Stand structure and deadwood amount influences saproxylic fungal biodiversity in Mediterranean mountain unmanaged forests
iForest - Biogeosciences and Forestry, Volume 9, Issue 1, Pages 115-124 (2015)
doi: https://doi.org/10.3832/ifor1304-008
Published: Sep 08, 2015 - Copyright © 2015 SISEF
Research Articles
Abstract
Old-growth forests are key elements of ecosystem diversity and conservation strategies, providing niche differentiation and trophic pathways that produce structural and compositional heterogeneity. In these forests, deadwood is particularly important for saproxylic and mycorrhizal fungi, sustaining forest productivity and environmental services. In this study, the saproxylic fungal diversity in Mediterranean mountain forests, characterized by different management histories and forest types (holm oak and beech), was analyzed. The relationships between saproxylic fungal biodiversity and structural attributes were considered in three forest stands of the Apennines (Italy). In addition, descriptive environmental parameters and forest traits were related to prevailing fungal communities, in order to analyze the species composition and distribution patterns of saproxylic fungi resulting from the ordination processes. The study sites were selected on the basis of their late-serial stage of development. Species frequency was analyzed through multivariate techniques to test the relationships between fungi, structural attributes and environmental variables. A Detrended Correspondence Analysis (DCA) was used to investigate the response of the overall fungal community structure to environmental gradients. Living tree volume, altitude, vegetation type, and the frequency of species with ephemeral sporocarp lifespan played a crucial role in diversifying species distribution patterns. Deadwood volume and decay classes were related to taxonomic and trophic community diversity. However, differences between the considered climatic regions exerted a major role on the occurrence of fungi with ephemeral sporocarps more than deadwood abundance, utilized primarily as fructification substrate.
Keywords
Apennines Forests, Forest Biodiversity, Old-growth Forest, Saproxylic Fungi, Sporocarp Lifespan, Structural Heterogeneity
Introduction
A general and unambiguous definition of old-growth forests has not been reached yet ([99]). Any definition should be supported by indicators easily applied for the identification of standard old-growth conditions, also in terms of structural and functional forest complexity ([81]). Some of the attributes often used to define old-growth forests are linked to the forest structure: they can be used as a proxy of compositional and functional features and are more readily measurable than biological indicators. Attributes include the large variation in tree size, big old trees and multilayered forest canopy ([29]).
Moreover, another typical criterion to identify old-growth traits is the presence of large amounts of standing and downed deadwood in all stages of decay, considering that large standing dead trees and woody detritus on the forest floor include indirect evidence of canopy mortality and gap phase dynamics ([99], [60], [39]). Structural indicators are important tools that can be conveniently used to understand, assess and manage forest ecosystems ([28], [53]).
Old-growth forests are key elements in the strategies for biodiversity conservation. As a consequence of their high structural heterogeneity levels, they provide special habitats and trophic niches necessary for the existence and development of many species, such as birds, invertebrates, lichens, bryophytes and particularly saproxylic fungi ([34], [91], [35], [88], [42], [44], [72], [13], [80], [48]). In this sense, forest ecosystems with a high degree of naturalness could provide a reference for implementing sustainable forest management techniques for conservation purposes, as well as a tool for better understanding the role of forests in preserving ecosystem services.
Among attributes and indicators often used to label a forest as old growth, deadwood is one of the most typical ([34], [94]). It plays significant roles in several ecological processes, creating the basis for cycling of photosynthetic energy, carbon, and nutrients stored in woody material ([41], [52]), contributing to soil formation, development and protection. Deadwood is also a major component of ecosystem carbon storage and cycling ([96]). Moreover, deadwood was included within the list of carbon pools (above-ground and below-ground biomass, litter, deadwood and soil) provided by Intergovernmental Panel on Climate Change ([79]), relevant for the estimation of carbon stocks and their changes under the UNFCCC and the Kyoto Protocol ([54]).
Beech (Fagus sylvatica L.) stands are crucially important forest ecosystems throughout the temperate and mountain areas of Europe ([69], [21]). Nevertheless, these ecosystems have been exploited by human activities for centuries and more recently have been affected by long-range air pollution ([76]), with the result that old-growth and structurally complex forests with consistent amount of decomposing wood can be found only in protected areas and remote patches of the continent ([19]). In the Mediterranean Europe, important tree species will probably be negatively affected by the expected increase in climatic extreme ([65]) and associated disturbances in addition to direct changes in habitat quality ([33]). Holm oak (Quercus ilex L.), in particular, is relatively sensitive to the reduction of soil moisture content caused by climate change, especially in the southern part of Italy and along coastal areas ([7], [97]). Currently, the importance of deadwood is emphasized for its function in the maintenance of biodiversity ([17]), providing habitats for a plenty of organisms. If deadwood might be considered a key factor for biodiversity and functioning of these forests ([51]), the same would apply to both saprotrophic and mycorrhizal fungi.
Fungi are known to be the principal wood decomposers in forest ecosystems and their activity makes deadwood available to other fungal species and wood-living organisms ([14]). In fact, wood-inhabiting fungi modify the necromass both chemically and structurally, and generate new habitats and food resources ([83], [14]). Many researchers have studied the effect of forest management on saproxylic fungal communities, focusing on the effect of necromass reduction. The results of these studies converge to indicate that saproxylic fungal biodiversity decreases in managed forests with impacts over the community composition ([50], [46], [68], [9], [2]). The amount and quality of deadwood, as well as the size and type of woody debris (in particular logs), and also the species, age and size of trees are structural attributes of forests that influence the saproxylic fungal community and its species richness ([70], [37], [73], [74], [45], [53]).
Most of the studies conducted on forest management and deadwood influence on saproxylic fungi have focused on central and northern forest ecosystems of Europe, with some exceptions (e.g., [13], [80], [86], [1], [2], [31]). Therefore, there is the need to widen and integrate the current European guidelines for sustainable forestry, which are mostly based on data from boreal forests. In particular, it is important to test how wood inhabiting fungi respond to the structural attributes selected to assess the degree of forest naturalness. This indirect approach could be useful to comprehend the role of biodiversity, considering the complex relationships between saproxylic fungi and habitat traits ([22], [55]).
This study aims at analyzing saproxylic fungal biodiversity in Mediterranean mountain forests - namely three stands of central Apennines (Italy) - characterized by different management histories and tree species (holm oak and beech). The investigation has been focused on the relationships between saproxylic fungal biodiversity and some structural attributes that characterize the study areas (including substrate type, deadwood biomass and living trees volume). In addition, descriptive environmental parameters, forest traits and morphological/ecological traits of the species were studied for the characterization of fungal community, in order to analyze the species composition and the distribution patterns of saproxylic fungi resulting from the ordination processes. The study sites were selected on the basis of their late-serial stage of development.
Material and methods
Study sites
Two out of three of the study areas are located in the Cilento, Vallo di Diano e Alburni National Park (PNCVD), in southern Italy (Campania Region). The Park was recognized as a UNESCO World Heritage Site in 1998 and as a Biosphere Reserve in 1997. Litho-morphological landscape is characterized by gentle hills and calcareous mountains. The hilly landscape is mainly constituted of stratified flysch formations of Tertiary age, namely Flysch del Cilento, that is a complex and chaotic sedimentation of sandstone rich in quartz, feldspar, and mica and other minor components widespread near the hydrographical basin of the river Alento and mountains of western Cilento ([64]).
The PNCVD is characterized by two climatic regions (Temperate and Mediterranean - [67], [84]). The bioclimate is mainly oceanic, ranging from thermo-Mediterranean to supratemperate, with an ombroclimate ranging from dry to hyperhumid. According to climatic zones, litho-morphology and soil features, the vegetation potential ranges from dry coastal garrigue to holm oak woodlands, mixed forests of oak, hornbeam, and alder, beech forests, and high-altitude grasslands. The two study sites in the PNCVD (“Ottati” and “Corleto”) are characterized by different ages of the old-growth forest. Particularly, the site of “Corleto” is a beech forest unmanaged since 1965, but still presenting the signs of past silvicultural activities. For example, the vertical and horizontal forest structure still reflects the past human activities, and stumps of artificial origin are evident. On the contrary, the “Ottati” study area is characterized by higher levels of naturalness ([64]), where the natural evolution has been in progress since decades. Marks of past human activities are scarce, considering that the stand is unmanaged since more than 170 years due to its difficult accessibility. The “Fonte Novello” site is located in the Venacquaro valley, in the Gran Sasso e Monti della Laga National Park (PNGSML), central Italy (Abruzzo Region). The study area is north facing, and the soils are classified as Lithic Rendolls, derived from the alteration of calcareous bedrocks. The territory falls within the temperate region, the bioclimate is oceanic while thermotype is inferior supratemperate and ombrotype is inferior humid ([11]). The vegetation of the Venacquaro Valley is dominated by beech forests. Particularly, the study area has not been subject to severe anthropogenic disturbances during the last centuries due to legal debates concerning the position of the administrative boundaries between Pietracamela and Fano Adriano municipalities.
Experimental design
A survey was conducted in spring and autumn 2012 on 3 plots, one for each site. The two sampling plots (ID51 and ID171) surveyed in the PNCVD were extracted from 22 separate forested areas considered as representative of the most common forest types in the Park. Detailed description of the survey design is reported in Corona et al. ([25]) and in Marchetti et al. ([64]). Each plot was previously defined as old-growth forest on the basis of stand structure and floristic information and former management history ([12], [85], [18], [58]). However, based on the last attribute, the plots differed allowing the distinction between potentially old growth (labeled with POG in Tab. 1) and old growth (labeled with OG in Tab. 1). These stands have been subjected to forest harvesting and anthropogenic disturbances until 40 years ago. These two plots were delimited (0.25 ha square plots), and a buffer area of equivalent size adjacent to the plots was considered to better integrate the original datasets.
Tab. 1 - Sampling sites in the central Apennines and their level of naturalness. (OG): old-growth; (POG): potentially old-growth), (physiognomy): type of forest; (Alt): altitude; (Exp): exposure; (DW): deadwood volumes ;(LT): total living tree volume.
| Plot | Years since unma. |
Naturalness | Physiognomy | Site | Alt. (m a.s.l.) |
Exp | DW (m3/ha) |
LT (m3/ha) |
|---|---|---|---|---|---|---|---|---|
| ID51 | 175 | OG | holm oak | Ottati | 633 | West | 44.26 | 316.13 |
| ID171 | 54 | POG | thermophilic beech | Corleto | 1298 | North | 14.40 | 907.40 |
| Fon | 300 | OG | microthermal beech | Pietracamela | 1340 | North | 89.00 | 1030.00 |
Moreover, the survey in the PNGSML, at the Fonte Novello (Fon) stand (Tab. 1), was realized on a 1-ha square plot; sampling was focused at the corners of the square plot, on a total of 0.36 ha. In this case, the buffer area was internal to the large plot, for a total surveyed area of 0.5 ha in order to have a similar sampling size.
Deadwood classification
Standing dead trees, downed dead trees, snags and stumps were measured when more than half base of their trunk was lying within the plot and buffer area. Coarse woody pieces were measured when more than half base of their thicker end was lying within the plot. A threshold height of 1.3 m was used to distinguish stumps (< 1.3 m) from snags (> 1.3 m). The position of each deadwood piece was obtained by measuring horizontal distances and azimuth from GPS points. The volume of standing and downed dead trees was estimated through volume equations ([95]), while the volume of snags, stumps and coarse lying deadwood was assessed using the cone trunk model (eqn. 1):
where V is the volume (m3), h is the height or length (m), D is the maximum diameter (m), d is the minimum diameter (m). The minimum diameter considered was 5 cm. Decay level classification of each deadwood piece was done following Hunter ([43]). The surveyed deadwood components and attributes are detailed in Tab. 2.
Tab. 2 - Deadwood components surveyed in the study sites. (Dbh): diameter at breast height; (H): height; (L): length; (Dmin): minimum diameter; (Dmax): maximum diameter; (Dbase): diameter at the base of the trunk; (Dtop): diameter at the top of the trunk; (Origin): natural or artificial.
| Deadwood component | Dimensional thresholds |
Assessed attributes |
|---|---|---|
| Standing dead trees | Dbh≥5 cm; H≥130 cm | Species, Dbh, H, crown height, crown projection on soil, decay class, position |
| Snags | Dbh≥5 cm; H≥130 cm | Species, Dbase, Dtop, height, decay class, position |
| Downed dead trees | Dbh≥5 cm; H≥130 cm | Species, Dbh, L, crown insertion distance, decay class, position, stem direction |
| Coarse woody debris | Dmin≥5 cm; L≥100 cm | Species, Dmin, Dmax, L, decay class, position, stem direction |
| Stumps | Dtop≥5 cm; H<130 cm | Species, Dbase, Dtop, height, decay class, origin, position |
Inventory of saproxylic fungi
Sporocarps of both Ascomycota and Basidiomycota were surveyed according to the sampling protocols used for similar studies in Europe ([8], [88]). Fungal intensive inventory was conducted at all the sites in late May and early June (S, spring) and October 2012 (A, autumn). All standing and downed deadwood with a minimum diameter of 10 cm was examined for the presence of fruit-bodies larger than 1 mm ([5]). The nomenclature adopted was based on the Dictionary of the Fungi ([47]) and the CABI Bioscience Database of Fungal Names found on internet - ⇒ http://194.131.255.3/cabipages/Names/names.asp?strGenus. The saproxylic fungi were described on the basis of their trophic role, as reported by various authors ([5], [38], [6], [98], [14], [57], [87]). Furthermore, the persistency of their sporocarps was described (annual: occurring for nearly one year; perennial: living for a number of years; ephemeral: occurring for some days). The species were also subdivided in morphological groups according to the anatomical features of their sporocarps (see Tab. S1 in Appendix 1), as described by Heilmann-Clausen ([35]).
Statistical analyses
Species frequency data (i.e., the occurrence of species at each plot) were analyzed by multivariate techniques to test the relationships between fungi, structural attributes and environmental variables. Detrended Correspondence Analysis (DCA) ([40]), performed by Multivariate Analysis of Ecological Data using the software package PC-ORD, version 5.0 for Windows® ([61]), was applied to investigate the response of the overall structure of the fungal communities to environmental gradients. The ordination axes of the DCA express similarities and dissimilarities in species composition between samples. DCA axes were rescaled with 26 segments.
The Mantel test was then performed by PC-ORD to test the hypothesis of independence between the community matrix and the variables (null hypothesis).
Pearson’s correlations were then calculated using the software package SPSS (version 18.0, PASW Statistics for Windows, SPSS Inc., Chicago, IL, USA), in order to interpret the community gradient and to evaluate the relationships between ordination scores and ecological features, as well as structural attributes and environmental variables.
Results
Structural attributes
Forest structure strongly differed among the studied sites, showing progressively greater amounts of deadwood proportionally with increasing years since last intervention (Tab. 1). The old-growth plot of Fonte Novello was also characterized by high volumes of living trees. Conversely, Ottati (PNCVD) showed relatively high amounts of deadwood and a lower volume of living trees. All the studied stands revealed also a multimodal diameter distribution of living trees (data not shown). Ten diameter classes (with a range of 10 cm) were detected over all the study sites, with a decreasing (though not regularly) frequency at the higher classes and a maximum diameter of 1.10 m. The larger frequency of small size classes suggests the occurrence of canopy mortality and gap dynamics likely due to natural disturbances, such as wind or snow.
Biodiversity of saproxylic fungi
A total of 83 species of saproxylic fungi, including 13 Ascomycota and 70 Basidiomycota were detected in the three studied plots, in spring and autumn 2012 (see Tab. S1 in Appendix 1).
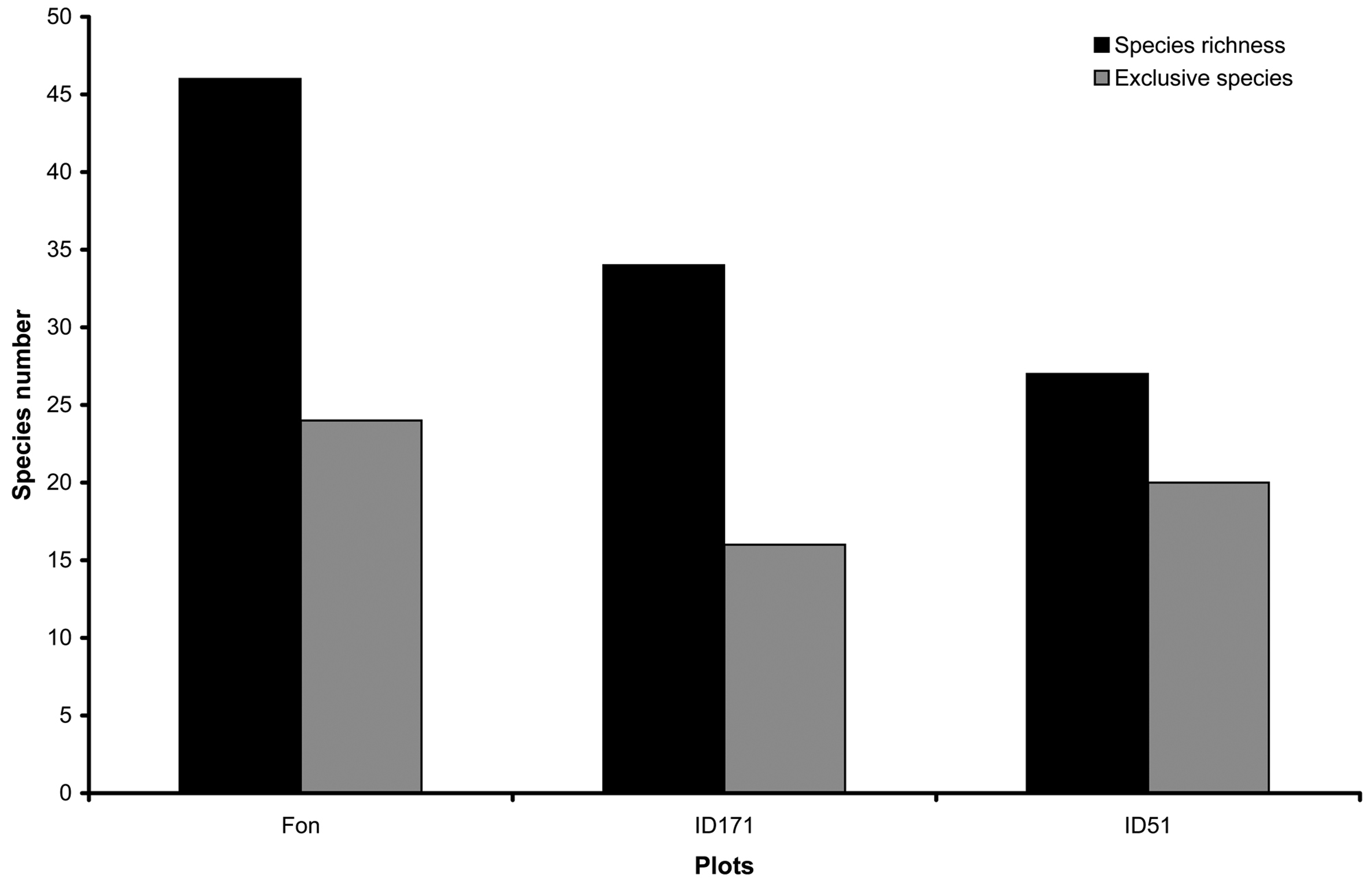
The most represented genera were Mycena with five species, Crepidotus, Phlebia, Pluteus and Trametes with four Phanerochaete, Polyporus and Stereum with three and Coprinellus, Diatrype, Ganoderma, Pholiota and Xylaria with two, while the remaining genera were represented by single species. The highest number of fungal species was recorded in Fonte Novello (Fon) that hosted 24 exclusive species, while in PNCVD, plots (ID171 and ID51) hosted 16 and 20 exclusive species, respectively (Fig. 1).
Fig. 1 - Total (spring and autumn) species richness and exclusive species found in the plots analyzed.
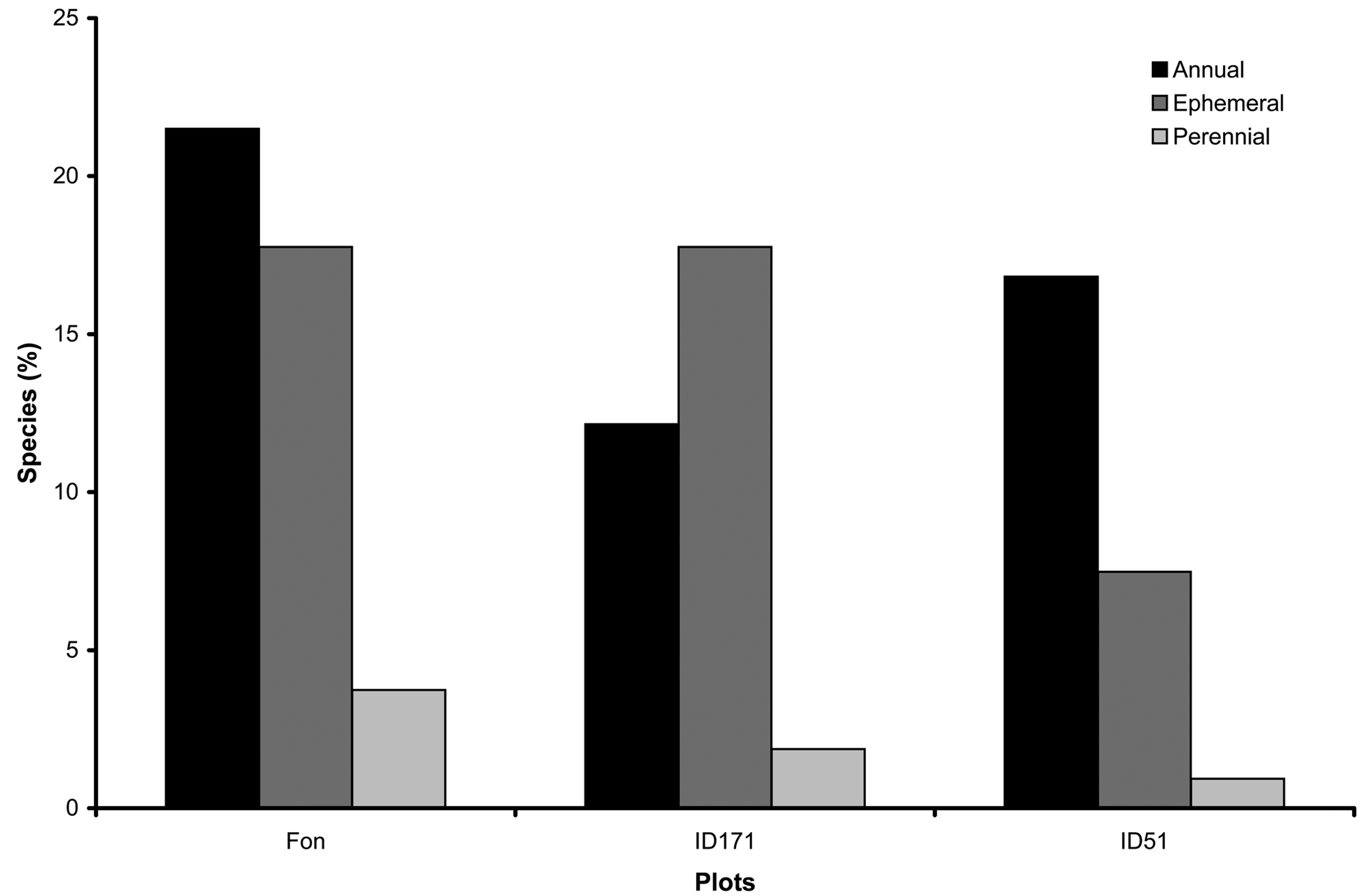
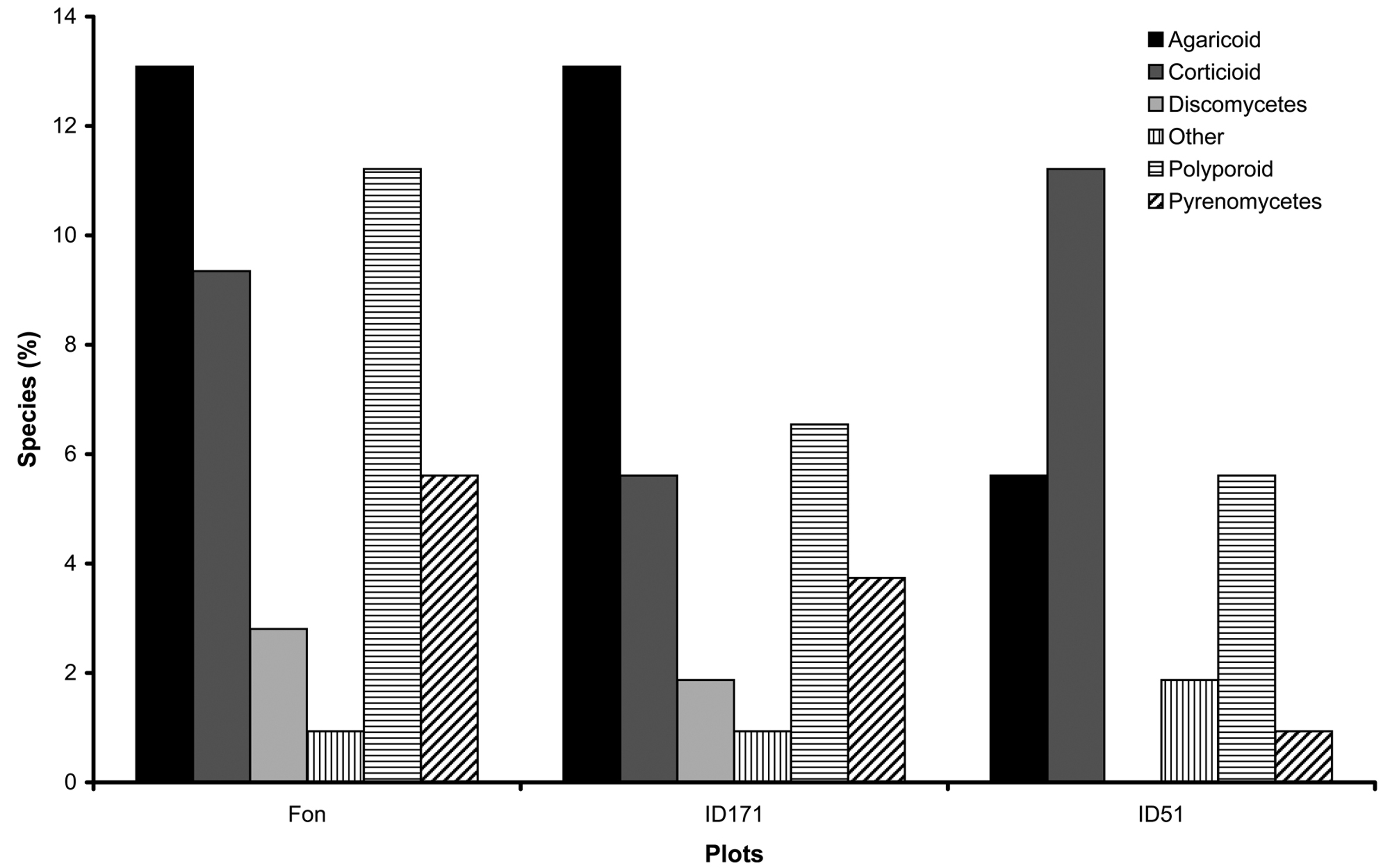
The species characterized by perennial sporocarps accounted for 7% of the total, species with annual sporocarps were 50%, while ephemeral species accounted for the remaining 43% (Fig. 2). From a trophic point of view, 72% of the species belonged to the wood saprotrophs (Ws), while 28% were necrotroph parasites (Np). Agaricoid species were the most represented morphogroup in beech plots, while corticioid characterized the Quercus ilex stand (Fig. 3).
Detrended Correspondence Analysis (DCA)
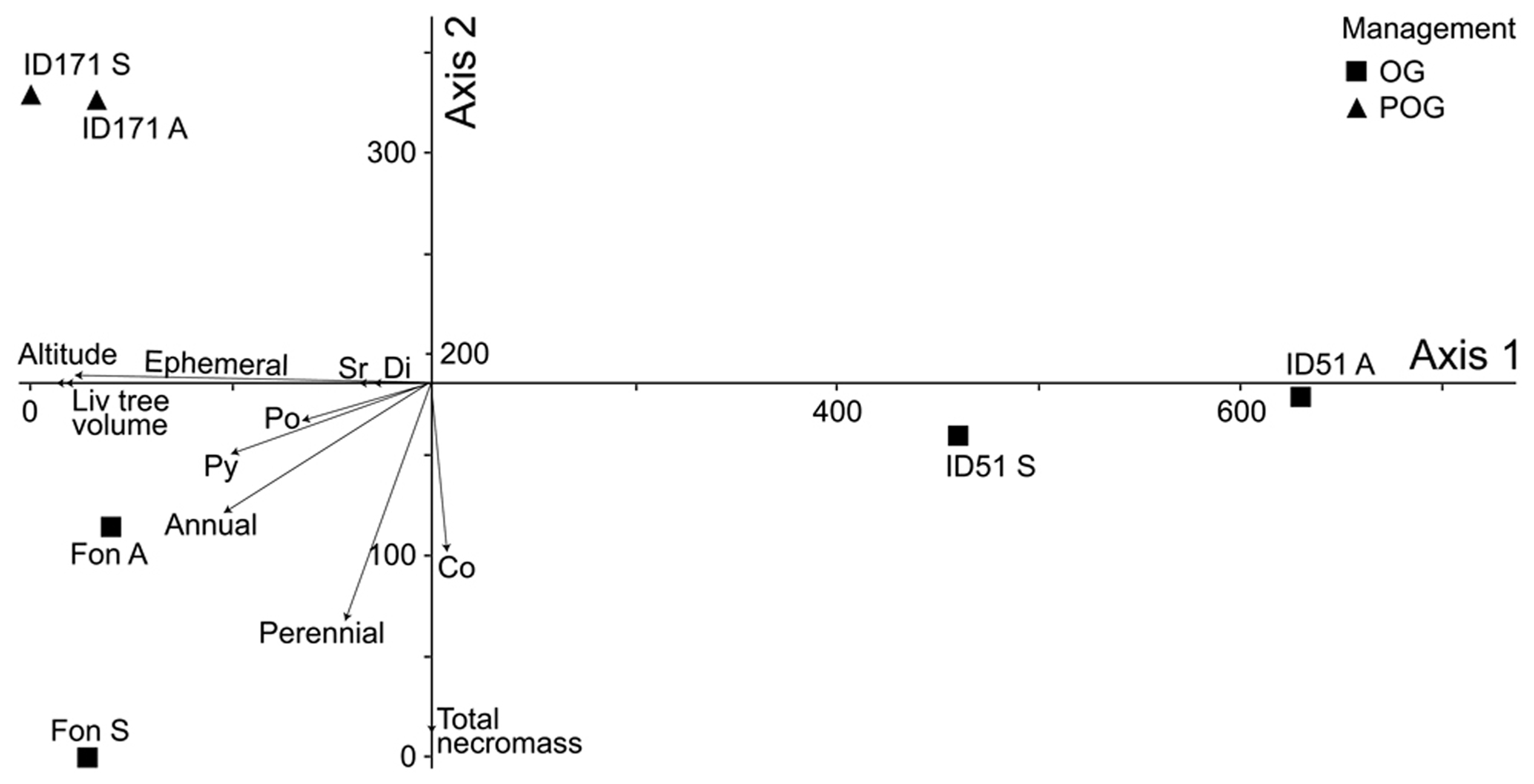
To further investigate the variables affecting the distribution of fungi species, data were processed through DCA to assess the presence of underlying gradients (Fig. 4).
Fig. 4 - Results of the Detrended Correspondence Analysis. Axes are scaled in SD units x 100. (OG): Old Growth; (POG): Potentially Old Growth; (A): Autumn; (S): Spring.
DCA results revealed two distinct community gradients, with an eigenvalue of 0.84 and 0.51 for Axis 1 and Axis 2, respectively, and gradient lengths of 6.29 and 3.30 SD units. These values confirmed a primary role of the gradient expressed by Axis 1. Axis 2 still represented a distinct beta-diversity gradient, though containing less information. The third ordination axis was considerably weaker (eigenvalue: 0.15; gradient length: 2.58 SD units), thus it was not considered in further evaluation.
Two groups of plots were clearly identified in the DCA sorting space displayed in Fig. 4. The first group included the two old-growth plots represented in the two quadrants below axis 1, while the other consisted of the potentially old-growth plot displayed in the quadrant above axis 1.
Mantel test
The Mantel test was used to assess the hypothesis of independence between the community matrix and the variables (null hypothesis). The test yielded the following results: the observed Z (3.15E+00) was larger than the expected Z (2.91E+00); the standardized Mantel statistic (r = 0.79) was statistically significant (p = 0.002) based on the randomization test.
Pearson’s Correlation
To obtain a complete interpretation of community gradients, a Pearson’s correlation was performed between the scores on the DCA axes and some environmental and ecological parameters (Tab. 3). The results of the Pearson’s correlation show that the gradient represented by DCA axis 1 was significantly and negatively related to living tree volume and altitude, and positively correlated with the vegetation type. DCA axis 1 was also significantly and negatively related to the frequency of species bearing short-lived fruit body. As regards to DCA axis 2, Pearson’s correlation analysis revealed that it was significantly and negatively related to the total necromass volume and positively to forest management. Moreover, the studied plots were quite evenly distributed along the second axis, with the two-old-growth stands located in its lower part (Fig. 4), indicating that DCA axis 2 represents a remarkable environmental gradient. By contrast, no significant correlations were found with DCA axis 3.
Tab. 3 - Pearson’s correlation between DCA axes scores (Ax1-Ax3) and recorded variables. (LT): Living tree volume; (Tn): Total necromass; (Alt): altitude; (Sr): species richness; (Man): Management; (Veg): vegetation; (Seas): season; (Po): polyporoid; (Co): corticioid; (Py): pyrenomycetes; (Di): discomycetes; (Ag): agaricoid; (Ot): others; (Per): Perennial; (Eph): Ephemeral; (Ann): annual; (*) p <0.05; (**): p <0.01.
| Variable | LT | Tn | Alt | Sr | Man | Veg | Seas | Po | Co | Py | Di | Ag | Ot | Per | Eph | Ann |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ax1 | -0.962** | -0.083 | -0.976** | -0.390 | -0.515 | 0.834* | 0.143 | -0.573 | 0.213 | -0.712 | -0.421 | -0.307 | -0.071 | -0.465 | -0.948** | -0.724 |
| Ax2 | -0.063 | -0.935** | 0.041 | 0.041 | 0.870* | 0.397 | 0.188 | -0.315 | -0.656 | -0.424 | 0.001 | 0.096 | 0.056 | -0.779 | 0.151 | -0.575 |
| Ax3 | 0.438 | -0.172 | 0.469 | -0.300 | 0.438 | -0.303 | -0.565 | -0.065 | -0.802 | -0.091 | -0.214 | -0.375 | -0.507 | 0.043 | 0.483 | 0.206 |
| LT | - | 0.272 | 0.994** | 0.462 | 0.354 | -0.935** | 0 | 0.674 | -0.070 | 0.830* | 0.500 | 0.406 | 0.140 | 0.633 | 0.969** | 0.802 |
| Tn | - | - | 0.166 | 0.262 | -0.804 | -0.595 | 0 | 0.606 | 0.688 | 0.596 | 0.318 | 0.194 | 0.203 | 0.917** | 0.056 | 0.733 |
| Alt | - | - | - | 0.444 | 0.453 | -0.891* | 0 | 0.622 | -0.149 | 0.784 | 0.476 | 0.394 | 0.120 | 0.545 | 0.987** | 0.739 |
| Sr | - | - | - | - | 0.031 | -0.482 | 0.700 | 0.876* | 0.515 | 0.632 | 0.993** | 0.977** | 0.943** | 0.402 | 0.417 | 0.446 |
| Veg | - | - | - | - | - | - | 0 | -0.785 | -0.195 | -0.913* | -0.535 | -0.410 | -0.191 | -0.866* | -0.830* | -0.939** |
| Seas | - | - | - | - | - | - | - | 0.376 | 0.424 | 0.233 | 0.655 | 0.828* | 0.781 | 0 | 0.070 | -0.153 |
| Po | - | - | - | - | - | - | - | - | 0.532 | 0.821* | 0.912* | 0.793 | 0.729 | 0.766 | 0.544 | 0.806 |
| Py | - | - | - | - | - | - | - | - | - | - | 0.640 | 0.592 | 0.400 | 0.823* | 0.721 | 0.872* |
| Di | - | - | - | - | - | - | - | - | - | - | - | 0.957** | 0.920** | 0.463 | 0.443 | 0.502 |
| Ag | - | - | - | - | - | - | - | - | - | - | - | - | 0.938** | 0.324 | 0.396 | 0.323 |
| Per | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.446 | 0.922** |
Discussion
Relationship between fungal species and decomposition processes
The species found belong to 56 genera, which supports the hypotheses that fungal diversity is high in late-serial and old-growth forests. In the studied stands, the high fungal diversity and abundance was expected because of the reduced anthro-pic disturbance. Nevertheless, an analogous trend in fungal diversity was also observed by Wubet et al. ([101]), which compared many European beech forest soils characterized by different management types. They found no differences in the fungal community structure, suggesting that soil fungal communities are region-specific, though composed of functionally diverse and complementary taxa. However, these site-specific studies suggest different patterns of co-existence in fungal diversity. In order to find a functional relationship between biotic and abiotic parameters and their association with forest management, a large-scale biogeographic approach should be conveniently applied.
Wood-inhabiting fungi abundance on coarse woody debris (CWD), showed that a consistent group of Ascomycota species, including Biscogniauxia nummularia (Bull.) Kuntze, Bisporella citrina (Batsch) Korf & S.E. Carp., Diatrype disciformis (Hoffm.) Fr., Hypoxylon fragiforme (Pers.) J. Kickx f., Kretzschmaria deusta (Hoffm.) P.M.D. Martin, Nemania serpens (Pers.) Gray, and Xylaria hypoxylon (L.) Grev., was associated with these plots.
Beside the above Ascomycota, it was noteworthy the occurrence in the Fonte Novello old-growth plot of Eutypa spinosa (Pers.) Tul. & C. Tul., an indicator species of beech woodlands ([3], [35]). Moreover, four Basidiomycota species such as Dentipellis fragilis (Pers.) Donk, Ganoderma pfeifferi Bres., Ischnoderma resinosum (Schrad.) P. Karst. and Phlebia nothofagi (G. Cunn.) Nakasone are reported among the 21 identified indicator species considered for the assessment of the conservation value of beech forests in Europe ([22]). The occurrence of these four indicator species in this plot, in addition to E. spinosa, was not quantitatively comparable to other European beech forest sites of high conservation value ([36]). Nonetheless, this peculiarity implies a low to moderate conservation value for the Fonte Novello beech forest, although it might be relevant as compared with the plot ID171, where only two indicator species were hosted, Ceriporiopsis gilvescens (Bres.) Domanski and P. nothofagi.
N. serpens is the only Pyrenomycetes species found in plot ID51. Along the environmental gradient represented by the DCA axis 1, beech stands were positioned at the opposite end compared with the holm oak plots. This could be due to the high content of tannins and other phenolic compounds in the bark of Quercus, as reported by Ciesla ([24]). In fact, such compounds are implied in suberization processes and inhibit the growth of fungi as well as other microorganisms ([90]). Moreover, plot OD51 shares a number of species with beech stands, but also showed a certain degree of specificity, hosting 20 exclusive species (Fig. 1). Among those, it is worth noticing the occurrence of the taxon Phanerochaete, which includes typical Mediterranean species, such as P. martelliana (Bres.) J. Erikss. & Ryvarden.
Furthermore, several environmental characteristics may explain the peculiarity of the plot ID51. It is located in the mid-upper hill belt at an altitude of 633 m a.s.l., and is characterized by thermophilous sclerophyllous vegetation, and well adapted to seasonal water availability fluctuations. Unlike the other two plots settled in the lower mountain belt, it has a very pronounced slope (55%) facing south-southwest, where xeric conditions are more frequent. Precipitation and temperature influence vegetation, and consequently soil properties, by affecting the type and rates of chemical, mineralogical, biological, and physical processes. Indeed, field experiments on soil warming have highlighted the sensitivity of decomposition process to temperature ([4]), which affects carbon sequestration and cycling, and consequently the level of biodiversity. Mean annual temperature has been indicated as one of the main drivers of CWD decay rates ([62]). Recently, Bradford et al. ([16]) has shown that local-scale factors influence more than climate the microbial activity and, consequently, the decomposition of deadwood.
DCA and Pearson’s correlation
Axis 1
DCA revealed a relatively high variation among plots along the first axis, that represents an important ecological gradient of differentiation among plots. As revealed by the Pearson’s correlation analysis carried out, living tree volume, altitude, vegetation, and the frequency of species with ephemeral sporocarp lifespan played a crucial role in diversifying species distribution patterns. Differences in CWD quality among stands may be due to different vegetation types and local microclimate, thus explaining the diverse arrangement of the plots along the DCA axes.
The beech forest plots located at higher altitudes were positioned on the left side along DCA axis 1, whereas the holm oak forest plot ID51 (both autumnal and spring inventories) was positioned on the far right of the same axis. Plot ID51 differed considerably from the other two plots, as it is settled at an altitude lower than any other plots considered in this study.
There is a scarcity of specific studies and consistent results concerning the relationships between elevation and wood-inhabiting fungi. Pouska et al. ([82]) outlined how elevation gradients and structural attributes might underlie and drive the shifts in species richness and composition patterns of saproxylic fungi. Sequences of fungal diversity along altitudinal gradients, enabling correlations of measured decay mechanisms with climate spatial variability (including altitude, exposure and slope), warrant further studies.
The investigated structural attributes, such as deadwood and living tree volume, represent some of the functional aspects of the old-growth forests providing special ecosystem values ([64]). In the surveyed forests, the long-time absence of logging led to an uneven-aged stand structure, although the effect of past management still influences the current structure of all sites. The heterogeneity of stands increased due to the occurrence of developmental gaps, probably related to small-scale disturbances, such as strong wind and heavy snow. It is likely that this feature influenced also the occurrence and diversity of wood-inhabiting fungi, as a consequence of the different microclimatic conditions on the forest floor. The living volume was significantly higher than in corresponding managed forests types ([64]), indicating that the occurrence of natural disturbances, rather than silvicultural practices, did not affect the overall forest productivity.
Differences between the considered climatic regions affects the occurrence of fungi with ephemeral sporocarps more than deadwood abundance, utilized primarily as fructification substrate. Moreover, based on the negative correlation of DCA axis 1 with the ephemeral sporocarp lifespan characteristics, we may infer that species with short-lived sporocarps displayed greater diversity, reflecting intra-annual climate variability ([10]), and variation in bioclimatic conditions.
The highly similar positioning in the DCA space of the two seasonal samplings from the same plots indicated that each stand has a specific fungal community based upon the presence of persistent species, on annual basis. The major drivers of such communities are ascribable to vegetation types, but also to differences between the considered climatic regions. This finding confirm that the composition of the wood-inhabiting macrofungal community is related to the stand type, as shown by Gates et al. ([30]). Heilmann-Clausen ([35]) also found that a pronounced shift in community structure could be driven by a complex gradient, including microclimate condition and decay rate, resulting in the diversification of CWD quality and classes.
Axis 2
The plot distribution along the DCA axis 2 may be interpreted as a response to a decreasing gradient of CWD availability (r= -0.935; p<0.01), also influenced by past forest harvesting. In fact, DCA axis 2 was also positively correlated with the management variable (r=0.870, p<0.05). Lindblad ([56]) showed that species richness of fruiting wood-inhabiting fungi was negatively affected by forest management and found higher species richness in decayed logs in a natural forest than similar logs in a managed forest.
As a consequence of the lower CWD availability, plots in the lower part of the DCA ordination space are expected to be characterized by a particular group of fungal species. This was the case of the plot in Fonte Novello, where species with perennial fruit body, such as Fomes fomentarius (L.) J. Kickx f., Fomitopsis pinicola (Sw.) P. Karst. and Ganoderma pfeifferi Bres., accounted for 57% of the total perennial species collected.
The strong relevance of the aforementioned polyporoid species agreed with the results obtained by Bader et al. ([8]) and Sippola et al. ([92]), which found that polyporoid species richness was related to the amount and quality of CWD. Likewise, high amounts of CWD, deadwood quality and the co-occurrence of diverse decay stages are among the ecological requirements of saproxylic fungal species, as demonstrated in a number of studies conducted in central and northern Europe ([83], [26], [71], [49]). The deadwood volume was previously considered an important variable in predicting species richness of several groups of species associated with deadwood habitats ([78]), but also a criterion to identify old-growth conditions ([58]) and a tool to understand the evolution phase of forests with low anthropic disturbances ([63], [58]). Particularly, deadwood volume was varying among the investigated stands, reflecting the past management legacy and the small-scale disturbance regime. These results were in accordance with a former study conducted within the PNCVD ([80]), which demonstrated how deadwood volume affected trophic roles and species diversity of saproxylic organisms in old-growth forests, including wood-inhabiting fungi.
The amount of deadwood observed in the study sites were significantly lower than those found in recently established reserves (reviewed by [23]), indicating a mean value of deadwood volume equal to 130 m3 ha-1.
The successional stage of each stand, the recent forest history and the disturbance regime can be considered as the main factors explaining the variation in deadwood amount observed in the investigated forests. Obviously, the amount of naturally occurring deadwood may have been reduced by the frequent collection of firewood by local people. Nonetheless, from a quantitative standpoint lying deadwood constituted the most significant deadwood component in the developmental phases close to old-growthness. Coarse woody debris and downed dead trees were the consequence of natural mortality occurring in the stands due to the natural processes established in the studied forests in the last decades. In our study sites, current mortality is mainly related to endogenous processes, such as competition, but also wind and heavy snow falls.
Although the investigated forests have been unmanaged for decades, signs of past removal of deadwood are still evident, and this was probably the reason why the deadwood in the most advanced decay classes was not frequent. Indeed, in Mediterranean ecosystems deadwood could decay five times faster than in other climatic regions ([59]). Therefore, the deadwood volumes occurring in the investigated forests did not necessarily indicate the presence of higher anthropic disturbance regimes. The presence of different decay stages in the examined stands supports the hypothesis that many saproxylic species occur in these forests, explaining the wood-inhabiting fungal richness.
orrelation of biological and structural variables
In this study, a high correlation was found between living tree volume and Pyrenomycetes morphogroup (r=0.830; p<0.05). Among the Pyrenomycetes collected, B. nummularia, E. spinosa, K. deusta showed necrotrophic attitude. These fungi would be considered either endophytes on the trunks or latent invaders ([15], [14], [77]). On the other hand, the correlation analysis revealed a very significant relationships of living tree volume with ephemeral sporocarps (r=0.969; p<0.01) and of the total necromass volume with perennial sporocarps (r = 0.917; p<0.01). These correlations might reflect a selected functional cross-connection between wood-decomposer fungal species and guild of mycorrhizal fungal species associated to living trees, occurring as active webs of extra radical hyphae in the soil, named “The Wood-Wide-Web” ([89]) referring to plant-to-plant carbon transfer throughout a common mycorrhizal network.
Martin & Nehls ([66]) explored the ecological aspects of ectomycorrhizal fungi from a genomics perspective in a French forest, and obtained evidence about the dominance of Agaricomycetes in the soil, including a large number of ectomycorrhizal species (EM). These results confirmed that EM fungi could be characterized by a dual lifestyle, living as symbiont with plant roots and as facultative saprotrophs in the soil. Ogura-Tsujita et al. ([75]) reported that Mycenaceae species are mycorrhizal of any plants and not exclusively of orchids, as previously reported for several Mycena species ([102]). Mycenaceae fungi are known as saprotrophic free living-litter and wood decomposers ([20]). In our study, the genus Mycena was represented with five species, usually occurring on wood in advanced decay stages. This evidence highlights the role of fungi in the nutrient and energy transport, connecting patches of different resources in the ecosystem ([27]), and support the high correlation observed between living tree volume and ephemeral sporocarps.
In this study, the high correlation observed between deadwood volume and perennial sporocarps, confirms the pivotal role in the wood decay process of a wide range of wood-rotting fungi, characterized by perennial sporocarps. The presence of large amounts of deadwood in forest ecosystems, and particularly in old-growth forests, has often been connected with high volume of living biomass ([93]). However, this relationship did not emerge from the correlation analysis carried out in this study. Nevertheless, Burrascano et al. ([19]) reported that old-growth forests host significantly higher amounts of living biomass than mature forests. In this sense, the presence of large living trees would be an important indicator of late successional forest structure; in particular, a minimum of 30 large living trees per hectare with diameter at breast height larger than 50 cm might be considered a suitable threshold density ([19]).
A lack of correlation between deadwood volume and living tree biomass in temperate broadleaved forests suggest that stand age and management, which also affect basal area and tree biomass, preclude the use of living biomass as a predictive variable of deadwood ([71]). However, it might be possible that correlations between living and deadwood biomass only emerge for stands with live tree stocking exceeding a certain threshold and/or when stands are experiencing self-thinning ([100]). When stands are under-stocked with a low number of small-sized trees, deadwood resources may be unpredictable and tree mortality may be a significant contributor of deadwood. Woodall & Westfall ([100]) found that the lowest levels of deadwood stocking were in stands with moderate levels of live tree stocking. Other studies showed that living tree volume alone failed to describe old-growth forest structure due to the high variation in tree size typical of these ecosystems ([64]).
Disturbance or self-thinning inducing tree mortality is the only way for deadwood stocking to increase, with live tree stocking shifting to either low or high levels. Deadwood accretion factors (e.g., disturbance, self-thinning, and senescence) would increase deadwood stocking in stands with high or low live tree stocking ([100]). This trend is shared by forests growing at high altitudes or latitudes (e.g., northern Europe or Alpine environments), where a more frequent incidence of windstorm-damages and natural disturbances, such as wind and ice storms, might explain the higher amount of deadwood generally reported in these contexts ([81]). Traditional harvest processes would reduce deadwood stocking in all forest stands, while decay progression diminishes deadwood amounts, especially in forests of temperate and warm climates. For example, the Mediterranean regions, with erratic precipitation regimes and temperature patterns, are generally characterized by faster decomposition rates due to higher temperatures when compared with northern Europe ([32]). However, no simple deadwood stocking recommendations can be applied, due to the inherent complexity of all the stand, site, and management factors that drive deadwood dynamics.
Conclusion
This paper provides a contribution to the analysis of saproxylic fungal biodiversity in relation to stand structural attributes in Mediterranean mountainous forests characterized by different management histories. Living tree volume, altitude, vegetation, and the frequency of species with ephemeral sporocarp lifespan played a crucial role in diversifying species distribution patterns. Particularly, the increase of the stand structural heterogeneity, related to the timespan from last management activities and to small-scale disturbances, deeply affects the occurrence and the diversity of wood-inhabiting fungi, as a consequence of the different microclimates occurring on the forest floor. The amount of deadwood at the study sites were related to the taxonomic and functional diversity of saproxylic organisms, including wood-inhabiting fungi, which were influenced by the deadwood volume and decay classes, according to decomposition processes ([74], [51]).
High CWD volumes and large living tree densities in old-growth forests influence the provision of habitat for ecologically important saproxylic organisms, thus supporting high levels of late-successional biodiversity. The study of old-growth and semi-natural forests was proved useful as reference tool to implement rehabilitation criteria in sustainable management, to maintain and increase biodiversity and other ecosystem services provided by managed forests. Forest management should mimic the natural thinning dynamics, increasing the amount of dead trees and the diversification of the vertical and horizontal tree layers, considering the good potential for restoring and increasing the diversity of saproxylic communities and their associated ecological functions.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Online | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Online | Gscholar
CrossRef | Gscholar
Online | Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Supplementary Material
Authors’ Info
Authors’ Affiliation
Dario Lunghini
Vito Mario Granito
Oriana Maggi
Silvia Pioli
Dipartimento di Biologia Ambientale, Sapienza Università di Roma, p.le Aldo Moro 5, I-00185 Roma (Italy)
Department of Agronomy, Mediterranean University of Reggio Calabria, loc. Feo di Vito, I-89060, Reggio Calabria (Italy)
Marco Marchetti
Dipartimento di Bioscienze e Territorio, Università degli Studi del Molise, c.da Fonte Lappone, I-86090 Pesche (Italy)
EFI Project Centre on Mountain Forests (MOUNTFOR), v. Edmund Mach 1, I-38010 San Michele all’Adige (Italy)
Corresponding author
Paper Info
Citation
Persiani AM, Lombardi F, Lunghini D, Granito VM, Tognetti R, Maggi O, Pioli S, Marchetti M (2015). Stand structure and deadwood amount influences saproxylic fungal biodiversity in Mediterranean mountain unmanaged forests. iForest 9: 115-124. - doi: 10.3832/ifor1304-008
Academic Editor
Giustino Tonon
Paper history
Received: Apr 03, 2014
Accepted: Jun 29, 2015
First online: Sep 08, 2015
Publication Date: Feb 21, 2016
Publication Time: 2.37 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2015
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 56898
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 45343
Abstract Page Views: 4425
PDF Downloads: 5711
Citation/Reference Downloads: 35
XML Downloads: 1384
Web Metrics
Days since publication: 3904
Overall contacts: 56898
Avg. contacts per week: 102.02
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2016): 33
Average cites per year: 3.30
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Review Papers
Linking deadwood traits with saproxylic invertebrates and fungi in European forests - a review
vol. 11, pp. 423-436 (online: 18 June 2018)
Research Articles
An index of structural complexity for Apennine beech forests
vol. 8, pp. 314-323 (online: 03 September 2014)
Review Papers
The forest biodiversity artery: towards forest management for saproxylic conservation
vol. 9, pp. 205-216 (online: 26 October 2015)
Research Articles
Saproxylic beetles in non-intervention and coppice-with-standards restoration management in Meerdaal forest (Belgium): an exploratory analysis
vol. 9, pp. 536-545 (online: 25 March 2016)
Research Articles
Consistency among forest structure and biodiversity potential index (IBP): an assessment of stand structural complexity for floodplain poplar woodlands
vol. 18, pp. 335-343 (online: 04 November 2025)
Research Articles
Diversity of saproxylic beetle communities in chestnut agroforestry systems
vol. 13, pp. 456-465 (online: 07 October 2020)
Research Articles
Do different indices of forest structural heterogeneity yield consistent results?
vol. 15, pp. 424-432 (online: 20 October 2022)
Book Reviews
National forest inventories: contributions to forest biodiversity assessments (2010)
vol. 4, pp. 250-251 (online: 05 November 2011)
Research Articles
The effects of forest management on biodiversity in the Czech Republic: an overview of biologists’ opinions
vol. 15, pp. 187-196 (online: 19 May 2022)
Research Articles
Investigating the effect of selective logging on tree biodiversity and structure of the tropical forests of Papua New Guinea
vol. 9, pp. 475-482 (online: 25 January 2016)
iForest Database Search
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword