) and Thymelaea lythroides (Barr. et Murbeck) in the western part of the forest and of rock rose (Halimium halimifolium [L.] Willk.) in the eastern part. These shrubs can form dense maquis vegetation that can almost completely eliminate the other shrub species ([3]). Dense stands (density > 200 trees ha-1) make up 7% of the cork oak surface area, moderately dense stands (100-200 trees ha-1) account for 29%, and stands with low stocking levels (<100 trees ha-1) make up the remaining 64% ([4]). Besides forest stands, open areas occupy about 5.6% of the surface area in the Mamora forest ([4]). These include degraded cork forests and natural clearings when soil conditions do not enable trees to grow (i.e., shallow soils close to the clayey subsoil, resulting in high water tables in winter).
Since 1914, the forest has been subjected to several management programs, including restoration attempts, introduction of exotic tree species (e.g., Eucalptus camaldulensis Dehnh. and Pinus pinaster Ait. in Soland var. atlantica), and management schemes which aim to implicate local users and to regenerate cork oak by seeding or replanting. Despite all these efforts, the actual cork oak stands represent only half of the former area and many stands have fairly low stocking levels (i.e., lower than 150-200 individuals per ha). Most remaining stems have grown tall and the coppice can be characterized as overstood; some even have several large stems on one stool.
Sampling strategy
A total of 21 stands were investigated in this study, all of them being in the western part of the forest (cantons A and B of the forest, between 6° 21′ and 6° 44′ W and between 33° 59′ and 34° 18′ N - see Fig. S1 in Appendix 1). All of these stands have not experienced any cut for 63 years and the total area with cork oak in these cantons is around 25 000 ha. The soils studied are sandy, devoid of rock and with a fine fraction (<2 mm) in the order of 100% as shown by the particle size analysis. A summary description is presented in Tab. 1. In addition to forest stands, one open area was also included in the investigation, so as to be able to compare the soil carbon storage in an area deforested for a long time. In each stand, all trees present in a central area of 1 ha were measured (tree girth at breast height: C130 in cm). This survey of 4963 trees enabled us to establish the diameter distribution in our study area. We selected individual trees for tree volume assessments and regression equations based on this inventory. A first allometric relationship was built between tree height and C130 using 117 trees representing the range of tree size encountered in the whole study region (Fig. S2 in Appendix 1). Out of these 117 trees, we selected 34 trees which were representative of our 21 studied stands. As Moroccan law does not allow trees to be sampled destructively in the protected cork oak stands in Mamora, we used an indirect method of geometrical cubing, considering that one stem belonged to several successive geometrical types ([72]). Tree volumes were thus determined by cubing all the subsequent boles. To this end, all of the 34 trees were accessed by climbing and the height and circumference of the boles were measured at 30 cm, 130 cm and then for each of the boles up to the height where the stem diameter reached 10 cm. The length and circumference of the branches were measured as well on each bole of 1 m length until the cutting diameter of 10 cm was reached.
Tab. 1 - Summary description of the study area and distribution of the stands per density class. (D): range of average tree diameters (at 130 cm) per stand using quadratic means to compute the mean diameter for each stand; (BA): Basal area; (a): of each density class in the cantons A and B, after [4]; (b): data from this study.
| Stands | Density class (stems ha-1) |
Surface area in Mamora (ha) a |
Soil pH b (H2O) |
D b (m) |
BA b (m2 ha-1) |
|---|---|---|---|---|---|
| Open area | - | 7350 | 6.1-6.9 | - | - |
| S1-S6 (n=6) | <100 | 37621 | 4.9-6.4 | 2.7-4.4 | 5.6-15.1 |
| S7-S12 (n=6) | 100-200 | 16665 | 4.9-6.4 | 3.1-4.3 | 7.8-14.4 |
| S13-S21 (n=9) | >200 | 4110 | 4.9-6.4 | 3.4-5.8 | 9.0-26.7 |
For the understorey vegetation, in each central area of 1 ha of each stand, 25 squares of 1 m2 were established at random. In each of these squares, the entire aboveground tissue of understorey species was harvested. Plants were weighed per species after drying at 65°C to constant weight. Finally, the plant material was cut into fine pieces and then ground before laboratory analyses. In order to estimate belowground biomass of the understorey vegetation, we harvested 10 individuals per understorey species completely (both aboveground and belowground). This was used to compute an average root/shoot ratio. The main understorey species were Chamaerops humilis, Thymelaea lythroides and Genista linifolia.
For the study of litter and soil, sampling was restricted to a plot of 0.25 ha located in the central 1 ha area used to survey the trees. This 0.25 ha plot was further subdivided into 25 subplots of 100 m2 each. In the middle of each one of these 25 subplots, a 1 m2 square was established for litter collection, followed by soil sampling down to a soil depth of 1 meter. The depth of 1 meter was based on preliminary observations showing that organic matter was mostly located in the uppermost horizons of the soil. For the litter, all of the litter from each 1 m2 square was collected, dried and weighed. For the soil, a soil core was taken and divided into five soil layers (0-20, 20-40, 40-60, 60-80, 80-100 cm). Both for the litter and the five soil depth layers, the 25 individual samples for the 0.25 ha plot were used to compose one composite sample per layer for the 0.25 ha plot (equal weight basis). These composite samples were oven-dried at 65°C to constant weight, then ground and sieved at 2 mm. Soil bulk density was assessed on one soil pit per 0.25 ha stand using density cylinders (Ø 5.5 cm, length 10.5 cm). To this end, a cylinder was inserted horizontally into each soil layer from the pit wall with a rubber hammer. The cylinder was then gently removed with a knife and excess soil was discarded. The density cylinders were emptied into sample bags and the weight was assessed for each sample after drying at 65 °C to constant weight, from which the soil bulk density (kg dm-3) was calculated.
For the soil samples, the organic carbon content was assessed by the acid oxidation method ([91]). It has been suggested that the above method used to determine soil carbon can lead to underestimates of organic carbon contents, but this bias has been shown to be lower for soils under dry conditions ([64], [89]). In order to check this potential bias, we first established a representative subset of soil samples and compared the Walkley & Black’s method with the method by Anne ([5]) and a method based on loss on ignition (600 °C for four hours). The three methods gave very comparable results for our conditions and consequently, for the entire set of soil samples, we applied the Walkley & Black’s method on a routine basis. For organic carbon analysis of the litter samples and plant samples, an aliquot of 2 g was combusted at 600 °C for four hours in a muffle furnace to estimate the organic matter content. The amount of carbon was then computed by dividing the organic matter content by a coefficient of 1.73 (after [15]).
Data handling and statistics
First, regression equations were used to estimate the aboveground tree volume and for this the circumference at breast height (C130 in cm) was used ([77], [25]). The equations were performed with SAS™ (JMP© 9.0.0 - SAS Institute, Cary, NC, USA) and included polynomial and logarithmic relationships with one (circumference C130) or two (circumference C130 and height H) entries (see Tab. S2 in Appendix 1 for the equations tested for the volume estimations). The choice of the final model was based on the following parameters: coefficient of determination (R2), residual standard error (Sy,x), Fisher’s F-test and Furnival’s indice (IF), mean relative errors for cubing (e) and standard deviation of error distribution for cubing (Se). The models were adjusted for the tree volume of wood, thick branches (Ø > 10 cm) and cork using the data from the 34 measured trees. In total eight equations were tested (Tab. S1 in Appendix 1). The statistical analysis of these models showed that the logarithmic models performed better, and furthermore they have the advantage of being easily transferable to other contexts and comparable to other studies. Therefore, in our study, the models retained were (eqn. 1):
for the one entry model, and for the two entry model (eqn. 2):
where Vtree is the tree volume (in m3), C the circumference at 130 cm (in m) and H the tree height (in m). Model performance for both equations was good and comparable (see Tab. S2 in Appendix 1 for the summary parameters for all eight equations, and in Fig. S3 in Appendix 1 for the residuals of eqn. 1 and eqn. 2).We finally preferred to retain the equation using only the circumference (eqn. 1) as the tree heights could not be assessed on a routine basis for all trees in all of the stands. As destructive measurements in the stand were not possible, we could not sample cork separately from wood. Likewise, we were unable to consider the small branches and leaves directly by the tree climbing and cubing method. Therefore, we chose to use an equation for cork biomass from the work by Makhloufi et al. ([61]) for nearby Quercus suber forests on hilly sites north of the study area in Morocco, and equations by Ruiz-Peinado et al. ([83]) from comparable ecoystems in Spain for the branches beyond our threshold of 10 cm diameter as well as foliage. The diameter and tree height ranges used in these two studies were well matched with those of this investigation. In particular, for the cork bark material we used the equation from Makhloufi et al. ([61]) - eqn. 3:
and (eqn. 4):
where Vcork is the volume of the cork (equation from [61], in dm3), C130 the circumference at 130 cm (in cm), Vstem-without-bark is the tree volume without bark (m3), and Vtree the tree volume of wood, thick branches and cork (in m3 from eqn. 1). The corresponding biomass values for both compartments were then computed as follows (eqn. 5):
and (eqn. 6):
Tab. 2 - Specific tissue density and mean carbon concentration of the tissues.
| Species | Plant component |
Mean carbon concentration (g kg-1) |
Specific tissue density (kg m-3) |
|---|---|---|---|
| Quercus suber | Wood of the trunk and thick branches (Ø > 10 cm) | 560 | 830 |
| Medium branches (2 ≤ Ø ≤ 10 cm) | 540 | - | |
| Small branches (Ø < 2 cm) | 530 | - | |
| Foliage | 520 | - | |
| Cork | 560 | 540 | |
| Roots | 540 | - | |
| Understorey | - | - | |
| Thymelea lythroides | Aboveground | 520 | - |
| Belowground | 450 | - | |
| Genista linifolia | Aboveground | 540 | - |
| Belowground | 530 | - | |
| Chamaerops humilis | Aboveground | 530 | - |
| Belowground | 550 | - |
where Bcork the biomass of cork (kg), Vcork the volume of cork (m3), Bstem-without-bark is the biomass of the stem and coarse branches without bark (kg), Vstem-without bark the volume of the stem without bark (m3) and D the specific tissue density (Tab. 2, kg m-3). For the medium branches and small branches including their foliage, we used the eqn. 7 and eqn. 8 from Ruiz-Peinado et al. ([83]):
and (eqn. 8):
where Bmedium-branch is the biomass of medium branches (diameter 2-10 cm) in kg, Bsmall-branch-foliage the biomass of small branches (< 2 cm diameter) and supported foliage (kg), D130 the diameter at breast height (in cm) and H the tree height (in m). For trees whose height was not measured directly, tree heights were derived from the relationship between H and C130 (Fig. S2 in Appendix 1). Then we computed tree biomass for all the aboveground tissues (eqn. 9) on an individual tree basis by adding the biomass of medium branches, small branches and foliage (from eqn. 7 and eqn. 8) to the biomass of trunk and branches without cork and the biomass of this cork (from eqn. 5 and eqn. 6), as follows (eqn. 9):
where Btree-ABG is the tree biomass (in kg), Bstem-without-bark the biomass of the stem and coarse branches without bark (kg), Bcork the biomass of cork (kg), Bmedium-branch is the biomass of medium branches (kg) and Bsmall-branch-foliage the biomass of small and supported foliage (kg). Then for the carbon contained in the aboveground biomass of the cork oak trees we applied the following equation (eqn. 10):
where Cst-ABG is the carbon stock (in kg) for an individual tree, 10-3 is a conversion factor, Btree-ABG is the aboveground biomass (in kg) for the respective components (i.e., cork, stem without bark, medium-branches, small branches and foliage) and Cconc-ABG is the mean carbon concentration of the respective aboveground tissues (in g kg-1 - values from Tab. 2). For the carbon content in the belowground tissues of trees, we used the equation developed by Ruiz-Peinado et al. ([83]) to estimate the root biomass, and multiplied this by the mean carbon concentration (eqn. 11):
where Btree-BLG is the belowground biomass of an individual tree in kg and D130 the diameter at breast height (130 cm) in cm. Combined with the mean carbon concentration, this can be written as (eqn. 12):
where Cst-BLG is the carbon stock in the belowground tissues of an individual tree (in kg), 10-3 is a conversion factor and Cconc-BLG the average carbon concentration in belowground tissues (in g kg-1 - Tab. 2).
Then for each stand, the total carbon content in aboveground (Cst-ABG-Stand) and belowground (Cst-BLG-Stand) tissues was calculated as the sum of the carbon contents of all the individual trees (eqn. 13):
where Cst-QUERCUS is the total carbon content in the Quercus suber trees of each entire stand (Mg ha-1), and Cst-ABG and Cst-BLG are the carbon contents in aboveground and belowground parts of each tree (kg), respectively.
For the understorey species, firstly dry weights per species for the fully harvested aboveground tissues were assessed after drying. Based on specific root/shoot values (see above), we estimated the corresponding root biomass. Then average biomass values were computed for each understorey species at the stand scale and multiplied by the average carbon concentration for each component to assess the carbon storage of the understorey (eqn. 14):
where Cst-Understorey is the carbon stock in the understorey vegetation (Mg ha-1), 10-3 is a conversion factor, BABG-sp1 is the biomass of the aboveground tissues of species 1 (kg ha-1), BBLG-sp1 is the biomass of the belowground tissues of species 1 (kg ha-1) and Cconc the average carbon concentration for the respective tissues (specific to species and component - g kg-1).
For the carbon stocks in the litter we used the following equation (eqn. 15):
where Cst-LITTER is the carbon stock (Mg ha-1), 10-3 is a conversion factor, MLITTER is the dry mass (g m-2) obtained from the harvested litter for each 1 m2 square, and Cconc-LITTER the mean carbon concentration for each plot (g kg-1). Carbon stocks in the different soil horizons were computed using three parameters ([12], [73], [40], [23] - eqn. 16):
where Cst-SOIL-layer is the carbon content in a given soil layer (Mg ha-1), 0.1 is a conversion factor, ThicknessLayer is the thickness of this soil layer (cm), BD is the bulk soil density (g cm-3) and Cconc-SOIL is the carbon concentration (g kg-1). Then, for each plot the total carbon content in the soil down to 1 m of soil depth was calculated as the sum of the carbon contents of all five soil depth layers (eqn. 17):
where Cst-SOIL is the carbon content of the soil down to 1 m of soil depth (Mg ha-1) and Cst-soil-layer is the carbon content of individual layers (Mg ha-1). In order to evaluate the carbon storage in our ecosystem, relationships between the overall parameters of stand density and basal area on the one hand, and carbon stocks in the trees (Cst-QUERCUS), the understorey (Cst-UNDERSTOREY), the soil (Cst-SOIL) and the litter (Cst-LITTER) on the other hand, were explored.
Results
Carbon storage in cork oak trees
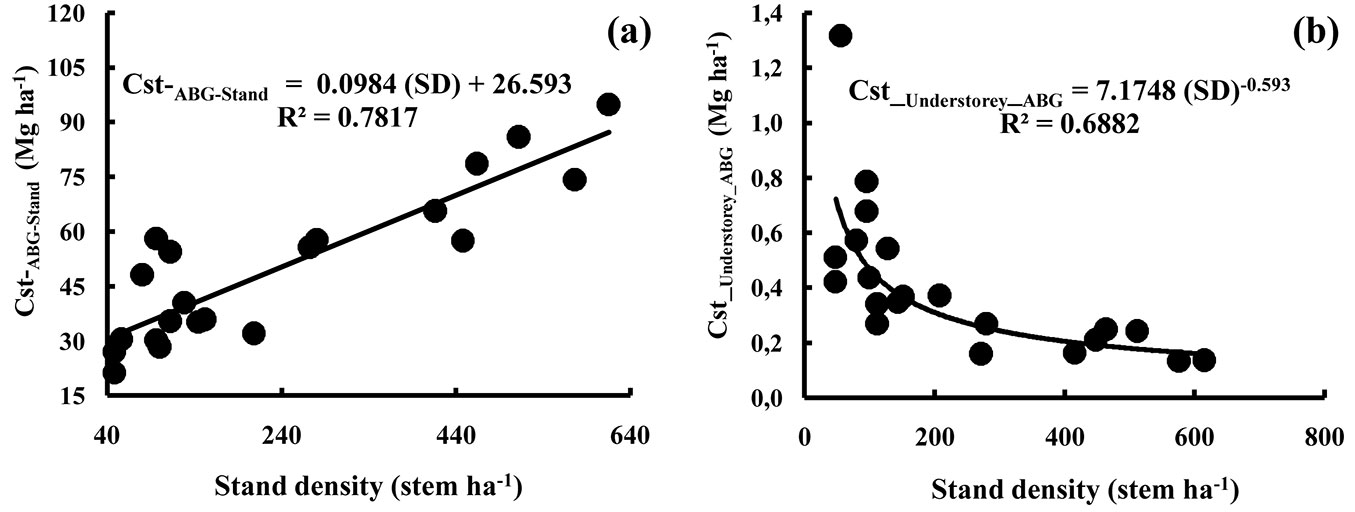
The structure of the cork oak stands considered in this work was regular with a modus occuring in the 70 cm circumference class, but no stems had a circumference less than 30 cm. This is representative of the overall structure of cork oak forest in the Mamora region. Small circumference individuals nearly only occur in regeneration plots protected by a fence, while very large individuals with diameters beyond 200 cm are uncommon (Fig. S4 in Appendix 1). The biomass at the stand scale ranged from 64.3 to 120.1 ha-1 for aboveground tissues and 10-20 Mg ha-1 for belowground tissues (Tab. 3). The overall averages were 89.5 Mg ha-1 for aboveground biomass (range of 38.1-170.2 Mg ha-1) and 14.6 Mg ha-1 for belowground biomass (range 5.9-28.2 Mg ha-1 - Tab. 3). This represented average carbon stocks of 35.8-66.9 Mg ha-1 for aboveground tissues and 5.4-10.8 Mg ha-1 for belowground tissues (Tab. 3). The lowest value for carbon stocks occurred in the rather open stand S4 (21.3 Mg ha-1 aboveground, 3.2 Mg ha-1 belowground), while the highest value was recorded in the dense stand S18 (94.8 Mg ha-1 aboveground, 15.2 Mg ha-1 belowground). For the aboveground tissues, on average 14.7% of all stored carbon was found within the cork, 74.1 % in the trunk and thick branches, 8.3 % in medium branches and 2.9 % in small branches and foliage (Tab. 3). The belowground biomass represented around 14% of total stand biomass. Fig. 1a shows the relationship between stand density and the total carbon stocks per stand present in the aboveground tissues of the cork oak trees (Cst-ABG-Stand). The correlation was fairly strong (R2 = 0.78). Despite some dimensional variation between individual trees, having a higher tree density correlated well with the total carbon stocks in the aboveground tissues. Had there been very young (small diameter, small height and small biomass) dense stands in Mamora forest, the relationship might not have been valid for such stands. The relationship can thus be used under our forest conditions (stand structure, age, dimensions) to estimate carbon storage in the aboveground parts of the cork oak trees.
Tab. 3 - Biomass and carbon stocks in the cork oak stands. (D): stand density of the cork oak stand (stem ha-1); (BA): basal area of the stand (m2 ha-1); (ABG): aboveground tissues; (BLG): belowground tissues; (C): carbon stock in the cork; (TTB): carbon stock in trunk and thick branches (Ø > 10 cm) without cork; (MB): carbon stock in medium branches (2 ≤ Ø ≤ 10 cm); (SBF): carbon stock in small branches (Ø < 2 cm) and foliage; (Σ): sum of carbon in the aboveground tissues.
| Density class (stem ha-1) |
Stand | BA (m2 ha-1) |
Stand biomass (Mg ha-1) | Stand carbon (Mg ha-1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ABG | BLG | Total | ABG | BLG | Total | |||||||
| C | TTB | MB | SBF | Σ | ||||||||
| D<100 | S1 | 8.04 | 54.63 | 8.49 | 63.12 | 4.2 | 23.5 | 2.1 | 0.7 | 30.5 | 4.6 | 35.1 |
| S2 | 15.14 | 104.00 | 15.99 | 119.99 | 8.0 | 45.0 | 3.7 | 1.3 | 58.0 | 8.6 | 66.6 | |
| S3 | 8.21 | 54.24 | 8.67 | 62.91 | 4.3 | 22.8 | 2.3 | 0.8 | 30.2 | 4.7 | 34.9 | |
| S4 | 5.63 | 38.13 | 5.95 | 44.08 | 3.0 | 16.3 | 1.5 | 0.5 | 21.3 | 3.2 | 24.5 | |
| S5 | 12.6 | 86.23 | 13.31 | 99.54 | 6.6 | 37.2 | 3.1 | 1.1 | 48.0 | 7.2 | 55.3 | |
| S6 | 7.15 | 48.41 | 7.56 | 55.97 | 3.8 | 20.7 | 1.8 | 0.7 | 27.0 | 4.1 | 31.1 | |
| Mean | - | 64.27 | 10.00 | 74.27 | 5.0 | 27.6 | 2.4 | 0.9 | 35.8 | 5.4 | 41.2 | |
| 100≤D≤200 | S7 | 9.75 | 63.14 | 10.30 | 73.44 | 5.2 | 26.1 | 2.9 | 1.0 | 35.2 | 5.6 | 40.8 |
| S8 | 7.77 | 50.88 | 8.21 | 59.09 | 4.1 | 21.2 | 2.2 | 0.8 | 28.3 | 4.4 | 32.8 | |
| S9 | 9.94 | 64.39 | 10.50 | 74.89 | 5.3 | 26.6 | 3.0 | 1.0 | 35.9 | 5.7 | 41.6 | |
| S10 | 8.04 | 72.55 | 11.69 | 84.24 | 5.9 | 30.3 | 3.1 | 1.1 | 40.4 | 6.3 | 46.7 | |
| S11 | 9.59 | 63.59 | 10.13 | 73.72 | 5.1 | 26.8 | 2.7 | 0.9 | 35.5 | 5.5 | 40.9 | |
| S12 | 14.45 | 97.62 | 15.26 | 112.88 | 7.6 | 41.7 | 3.8 | 1.3 | 54.4 | 8.2 | 62.6 | |
| Mean | - | 68.70 | 11.02 | 79.71 | 5.5 | 28.8 | 2.9 | 1.0 | 38.3 | 5.9 | 44.2 | |
| D>200 | S13 | 12.11 | 103.56 | 16.97 | 120.53 | 8.5 | 42.6 | 4.9 | 1.7 | 57.7 | 9.2 | 66.9 |
| S14 | 18.56 | 117.78 | 19.67 | 137.45 | 9.9 | 47.6 | 6.0 | 2.1 | 65.6 | 10.6 | 76.2 | |
| S15 | 24.3 | 154.32 | 25.66 | 179.98 | 12.9 | 62.6 | 7.7 | 2.7 | 85.9 | 13.9 | 99.9 | |
| S16 | 16.57 | 103.28 | 17.50 | 120.78 | 8.8 | 41.1 | 5.6 | 2.0 | 57.5 | 9.5 | 67.0 | |
| S17 | 15.49 | 100.01 | 16.36 | 116.37 | 8.2 | 41.2 | 4.7 | 1.7 | 55.8 | 8.8 | 64.5 | |
| S18 | 26.73 | 170.19 | 28.23 | 198.42 | 14.2 | 69.1 | 8.5 | 3.0 | 94.8 | 15.2 | 110.0 | |
| S19 | 22.29 | 141.11 | 23.54 | 164.65 | 11.8 | 57.1 | 7.1 | 2.5 | 78.5 | 12.7 | 91.3 | |
| S20 | 21.32 | 133.31 | 22.52 | 155.83 | 11.4 | 53.2 | 7.1 | 2.5 | 74.2 | 12.2 | 86.4 | |
| S21 | 8.99 | 57.53 | 9.56 | 67.09 | 4.8 | 23.3 | 2.9 | 1.0 | 32.0 | 5.2 | 37.2 | |
| Mean | - | 120.12 | 20.00 | 140.12 | 10.1 | 48.6 | 6.0 | 2.1 | 66.9 | 10.8 | 77.7 | |
Fig. 1 - Relationships between stand density and total carbon stocks per stand present in the aboveground tissues. Diagrams refer to: (a) Quercus suber; (b) understorey. (Cst_ABG_Stand): total carbon stock per stand present in the aboveground tissues of the cork oak trees (Mg ha-1); (Cst_Understorey_ABG): total carbon stock per stand present in the aboveground tissues of understorey (Mg ha-1); (SD): stand density (stem ha-1).
Carbon storage in the understorey of the cork oak stands
Understorey vegetation was present in variable quantities in the 21 stands investigated, i.e., very degraded stands where understorey was nearly absent co-occurred with stands still having a fairly well developed understorey. Over larger areas typically the understorey vegetation was either nearly absent or rather homogenous as distribution and dimensions. Three species were dominant in the inventory, and these represented in total between 0.5 and 4.8 Mg ha-1 of biomass (Tab. 4), with an average of 1.6 Mg ha-1. The average was 0.85 Mg ha-1 for the densest stands and 2.62 Mg ha-1 for the most open stands. Flax-leaf broom was the species contributing most to the total biomass. Understorey represented 0.4 to 2.3 % of total stand aboveground biomass for cork oak stands with stand densities over 200 stems ha-1 or less than 100 stems ha-1, respectively. The respective carbon stocks in the understorey (aboveground and belowground) ranged from 0.3 to 2.9 Mg ha-1 with an average of 1.0 Mg ha-1 (Tab. 4). Fig. 1b shows the relationship between stand density and the total carbon stocks per stand present in the aboveground tissues of the understorey (Cst-Understorey-ABG). This shows that the stock of carbon in the aboveground biomass of the understorey is inversely correlated to the cork oak stand density (with R2 = 0.69).
Tab. 4 - Biomass and carbon stocks in the understorey of the cork oak stands. (ABG): aboveground tissues; (BLG): belowground tissues; (D): stand density of the cork oak stand (stem ha-1); (*): belowground biomass was computed with a mean root : shoot ratio (R/S) established on 10 uprooted individuals for each main understory species, yielding values of R/S of 35% for G. linifolia, 49% for T. lytrhoides and 93% for C. humilis.
| Density Class (stem ha -1) |
Stand | Total biomass ABG + BLG (Mg ha-1)* | Total carbon ABG + BLG (Mg ha-1) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Genista linifolia | Thymelaea lythroides | Chamearops humilis | Σ understory |
Genista linifolia | Thymelea lythroides | Chamearops humilis | Σ understory |
||
| D<100 | S1 | 2.50 | 1.62 | 0.71 | 4.83 | 1.34 | 1.14 | 0.39 | 2.87 |
| S2 | 1.49 | 0.97 | 0.43 | 2.89 | 0.80 | 0.68 | 0.23 | 1.72 | |
| S3 | 1.29 | 0.83 | 0.37 | 2.49 | 0.69 | 0.59 | 0.20 | 1.48 | |
| S4 | 0.97 | 0.63 | 0.28 | 1.87 | 0.52 | 0.44 | 0.15 | 1.11 | |
| S5 | 1.09 | 0.70 | 0.31 | 2.10 | 0.58 | 0.50 | 0.17 | 1.25 | |
| S6 | 0.80 | 0.52 | 0.23 | 1.55 | 0.43 | 0.36 | 0.13 | 0.92 | |
| Mean | 1.36 | 0.88 | 0.39 | 2.62 | 0.73 | 0.62 | 0.21 | 1.56 | |
| 100≤D≤200 | S7 | 0.00 | 1.29 | 0.50 | 1.79 | 0.00 | 0.91 | 0.27 | 1.18 |
| S8 | 0.00 | 1.62 | 0.62 | 2.24 | 0.00 | 1.14 | 0.34 | 1.49 | |
| S9 | 0.94 | 0.00 | 0.97 | 1.92 | 0.51 | 0.00 | 0.53 | 1.04 | |
| S10 | 1.47 | 0.00 | 0.72 | 2.19 | 0.79 | 0.00 | 0.39 | 1.18 | |
| S11 | 0.73 | 0.00 | 0.36 | 1.08 | 0.39 | 0.00 | 0.20 | 0.59 | |
| S12 | 0.92 | 0.00 | 0.45 | 1.37 | 0.49 | 0.00 | 0.25 | 0.74 | |
| Mean | 0.68 | 0.49 | 0.60 | 1.77 | 0.36 | 0.34 | 0.33 | 1.04 | |
| D>200 | S13 | 0.08 | 0.92 | 0.15 | 1.15 | 0.04 | 0.65 | 0.08 | 0.78 |
| S14 | 0.05 | 0.56 | 0.09 | 0.70 | 0.03 | 0.39 | 0.05 | 0.47 | |
| S15 | 0.07 | 0.83 | 0.14 | 1.04 | 0.04 | 0.58 | 0.08 | 0.70 | |
| S16 | 0.06 | 0.72 | 0.12 | 0.90 | 0.03 | 0.51 | 0.07 | 0.61 | |
| S17 | 0.05 | 0.55 | 0.09 | 0.69 | 0.03 | 0.38 | 0.05 | 0.46 | |
| S18 | 0.04 | 0.47 | 0.08 | 0.59 | 0.02 | 0.33 | 0.04 | 0.39 | |
| S19 | 0.64 | 0.07 | 0.13 | 0.85 | 0.34 | 0.05 | 0.07 | 0.47 | |
| S20 | 0.34 | 0.04 | 0.07 | 0.45 | 0.18 | 0.03 | 0.04 | 0.25 | |
| S21 | 0.93 | 0.15 | 0.20 | 1.28 | 0.50 | 0.11 | 0.11 | 0.71 | |
| Mean | 0.25 | 0.48 | 0.12 | 0.85 | 0.14 | 0.34 | 0.07 | 0.54 | |
Carbon storage in the litter and soil of the cork oak stands
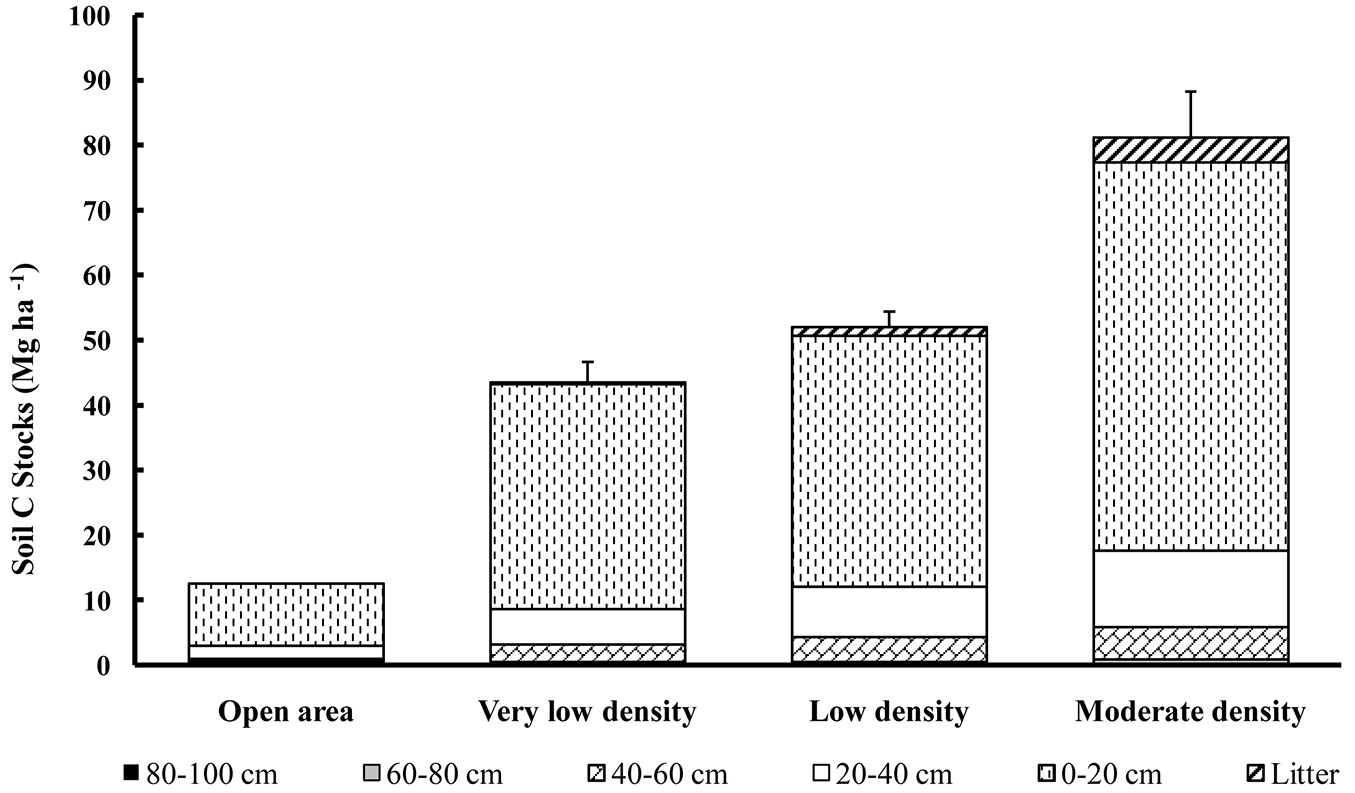
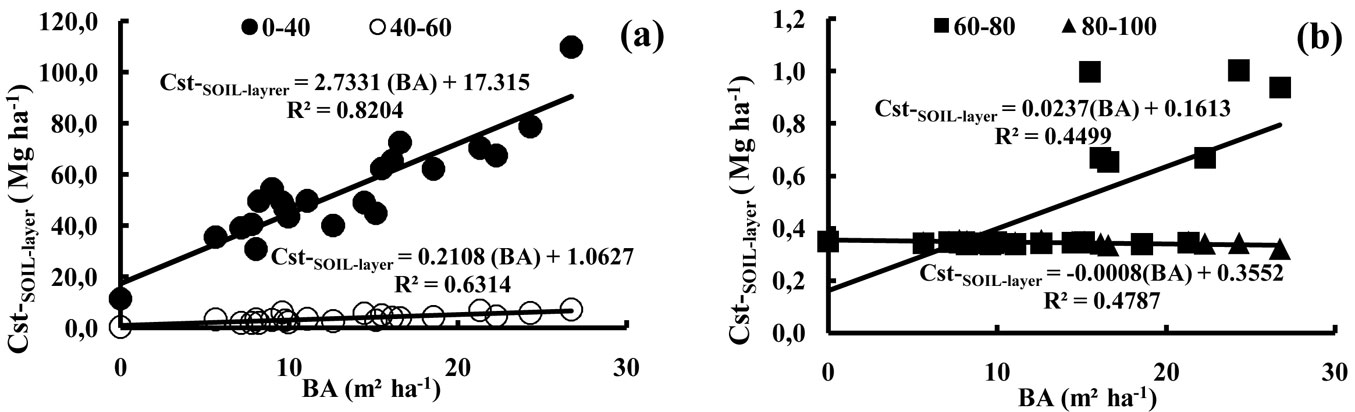
The carbon concentrations in the litter and soils varied from 540 to 560 g kg-1 for the litter and between 0.1 to 32 g kg-1 for the soil (Tab. 5). The lowest organic carbon concentrations were observed at the deepest soil depth (i.e., 80-100 cm soil depth), although these low values of 0.1 g kg-1 were attained in some stands from 60 cm downwards and in the open area from 40 cm downwards (Tab. 5). The lowest carbon concentrations in the top soil layers (i.e., 0-20 cm soil depth) were found in the stands with the lowest stand density (with values ranging between 9.7-14.3 g kg-1 and a mean value of 11.6 g kg-1). Elsewhere, the carbon concentrations of this soil depth layer were intermediate (18.5 g kg-1). The values for the open area were very low, even in the top soil layer (3 g kg-1 or lower). The minimum and maximum organic carbon stocks at the stand scale ranged between 0.2-8.1 Mg ha-1 of carbon for the litter and 34.8-118.2 Mg ha-1 for the soil (0-100 cm). Fig. 2 displays the average carbon stocks per ha for each stand density class. The total stocks of organic carbon in the soils of our study (total of 0-100 cm soil depths) varied on average from 12.5 to 81.2 Mg ha-1 for the open area and the highest stand density class, respectively (Fig. 2), with an overall average of 58 Mg ha-1. With the exception of stand S18 (118 Mg ha-1), the organic carbon stocks in the first 100 cm of the soil did not exceed 86 Mg ha-1. The relative importance of the different soil layers is given in Tab. 6. Soil depth appeared to be the most important factor explaining the observed differences. With the exception of stand S18, the carbon stocks within the first 40 cm of the soil profile did not exceed 78 Mg ha-1. In all stands, more than 72% of the total carbon stocks were present within the first 20 cm of the soil. Then, for the first 40 cm of soil, this value was over 88% of the total carbon stocks in the soil (Tab. 6). For the investigated stands, the total organic carbon stocks (in 0-40 cm and in 40-60 cm soil depths) were dependent on the total basal area of the stands (Fig. 3a). In contrast, the carbon stocks of the 60-80 and 80-100 cm soil depths did not show any significant relationship with basal area (Fig. 3b).
Tab. 5 - Carbon concentrations in the litter and the soil at different soil depths. (D): stand density of the cork oak stand (stem ha-1).
| Density class (stem ha-1) |
Stand | Litter (g kg-1) |
Soil depth (cm) | ||||
|---|---|---|---|---|---|---|---|
| 0-20 (g kg-1) |
20-40 (g kg-1) |
40-60 (g kg-1) |
60-80 (g kg-1) |
80-100 (g kg-1) |
|||
| D=0 | Open area | - | 3.0 | 0.6 | 0.1 | 0.1 | 0.1 |
| D<100 | S1 | 550 | 9.7 | 0.7 | 1.0 | 0.1 | 0.1 |
| S2 | 560 | 12.0 | 2.5 | 0.9 | 0.1 | 0.1 | |
| S3 | 550 | 14.3 | 2.9 | 0.6 | 0.1 | 0.1 | |
| S4 | 540 | 10.8 | 1.1 | 1.0 | 0.1 | 0.1 | |
| S5 | 550 | 11.0 | 2.2 | 0.8 | 0.1 | 0.1 | |
| S6 | 550 | 12.0 | 0.8 | 0.6 | 0.1 | 0.1 | |
| Mean | 550 | 11.6 | 1.7 | 0.8 | 0.1 | 0.1 | |
| 100≤D≤200 | S7 | 550 | 12.5 | 3.4 | 0.9 | 0.1 | 0.1 |
| S8 | 540 | 11.3 | 2.1 | 0.6 | 0.1 | 0.1 | |
| S9 | 550 | 12.1 | 3.2 | 0.7 | 0.1 | 0.1 | |
| S10 | 550 | 15.3 | 1.9 | 1.1 | 0.1 | 0.1 | |
| S11 | 550 | 14.8 | 2.3 | 1.8 | 0.1 | 0.1 | |
| S12 | 550 | 14.6 | 2.1 | 1.7 | 0.1 | 0.1 | |
| Mean | 550 | 13.4 | 2.5 | 1.1 | 0.1 | 0.1 | |
| D>200 | S13 | 550 | 21.0 | 2.6 | 1.3 | 0.2 | 0.1 |
| S14 | 550 | 17.8 | 4.5 | 1.4 | 0.1 | 0.1 | |
| S15 | 560 | 23.7 | 4.1 | 1.8 | 0.3 | 0.1 | |
| S16 | 550 | 23.4 | 3.7 | 1.3 | 0.2 | 0.1 | |
| S17 | 540 | 20.1 | 2.5 | 1.6 | 0.3 | 0.1 | |
| S18 | 540 | 32.3 | 9.2 | 2.5 | 0.3 | 0.1 | |
| S19 | 550 | 21.4 | 2.9 | 1.5 | 0.2 | 0.1 | |
| S20 | 540 | 20.6 | 4.3 | 2.1 | 0.1 | 0.1 | |
| S21 | 550 | 17.0 | 2.3 | 1.0 | 0.1 | 0.1 | |
| Mean | 550 | 21.9 | 4.0 | 1.6 | 0.2 | 0.1 | |
Fig. 2 - Mean organic carbon stocks (Mg ha-1) in the litter and in the first 100 cm of the soil. Error bars represent the standard error.
Tab. 6 - Relative contribution to the total soil organic carbon pool of the five soil layers studied.
| Density class (stem ha-1) |
Stand | Relative contribution per soil depth (cm) | ||||
|---|---|---|---|---|---|---|
| 0-20 (%) |
20-40 (%) |
40-60 (%) |
60-80 (%) |
80-100 (%) |
||
| D=0 | Open area | 75.8 | 15.8 | 2.7 | 2.8 | 2.8 |
| D<100 | S1 | 81.9 | 6.6 | 9.5 | 1.0 | 1.0 |
| S2 | 75.6 | 16.7 | 6.3 | 0.7 | 0.7 | |
| S3 | 77.7 | 17.2 | 3.7 | 0.6 | 0.7 | |
| S4 | 80.8 | 9.0 | 8.5 | 0.9 | 0.9 | |
| S5 | 75.9 | 16.3 | 6.2 | 0.8 | 0.8 | |
| S6 | 87.1 | 6.4 | 4.8 | 0.8 | 0.8 | |
| Mean | 79.8 | 12.0 | 6.5 | 0.8 | 0.8 | |
| 100≤D≤200 | S7 | 71.6 | 21.1 | 5.9 | 0.7 | 0.7 |
| S8 | 77.9 | 15.8 | 4.7 | 0.8 | 0.8 | |
| S9 | 72.6 | 21.1 | 4.8 | 0.7 | 0.7 | |
| S10 | 81.0 | 11.0 | 6.7 | 0.6 | 0.6 | |
| S11 | 75.3 | 12.6 | 10.9 | 0.6 | 0.6 | |
| S12 | 76.3 | 12.0 | 10.4 | 0.6 | 0.6 | |
| Mean | 75.8 | 15.6 | 7.2 | 0.7 | 0.7 | |
| D>200 | S13 | 81.8 | 11.1 | 5.7 | 0.9 | 0.5 |
| S14 | 73.2 | 19.4 | 6.3 | 0.5 | 0.5 | |
| S15 | 77.7 | 13.9 | 6.8 | 1.2 | 0.4 | |
| S16 | 79.8 | 13.9 | 5.1 | 0.8 | 0.4 | |
| S17 | 79.6 | 11.1 | 7.4 | 1.4 | 0.5 | |
| S18 | 71.0 | 21.8 | 6.1 | 0.8 | 0.3 | |
| S19 | 80.9 | 11.4 | 6.3 | 0.9 | 0.5 | |
| S20 | 73.5 | 16.8 | 8.8 | 0.4 | 0.4 | |
| S21 | 81.3 | 12.1 | 5.4 | 0.6 | 0.6 | |
| Mean | 77.6 | 14.6 | 6.4 | 0.8 | 0.5 | |
Fig. 3 - Relationships between basal area (BA, m² ha-1) and stocks of organic carbon in the soil layers (Cst-SOIL-layer , Mg ha-1); (BA): basal area (m² ha-1).
Discussion and conclusions
General considerations
When evaluating ecosystem stocks and fluxes, the computed values probably include both spatial and temporal variations ([93]). Temporal variation - such as differences in the ecology between seasons or between years for some fluctuating parameters - seems of minor importance in our study, relative to the biomass of the cork oak stands. In the longer term, events such as cuttings or annual growth will of course change the biomass of our stands slightly, but the data of the present study are robust for the year of our inventory. The sampling effort in the present study included the complete inventory of 21 stands (central parts of 1 ha each) in the western part of Mamora forest, having a total surface area of around 25 000 ha, where cork oak is still present. The inventory consisted of all trees present in these plots, and the structure (diameter distribution) was representative of the forest area in these cantons. The equation used for assessing the tree volume gave very comparable results to the outcome reported by Makhloufi et al. ([61]) in the mountainous regions of Morocco, even though we had to restrict the entry to our model to only one parameter, i.e., the circumference at breast height. The equations used to estimate either aboveground or belowground tissues of the cork oak included several equations obtained from the literature ([83]), but these were from a similar context, were based on a similar diameter range and the values for stem and trunk biomass in both their and our equations were similar. However, as most stems in our study forest were growing from stools, it is not impossible that the equation used might lead to some underestimation of total root biomass ([90]). Overall, given that 21 stands of 1 ha were sampled in the area, we are confident that the biomass values obtained for these stands are valid in the experimental context of the Mamora forest, and that the estimates do not suffer from a strong spatial variation.
As stated by Augusto et al. ([8]), rather than choosing allometric relationships from the literature or established for the study, there is a need to ensure that nutrient concentration values obtained locally are used, due to the higher uncertainty when using those from literature. Accordingly, we used locally-obtained carbon concentrations to compute our carbon stocks. Overall, the sampling effort and prediction for the cork oak volume, biomass and carbon content seem adequate and the obtained data for biomass and carbon storages were in line with those from the literature (see below). The understorey was inventoried on a complete basis (extensively in all the 21 stands) for the aboveground biomass, and 10 individuals per species were harvested to establish species-specific root/shoot ratios. Likewise, carbon concentrations were assessed for all of the plant species-compartment combinations. As a consequence, the sampling effort seems appropriate for this compartment of the ecosystem in our 21 stands. Outside our stands, variation in the understorey may be somewhat larger; in particular, some more species can be expected, such as the Mamora pear or the mastic tree.
Soils and litter were sampled with a two-step sampling strategy, with 1 sample every 100 m2 subplot before generating the composite samples used for analysis, yielding 25 individual samples and one composite sample per layer. This should overcome problems of spatial variability ([93]), and seems an adequate sampling intensity for our conditions ([92]). However, some temporal variability must occur, notably for the litter samples, as this layer is the most exposed to microclimatic effects (including fast oxidation of organic matter - [57]). Overall, we feel confident in the values produced for the carbon stocks in the ecosystem of our experimental context.
Carbon in trees and understorey vegetation
In our study the values of aboveground biomass per tree ranged from 26 to 2177 kg (mean value of 379 kg). Such range fairly overlaps those reported by Makhloufi et al. ([61]) for Morocco (range: 11-1255) and by Ruiz-Peinado et al. ([83]) for Spain (range: 11-2412 kg). In this study, the 38.1-170.2 Mg ha-1 for the aboveground biomass of cork oak stands is fairly comparable to the mean biomass of 34 and 183 Mg ha-1 for Quercus ilex stands respectively in the high and middle Atlas in Morocco ([18]). The respective carbon stocks in the aboveground parts of the cork oak vary from 21.3 to 94.8 Mg ha-1. To our knowledge, this is the first work reporting carbon stocks in cork oak forests. Indeed, Quercus species, such as Q. canariensis, Q. faginea and Q. suber have been poorly studied so far, despite their great ecological and economic importance ([83]). The observed range compares well with species of the same genus, such as Quercus ilex (17-91 Mg ha-1 of carbon - [19]), though these figures are much lower than those reported for mature stands of exotic fast growing species such as Pinus pinaster Ait. in the same region (166-249 Mg ha-1 of carbon - [84]).
Root biomass was reported to account for 10-40% of total tree biomass ([60]), between one fifth and one third for coniferous species ([82]) and between 15-49% for deciduous species ([83]). Commonly, root biomass is supposed to be scaled to the aboveground dimensions rather than tree age or the growing site ([31], [56]), although trees growing in dense or open stands were shown to have different root/shoot ratios ([78]). Root biomass values in our study ranged between 5.9-28.2 Mg ha-1, representing about 14% of the total tree biomass, thus close to the lower values reported for deciduous species (10-49% - [60], [83]). Regarding the carbon content in trees, roots were reported to make up between 13-15% of the carbon content of trees, though most studies state that the estimate for the belowground part is rather inprecise ([58], [7], [34], [77], [66], [71]). Indeed, despite the large size of root biomass as a carbon pool, roots remain relatively poorly studied, as the methods for their assessment is always complex and very time consuming ([81], [60], [65]). Concerning the soil, roots were also credited as being a large carbon pool ([67]), representing between 20 to 40 % of the total carbon stocks in forest soils ([49]). Here, the amount of carbon stored in the roots of cork oak varied between 3.2-15.2 Mg ha-1 for the lowest and highest stands, respectively. Such content is rather low compared with the recorded values for tropical and temperate forests (21-22 Mg ha-1 - [49]) or even boreal forests (14 Mg ha-1 - [49]).
The understorey was reported to represent between 1-7% on average of the total ecosystem aboveground biomass for temperate forests ([35], [74], [44]). Under high light conditions due to low stand density or more canopy openings, understorey biomass would be expected to be higher ([86], [6]), as more light and more resources such as water and nutrients are available for understorey primary productivity ([44]). For our cork oak stands, despite a low stand density and the presence of openings throughout the stands, the part of understorey vegetation in the total aboveground ecosystem biomass varied on average between only 0.4 to 2.3% of total stand aboveground biomass for stand densities over 200 stems ha-1 and below 100 stems ha-1, respectively. This rather low value probably reflects the low fertility and the overexploitation in this area for decades, if not centuries. In addition, part of the understorey has probably been harvested for fuel wood or charcoal production. The total carbon storage was only 0.3-2.9 Mg ha-1, with an average of 1.0 Mg ha-1. These values are lower than those obtained for Pinus pinaster stands in the south west of France, with mean carbon stocks for the understorey of 1.7 Mg ha-1 and a range of 0.7-4.1 Mg ha-1 ([75]). Under our conditions, the aboveground carbon stocks in the understorey decrease with increasing cork oak stand density, which can be explained by the competition for light between trees and the understorey.
Carbon storage in the litter and soil of the cork oak stands
The litter of the western area of the Mamora forest contained between 0.2 and 8.1 Mg ha-1 of organic carbon. These values are close to the 3.5-8.6 Mg ha-1 reported for Quercus ilex stands in Morocco ([19]). In comparison, the arenosols of European forests show higher carbon stocks in the litter layer ([34], [10]), except under a Mediterranean climate where this pool is similar in size ([10]). The rather low values for the Mamora forest can be explained by the human pressure on the area, including pasture with cattle and collection of dead woody debris (branches, twigs) as fire wood by the local population.
Carbon concentration generally decreases with increasing depth of the mineral soil ([9], [28]). This general pattern was also observed in all our stands. Furthermore, the lowest values for the topsoil were observed in the stands with the lowest stand density. As a result, both soil depth and stand density explained differences in organic carbon stocks in our stands. With the exception of 1 out of the 21 stands investigated, the carbon stocks in the top 40 cm did not exceed 78 Mg ha-1. Therefore, our values were close to those (40-70 Mg ha-1) observed by Diaz-Pinés et al. ([28]) in 50 cm of soil underneath a Quercus pyrenaica Willd. stand in the center of Spain, but inferior to the 100 Mg ha-1 reported by Sabir & Roose ([85]) for a 30 cm deep soil profile in a weakly degraded cork oak forest in the Rif mountains in Morocco. In our study, a strong decrease of carbon content with depth occurred in all stands. This result is consistent with the scientific literature, as topsoil is shown to contain the major part of the soil organic carbon pool in Mediterranean forest ecosystems ([9], [17], [19], [20], [24], [76]). The relative contribution of topsoil to the soil pool of organic carbon of our degraded Mediterranean forest (here as much as 88% of all the organic carbon was contained in the top 40 cm) was notably higher than values commonly reported for the first 100 cm of soils throughout the world ([11]). Presumably, some further carbon is present below the first 100 cm of soil, but concentrations were at least 100 times lower than in the top soil, so that likely this share of carbon overlooked in our calculations is overall very low.
The total stocks of organic carbon in the soils of our study (total of 0-100 cm soil depths) ranged from 35 to 118 Mg ha-1, with an overall average of 58 Mg ha-1. The entire forest of Mamora has been disturbed by human and animal pressure, sparing no stand density class. Therefore, the total organic carbon stocks for the top 60 cm were correlated to the total stand basal area, underlining the relationship between the soil and the trees (notably by inputs of organic carbon by aboveground- and belowground litter, which protect against erosion and leaching). The open areas have even lower organic carbon stocks, as no new organic matter inputs from trees to soil occur, and existing organic matter degrades rapidly. The average value of 58 Mg ha-1 for organic carbon stocks in the first 100 cm of the soil is clearly lower than the 136 and 152 Mg ha-1 given by Eglin ([36]) and Lecointe et al. ([55]), respectively. Soils from the Mediterranean region are relatively poor in carbon ([80]) and studies on carbon stocks in soils have shown that the organic carbon stocks may vary considerably with the soil type ([11], [32], [21], [10]). For a soil depth of 100 cm, values range from 12 Mg ha-1 for Xerosols to more than 100 Mg ha-1 for Podzols.
Total carbon storage of the cork oak ecosystems
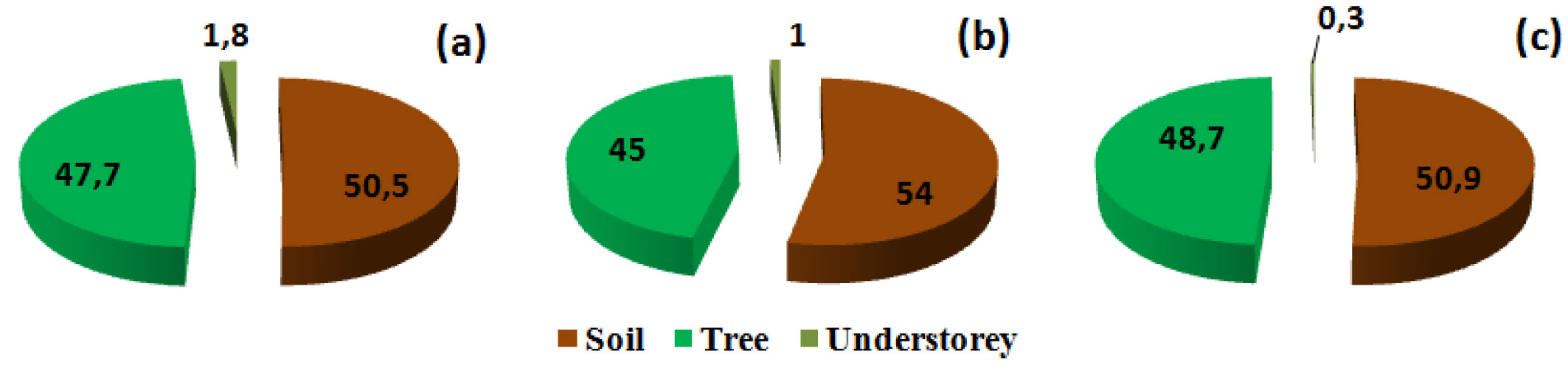
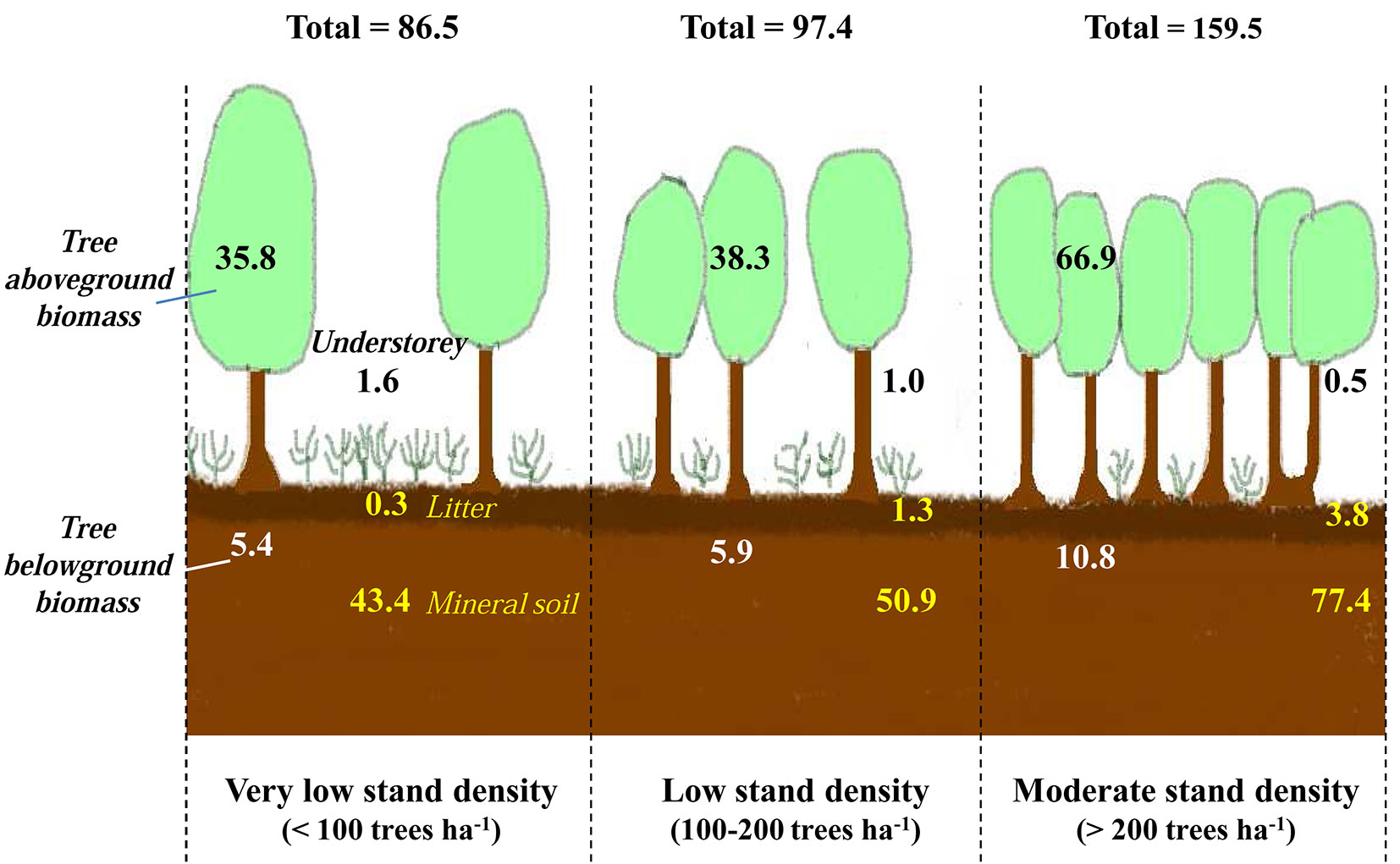
The soil is a key reservoir for carbon and may contain up to two-thirds of the total ecosystem carbon stocks ([30], [63]). This has also been demonstrated in Mediterranean forest ecosystems ([84] and references therein). In our context, for the three stand density classes considered, the first 100 cm of the soil (including the forest floor) represented the major storage compartment of this ecosystem, with roughly 51% of the total organic carbon stocks (Fig. 4). The biomass of the cork oak, including aboveground and belowground tissues, constituted the second largest pool of carbon (more than 47 %) and the contribution of the understorey to the total organic carbon stocks was less than 2% (Fig. 4).
Fig. 4 - Contribution of the three components of the ecosystem (tree, understorey and soil) to total carbon stocks. (a): lowest stand density class (D<100 stems ha-1); (b): moderate density class (100<D< 200 stems ha-1); (c) highest density class (> 200 stems ha-1).
In holm oak (Quercus ilex) ecosystems from the middle Atlas Mountains in Morocco the stock of organic carbon in litter and soils represents between 40 and 55% of the total organic carbon stocks ([18], [20]). Such distribution over pools depends apparently on the biome. Indeed, tropical forests were reported to have 56% of carbon stored in biomass and 32% in soil, whereas boreal forests had only 20% in biomass and 60% in soil, the remaining carbon being withheld in dead wood and litter ([70]).
According to the FAO ([41]), carbon stocks in forests in North Africa (the total carbon in biomass, dead wood, forest floor and the first 30 cm of the soil profile) were on average 64.9 Mg ha-1. Here, the total stock of organic carbon in the first 20 cm of soil ranged from 58 to 202 Mg ha-1, with an average stock of 107 Mg ha-1, which is somewhat higher than the overall FAO estimate.
Based on our results, the deforestation of 1 hectare of cork oak would lead to a loss of 183 Mg ha-1 of CO2. Such deforestation would eventually induce a loss of organic carbon from the soil organic carbon stocks of more than 79%, underlining the strong relationship between stand biomass and soil carbon. The extremely dry soil conditions may actually exacerbate this loss of organic soil carbon ([57]), as organic material can be oxidized rapidly whenever soils are bare or lose their cover. Boulmane ([18]) showed that this loss could even be as high as 90%. This confirms that forest is the land use having the highest stock of soil carbon, thereby human activities that modify these ecosystems can profoundly affect both carbon reservoirs in biomass but also in soils ([80]). The stock of organic carbon within all of the compartments studied (tree biomass, understorey biomass, forest floor and the first 100 cm of soil) are very well correlated with the density of the cork oak stands. In particular, with the exception of the understorey vegetation, the higher the stand density, the higher the carbon stocks in these stands (Fig. 5). This tight link may be the result of the high dependency of soil carbon stocks on litterfall fluxes, provided that the litter on soil remains undisturbed, as suggested by the location of almost all of the soil carbon pool in the upper-most layer of soil profiles. According to Fischlin et al. ([43]), in the context of climate change, Mediterranean forests are considered vulnerable with a risk of biodiversity loss and a decreased function of carbon sequestration. As a consequence, a sustainable forest management scheme should be applied in line with multi-criterion objectives aimed at optimizing both the increase of biomass and the sequestration of carbon ([22]).
Fig. 5 - Change in the average carbon stocks (Mg ha-1) in different compartments, depending on the cork oak stand density.
In this context and taking into consideration the results of this study with the objective of improving the carbon storage capacity in this ecosystem, there is a need to encourage management strategies that favor the restoration of cork oak stands. To this end, we propose:
- to temporarily prevent the access to low-density stands (less than 200 stems ha-1) with fences, followed by seeding or plantations of cork oak seedlings from local nurseries, with the aim of increasing the stand density up to more that 200 stems ha-1;
- to fence the open areas within the stands which have resulted from the degradation of the cork oak forest, where Quercus suber plantations should be established, with the aim of locally restoring the canopy cover by this species.
- to promote among local users and stakeholders the awareness of the numerous functions of such forest (notably ecological) and of the negative impact of its irrational exploitation.
Conclusions
Regarding our first objective, the present study showed that the total organic carbon stocks in the cork oak forests investigated range from 65 to 237 Mg ha-1, with an average total organic carbon stock of 121 Mg ha-1. These stocks, which are relatively low, can be explained by the widespread degradation and the high pressure the Mamora forest is subjected to. The soil is the major reservoir of carbon in such ecosystem: more than 88 % of the organic carbon was found within the first 40 cm of the soil profile. Carbon stocks estimates varied consi-derably with the basal area of the cork oak stands. The biomass of cork oak trees represents the second largest carbon pool, whereas the understorey biomass poorly contributes (less than 2%) to the total organic carbon stocks. Concerning our second objective, the main parameter correlated with carbon stocks in this cork oak forests was the tree density of stands. Indeed, the stand density was strongly related to the total carbon sequestration within the three main compartments: tree biomass, soil and understorey vegetation. As such, a shift from dense to open cork oak stands due to overexploitation implies a calculated loss in total organic carbon stocks of around 47% on average. Loss of carbon in tree biomass and in the first 100 cm of soil in low-density cork oak stands is not compensated by an increase of carbon in the understorey vegetation (representing less than 2% of the total carbon pool). Regarding our third objective, forest managers should implement silvicultural practices aimed at increasing the stand density and enhancing the public awareness among forest users to avoid continued over-exploitation.
Acknowledgements
The authors express their gratitude to the technicians Ms Bounimi Zhour, M Amrani and M Aaziz at the Centre de Recherche Forestière in Rabat (Morocco), for their valuable contributions in the field and in the chemical analysis of samples. We also acknowledge the bilateral project between France and Morocco PRAD 01/13 “Projet de Recherche Agronomique pour Developpement” for the financial support, permitting the exchange of staff and constructive work sessions.
References
Gscholar
Online | Gscholar
Online | Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Online | Gscholar
Gscholar
Online | Gscholar
Online | Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Online | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Online | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Online | Gscholar
CrossRef | Gscholar
Authors’ Info
Authors’ Affiliation
Mohammed Halim
Université Mohammed V Agdal - Faculté des Sciences- Rabat, Address: 4 Avenue Ibn Battouta B.P. 1014 RP, 10000 Rabat (Morocco)
Laboratoire de Pédologie, Centre de Recherche Forestière, B.P. 763, Av. Omar Ibn Khattab Agdal-Rabat, 10000 Rabat (Morocco)
Corresponding author
Paper Info
Citation
Oubrahim H, Boulmane M, Bakker MR, Augusto L, Halim M (2015). Carbon storage in degraded cork oak (Quercus suber) forests on flat lowlands in Morocco. iForest 9: 125-137. - doi: 10.3832/ifor1364-008
Academic Editor
Giorgio Matteucci
Paper history
Received: May 30, 2014
Accepted: Mar 30, 2015
First online: Aug 08, 2015
Publication Date: Feb 21, 2016
Publication Time: 4.37 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2015
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 61449
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 47670
Abstract Page Views: 5413
PDF Downloads: 6679
Citation/Reference Downloads: 29
XML Downloads: 1658
Web Metrics
Days since publication: 3935
Overall contacts: 61449
Avg. contacts per week: 109.31
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2016): 26
Average cites per year: 2.60
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Soil fauna communities and microbial activities response to litter and soil properties under degraded and restored forests of Hyrcania
vol. 14, pp. 490-498 (online: 11 November 2021)
Research Articles
The manipulation of aboveground litter input affects soil CO2 efflux in a subtropical liquidambar forest in China
vol. 12, pp. 181-186 (online: 10 April 2019)
Research Articles
Estimating changes in soil organic carbon storage due to land use changes using a modified calculation method
vol. 8, pp. 45-52 (online: 17 June 2014)
Research Articles
Effects of traditional forest management on carbon storage in a Mediterranean holm oak (Quercus ilex L.) coppice
vol. 11, pp. 344-351 (online: 18 April 2018)
Research Articles
Soil stoichiometry modulates effects of shrub encroachment on soil carbon concentration and stock in a subalpine grassland
vol. 13, pp. 65-72 (online: 07 February 2020)
Research Articles
Soil respiration along an altitudinal gradient in a subalpine secondary forest in China
vol. 8, pp. 526-532 (online: 01 December 2014)
Research Articles
Carbon storage and soil property changes following afforestation in mountain ecosystems of the Western Rhodopes, Bulgaria
vol. 9, pp. 626-634 (online: 06 May 2016)
Research Articles
The effect of clear-cut age on soil organic carbon and nitrogen indices in Scots pine (Pinus sylvestris L.) stands
vol. 18, pp. 146-153 (online: 09 June 2025)
Research Articles
Controlling soil total nitrogen factors across shrublands in the Three Rivers Source Region of the Tibetan Plateau
vol. 13, pp. 559-565 (online: 29 November 2020)
Research Articles
Soil respiration and carbon balance in a Moso bamboo (Phyllostachys heterocycla (Carr.) Mitford cv. Pubescens) forest in subtropical China
vol. 8, pp. 606-614 (online: 02 February 2015)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword