Dead wood and stand structure - relationships for forest plots across Europe
iForest - Biogeosciences and Forestry, Volume 7, Issue 5, Pages 269-281 (2014)
doi: https://doi.org/10.3832/ifor1057-007
Published: Apr 14, 2014 - Copyright © 2014 SISEF
Research Articles
Abstract
Dead wood and stand structural parameters were sampled in eleven countries using standardized methods at about 90 intensive forest monitoring sites across large parts of Europe. Besides descriptions and correlation analyses of dead wood and stand structure parameters, a joint evaluation of both fields was performed by principal component analysis (PCA). The extracted principal components were subsequently regressed against important numerical and categorical site-related parameters like soil pH, altitude, or forest type. Dead wood volumes varied largely across plots, however, 77 percent of them had volumes below 25 cubic meter per hectare. While all fractions of dead wood - except cut stumps - reveal high intercorrelation, different aspects of stand structure varied more independently. Clark-Evans index, number of tree species and standard deviation of tree trunk diameters revealed as most self-contained. The 1st PCA axis covered 46 percent of the total variance and was mostly loaded by total dead wood volume denoting it as the feature differentiating forests most. The 2nd axis was primarily loaded by tree species diversity together with stem density and the Clark-Evans index. On the 3rd axis diameter differentiation of trees together with the volume of cut stumps prevailed, while the 4th was mainly related to the decay class of woody debris. Bivariate ex post analyses revealed country as a significant predictor of all PCA axes, underlining national forest legislations and management rules as crucial for all investigated structural features of forests. Forest type was related only to the 3rd and 2nd axis. Only the 3rd axis revealed significant relationships with some ecological site factors (age, number of tree layers, latitude, altitude). The outcome underlines the significance of nationally enacted forest legislations for both important structural and biodiversity-relevant features of forest ecosystems and encourages similar approaches with data from national forest inventories or monitoring systems.
Keywords
Structural Diversity, Principle Component Analysis, Forest Monitoring, ICP Forests, ForestBIOTA
Introduction
Both, living trees and its remains, decaying wood, constitute the main structural compounds of any forests. Stand structure has often been mentioned as an important driving force for species diversity in forests and close relationships between structural attributes of tree stands and their faunal diversity have been shown for several systematic groups or ecological guilds (e.g. carabid beetles: [26]; bats and birds: [35]; birds, saproxylic beetles and fungi: [80]; oscine birds: [81]; a felid: [104]; general habitat functions: [31]). Diversity of ground vegetation may also be linked to aspects of stand structure ([45], [86]). Berg et al. ([9]) identified in particular structural features of old forests as critical elements for threatened species in Sweden. Moreover, structural diversity may influence productivity, stability and resilience of forests ([60], [114], [93], [94]) and living trees are carrying for the most part carbon sequestration of forests.
Dead wood constitutes habitats for many species of invertebrates like saproxylic beetles, amphibians, mammals, birds, fungi, lichens, bryophytes, and even tree saplings (e.g., [44], [95], [112], [77], [68], [108], [84], [49]). Dead wood also represents a notable, albeit in large part transient carbon pool ([4]), as well as a source of mineral elements in soils ([40]), since decomposing tree trunks are true slow-releasing fertilizer pools ([15], [101]).
Forest management has a direct effect on stand structures and dead wood by planting, tending and harvesting trees. Compared to unmanaged stands this may result in changes of tree species composition, vertical layering, dbh distribution, dead wood amount and other structural attributes ([109], [122], [103]). Historical and recent forest management interventions exert negative and/or positive effects on biodiversity depending on forest type, species group considered, or management measures applied ([8], [85], [89], [11]). The number of old big trees is usually lower in managed than unmanaged forests, because classical forest management is based on rotations shorter than species’ longevity ([41]). In managed stands dead wood is often removed to avoid outbreaks of pest insect populations, to eliminate or reduce physical obstacles to silvicultural activities, or to reduce the risk of forest fires ([78]). As a result, the amount of dead wood typically is about 70% to 98% lower in managed forests than in comparable unmanaged forests ([39], [38], [56], [32]).
Since the United Nations Conference on Environment and Development (UNCED) held in 1992, the concept of sustainable multi-purpose forest management is internationally recognized and fostered. On the European level, the implementation of sustainable forest management by specific criteria and indicators is strongly promoted by the Ministerial Conference on the Protection of Forests in Europe (MCPFE, now Forest Europe). Within this context, dead wood and certain features of stand structure are recognized as important preconditions for biological diversity of forests at stand level ([102], [61], [73]). In the last decades, biodiversity-oriented management practices have been proposed to increase the quantity of dead wood and the presence of veteran trees; these include prolonging the rotation period, leaving dead trees in forests or even creating artificial high stumps from living trees (e.g., [54], [96], [1], [55], [7]).
Because of its ecological importance and its role in C sequestration, surveys of dead wood have recently been included in most National Forest Inventories (NFIs) and monitoring programmes (e.g., [34], [20], [119]). However, protocols for dead wood assessment differ among European NFIs, and data are difficult to compare ([46]). Schuck et al. ([105]) analysed inventory methods for 22 countries and found differences among attributes measured, diameter thresholds and sampling design. Furthermore, there is also a need for standardized indices of stand structure, that have been shown by Neumann & Starlinger ([82]) to be suitable indicators to asses biodiversity- related aspects of forest management. Since, similar investigations with the aim of proposing methods for harmonizing results from NFIs and probably adjusting NFIs’ field methods have been undertaken. These activities resulted in bridging functions for parameters among NFIs ([121], [75], [123], [98]).
Up to now, the relationships between silviculture, stand structure, dead wood and other aspects of biodiversity have rarely been studied in an integrated manner on a large scale. This study investigates the relationships between dead wood and stand structure based on a survey at 90 intensive monitoring plots across Europe. The survey was carried out in 2004 on plots in the Czech Republic, Denmark, Finland, Germany, Greece, Italy, The Netherlands, Slovakia, Spain, Switzerland, and Ukraine. The objectives of this large-scale evaluation were to: (i) identify a sets of variables suitable to characterise dead wood structures and a set to characterise stand structures; (ii) detect the relationships between dead wood and stand structure characteristics; (iii) investigate the relationships between PCA factors derived from both, dead wood and stand structure, with influencing factors such as forest type, latitude, altitude or atmospheric deposition, cutting activities and silvicultural system.
This evaluation therefore interlinks features of dead wood and stand structure with general features of different forest ecosystems and their management across Europe.
Methods
Material
This study has been carried out on a subset of the ICP Forests (International Co-operative Programme on Monitoring and Assessment of Air Pollution Effects on Forests) / EU Forest Focus permanent intensive monitoring (Level II) plots that had been selected by countries participating in the ForestBIOTA (Forest Biodiversity Test phase Assessments) project. Both surveys included 91 (dead wood) and 89 (stand structure, a subset of the dead wood plots) plots in 11 countries. According to ICP Forests ([51]) one of the selection criteria for Level II plots was to have typical forests, and thus the plots are mainly located in managed forests. However, in a few cases stands recently abandoned or even unmanaged for some decades (e.g., [117]) have been included.
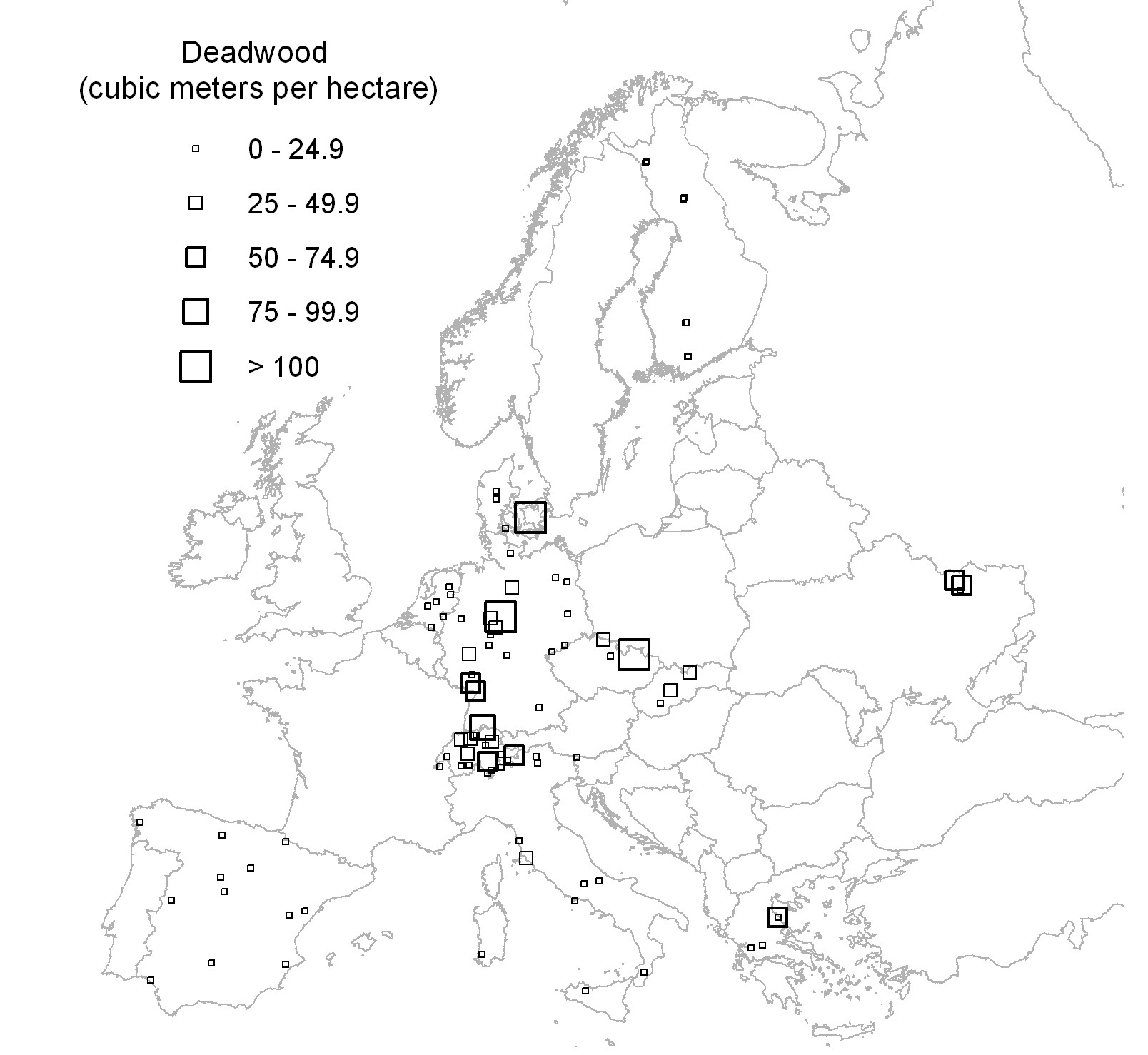
Data on dead wood and stand structure were collected according to the ForestBIOTA field protocol ([29]). The survey scheme consisted of a magnetic north-oriented 50 x 50 m square plot with a cluster of four circular subplots with 7 m radii and centred on the corners of a magnetic north-oriented 26 x 26 m square, thus summing up to a total of 616 m2 ([28] - Fig. 1).
Fig. 1 - Distribution of the ForestBIOTA plots, labelled with classes of their total dead wood volume (m3 ha -1).
A complete inventory of all standing (Vst) and downed dead trees (Vdt) along with broken snags (Vsn) in the 50 x 50 m plot was performed, whereas cut stumps (Vsm) and lying dead wood pieces (Vol) were surveyed within the four circular subplots ([116]). Tab. 1 lists the attributes measured for each dead wood element and methods to assess single volumes. On the four circular plots, both fine (elements with a bigger diameter ≤ 10 cm) and coarse (elements with a bigger diameter > 10 cm) woody debris were recorded down to a minimum diameter of 3 cm, whereas neither litter nor attached dead wood and hollows were noted. The total amount of dead wood (V) within the plots was calculated as the sum of all mentioned components on a volume per ha basis [m3 ha-1], whereas the volume of cut stumps and lying dead wood pieces were summed over the circular subplot level and transformed into volume per hectare. Variance, standard deviation, and coefficient of variation [%] of woody debris were computed as well. For each piece of dead wood the decay class was determined after Hunter ([50]) and respective amounts calculated for each plot (Vd1 to Vd5). The distributions between coarse and fine woody debris of total dead wood volume were computed as well.
Tab. 1 - List of attributes surveyed in the field for dead wood and stand structure assessment. (*): References: Finland, [59]; Greece, [3]; Italy, [16]; Spain, NFI double entry volume equation; Germany, [58]. Volumes for Czech Republic, Denmark, Slovak Republic, Switzerland, The Netherlands and Ukraine were computed by means of German tables. (**): Length is measured from the thicker end of the piece to the point where the diameter is less than 3 cm; d1.3m equates DBH, however is more suitable for lying dead wood; (d1/2h): diameter at half height.
| Attribute | Survey unit |
Height/length | Diameter (d) | Coordinates | Volume (m3 ha-1) |
Species/Decay |
|---|---|---|---|---|---|---|
| Living trees |
Plot | h: tree height | d1.3 m ≥ 5 cm | Yes | Vlt: NFI double-entry volume tables* | Species |
| Standing dead trees |
Plot | h: tree height | d1.3 m ≥ 5 cm | No | Vst: NFI double-entry volume tables* | Decay: Hunter ([50]) |
| Broken snags |
Plot | h: Height of stem truncation |
h>4 m: d1.3 m ≥ 5 cm | No | Vsn: Applying a reduction factor to NFI double-entry volume tables* | Decay: Hunter ([50]) |
| h≤ 4 m: d1/2h ≥ 5 cm | No | Huber’s formula | Decay: Hunter ([50]) |
|||
| Downed dead trees |
Plot | l: Total tree length | d1.3 m ≥ 5 cm | No | Vdt : NFI double-entry volume tables* | Decay: Hunter ([50]) |
| Lying dead wood pieces |
Subplots (thicker end) |
l: Length of the piece** | Diameter at half length (when diameter at thicker end is ≥ 5 cm) and its thicker end lies within the boundary of the subplot) | No | Vol: Huber’s formula | Decay: Hunter ([50]) |
| Cut stumps | Subplots | h: Height at the level of cut |
Diameter at cut ≥ 10 cm | No | Vsm: Huber’s formula | Decay: Hunter ([50]) |
The assessment of the forest stand structure referred to living trees only. On the 50 x 50 m square plot the survey of trees included tree coordinates in addition to the parameters of the ICP Forests Growth survey ([22] and earlier versions), which are diameter at breast height (dbh), tree species and tree heights (h, only subsamples). Measurements were carried out on all standing trees with dbh ≥ 5 cm in 2004 (Tab. 1) or derived from previous comparable inventories ([21], [27]). Three plots deviated slightly from this standard design with regard to orientation and outer alignment forced by local orography and/or boundaries of management units.
All the indices chosen to describe the stand structure are described in Appendix 1. The first group consists of spatially explicit indices (Clark Evans index: CE; contagion index: W; mingling index: MI; diameter differentiation: T). CE and W are measures of the regularity of the horizontal distribution of tree specimen, MI refers to separation of the different tree species in space at a scale of small groups of trees. Also T reflects the degree of diameter differentiation at small spatial scales. While CE refers to the total plot area, W, MI, T are based on the “structural group of four (trees)”. As a consequence of a pilot study, systematic samples of the “structural groups of four” were not taken in the field, but computed on the basis of tree coordinates. Indices without regard to explicit spatial relationships of trees are standard deviation of dbh (SD), number of tree species (SN), Shannon index (H’), evenness (J’), and Simpson index (D’). The latter three were calculated for both, numbers and basal areas of individual trees. Density of living tree stems (DE), basal area (BA) and volume of living trees (Vlt) were calculated for all trees with dbh ≥ 5 cm.
At the base of the 50 x 50 m quadrats the following parameters were additionally registered in the field: number of tree layers with heights greater than 5 m, type of tree species mixture, visually estimated canopy closure, ancient forest site, ranked cutting activity index, silvicultural and cutting system according to definitions given in Tab. 2. Each plot was assigned to the forest type classification scheme of the BEAR project ([5], see also [28]), which is almost congruent with the EUNIS classification ([23]).
Tab. 2 - List of simple estimates recorded in the field and used in final bivariate regression and main effect models (cf. Tab. 6).
| Parameter | Estimation/classes | N of cases per class / quartiles etc. |
|---|---|---|
| Main tree species |
Main tree species with coverage (10% steps) plus additional tree species |
Q25 : 1, Med: 2, Q75 : 4, Max.: 13 |
| Number of tree layers > 5 m |
one layer | 43 (52.4%) |
| two layers (each min. of 10% coverage) | 39 (47.6%) | |
| multilayered (each min. of 10% coverage) | 0 | |
| irregular | 0 | |
| Canopy closure |
percentage coverage of tree layer > 5 m (estimated in 5% steps) |
Min: 0, Q25 : 55, Med: 70, Q75 : 80, Max: 100 |
| Ranked index of cutting activities |
no sign of cuttings, natural development (0) | 6 (7.4%) |
| signs of past cuttings, however, abandoned to natural development (1) | 36 (44.4%) | |
| signs of recent and/or older cuttings visible (2) | 39 (48.2%) | |
| Silvicultural system |
high forest | 62 (79.5%) |
| coppice without standards | 5 (6.4%) | |
| coppice with standards | 7 (9.0%) | |
| plantation | 4 (5.1%) |
The pHCaCl2 of the organic and upper mineral soil layers and the annual wet throughfall deposition of sulphur and nitrogen had been determined for these plots within the regular ICP Forests surveys according to national protocols meeting the requirements described in the ICP Forests Manual ([51]). Values had been taken from the ICP Forests database available in 2006 and referring approximately to the time span from 1995 to 2004 (Ukraine delivered pH information separately).
Evaluations
All dead wood related parameters were log-transformed to approximate normal distributions. To give answers to the questions, which of the parameter from both domains are most distinguishing within this geographically broad-scaled dataset, all dead wood parameters and, separately, all stand structural parameters (and indexes) were correlated with each other (PROC CORR, SAS 9.2).
As the domain-related correlation analyses with high numbers of parameters and comparatively low numbers of cases revealed a complex dependency structure, multiple regression or respective mixed models seemed not to be suitable approaches. Therefore, principal component analysis (PCA, PROC FACTOR, SAS 9.2) was applied as an appropriate means to combine information from the dead wood and the stand structure survey (e.g., [124]). In order to avoid distortion within the ordination, only the statistically more self-standing (less intercorrelated) parameters with correlation coefficients < |0.7| in the above mentioned domain- related correlation analyses were used. For dead wood, generally preference was given to volumes over highly correlated densities of pieces.
Finally, we investigated the general relationships of extracted PCA axis derived from dead wood and stand structural features with additional stand characteristics, site factors or proxies reflecting climate or general country-specific forest management practices, usually fixed by forest legislations. To this purpose the axis-related PCA plot scores were regressed against such additionally assessed parameters (Tab. 2) or parameters drawn from the ICP Forests programme like stand age, soil pH, S and total N throughfall deposition, absolute yield, latitude, and altitude ([51]). If categorical parameters were used as predictors, main effects models were applied (PROC GLM, SAS 9.2). All p-values were adjusted post-hoc for multi-comparisons with the Bonferroni-Holm procedure. The selection of the parameter was based on both availability and hypothetical importance of processes in forest ecosystems. Other available parameters, like longitude or discrete N-compounds, had tentatively been tested. However, they have been excluded from this family of bivariate analyses in order to avoid overload of the Bonferroni-Holm procedure.
Results
Dead wood
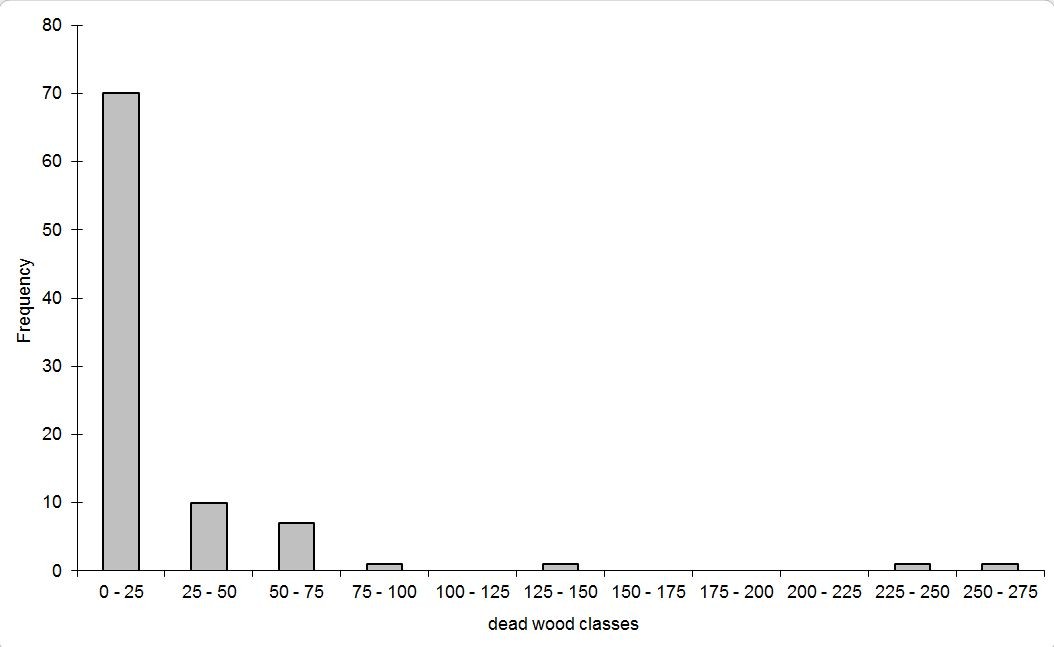
Total dead wood volume of the 91 plots varied between 0 and 258 m3 ha-1. The frequency distribution was inverse J-shaped, as 77% of the examined plots had dead wood volumes less than 25 m3 ha-1 (Fig. 2). The highest values with total dead wood volumes greater than 100 m3 ha-1 occurred in three plots from central Europe (Fig. 1). The plot with the highest amount of dead wood was a beech stand in a strict forest reserve ([25]). The high amount of dead wood on a German site with spruce forest ([24]) was due to windthrow in combination with an ongoing infestation by stem boring insects. The third plot with a dead wood volume > 100 m3 ha-1, also stocked by spruce, was exposed to extreme weather conditions close to the upper timber line of the Jeseníky mountains (Czech Republic), which suffered in the second half of the last century from heavy air pollution and was nearly without management (Buriánek, personal communication).
Fig. 2 - Frequency distribution (number of plots) against classes of total dead wood volume [m 3 ha -1] at the monitoring plots.
The main share of woody debris consisted of coarse dead wood pieces. The coefficient of variation of the volumes of all lying and standing dead wood pieces varied at plot level between 0% and 100% with a mean of 32% and a median of 25%. The 1st and 3rd quartiles were 12% and 48% respectively. Volumes of lying dead wood (downed dead trees, lying dead wood pieces) and cut stumps exceeded the volume of the standing dead wood (standing dead trees and broken snags). The density of dead wood pieces per plot varied between 0 and 4000 per ha. Volumes and densities of dead wood pieces per plot in general and within the different types were strongly intercorrelated (Tab. 3), thus only volumes were used for subsequent calculations. 38% of the monitoring plots were dominated by dead wood decay class 3, while decay classes 2 and 1 dominated at 24% and 12%, respectively. More decomposed dead wood of decay classes 4 and 5 dominated at 19% and 7% of the plots, respectively.
Tab. 3 - Pearson correlation coefficients and significance levels of correlation between parameters referring to dead wood. All parameters have been log-transformed, n = 89. (Vto): total volume of dead wood; (nto): density of all dead wood pieces; (vol): volume of lying dead wood pieces; (nol): density of lying dead wood pieces; (vsn): volume of broken snags; (vdt): volume of downed dead trees; (ndt): density of downed dead trees; (vst): volume of standing dead trees; (nst): density of standing dead trees; (vsm): volume of stumps; (nsm): density of stumps; (v<10): fine necromass volume; (n<10): density of fine necromass pieces; (v>10): coarse necromass volume; (n>10): density of coarse necromass pieces; (vd1-vd5): volume of decay classes 1 to 5. (*): p < 0.05; (**): p < 0.01; (***): p < 0.001.
| Var. | Nto | vol | nol | vsn | Vdt | ndt | Vst | nst | vsm | nsm | v<10 | n<10 | v>10 | n>10 | vd1 | vd2 | vd3 | vd4 | vd5 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vto | 0.71*** | 0.64*** | 0.54*** | 0.45*** | 0.60*** | 0.52*** | 0.47*** | 0.36*** | 0.13 | 0.02 | 0.60*** | 0.52*** | 0.98*** | 0.66*** | 0.37*** | 0.65*** | 0.73*** | 0.512*** | 0.49*** |
| Nto | - | 0.75*** | 0.78*** | 0.36*** | 0.32** | 0.30** | 0.33** | 0.33** | 0.33** | 0.34** | 0.74*** | 0.79*** | 0.64*** | 0.78*** | 0.33** | 0.57*** | 0.69*** | 0.475*** | 0.36*** |
| vol | - | - | 0.94*** | 0.25* | 0.30* | 0.20 | 0.31** | 0.24* | 0.28** | 0.28** | 0.75*** | 0.76*** | 0.59*** | 0.51*** | 0.31** | 0.43*** | 0.62*** | 0.455*** | 0.26* |
| nol | - | - | - | 0.20 | 0.25* | 0.18 | 0.29** | 0.26* | 0.26* | 0.28** | 0.79*** | 0.86*** | 0.47*** | 0.46*** | 0.24* | 0.42*** | 0.60*** | 0.428*** | 0.21* |
| vsn | - | - | - | - | 0.28** | 0.31** | 0.27* | 0.26* | 0.04 | 0.04 | 0.36*** | 0.30** | 0.44*** | 0.37*** | 0.21 | 0.29** | 0.37*** | 0.344*** | 0.32** |
| Vdt | - | - | - | - | - | 0.92*** | 0.46*** | 0.40*** | -0.15 | -0.19 | 0.44*** | 0.34** | 0.59*** | 0.29** | 0.44*** | 0.36*** | 0.42*** | 0.317** | 0.34** |
| ndt | - | - | - | - | - | - | 0.51*** | 0.52*** | -0.17 | -0.19 | 0.41*** | 0.32** | 0.48*** | 0.28** | 0.39*** | 0.34** | 0.42*** | 0.261* | 0.25* |
| Vst | - | - | - | - | - | - | - | 0.93*** | -0.27** | -0.19 | 0.48*** | 0.41*** | 0.41*** | 0.15 | 0.32** | 0.42*** | 0.38*** | 0.145 | 0.08 |
| nst | - | - | - | - | - | - | - | - | -0.23* | -0.14 | 0.45*** | 0.41*** | 0.28*** | 0.12 | 0.36*** | 0.37*** | 0.30** | 0.061 | -0.03 |
| vsm | - | - | - | - | - | - | - | - | - | 0.92*** | 0.02 | 0.08 | 0.14 | 0.48*** | 0.06 | 0.21* | 0.11 | 0.106 | 0.07 |
| nsm | - | - | - | - | - | - | - | - | - | - | 0.02 | 0.10 | 0.02 | 0.44*** | 0.11 | 0.17 | 0.01 | -0.003 | 0.02 |
| v<10 | - | - | - | - | - | - | - | - | - | - | - | 0.94*** | 0.48*** | 0.39*** | 0.25* | 0.43*** | 0.70*** | 0.477*** | 0.35*** |
| n<10 | - | - | - | - | - | - | - | - | - | - | - | - | 0.42*** | 0.41*** | 0.26* | 0.42*** | 0.62*** | 0.480*** | 0.29** |
| v>10 | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.67*** | 0.35*** | 0.620*** | 0.69*** | 0.524*** | 0.49*** |
| n>10 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.17 | 0.49*** | 0.57*** | 0.517*** | 0.48*** |
| vd1 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.33** | 0.09 | 0.046 | 0.08 |
| vd2 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.55*** | 0.264* | 0.17 |
| vd3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.562*** | 0.27** |
| vd4 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.46*** |
Stand structure
Tab. 4 summarizes all univariate properties of stand-related parameters and calculated indexes. Additionally, forest type for plots with maximum and minimum values was given. SN, H’, D’, and MI were typically right-skewed. Maximum SN was reached in a plot with “Mediterranean broadleaved woodlands”, while SH, SI and MI, all reached their maximum values in plots with “meso- to eutrophic oak forest”. All indexes related to diversity including J’ were strongly intercorrelated (Tab. 5). Therefore, only SN was used for PCA.
Tab. 4 - Univariate characteristics of stand-related parameters and indices; additionally forest type per plot with the respective minimum and maximum values are given. (*): coniferous plantation misclassified as Hemiboreal forest.
| Index | Forest type at minimum |
min | Q25 | med | mean | std | rV [%] |
Q75 | max | Forest type at maximum |
|---|---|---|---|---|---|---|---|---|---|---|
SN
|
a few | 1 | 1 | 2 | 3.06 | 2.31 | 75 | 4 | 13 | Med. broadleaved |
H’
|
a few | 0 | 0 | 0.22 | 0.42 | 0.48 | 116 | 0.73 | 1.8 | meso-/eutroph oak |
D’
|
a few | 0 | 0 | 0.1 | 0.22 | 0.25 | 116 | 0.43 | 0.79 | meso-/eutroph oak |
J’
|
Taiga | 0 | 0.09 | 0.35 | 0.37 | 0.29 | 79 | 0.59 | 0.99 | fir/spruce |
MI
|
Hemiboreal* | 0 | 0 | 0.07 | 0.18 | 0.22 | 122 | 0.33 | 0.75 | meso-/eutroph oak |
W
|
a few | 0.5 | 0.57 | 0.58 | 0.59 | 0.04 | 7 | 0.61 | 0.75 | Med. broadleaved |
CE
|
Med. broadleaved | 0.39 | 0.96 | 1.14 | 1.13 | 0.29 | 26 | 1.37 | 1.79 | Taiga |
T
|
Taiga | 0.11 | 0.23 | 0.32 | 0.32 | 0.11 | 36 | 0.4 | 0.64 | lowland beech |
SD
|
Taiga | 3.19 | 6.76 | 10.26 | 10.87 | 5.02 | 46 | 15.3 | 24.3 | lowland beech |
Dlt
|
Med. broadleaved | 120 | 316 | 492 | 672.4 | 628.5 | 93 | 785 | 4240 | Med. broadleaved |
BA
|
Med. broadleaved | 6.04 | 27.77 | 33.24 | 11.78 | 11.77 | 35 | 41.18 | 34.03 | fir/spruce |
V (78) |
Med. braodleaved | 11.9 | 202.4 | 355.9 | 363.7 | 211 | 58 | 499.8 | 849.9 | fir/spruce |
Tab. 5 - Pearson correlation coefficients and significance levels of correlation between stand structural indices. Values in brackets indicate the number of valid cases per variable. Indices covering the same aspect of structure are labeled with the same letter (a, b, c, d). (SN): tree species number; (H’): Shannon index; (D’): Simpson index; (MI): mean mingling; (J’): evenness; (W): contagion index; (CE): Clark Evans index; (T): diameter differentiation; (SD): standard deviation of dbh; (BA): stand basal area; (DE): density of living trees. (*): p < 0.05; (**): p < 0.01; (***): p < 0.001.
| Index | H’ (88) a |
D’ (88) a |
MI (89) a |
J’ (60) a |
W (89) b |
CE (89) b |
T (89) c |
SD (89) c |
BA (89) d |
DE (89) d |
|---|---|---|---|---|---|---|---|---|---|---|
SN (88) a |
0.87*** | 0.80*** | 0.71*** | 0.42*** | 0.39*** | -0.37*** | 0.28** | 0.13 | 0.09 | 0.40*** |
H’ a |
- | 0.99*** | 0.93*** | 0.85*** | 0.31** | -0.35*** | 0.41*** | 0.36*** | 0.14 | 0.02 |
D’ a |
- | - | 0.96*** | 0.92*** | 0.29** | -0.34** | 0.45*** | 0.40*** | 0.17 | 0.22* |
MI a |
- | - | - | 0.90*** | 0.19 | -0.26* | 0.49*** | 0.47*** | 0.21* | 0.12 |
J’ a |
- | - | - | - | 0.06 | -0.20 | 0.47*** | 0.54*** | 0.14 | -0.08 |
W b |
- | - | - | - | - | -0.77*** | 0.20 | -0.03 | -0.08 | 0.55*** |
CE b |
- | - | - | - | - | - | -0.37*** | -0.16 | 0.05 | -0.45*** |
T c |
- | - | - | - | - | - | - | 0.79*** | 0.26 | 0.00 |
SD c |
- | - | - | - | - | - | - | - | 0.40*** | -0.33** |
BA d |
- | - | - | - | - | - | - | - | - | 0.19 |
CE reached its minimum (clustered horizontal tree distribution) in a plot with “Mediterranean broadleaved woodland”, while in a “taiga woodland” plot a remarkable tendency towards regularly spaced trees was found. The overall range of the contagion index Wi was rather small and has not been used for PCA due to its close correlation with CE. T and SD behaved rather similar, with the latter being broader ranged. BA varied considerably between around 6 m2 ha-1 for a plot in Spain and almost 70 m2 ha-1 for a plot in Slovakia. Vlt was closely related to BA (r = 0.76) and had therefore been excluded from PCA. Tree density covered a wide range from 120 to 4240 trees ha-1.
Integrated evaluations
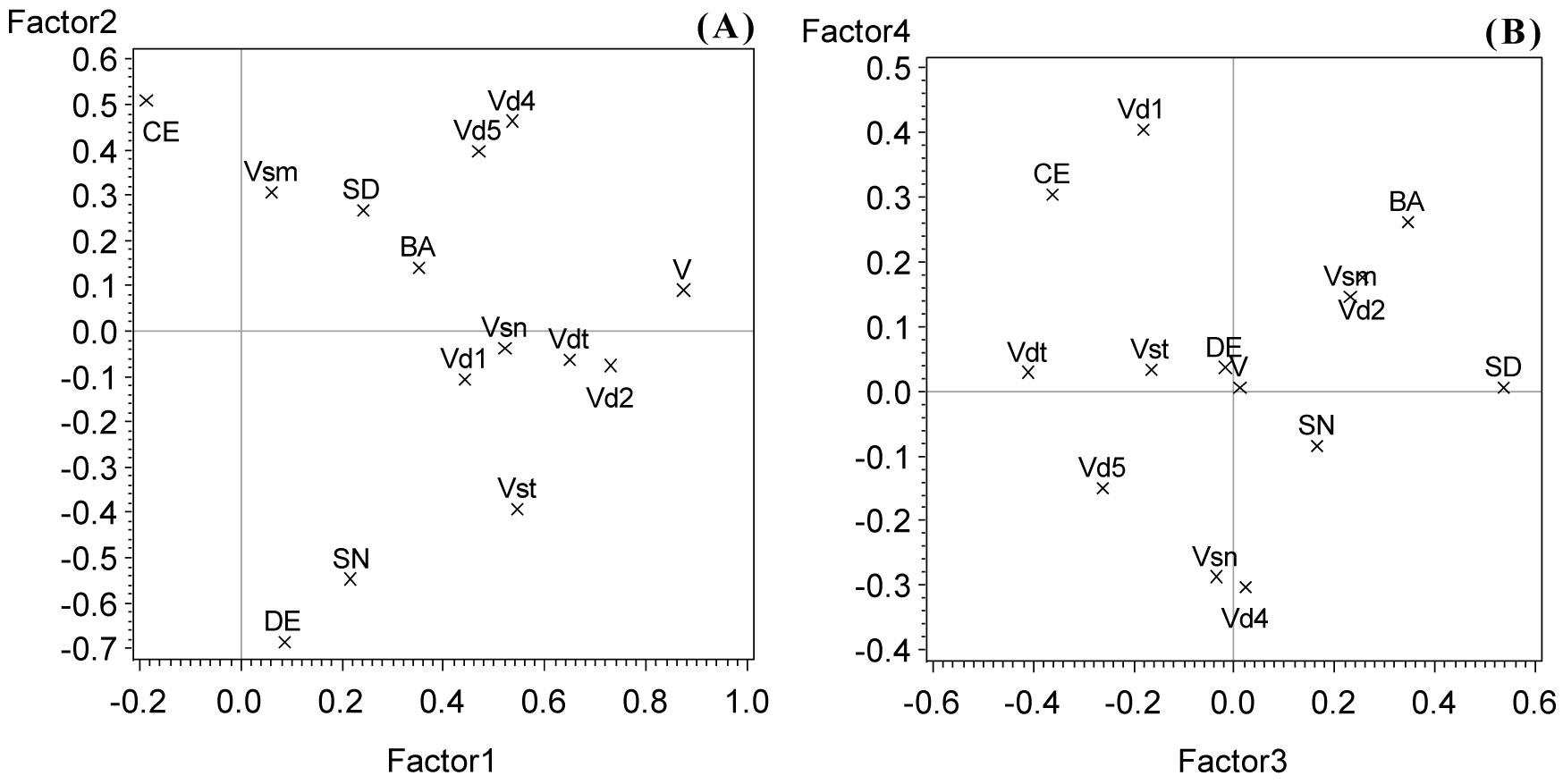
After the domain-related correlation analyses (Tab. 3 and Tab. 5) 14 meaningful and well differentiated parameters from both domains - dead wood and stand structure - were used for a combined PCA (see legend of Fig. 3). The resulting model accounted for 46% of the total variance on the first axis and 25%, 14%, 8%, and 7% on the second, third, fourth, and fifth axis, respectively.
Fig. 3 - Axis 2 against axis 1 (A) and axis 4 against axis 3 (B) of a PCA with dead wood and stand structural parameters at plot level. (CE): Clark-Evans index; (SD): standard deviation of stem diameters; (SN): tree species number; (BA): basal area; (DE): tree density; (V): log volume of total dead wood; (Vst): log volume of standing dead wood; (Vsm): log volume of cut stumps; (Vsn): log volume of broken snags; (Vdt): log volume of downed dead trees; (Vd1), (Vd2), (Vd4), (Vd5): log volume of dead wood decomposition class 1, 2, 4, and 5, respectively.
The 1st axis was mainly loaded by the total amount of dead wood volume, which was correlated with almost all types of dead wood reaching also higher scores on the first axis, except for the volume of cut stumps (Fig. 3a). This means that the amount of dead wood was the most differentiating feature of those investigated forest stands. The highest loading on the 2nd axis was on the negative side reached by stem density associated with tree species diversity and - on the positive side - the Clark-Evans index (Fig. 3a). This implies that low density of stems and low numbers of tree species coincide with regular spacing of tree individuals and vice versa. The mass of more to highly decayed dead wood (classes 4 and 5) reached also higher positive scores on the 2nd axis. The 3rd axis was mainly loaded by standard deviation of stem diameters (Fig. 3b) and volume (and density) of cut stumps, at which more differentiated stands go along with low volumes of cut stumps. The 4th axis was mainly loaded by the volume of least decayed woody debris (class 1) on one side, and on the other side - but less distinctly - the amount of woody debris belonging to decay class 4. The 5th axis (not shown) was mainly loaded by stem density and the volume of cut stumps. Both parameters are already involved in significantly loading the 2nd axis. The 5th axis seems to bind remaining fractions of the total variance not consumed by the more important axes and might not be of any substantial self-contained importance. Therefore, it was not regarded any further.
Finally, family-wise bivariate ex post analyses by regression and main effect models between plot scores of the PCA axes and additional factors or categories were performed (Tab. 6). With the 1st axis only country revealed significant coincidences. This obvious lack of relationships found was in distinct contrast to the high importance of this PCA axis in terms of explained total variance. Also the 2nd PCA axis lacked significant relationship with any of the numerical parameters. Instead, high amounts of the variation of this axis could again be explained by country and additionally by forest type and the silvicultural system. Regression analyses between the 3rd PCA axis and stand and site factors revealed closer relationships with the number of tree layers, stand age, latitude, and altitude. Also almost 50% of the variance of the plot scores on the 3rd axis could be explained by forest type, which was an even stronger relationship as with country. The 4th axis - mainly related to the decay status of woody debris - was again only related to country. A number of additional, ecologically-relevant factors like canopy closure, absolute yield, soil pH-value, S or N throughfall deposition did not show any significant relationship with plot scores at any of the PCA axes.
Tab. 6 - Significant relationships of a family of bivariate regression and main effect models (R2 values are given with p > F, p-values adjusted for multi-comparisons by Bonferroni-Holm procedure) between plot-related factors from the combined PCA of deadwood and stand structures on one side and selected stand and site-related numeric or categorical variables as predictor on the other side.
| Predictants | PCA Factor (axis) | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|
| Categorical predictands (main effect models) |
Country | 0.473** | 0.442** | 0.440** | 0.435** |
| Forest type | - | 0.473** | 0.491*** | - | |
| Sylvicultural system | - | 0.287** | - | - | |
| Main tree species | - | - | - | - | |
| Numeric predictands (linear regression models) |
Mean stand age | - | - | 0.221** (+) | - |
| Number of tree layers | - | - | 0.226** (+) | - | |
| Canopy closure | - | - | - | - | |
| Index of cutting activities | - | - | - | - | |
| Absolute yield | - | - | - | - | |
| Latitude | - | - | 0.226** (-) | - | |
| Altitude | - | - | 0.150* (+) | - | |
| pH mineral layer 0-10 cm depth | - | - | - | - | |
| S throughfall deposition | - | - | - | - | |
| Ntot throughfall deposition | - | - | - | - |
Discussion
Dead wood
Dead wood volumes within the 91 plots were generally low when compared with the amounts found in European forest reserves. Hahn & Christensen ([41]), Ódor et al. ([85]), and Christensen et al. ([17]) give 130 m3 ha-1 as average for beech stands in European forest reserves. In boreal spruce forests Siitonen et al. ([109]) found 111 m3 ha-1 of coarse woody debris in old-growth forests, but only 14 and 22 m3 ha-1 in mature and over-mature managed forests, respectively. The three ForestBIOTA plots with the highest amount of dead wood were influenced by extreme weather conditions, hit by insect infestations or suffered from heavy historic air pollution. Even if two of these plots experienced a restricted management, no general relationship between signs of cutting activities and the amount of dead wood could be corroborated (Tab. 6). This does not necessarily mean that such a relationship does not exist, however, the dataset was too small to support models with nested dependency structures or interaction effects between country and management intensity. For instance, Fridman & Walheim ([32]) calculated 7 m3 ha-1 for managed and 30 m3 ha-1 for unmanaged forests in Sweden.
As already mentioned, the ICP Forests manual recommends that intensive monitoring plots should be located in managed forests ([51]), thus the generally low quantity of dead wood is not surprising. Mean dead wood volumes are in the order of magnitude known from national forest inventories in Europe, even if harmonization efforts between dead wood inventories of different states have not yet been finalized ([98]). For Switzerland 19 m3 ha-1 were estimated on average ([12]), for Italy 9 m3 ha-1 ([91]). For Germany, after severe storm events occurred in the last years, Bolte & Polley ([13]) reported that federal states forests contained in 2008 on average 31.7 m3 ha-1 of dead wood and privately owned forests 18.2 m3 ha-1. Some years earlier 11.5 m3 ha-1 were calculated for this country ([10]). Such a difference might be climatically caused (storms) or methodological shifts may have occurred between such subsequent inventories. In general, it has been estimated that only 2 to 30% of the dead wood found in unmanaged forests occurs in managed forests ([39], [38], [56], [32]). However, this relation may change in the future as dead wood might be kept within the forests due to nature-like forest management practices in many countries. Also increasing adverse weather condition like storms and insect calamities may locally or regionally lead to higher the amounts of dead wood in forests. Contrary to this development, more (dead) wood might be extracted from forests in the near future due to enhanced societal demands for woody biomass ([47]).
Direct comparisons of dead wood estimates, however, need to be interpreted with care as results depend considerably on definitions of dead wood fractions and on the sampling design applied ([12], [83], [98]). For instance, the size of sampled plots is of distinctive relevance ([19]) and may vary considerably. Smaller plots are always prone to sampling inaccuracies if no probabilistic repeated sampling is adopted, and even in this case there is the risk to create artefacts, as shown by Rubin et al. ([100]). In this study, for most fractions 2500 m2 plots were used, almost 4 times larger than the 672 m2 units used by McRoberts et al. ([74]). As the spatial heterogeneity of dead wood in forest can be considerable, a meta-analysis including all methodological aspects ([71]) should be of great value.
Most categories of dead wood - with the exception of cut stumps - were found to be considerably intercorrelated, suggesting that recent forest management practices is influencing the amount of dead wood in general and less compartmentalized fractions of it. Country as a categorical parameter should integrate both, recent and historic forest management practices as von Oheimb et al. ([118]) have shown that also historical conditions can influence the amount and pattern of dead wood occurrence. This finding is supported by the exclusiveness of the relationship between country and PCA factor 1 (Tab. 6), which is mainly loaded by dead wood volume (Fig. 3a). This result seems to underline the importance of nationally enacted recent but also historic forest legislations. For instance, in many Mediterranean countries stand cleaning after harvesting is strictly recommended as a measure to prevent wildfire by reducing the amount of dead wood ([78], [6]). As none of the geographically or ecologically relevant factors like latitude (mainly a proxy for climatic differences) or pH (as an indicator for soil properties) are correlated to this axis, such factors seem to be of minor importance against national forest legislations and respective management practices.
Under natural conditions the probability that trees die of old age or fall prey to pathogens or parasites should increase with aging. If not removed, this should lead to larger amounts of dead wood with increasing stand age ([109], [43]). Meyer & Schmidt ([76]) showed that in unmanaged beech forests dead wood increased by a mean net rate of about 1 m3 ha-1 y-1. However, such an intuitionally obvious relationship could not be corroborated by this study, most likely due to recent or earlier forest management interventions and/or the strength of the last severe natural disturbance.
The 4th axis was mainly loaded by decay class 1 and less distinctly the volume of cut stumps on the positive and - also less distinctly - decay class 5 on the negative side. This implies that higher amounts of freshly accrued dead wood hardly occur together with greater amounts of rather decomposed dead wood. This finding points at a primarily discontinuous formation of dead wood. However, none of the external numeric parameters were related to this axis respectively process. It can be assumed that it is mainly the elapsed time since a major disturbance that determines the predominant decay class of dead wood at plot level, even if decomposition rates have to be considered, which depend on tree species, size and other features of dead wood as well as climatic and soil conditions ([44], [66]).
As the volume or density of dead wood pieces are under the recent conditions of forest in Europe largely independent from natural drivers and strongly influenced by management practices, dead wood related parameters (exclusive cut stumps) could substantially contribute to or could even be used directly as an indicators of naturalness ([105], [65], [6]). In contrast, the density (and volume) of cut stumps might be taken as an indicator or become part of an indicator of forest management intensity ([109], [72]). However, a respective bivariate inference approach (not shown) did not reveal a significant result.
Stand structure
The quantification of different aspects of stand structure has been discussed for a long time ([33], [92], [71]). Many parameters have been used so far, ranging from simple measures to sophisticated indexes ([30], [71]) or spatial statistics ([97], [52]). It is still a matter of debate which of the indexes provide the most reasonable, adequate, and comparable results. Selected parameters or derived indexes should reveal meaningful results in ecological terms, be sensitive to structural differences between stands, and to structural dynamics as well. Furthermore, it is desirable that data assessments are widely compatible with standards of contemporary forest inventories. The parameters or indexes applied in this study address three aspects, namely: (i) horizontal distribution of trees; (ii) differentiation of tree dimensions; and (iii) tree species composition:
- The widely-used Clark-Evans index ([69]) performed well in respect to univariate statistical criteria and revealed a wider spread of values than the contagion index
Wi, is easier to compute than theWi and is based on all specimens of a plot, whileWi is based on structural groups of 4 trees only. Moreover,CErevealed in the PCA the highest loading among the stand structural parameter. AsWi andCEare highly correlated with each other (r = 0.77), the latter should be given preference. This might not be valid, if one wants to focus on small-scale neighborhood conditions of trees ([2]) or perform detailed analyses of spatial patterns (e.g., Ripley’s L function, see [92]), which both might be of interest for up-scaling studies ([88]) or planning of local multi-purpose management measures. - Standard deviation of
dbhis well suited to describe the differentiation of tree dimensions.SDwas used as a diversity measure of forests ([79]) and revealed in this study a good self-contained performance within the PCA. - Tree species diversity has at least three aspects: (a) species number; (b) relative quantitative partitions of species (in terms of stem number or basal area); and (c) the spatial separation or mixture of specimen belonging to different tree species. While species number is a simply defined parameter, Simpson’s and Shannon’s indexes integrate both species number and the dominance aspect, whereas evenness exclusively refers to equitability of species within stands ([64]). Theoretical considerations ([90], [115]) and the presented results underline a gradual similarity of these diversity indexes ([82]). Therefore the number of tree species in combination with evenness may give a rather comprehensive picture of both qualitative and quantitative aspects of tree diversity ([64], [67]). However, if an indicator integrating both species richness and species abundance is aspired, Shannon’s or Simpson’s index has to be used. The latter gives higher importance to the more abundant species ([115]). Mean mingling is given less priority. In our dataset it was closely correlated with the other diversity measures. As it refers to small-scale mingling of species, it might describe spatially differentiated mixtures of tree species at respective scales more efficiently.
Basal area or the closely related volume of living trees per area, along with stem density, are doubtlessly important features of forest stands and should by all means be reported. For instance, the exceptionally high BA of almost 70 m2 ha-1 for a “fir and spruce woodland” plot in Slovakia, which is well above values found in conventionally managed forests ([106] - Fig. 15.5) and is an important information.
The selected indexes cover relevant aspects of forest structure. For example, Seidling & Fischer ([107]) found PCA scores derived from the ground vegetation at these monitoring plots to be related to tree diameter differentiation and basal area, while epiphytic lichen diversity was strongly correlated with the Clark-Evans index ([36]). However, the assessed indexes of forest structure may be limited in describing the complex requirements of different biota, which is still a major issue ([82], [48]). Canopy layering and canopy closure were found to be not related to factors dominated by stand structural parameters measured within this project (Tab. 6). They should therefore be seen as independent features and considered as potentially independent predictors for any biological response ([37]). Even other features might be needed for an adequate description of habitat structures of specific organisms, in addition to those parameters collected by this project. For instance, micro-structures like hollows in stems ([120]) are known to be important parts of forest habitats for various species or even whole guilds ([70], [14]).
Integrated evaluations
The joint evaluation of stand structure and dead wood was based on the assumption that both are closely linked at each site by common historic and recent forest management practices and natural processes, especially climatic events. Such mostly instantaneous interventions might be superimposed by soil conditions and unintentional anthropogenic factors like air pollution impacts. PCA is an efficient method to reduce the dimensionality of a multidimensional response structure composed in this case of different features of dead wood and the horizontal stand structure ([53]). Reductions of dimensionality are especially necessary for datasets with a comparatively low numbers of cases and large plenty of parameters, typical for networks composed of case studies like the Level II network of ICP Forests ([21]). McElhinny et al. ([72]) followed a similar approach in aiming at an additive index of stand structural complexity, based on an even broader variety of features of forest stands, like hollow-bearing trees, litter cover, or properties of the ground floor vegetation. As closely intercorrelated parameters strain the resulting component structure, only those parameters were used for the final PCA which revealed a certain self-reliance as expressed by correlation coefficient lower than 0.7. This is almost the same value applied by McElhinny et al. ([72]) for a similar purpose.
The stepwise approach - firstly applying domain-specific correlation analyses, followed by PCA with the most self-contained variables from both domains, and finally regression and main effects analyses between PCA scores and external predictor variables - assures a comprehensive consideration and evaluation of all available parameters. Against an intuitive or a purely hypothesis-guided pre-selection of considered parameters, which may have advantages of its own ([57]), this approach makes it highly probable that all important statistical relationships are considered, which is especially important for studies with a more exploratory character.
The dataset presented originates from a huge geographic area, covering macro-ecological gradients across Europe and a wide range of management practices between countries, which both inevitably influences the relationships between forest ecosystem components. Therefore, categorical parameters like country or forest type reveal relationships to more than one PCA axes. Especially the amount of dead wood and most of its fractions, which differentiated according to the PCA the plots best, seems largely be influenced by country-specific forest legislations and management practices, like the above mentioned removal of woody debris in the Mediterranean countries. Even the independently varying decay classes of dead wood (PCA axis 4) seem determined by country-specific management practices, and not by any of the available ecological factors. Country also explained almost 45% of the variation of PCA axis 2 and axis 3, both mainly loaded by stand related features (tree density and standard deviation of dbh). However, especially the two axes governed by stand related factors revealed its strongest relationships with the applied forest type classification. This finding is due to the fact that important stand structural features are intrinsic properties of even these forest types and may underline the existence of complex trade-offs between harvest and other management operations on one side and natural processes of stand development on the other side.
Only PCA axis 3 is related to some of the basic environmental parameters. It can be concluded that the original parameter “diameter differentiation of trees” loading this axis might be higher (and the likewise original parameter “volume of downed dead trees” lower) at plots with high numbers of tree layers, high stand age, high altitudes or low latitudes. According to Rouvinen & Kuuluvainen ([99]) unmanaged forests have generally higher diameter spreads than managed forests, even if a considerable between-stand variation does exist. However, diameter spread might not only be sensitive to forest management. According to theoretical considerations - as long as no major disturbance interferes with stand development - diameter differentiation should generally increase with time ([42], [113]), even if this has rarely been empirically corroborated (however, see [110], p. 95). This might be due to a lack of unmanaged forests, and due to often purely descriptive approaches ([63], [62], [106]). The positive relationship between the number of tree layers and PCA axis 3 may refer to the same complex of stand features, as forest stands more differentiated in tree height should also reveal a higher differentiation of tree diameters.
Latitude and altitude are proxies for the major climatic drivers, e.g., temperature and precipitation. Country does also not vary independently from climatic conditions due to their geographically fixed positions. Therefore, relationships with both variables partly include influences of climatic factors. Any statistical separation of respective climatic and management effects needs either much larger or more systematic datasets.
Conclusions
The present large-scale assessment of dead wood and stand structures with general features of different forest ecosystems and their management across Europe underlines the high importance of country-wise forest management regulations. Such legislation - commonly enacted on national level - seem to have a large impact especially on the formation of dead wood, and on forest stand structures as well, than naturally driven processes. As a result, dead wood and stand structure appear uncorrelated with each other on a large scale. This is consistent with the finding by Neumann & Starlinger ([82]) that ground vegetation in Austria is widely independent from stand structure, even if respective relationships can be observed at local scales between adjacent forest stands.
Due to the fact that plot selection was carried out by the countries following different national interests, the results provided cannot be assumed to be representative neither at a European nor at a national, regional or forest type related level. This can only be achieved by inventories based on probabilistic sampling schemes ([121], [98]) possibly combined with remote sensing techniques ([111], [18]). Thus, the results described above apply primarily for the evaluated set of plots. However, considering the ForestBIOTA dataset as “found” data ([87]), which can be taken as a random sample with respect to the questions asked, the detected relationships might be valid beyond this mere sample. This study may be also of particular importance as a reference to which different national methodologies can be linked for harmonization or bridge-building between different national forest inventories ([121], [98]).
Acknowledgments
We dedicate this work to Matthias Dobbertin (†: 31st October 2012) remembering him and the stimulating discussions on this and related subjects.
This work was supported by national experts, who contributed to method adaptation, were responsible for field work, and stimulated the evaluations. The teams were led by Václav Buriánek (Czech Republic), Lars Vesterdal, Annemarie Bastrup-Birk and Jacob Heilmann-Clausen (Denmark), Maija Salemaa and Tiina Tonteri (Finland), Ulrich Matthes, Joachim Block, Marcus Schmidt, Henning Meesenburg, Gerhard Raben, Claus Schimming, Angela Steinmayer, and Lutz Genssler (Germany), George Baloutsos and Evangelia Daskalakou (Greece), Bruno Petriccione (Italy), Gerard Grimberg and Han van Dobben (The Netherlands), Jozef Vladovic, Roman Longauer, and Jozef Istona (Slovak Republic), Gerardo Sanchez Peña, and Mayte Minaya (Spain), Norbert Kräuchi, Franziska Heinrich and Nadine Hilker (Switzerland), and Igor Buksha (Ukraine). Reviewers are acknowledged for their constructive comments. The project was co-financed by the European Commission under the Forest Focus regulation (EC no. 2152/ 2003).
References
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Online | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Online | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Supplementary Material
Authors’ Info
Authors’ Affiliation
Thünen Institute of Forest Ecosystems, Eberswalde (Germany)
Dipartimento di Gestione dei Sistemi Agrari, Alimentari e Forestali, University of Florence (Italy)
Northwest-German Forestry Research Station, Goettingen (Germany)
WSL, Swiss Federal Institute for Forest, Snow and Landscape Research, Birmensdorf (Switzerland)
Thünen Institute of International Forestry and Forest Economics, Hamburg (Germany)
DigSyLand, Husby (Germany)
Dipartimento di Bioscienze e Territorio, University of Molise, Pesche (Italy)
Consiglio per la ricerca e la sperimentazione in agricoltura, Forestry Research Centre, Arezzo (Italy)
Corresponding author
Paper Info
Citation
Seidling W, Travaglini D, Meyer P, Waldner P, Fischer R, Granke O, Chirici G, Corona P (2014). Dead wood and stand structure - relationships for forest plots across Europe. iForest 7: 269-281. - doi: 10.3832/ifor1057-007
Academic Editor
Enrico Marchi
Paper history
Received: Jun 24, 2013
Accepted: Feb 27, 2014
First online: Apr 14, 2014
Publication Date: Oct 01, 2014
Publication Time: 1.53 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2014
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 62532
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 49895
Abstract Page Views: 4679
PDF Downloads: 6226
Citation/Reference Downloads: 48
XML Downloads: 1684
Web Metrics
Days since publication: 4413
Overall contacts: 62532
Avg. contacts per week: 99.19
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2014): 22
Average cites per year: 1.83
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Background, main results and conclusions from a test phase for biodiversity assessments on intensive forest monitoring plots in Europe
vol. 2, pp. 67-74 (online: 18 March 2009)
Research Articles
Using self-organizing maps in the visualization and analysis of forest inventory
vol. 5, pp. 216-223 (online: 02 October 2012)
Review Papers
Integration of forest mapping and inventory to support forest management
vol. 3, pp. 59-64 (online: 17 May 2010)
Review Papers
Biodiversity assessment in forests - from genetic diversity to landscape diversity
vol. 2, pp. 1-3 (online: 21 January 2009)
Research Articles
An index of structural complexity for Apennine beech forests
vol. 8, pp. 314-323 (online: 03 September 2014)
Commentaries & Perspectives
Benefits of a strategic national forest inventory to science and society: the USDA Forest Service Forest Inventory and Analysis program
vol. 1, pp. 81-85 (online: 28 February 2008)
Research Articles
Do different indices of forest structural heterogeneity yield consistent results?
vol. 15, pp. 424-432 (online: 20 October 2022)
Research Articles
Availability and evaluation of European forest soil monitoring data in the study on the effects of air pollution on forests
vol. 4, pp. 205-211 (online: 03 November 2011)
Research Articles
Relationships between overstory and understory structure and diversity in semi-natural mixed floodplain forests at Bosco Fontana (Italy)
vol. 9, pp. 919-926 (online: 21 August 2016)
Technical Advances
A meta-database comparison from various European Research and Monitoring Networks dedicated to forest sites
vol. 6, pp. 1-9 (online: 14 January 2013)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword