Stand structure and regeneration of Cedrus libani (A. Rich) in Tannourine Cedar Forest Reserve (Lebanon) affected by cedar web-spinning sawfly (Cephalcia tannourinensis, Hymenoptera: Pamphiliidae)
iForest - Biogeosciences and Forestry, Volume 11, Issue 2, Pages 300-307 (2018)
doi: https://doi.org/10.3832/ifor2502-011
Published: Apr 13, 2018 - Copyright © 2018 SISEF
Research Articles
Abstract
The analysis of forest structure is a useful tool to understand stand biodiversity characterizing forest ecosystems, and could help in suggesting appropriate management plans. Cedar forests in Lebanon are remnant patches that survived past human activities but are still threatened by other different anthropogenic and natural disturbances. Among these threats, the cedar web-spinning sawfly (Cephalcia tannourinensis) discovered in Tannourine Cedar Forest Nature Reserve in 1997, which is able to cause the death of trees. The aim of this study is to investigate the impact of this pest on the stand structure and regeneration of Cedrus libani in Tannourine Cedar Forest Nature Reserve located in North Lebanon. The dependence of stand structural attributes (diameter at breast height, total height and basal area) on the presence of infestation by the cedar web-spinning sawfly was identified using the Student’s t-test. The Ripley’s K(d) function was used to analyse the spatial pattern of cedar stands. In addition, the diameter, the vertical structure and the crown projection were characterized using the Weibull function and graphic representations. The results showed that stand structure and regeneration are significantly different between infested and non-infested stands. The cedar of Lebanon remains as the dominant species, with abundant young individuals and a good regeneration status (c = 1.0). The analysis of the spatial pattern showed a positive spatial relationship between mature Lebanese cedar trees as well as between mature and juvenile cedars, with a bigger aggregation in infested plots (6 to 10 meters) than in non-infested quadrates (2 to 7 meters), reflecting the impact of the cedar web-spinning sawfly on the stand structure and regeneration of Cedrus libani stands.
Keywords
Cedrus libani, Stand Dynamic, Pest Damage, Spatial Pattern, Vertical Structure, Cephalcia tannourinensis
Introduction
Forest structure is the description of the forest composition and the spatial distribution of different species with various shapes, sizes, ages and abundance ([18]). A cause-effect relationship exists between forest structure and the surrounding environment. Forest structure and composition are affected by environmental gradients, climatic conditions (temperature, moister, heat, etc.) and topographic variables (elevation gradients, altitude, latitude, and slope - [23], [29]). In addition to that, they are influenced by disturbances (windstorms, wild fires, air pollution, etc.) and local environment features ([29], [16], [12]). Human activities, like forest products extraction, grazing, infrastructure development, or tree cutting, also alter and change the structure and the composition of forest ecosystems ([18], [14]). Furthermore, forest structure components represent an important factor influencing stand biodiversity, habitat suitability, niche diversification, distribution and mobility of living biota and wildlife, as well as other dynamic ecological processes and functions within a forest, such as germination and growth of other plants, fire behaviour, stand productivity and stand dynamics ([15], [11], [9]). Therefore, forest structure represents a major element in forestry and for est ecology. The purpose of forest structure analysis is to understand several ecological processes, identify the impacts of management activities and environmental factors on forests and to guide management needs in ecosystems ([13], [28], [20]).
The cedar of Lebanon (Cedrus libani A. Rich) is one of four species found in genus Cedrus ([3], [25]). The cedar of Lebanon is a drought resistant and light demanding species, but it is also capable of surviving in partial shade conditions at a young age. Moreover, it can grow old (over 1000 years) and reach impressive sizes and appearances ([7], [3]). The genus Cedrus is endemic or sub-endemic to the Mediterranean basin ([26]), and cedar forests are naturally distributed in Syria, Lebanon and Turkey ([25]). Unfortunately, the continued historical human activities such as grazing, cutting, burning, urbanizing etc. have caused the degradation of the Lebanese cedar forests throughout the world ([26]). Today, C. libani is listed in The World Conservation Union Red List among the low-risk and nearly threatened species ([6]). Therefore, it is important to conserve this species for its historical, cultural, aesthetic, scientific and economic reasons ([3], [26]). In Lebanon, cedar forests cover some relict forest patches that survived anthropogenic activities over thousands of years. They are mainly distributed on the western slope of the Mount Lebanon chains, in the mountainous Mediterranean bioclimatic zone, at altitudes ranging between 1400 and 2000 m a.s.l., with a southernmost limit in Al-Shouf Cedar Reserve ([7], [25]). The cedar of Lebanon is found in mixed stands with other tree species (Juniperus excelsa M. Bieb, Quercus calliprinos Webb, Q. infectoria Oliv.), but the majority of its remnant forests consist of pure cedar stands sheltering sporadic tree species such as Cilician fir (Abies cilica Antoine et Kotschy, Carrière 1855), wild apple (Malus sylvestris Miller.), maple (Acer hermoneum Bornm & Schwer), oak and juniper ([25]). Recently, the Lebanese cedar forests are threatened by a new insect, the cedar web-spinning sawfly (Cephalcia tannourinensis Chevin). It was first discovered in 1997 in the Tannourine Cedars Forest Nature Reserve (TCFNR) which is one of the largest cedar forests in Lebanon. The larvae of this insect feeds on the needles of cedar trees and causes their defoliation, which may lead to the death of trees in case of severe infestation ([21]). Based on the life cycle of this insect, the adults emerge and fly between mid-April and mid-June and lay their eggs on the new needle buds of the cedar trees. Then, the eggs hatch, the larvae pass through three instars during the months of June and July before hibernating in the soil. While developing, the larvae feed on the needles of the new cedar buds ([26], [22]). The feeding of the larvae on the new shoots during spring and early summer infers the severity of the damage that is done by the cedar web-spinning sawflyand its potential impact on the cedar growth.
The aim of this study consists of a stand-based assessment of structural attributes and the dynamics of the tree regeneration of Cedrus libani in TCFNR in relation to the cedar web-spinning sawfly infestation. This objective was fulfilled by: (i) quantifying the present composition and structure of the forests, based on diameter distribution and regeneration of trees population of cedar forests in Central Lebanon; (ii) evaluating the impact of Cephalcia tannourinensis on the regeneration and growth of cedar trees; and (iii) characterizing the spatial pattern of Cedrus libani forests in undisturbed and disturbed stands caused by Cephalcia tannourinensis. The results of this study will improve our understanding of the dynamics of C. libani forests in Lebanon based on a comprehensive understanding of the structures and regeneration processes in stands affected by Cephalcia tannourinensis.
Materials and methods
Study area
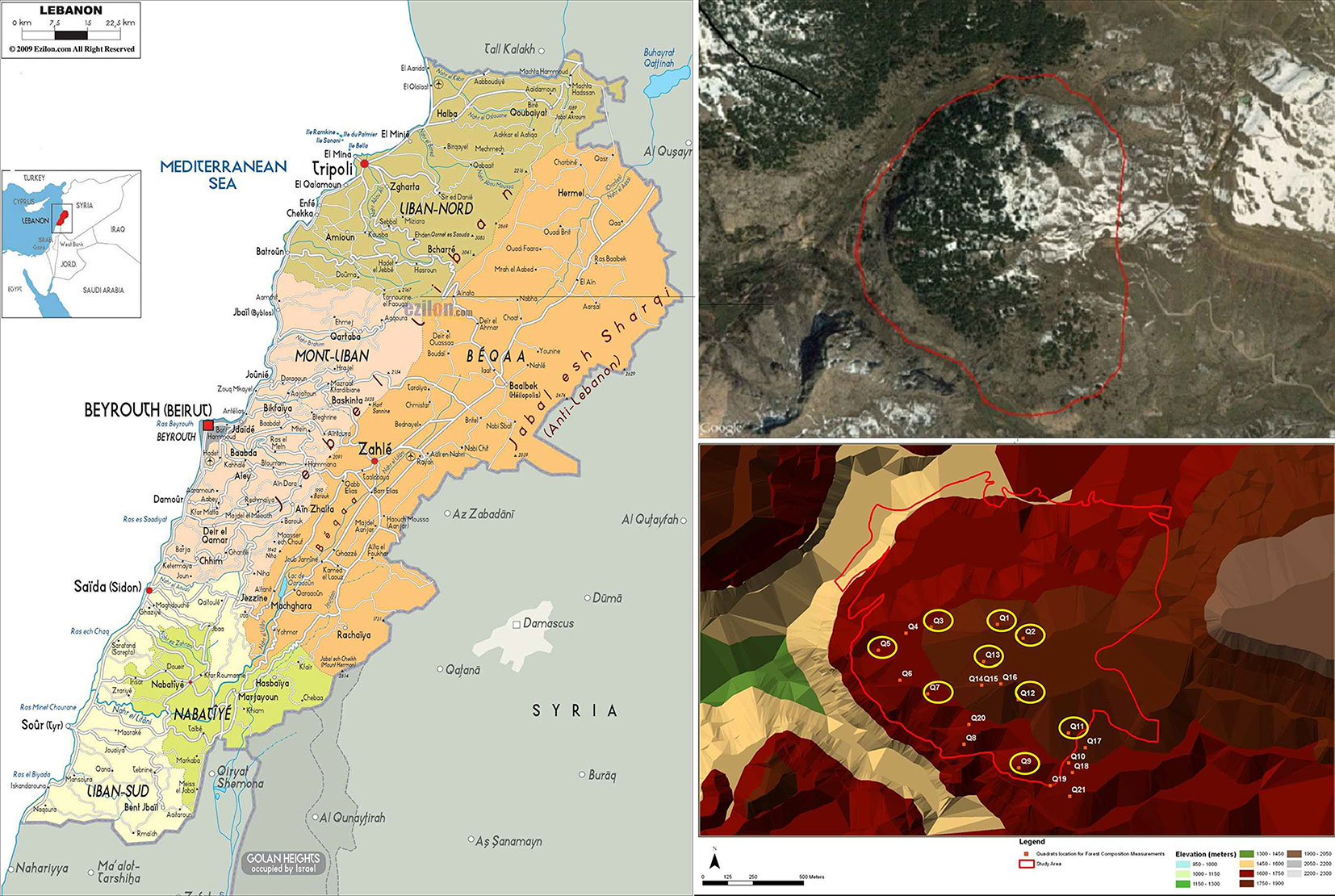
This study was carried out in the Tannourine Cedar Forest Nature Reserve (TCFNR), which was created by the Lebanese Ministry of Environment under the Law no. 9 on February 1999 ([26], [19], [6]). The reserve is located in the Caza of Batroun (North Lebanon) and it constitutes a part of Tannourine-Hadath El-Jebbeh protected area, the largest cedar forest in Lebanon ([8], [25]). Annual precipitation averaged between 1000 and 1200 mm, with scarce precipitations between June and August, resulting in late-summer drought. Snow cover usually lasts 15-30 days between November and March, and the average temperature registered in January is 3 °C, while in August the minimum average temperature registered is 18 °C. The TCFNR is located on calcareous, sandy and volcanic soils and spreads over an area of 1.5 km2 on the north-eastern and south-western slopes, at altitudes ranging between 1300 and 1900 m a.s.l., the latitude varying between 34° 12′ and 34° 15′ and the longitude between 35° 54′ and 35° 56′ ([26], [6]). It is considered as one of the remnant patches of the old cedar forest. The cedar of Lebanon is the main and dominant species, with 90% relative density in TCNFR ([19]). Besides the cedar trees, the forest contains sporadic trees, shrubs and herbs species, such as Juniperus excelsa, J. oxicedrus L., Cupressus sempervirens L, Quercus calliprinos, Q. infectoria Q. cerris L, Q. cedrorum Ky., Q. brantii Lindl., Berberis libanotica Ehrenb , Prunus ursina Ky., and Pirus syriaca Boiss ([26]). The reserve is one of the richest forests in biodiversity in the region: two hundred and twenty-nine plant species were inventoried therein, and twenty-five of these species are endemic and constitute more than 50% of the total endemic plant species identified in Lebanon ([8]).
Data collection
The web-spinning sawfly impacts on the forests in TCFNR are spatially heterogeneous. In 1999, the infestation of the Tannourine Hadath el Jebbeh cedar forest reached a very high level and it was spreading and threatening the surrounding cedar forests in Lebanon. Consequently, aerial treatments with the insect growth regulator diflubenzuron (Dimilin SC 48® mixed with the oil adjuvant, Citrole) were carried out between 1999 and 2004, and resulted in considerable suppression of the cedar web-spinning sawfly population ([22], [25]). Following the outbreak in 1999, the adult density of web-spinning sawfly was assessed on a yearly basis using yellow sticky traps. The procedure consisted of a weekly recording of the number of sawfly adults caught on ten 20 × 20 cm2 yellow sticky traps distributed in the forest between the end of April and the end of June. The population of the insect was estimated in each location by calculating the cumulative average of the yellow traps during the catching period. Based on this evaluation of the insect population, six 20 × 20 m2 square plots with Cephalcia infestation and three 20 × 20 m2 square plots with no infestation were established throughout the reserve. In addition to the level of infestation by Cephalcia, the nine plots were selected based on the following criteria: (i) at least ten adult trees were present; (ii) the forest patch in which the stand was located was dominated by cedar trees; and (iii) the site was accessible (Fig. 1). A full inventory of the nine plots was completed during the summer of 2011. All trees were projected on settled Cartesian axes and their spatial coordinates (x, y) were recorded. Along with the species identification, diameter at breast height (DBH, in cm) and height (in m, using a hypsometer) of all trees greater than 5 cm were measured. Tree competitive status of all trees was visually assessed as dominant, codominant, or suppressed based on the total height of their crowns and the percentage of crown projection. Regeneration of tree species was measured in three subplots of 4 m2 set equidistantly along the middle line parallel to y-axis and was categorized by height classes as: (i) seedlings (h < 50 cm); (ii) short saplings (50 ≤ h < 150 cm); and (iii) tall saplings (≥ 150 cm and DBH< 5 cm - [4]).
Fig. 1 - Location of the study area on Tannourine Cedar Forest Nature Reserve on the Lebanese map (TCFNR - left), a satellite image of the extent of TCFNR (upper right - Google Earth®, 2015), and the plot distribution within TCFNR (lower right).
Stand structure
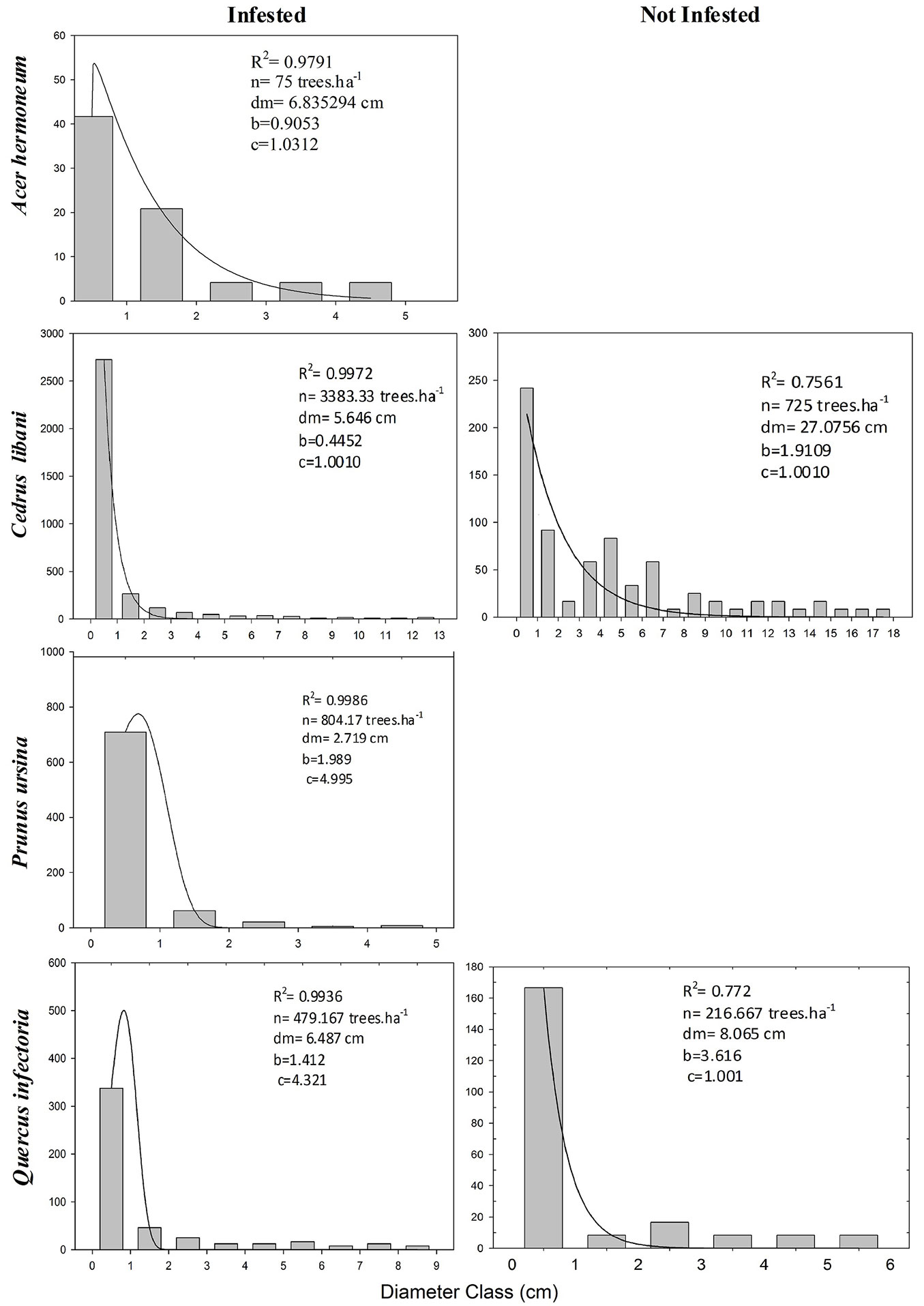
Prior to statistical analysis, the dasometric data were examined to check their univariate distribution and the validity of assumptions of the analysis of variance, using the Kolmogorov-Smirnov’s and Levene’s tests, respectively. When the data distribution did not fit a normal curve, the data were subjected to a square root transformation. The results in tables are presented as means with their standard deviation for the untransformed variables. Once it was identified that the basic requirements were met, the data were analysed statistically using the Student t-test to compare the means of DBH, basal area and height between the two levels of infestation for all identified species. Chi-square test was applied to check the dependence between the infestation and the regeneration of the different species encountered in the plots ([27]). Null hypotheses were rejected at the p < 0.05 level and all analyses were run using the package SPSS® ver. 15.02 (⇒ http://www.spss.com/). It should be noted that Pirus syriaca was not included in the analysis because it was found only in one infested quadrate. The frequency distribution among diameter classes was plotted for Cedrus libani, Prunus ursina and Quercus infectoria. The four-parameter Weibull function was applied to compare quantitatively the forest structure parameters between infested and non-infested plots ([20]). The dynamic curve fitting method of the statistical package SigmaPlot® (Systat Software, Inc. - ⇒ http://systatsoftware.com/products/sigmaplot/) was used to calculate the Weibull peak fitting, and the c parameter values were used for the interpretation as follows: (i) when 0<c<1, it shows an exponential distribution and indicates that there are abundant young individuals in the stand plot and the regeneration status of the species is very good; (ii) when 1<c<2.6, a positively skewed Weibull pdf distribution (with a right tail) is suggested. It reflects a right-skewed distribution of age and indicates a few young individuals compared to the individuals of average age, meaning that the regeneration status is medium; (iii) when 2.6<c<3.7, the coefficient of skewness is nearly zero and the Weibull pdf distribution may approximate the normal pdf (with no tail); (iv) when c>3.7, the distribution is negatively skewed (with left tail) and reflects a poor regeneration status.
Spatial pattern of association
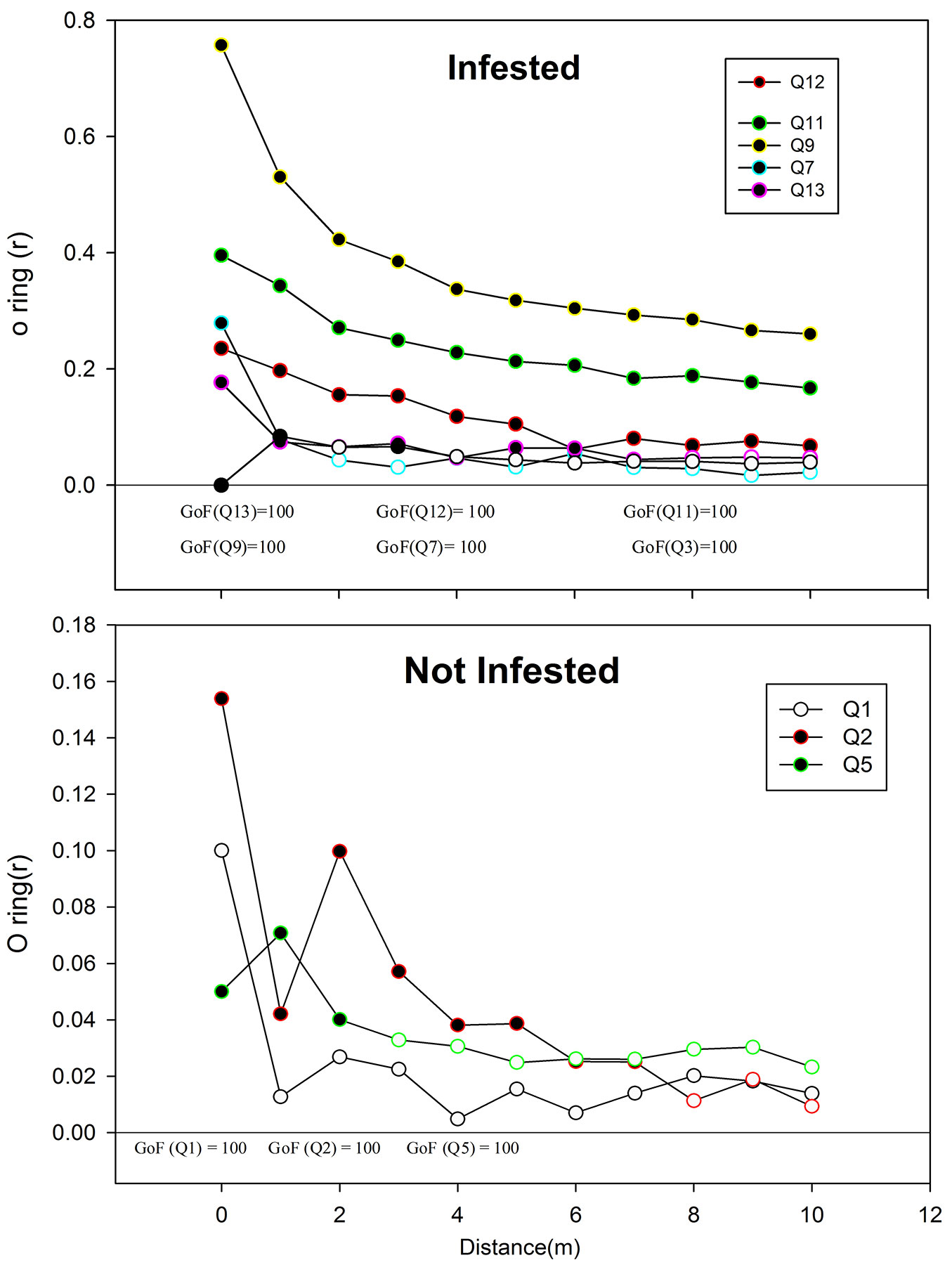
A crown projection map of all stands and the vertical structure of two selected transects were sketched for visual representation of the spatial distribution of the Lebanese cedars and the associated species. Ripley’s K-function and the O-ring statistic were applied to quantify the relationships along spatial patterns ([32]). The univariate form of the Ripley’s K function was used to describe the general patterns of points, whereas the bivariate form was used to describe the relationships between two different patterns of points, e.g., between regeneration and adult trees, and between Cedar trees and other species ([10]). Because Ripley’s K is a cumulative function and requires to be interpreted with care, L(d) the square root transformation of K(d) was used ([31]). The O-ring function, which is a transformation of the pair correlation function [g(r)], was also used. Edge effects were corrected in both analyses using the toroidal correction method ([10]). Analyses were performed for a distance of one to 10 meters at 0.5 m intervals (the lag distance). Tests were run separately for each of the two levels of infestation. Confidence boundaries (simulation envelopes) were calculated using a Poisson’s process model for the number of points in the sample ([31]). Models were randomly simulated 999 times using a Monte Carlo method, and the minimum and maximum values were set as the 99-percentile confidence boundaries. Values beyond these boundaries were considered significant and plotted as filled dots in the figures ([24]). Spatial patterns were evaluated using the software Programita ([2]) and goodness-of-fit (GOF) tests were performed to assess the potential underestimation of type I error.
Results
Stand structure
Cedrus libani was the dominant species in all the plots. The other species found in the reserve area were Acer hermoneum, Juniperus excelsa, Juniperus oxcycedrus, Pirus syriaca, Prunus ursina and Quercus spp., mixed with shrub species such as Loniceranum mularifolia Jaub. & Spach, Berberis libanotica, Cotoneaster nummularia Fisch & Mey, and Rosa canina Boiss. The total density of trees with DBH > 5 cm was 975 stems ha-1 in the infested stands, distributed among Cedrus libani (67.5%), Quercus infectoria (14.5%), Prunus ursina (9.8%), Acer hermoneum (3.4%), Juniperus oxycedrus (2.1%), J. excelsa (1.7%), and Pirus syriaca (0.8%). Furthermore, the density was lower (600 stems ha-1) in the non-infested stands with the presence of Cedrus libani (80.5%), Quercus infectoria (8.3%), Prunus ursina (6.9%), and Juniperus excelsa (4.2%). Lebanon cedar total density (including trees with DBH < 5 cm) was significantly higher (p<0.001) in the infested than in the non-infested stands (3383 vs. 725 stems ha-1, respectively). Mean diameters of Lebanon cedar ranged from 28.78 cm in non-infested stands plots to about 5.65 cm in infested stands (p<0.001 - Tab. 1). Tree basal area and total height were also significantly different (p<0.001) among infested (0.47 m2 ha-1 and 2.54 m, respectively) and non-infested stands (5.52 m2 ha-1 and 9.60 m, respectively). Acer hermoneum, Prunus ursina, Juniperus sp., and Quercus sp. showed no significant difference in the studied dasometric variables (Tab. 1). The Chi-square test showed a significant dependence of the regeneration of Lebanon cedar on the level of infestation (p<0.001), but the dependence was not significant for other species (Tab. 2). Both infested and non-infested stands showed a reversed-J curve with decreasing density of larger diameter classes. The infested stands held more trees in the three smallest diameter classes, while the non-infested stands had more trees in larger diameter classes, and trees with DBH>70 cm that were missing in the infested stands (Fig. 2). The c parameter of the cedar curve had a value of c=1.00 for both levels of infestation which indicates a very good regeneration status of Cedrus libani in both situations. Quercus infectoria, Prunus ursina and Acer hermoneum showed a diameter structure similar to Lebanon cedar, though showing less diameter classes and smaller mean diameter (< 25 cm for Prunus ursina and Acer hermoneum, and < 45 cm for Quercus infectoria). Acer hermoneum in infested stands and Quercus infectoria in non-infested stands had a good regeneration with c values close to 1.0 (c = 1.03 and c = 1.00 respectively). Both Quercus infectoria and Prunus ursina in infested stands showed poor regeneration status with c = 4.32 and c = 4.99, respectively. It is worth to mention that Prunus ursina and Acer hermoneum did not have enough individual trees in the non-infested stands to be analysed.
Tab. 1 - Tree density, DBH, basal area and height of the main tree species found in the studied plots in Tannourine Cedar Forest Nature Reserve. Values are means ± standard deviation. (Lower row): Student t-test statistics and its p-value (in parentheses); (*): significantly different values; (‡): density of trees with DBH>5 cm.
| Species | DBH (cm) |
Basal area (m2 ha -1 ) |
Total Height (m) |
Density (‡ ) (stems ha -1 ) |
||||
|---|---|---|---|---|---|---|---|---|
| Infested | Not-infested | Infested | Not-infested | Infested | Not-infested | Infested | Not-infested | |
| Acer hermoneum (df = 17) |
6.84 ± 7.83 | 2.6 ± 1.13 | 0.2 ± 0.44 | 0.01 ± 0.01 | 2.1 ± 1.52 | 1.64 ± 1.29 | 33 | 0 |
| -0.75 (0.466) | -0.6 (0.556) | -0.41 (0.687) | ||||||
| Cedrus libani (df=868) |
5.65 ± 14.48 | 28.78 ± 44.79 | 0.47 ± 4.68 | 5.52 ± 21.92 | 2.54 ± 3.72 | 9.6 ± 7.77 | 658 | 483 |
| 10.17 (<0.001)* | 5.38 (<0.001)* | 14.07 (<0.001)* | ||||||
| Prunus ursina (df = 243) |
2.72 ± 3.35 | 2.6 ± 2.08 | 0.04 ± 0.11 | 0.02 ± 0.04 | 1.18 ± 1.02 | 0.95 ± 0.61 | 96 | 42 |
| -0.18 (0.856) | -0.95 (0.342) | -1.59 (0.114) | ||||||
| Quercus sp. (df = 146) |
6.2 ± 9.94 | 8.07 ± 14.88 | 0.27 ± 0.67 | 0.55 ± 1.62 | 2.43 ± 3.16 | 2.87 ± 4.04 | 142 | 50 |
| 0.79(0.430) | 1.42 (0.158) | 0.6 (0.549) | ||||||
| Juniperus sp. (df=20) |
11.12 ± 14.92 | 24.63 ± 27.78 | 0.66 ± 1.72 | 2.33 ± 3.76 | 2.96 ± 2.90 | 5.29 ± 4.45 | 38 | 25 |
| 1.4 (0.177) | 1.41 (0.175) | 1.32 (0.200) | ||||||
Tab. 2 - Chi-square test for independence between tree species regeneration and Cephalcia tannourinensis infestation level for the main tree species in the Tannourine Cedar Forest Nature Reserve.
| Species | Pearson’s Chi-square | Prob. |
|---|---|---|
| Acer hermoneum | 2.012 | 0.156 |
| Cedrus libani | 35.055 | 0.000 |
| Juniperus sp. | 0.512 | 0.474 |
| Prunus ursine | 3.270 | 0.071 |
Fig. 2 - Diameter class histograms and corresponding fitted of four-parameter Weibull distributions (solid line) for Cedrus libani in two Cephalcia tannourinensis infestation level. The x-axis is uniform and covers 5 cm diameter intervals, while y-axis represents the frequency of individuals (stems ha-1). R2, sample size (n), mean diameter (dm) and Weibull shape statistics (b, c) are reported in each diagram.
Spatial pattern
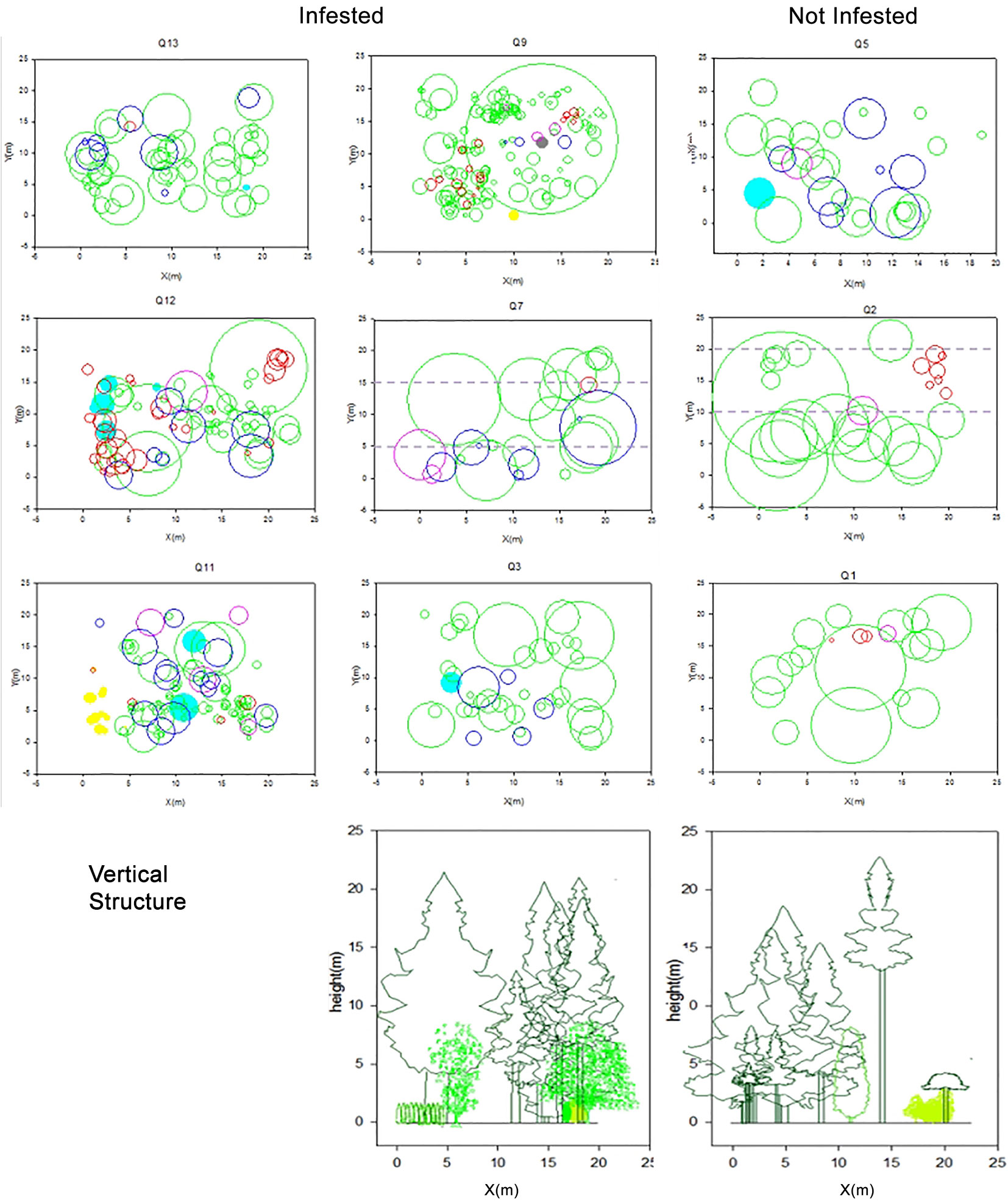
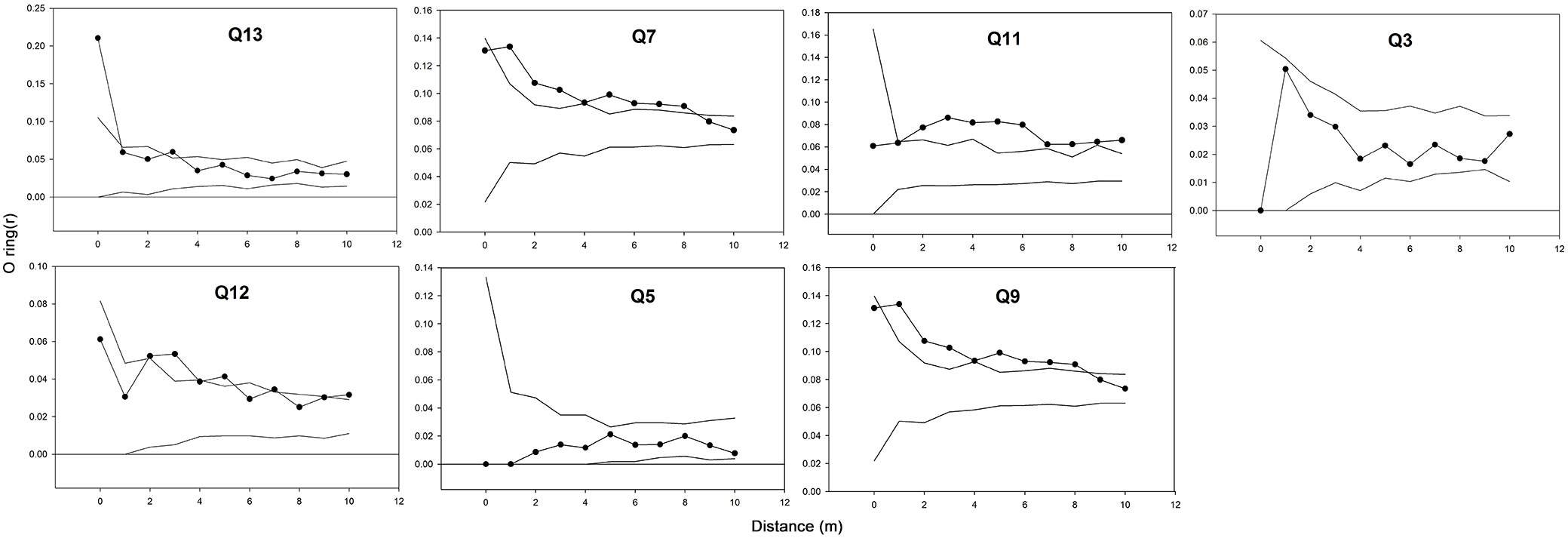
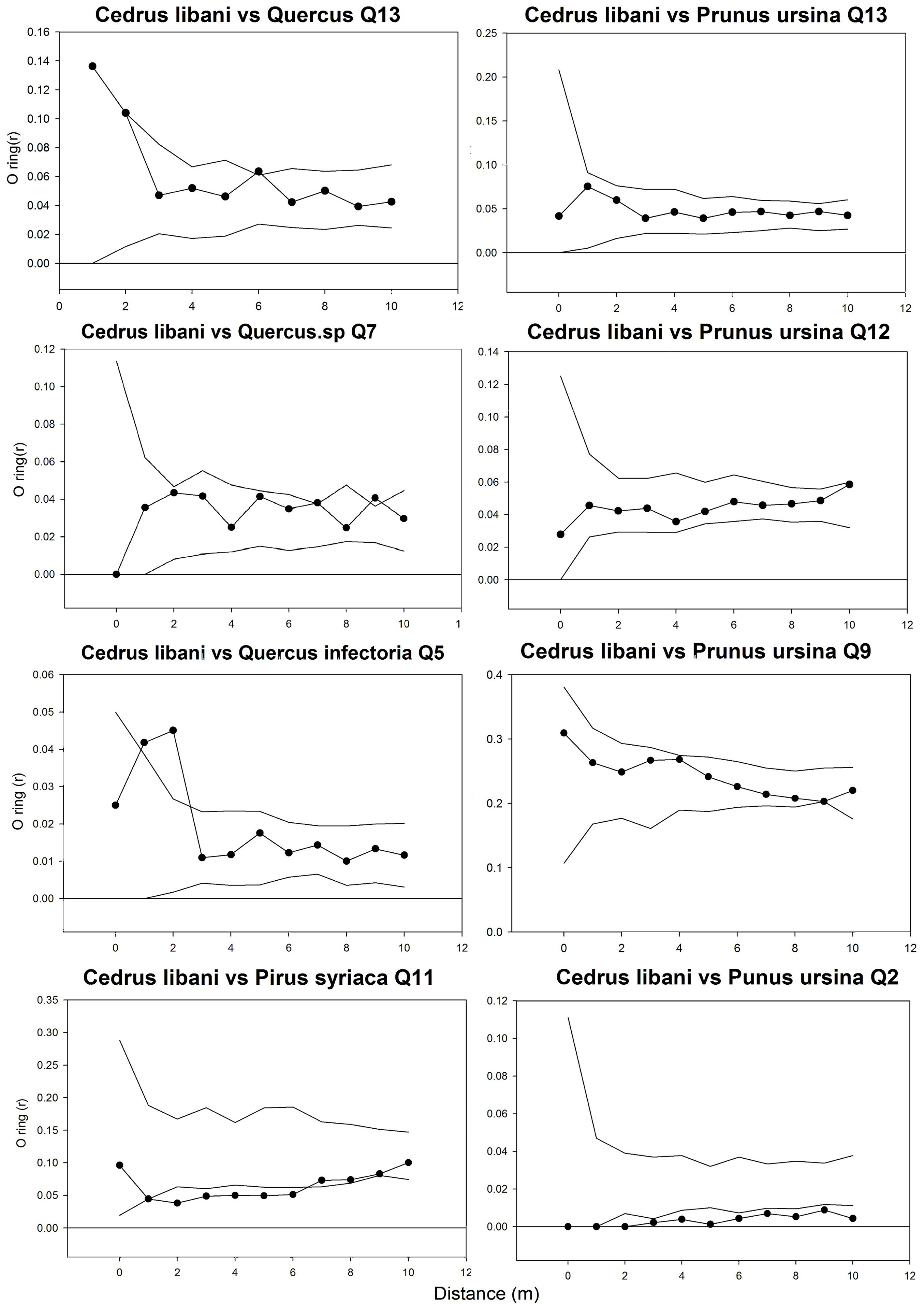
The O-ring (r) and L(r) statistics of the cedar of Lebanon were always above zero in all plots (Fig. 3), which reflects a general tendency of positive spatial dependence among Cedrus libani trees. In both infested and non-infested stands, values decreased with larger distance and approached a value of zero. However, the infested stands reached larger values in comparison with the non-infested ones. Moreover, the trees were clustering up a distance of 6 to 10 meters in infested stands, as compared to a distance of 2 to 7 meters in non-infested ones (Fig. 3). The tendency to aggregation is also visually observed in the vertical and crown projection map in Fig. 4. A similar trend can be noticed in Fig. 5, where a positive spatial relationship between mature and juvenile individuals of Lebanon cedar is shown. The spatial correlation peaks at a distance < 2 m and then decreases and becomes irregular for larger distances. The (r) statistic varies between a minimum of 0.02 and a maximum of 0.2 over the studied plots. The spatial relationship between the cedar of Lebanon and other species (Quercus sp., Prunus ursina, and Pirus syriaca) did not show a consistent trend with the distance (Fig. 6). The L(r) for the cedar of Lebanon and the oak species was always above zero, and the O-ring (r) peaks at a distance of 2 m in most of the plots. These observations were explained by the presence of a significant positive spatial correlation between Cedrus libani and Quercus infectoria for distances lower than two meters. L(r) values were negative at all the distances between the cedar of Lebanon and Pirus syriaca with (r) peaking at a distance as small as zero (Fig. 6), showing a significant negative spatial association between the cedar of Lebanon and Pirus syriaca. L(r) value was highly variable between the cedar of Lebanon and Prunus ursine (Fig. 6). Thus, the spatial association between Cedrus libani and Prunus ursina showed three different tendencies: a non-significant positive relationship, a significant negative relationship and a none significant relationship between these two species. Finally, the analysis of the crown projection maps and the vertical structure (Fig. 4) showed that the TCFNR presents dense cedar stands where the overstory is mainly constituted by big old cedar trees with wide canopies. whereas the understory is composed by younger cedar individuals and other species.
Fig. 3 - Spatial pattern of Cedrus libani in the two Cephalcia tannourinensis infestation level using the univariate form of the Ripley’s K(d) function. The x-axis shows the distance in meters between trees and the y-axis shows the (r) and L(d) values.
Fig. 4 - Crown projection map of studied plots. Cedrus libani trees are coloured in green, Acer hermoneum in cyan, Pirus syriaca in yellow, Prunus ursina in red, Juniperus spp. in pink and Quercus spp. in blue. The two vertical transects show the vertical structure in the two levels of infestation.
Fig. 5 - Spatial patterns and relationships between mature and juvenile trees of Cedrus libani using the bivariate form of the Ripley’s K(d) function. The x-axis represents the distance in meters and y-axis represents the (r) and L(d) values.
Fig. 6 - Bivariate spatial analysis between Cedrus libani and each of Quercus sp., Prunus ursina, and Pirus syriaca in different plots. The x-axis shows the distance in meters between individual trees and the y-axis shows L(d) and (r).
Discussion
The cedar web-spinning sawfly is a specific pest of cedar species. The larvae cannot feed on needles of other coniferous species and they die when placed on the needles of Juniperus excelsa, Abies silica, Pinus pinea and Pinus halepensis. Also, the fe male sawfly is not capable of laying eggs on tree species other than the cedar of Lebanon ([21]). This specificity of the web-spinning sawfly to cedars determines the amount of growth damage this pest can cause to the cedar of Lebanon and explains the significant dependence of its dasometric variables (DBH, basal area and total height) on the level of infestation. Contrastingly, there was no significant differences in dasometric parameters for the other species, especially the Juniperus sp. The severe outbreak of Cephalcia tannourinensis in 1997 in TCNFR caused the death of a high number of adult trees ([22], [25]). This seems to be clearly related to the higher number of young individuals and the lower DBH value of infested stands (DBH = 5.65 cm) compared to larger trees (DBH = 27 cm) in non-infested stands. The cedar of Lebanon is a light demanding species, but it is also characterized by its capability of surviving in partial shade during its early growth stages ([5]), which explains the good regeneration status and the high number of young individuals in all stands. Furthermore, the loss of large trees in the infested plots after 1999 has created large canopy openings with high levels of light in the understory. These new conditions produced by the cedar web-spinning sawfly are similar to those created by shelter-wood harvesting which are more favourable for the regeneration, survival and growth of young cedar trees ([3]). Therefore, the dependence of cedar regeneration on the level of infestation was expected. The abovementioned arguments also explain the increase of Lebanon cedar’s relative density from 50% to 68% when trees with a DBH smaller than 5 cm are included. It also explains the higher relative density (70%) in the trees that are larger than 5 cm DBH in non-infested stands versus the 50% in the infested ones. The reversed J-shape distribution curve is typical of old-growth and uneven-aged stands where the number of trees decreases gradually with larger DBH as the results of competition for space and resources, especially for light ([26]). The smaller DBH classes of the other species growing in the cedar stands are caused by the dominance of Cedrus libani in the Tannourine Cedar Forest Nature Reserve ([26]), and its capacity to grow old and reach very large dimensions ([3]). The clustering of mature and juvenile cedar trees at a maximum distance of 2 meters seems to be related to the production of seeds from mature trees (> 30 years) and their localized dissemination under the dense canopy ([7]). The 2-meters distance is very reasonable based on the width of canopy projection that was observed in this study. The positive spatial dependence among the cedar of Lebanon has been related to the general competition that exists in any forest type. However, the higher and more widespread aggregation in infested stands versus non-infested ones can be explained by the better regeneration in infested stands coupled with the discovery of a positive spatial relationship between juvenile and mature cedars. The spatial relationship of the cedar of Lebanon with other species cannot be analysed since the results were not consistent as in the case of Prunus ursina, or because the results were limited to one plot as in the case of Pirus syriaca. In addition to that, the only consistent positive correlation found between Cedrus libani and Quercus infectoria might be due to the fact that Quercus infectoria is mostly found in mixture with other species like Quercus calliprinos and cedar ([7]).
Comparison of the structures and dynamics of C. libani under different levels of pest damage may produce significant changes in the structure and dynamics of population sizes and in the dynamic of the regeneration processes. Attention should be dedicated to stand structure of different forest types in relation with various climatic and site conditions, on both small and large scales, and the impact of recent and historical anthropogenic interventions, as well as the impact of climate change on different forest species, and more specifically, on endemic and threatened ones.
Silviculture is a useful strategy to decrease the range of the pest as well as preventing further spread by altering stand structures, tree species composition and improving tree vigor across the landscape ([30]). These silvicultural treatments should be based on new silvicultural strategies that integrate maintenance and restoration of stand structures with individualized stand prescriptions within the context of broad-scale pest management systems.
Conclusion
The results of this study show a clear relationship between structures and dynamics of C. libani with the infestation level of Cephalcia tannourinensis, adding complexity to forest ecosystem management. However, forest stand structure is also known to be affected by local environmental and sites conditions, climate variables and human activities and disturbances. The lack of seedlings and small saplings reflects the perturbation of C. libani regeneration in relation to a gradient of Cephalcia tannourinensis infestation levels. The prohibition of tree cutting, controlled fire use and sustainable grazing in those forests in nature reserves and protected sites (Lebanese Ministry of Environment 2004), as the Tannourine Cedar Forest Nature Reserve, can exacerbate these problems conducting to very dense forests. Silviculture is an important part of an integrated pest management program and a decrease in silvicultural and anthropic pressure could lead to a change in the further development of the forest. The mismanagement of the forests, in addition to outdated and weak forest policies and laws, represents a serious issue in the field of forestry in Lebanon. Based on the result of this study, long-term forest management plans can contribute to reduce pest outbreak risk. Furthermore, there is an urgent need of updating Lebanese forest laws which allow cleanings, controlled grazing, and thinning as tools for forest management and biodiversity conservation.
Considerable additional research is required into silvicultural management modelling, such as dendrochronology studies ([17]). In particular, those related to pest and climate change impacts on cedar forest in Lebanon ([1]) may be useful in providing data on the relations between pest outbreaks, stand structure, species diversity, site history (fire control and logging), and climatic events.
Acknowledgments
Many thanks to the Centre for Applied Research in Agroforestry Development (IDAF) and ERSAF research group at the University of Cordoba (Spain), for their assistance and guidance in the elaboration of the methodology used on this research, in data analysis and processing, and in providing the needed materials to achieve field data collection. A debt of gratitude is also owed to Dr. Nabil Nemer and all the staff at Tannourine Cedar Forest Nature Reserve for allowing the access and helping in the fieldwork. This study would not have been possible without their diligent efforts and expertise.
References
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Online | Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Salim Kattar
Lebanese University, Faculty of Agricultural and Veterinary Sciences, Dekwaneh, Beirut (Lebanon)
Department of Forestry, Faculty of Agricultural and Forestry, Dendrodat Lab, University of Córdoba (Spain)
Guillermo Palacios Rodríguez
Center for Applied Research in Agroforestry Development (IDAF), University of Córdoba (Spain)
Department of Agricultural Sciences, Faculty of Agricultural and Food Sciences, Holy Spirit University of Kaslik, Jounieh (Lebanon)
Tannourine Cedar Forest Nature Reserve, Scientific Coordinator, Tannourine El Fawka (Lebanon)
Corresponding author
Paper Info
Citation
Bassil S, Kattar S, Navarro-Cerrillo RM, Navarrete Poyatos MA, Nemer N, Palacios Rodríguez G (2018). Stand structure and regeneration of Cedrus libani (A. Rich) in Tannourine Cedar Forest Reserve (Lebanon) affected by cedar web-spinning sawfly (Cephalcia tannourinensis, Hymenoptera: Pamphiliidae). iForest 11: 300-307. - doi: 10.3832/ifor2502-011
Academic Editor
Susanna Nocentini
Paper history
Received: May 25, 2017
Accepted: Jan 12, 2018
First online: Apr 13, 2018
Publication Date: Apr 30, 2018
Publication Time: 3.03 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2018
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 52746
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 45169
Abstract Page Views: 3542
PDF Downloads: 3027
Citation/Reference Downloads: 16
XML Downloads: 992
Web Metrics
Days since publication: 2775
Overall contacts: 52746
Avg. contacts per week: 133.05
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2018): 6
Average cites per year: 0.75
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Estimation of above-ground biomass and sequestered carbon of Taurus Cedar (Cedrus libani L.) in Antalya, Turkey
vol. 6, pp. 278-284 (online: 01 July 2013)
Research Articles
Case study of a new method for the classification and analysis of Dryocosmus kuriphilus Yasumatsu damage to young chestnut sprouts
vol. 5, pp. 50-59 (online: 10 April 2012)
Research Articles
Spatial structure of the vertical layers in a subtropical secondary forest 57 years after clear-cutting
vol. 12, pp. 442-450 (online: 16 September 2019)
Research Articles
Monitoring of damage from cedar shoot moth Dichelia cedricola Diakonoff (Lep.: Tortricidae) by multi-temporal Landsat imagery
vol. 7, pp. 126-131 (online: 13 January 2014)
Research Articles
Discovering interaction between oaks and carabid beetles on a local scale by point pattern analysis
vol. 9, pp. 618-625 (online: 06 May 2016)
Research Articles
Long-term effects of thinning and mixing on stand spatial structure: a case study of Chinese fir plantations
vol. 14, pp. 113-121 (online: 08 March 2021)
Research Articles
Large-scale spatial distribution of deer browsing damage to young tree plantations
vol. 14, pp. 34-40 (online: 14 January 2021)
Research Articles
Consistency among forest structure and biodiversity potential index (IBP): an assessment of stand structural complexity for floodplain poplar woodlands
vol. 18, pp. 335-343 (online: 04 November 2025)
Research Articles
Long-term dynamics of stand structure and regeneration in high-stocked selection fir-beech forest stand: Croatian Dinarides case study
vol. 14, pp. 383-392 (online: 24 August 2021)
Research Articles
Rapid assessment of wind storm-caused forest damage using satellite images and stand-wise forest inventory data
vol. 6, pp. 150-155 (online: 08 April 2013)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword