Sap flow, leaf-level gas exchange and spectral responses to drought in Pinus sylvestris, Pinus pinea and Pinus halepensis
iForest - Biogeosciences and Forestry, Volume 10, Issue 1, Pages 204-214 (2016)
doi: https://doi.org/10.3832/ifor1748-009
Published: Nov 01, 2016 - Copyright © 2016 SISEF
Research Articles
Abstract
In a climate change scenario, Mediterranean forest species such as pines may be endangered by rising temperatures and reduced precipitation, thus calling for studies on the transpiration and water balance in pines. In this paper, the response of young plants of Pinus sylvestris L., Pinus pinea L. and Pinus halepensis Mill. to different irrigation treatments has been studied. Significant differences were found in water potential, sap flow, leaf-level gas exchange and spectral variables. P. sylvestris had higher pre-dawn and midday water potentials, sap flow rates and leaf-level gas exchange rates compared to the other two species in well-watered conditions. Vapor pressure gradient correlated with stomatal conductance, net assimilation and transpiration, but the association between stomatal conductance and sap flow was weak. The environmental variables more strongly associated with sap flow were solar radiation and reference evapo-transpiration, especially in the well-watered plants, but those associations were weaker in the stressed plants. All three pine species showed the isohydric, drought-avoiding strategy common in the genus Pinus, maintaining relatively high water potentials in dry conditions. Nevertheless, P. halepensis showed a water-saving strategy, with a stomatal closure behavior under drought. Stomatal regulation was less strict in P. sylvestris, closer to a water-spending pattern, while P. pinea showed an intermediate behavior. Significant differences were recorded among species in spectral reflectance in the visible and infra-red regions. Photochemical Reflectance Index, Normalized Difference Vegetation Index and combinations of other ratios permitted the discrimination among the three pine species. These spectral variables showed association with sap flow rate, water potential and leaf-level gas exchange variables. Both cluster analysis and k-means classification discriminated Scots pine and Aleppo pine in two different groups. On the other hand, Stone pine showed differences in spectral behavior depending on the hydric status of the plants. Well-watered Stone pine plants had the same spectral behavior as Scots pine, while the plants subjected to drought stress were closer to Aleppo pine plants in spectral response. These findings may help to quantify the impacts of early and mid-summer water deficit on Mediterranean pines in future climate regimes.
Keywords
Carbon Assimilation, Aleppo Pine, Hydric Relations, Reflectance, Scots Pine, Stone Pine, Transpiration
Introduction
In a global warming scenario, many Mediterranean forest species may be endangered by a temperature rise and a decrease in precipitation. This trend has already been described and assessed for some pine forests in North-Eastern Spain ([19]), where severe drought-induced mortality has occurred during recent dry episodes. Similar climate change scenarios have been described in other parts of the world, for instance in the eastern Mediterranean basin ([37]) and in the Southern United States ([33]). This situation underlines the need for studies on the water balance in plants from endangered ecosystems, in particular, the regulation of the hydric status and transpiration of the dominant woody vegetation. In other Mediterranean and semi-arid ecosystems, hydric and gas exchange parameters at the leaf level have successfully been used as indicators of the ecophysiological competence of forest species in a changing environment ([16]).
Plant resistance to drought has been hypothesized to reflect two contrasting physiological strategies: drought avoidance and drought tolerance. The mechanisms of either approach, respectively called isohydric and anisohydric, involve control of stomata and water status ([36], [21]). Isohydric plants reduce stomatal conductance in dry conditions, maintaining relatively constant leaf water potential regardless of drought intensity, for instance in Prunus sp. ([36]). In contrast, anisohydric species allow leaf water potential to decrease with water availability; there are good examples among the Cupressaceae ([20]). Both strategies enable plant survival under drought conditions. However, each has inherent drawbacks that can be fatal in certain conditions. Prolonged reductions of stomatal gas exchange expose isohydric species to carbon depletion, while extreme water potential gradients in anisohydric species result in xylem cavitation and embolism and subsequent hydraulic failure ([21]).
Scots pine (Pinus sylvestris L.) is one of the most widely distributed forest species in the world, extending from the Siberian taiga to mountainous ecosystems in South-western Europe, where it reaches the boundary with semi-arid Mediterranean climate and may be more at risk of aridity in a climate change scenario ([19]). Stone pine (Pinus pinea L.) is a Mediterranean pine species, present in many south European countries, adapted to semi-arid continental and coastal locations, from the sea-level to about 1000 m altitude. Stone pine prefers sandy acidic soils with good water drainage, and a local annual precipitation between 400 and 800 mm. Aleppo pine (Pinus halepensis Mill.) is another characteristic species of the Mediterranean basin, with preference for basic soils, which shows more resistance to drought, being capable of living with as little as 250 mm annual precipitation. In spite of the environmental importance of these pine species, there have been few comparative studies aimed at relating the different ecophysiological responses in whole-plant transpiration and in their adaptation to drought ([23]). Moreover, transpiration of whole-tree water use may be estimated by means of sap flow measurements. One of the most common methods for measuring sap flow is the Thermal Dissipation Probe (TDP) technique, which measures sap flux density in the stem xylem and can then be converted to volumetric flow rate ([10]). Several studies have related the whole plant transpiration with gas exchange variables at the plant level and with micro-meteorological factors ([26]). Furthermore, the spectral features of radiation reflected or transmitted by plants have been used to retrieve their biophysical properties ([38]), and the spectral behavior of the forest canopy has been successfully linked to physiological variables, for instance detecting forest decline before damage is visible ([12]).
The spectrum in the visible region (VIS: 400-700 nm) has been associated to high absorption of the photosynthetically active radiation (PAR) by plant pigments, mainly the chlorophylls, while reflectance in the near infrared wavelengths (NIR: 700-1350 nm) is determined by the structure of the crown. Water content is the main factor affecting the reflectance in the shortwave infrared wavelengths (SWIR: 1350-2500 nm - [14]). Spectral methods are based on the association of ecophysiological traits with spectral signatures from which the plant status is inferred, for instance through the inversion of radiative transfer models ([14]), or using spectral indices ([40]). In this study, we followed the second approach. Several remotely sensed vegetation indices have been used as ecophysiological indicators ([17]). These indices are associated with plant water stress ([35]) or radiation use efficiency ([8]). However, few studies have tried to relate sap flow rates and leaf-level gas exchange variables with spectral indices. Those studies have been restricted to the agricultural sector ([18]) and only very few are focused on the forestry and environmental sector.
A better understanding of the ecophysiology of young plants is important for successful nursery management and for the establishment of new plantations. Therefore, our objectives were: (1) to measure daily sap flow in plants from three pine species present in different ecosystems of the Mediterranean area, namely Scots pine (Pinus sylvestris), stone pine (P. pinea) and Aleppo pine (P. halepensis), and to examine their relationship with environmental conditions such as water availability and evaporative demand; (2) to describe and compare gas exchange responses to different water availabilities, defining and comparing the ecophysiological strategies to face drought adopted by the three pine species; (3) to measure hyperspectral reflectance and compare the spectral behavior of all three species in relation to water availability and to ecophysiological variables such as sap flow rate, water potential and leaf-level gas exchange variables; (4) to predict ecophysiological variables by means of the plant spectral behavior; and (5) to identify possible differences between ecophysiological attributes to explore management implications in the context of a climate-change scenario of increasing aridity.
Materials and methods
Plant materials
In all experiments, three pine species were used, i.e., Pinus halepensis, P. pinea and P. sylvestris. All the plants were ca. 10 years old, collected from a nursery. Eight plants per species were used, four of which were subjected to a high irrigation regime and four to a low irrigation regime. Thus, a total of 12 plants received a high irrigation dose and 12 a low irrigation dose. Besides, plants also received natural rainfall during the measurement period, although the amount was negligible (25.4 mm from May 10 to September 11). All plants were transferred to 15 L plastic pots (7 dm3 area) with a mixture (1:1) of peat and soil and placed in a nursery. Plants were allowed to establish good root contact with the substrate for 15 days before the experiment started. The experiment started on May 10, and ended on September 11. For each plant, the diameter was measured at the base of the stem and used to calculate the mean cross sectional area at the base of the stem. The total leaf area (Al, m2) of each plant was estimated using the diameter as independent variable in the allometric equations developed by Montero et al. ([25]) for P. sylvestris and P. pinea, and by Mitsopoulos & Dimitrakopoulos ([24]) for P. halepensis. Plant size at the beginning of the experiment is given in Tab. S1 (Supplementary material).
Experimental design and measurements
Two irrigation doses were applied throughout the experiment duration. The high dose consisted of 7 mm day-1 through an automatic dripping system. One irrigation event was given per day, at 6 a.m. solar time. The low dose was 3.5 mm day-1, in the same conditions.
Sap flow was determined using the thermal dissipation probe (TDP) technique ([10]). Briefly, this system consists of two thermocouple probes which are inserted into the plant sapwood, one directly above the other, at a distance of 4 cm each other. The upper probe contains an electrical heater. The thermocouples measure the temperature difference (dT) between the heated probe and the sapwood temperature below. Then, dT will decrease as the flowing sap cools the heated needle. The dT variable and the daily maximum (dTm) at zero flow are used to estimate sap flux density and therefore whole-tree transpiration. In our experiment, a TDP30 Dynamax® (Houston, TX, USA) thermal dissipation probe was inserted in the stem of each plant, at about 10 cm height from the base. We checked that the sensing zone of the probe was in full contact with the xylem of the plant. Then the area between the upper and lower probe was isolated with styrofoam eggs and shielded with aluminium reflective bubble foil cover to minimize natural temperature gradients and provide thermal insulation and mechanical protection, according to the vendor instructions. The TDP probes were connected to a Dynamax Probe 12 biomonitor. The upper probe of each sensor was supplied permanently with a constant power of 0.15 to 0.2 W, while the lower probe was unheated for reference. Sap flow data were recorded and estimated according to Granier ([10]), from May 10th to September 11th 2012. From the hourly recorded dataset, two sap flow-based variables were calculated: mean sap flow of the growing season (F, g h-1 - eqn. 1) and mean sap flow velocity (v, cm s-1 - eqn. 2).
where F is the sap flow (g h-1) and As is the cross-sectional area of sap conducting wood (cm2). Given the age of the plants, we considered that the sapwood area equaled the whole cross section. The sap flow velocity v (cm s-1) was estimated according to the empirical formula by Granier (1985 - eqn. 2):
where dT is the measured difference in temperature between that of the heated probe, referenced to the lower non-heated probe, and dTm is the daily maximum dT at zero flow.
Water potential was measured in all plants with a PMS pressure bomb at dawn and at midday. Three needles were measured per plant and the average water potential was calculated per plant. Measurements were taken on June 26th, August 17th and September 24th 2012, trying to represent the hydric status in the first month, mid-season and the end of the dry period, respectively.
Leaf-level gas exchange variables, i.e., net assimilation (An), transpiration (E), stomatal conductance (gs), internal CO2 concentration (Ci), water use efficiency (WUE), intrinsic water use efficiency (IWUE), leaf-to-air vapor pressure gradient (VPG) and photosynthetic photon flux density (PPFD) were recorded with a Delta T Infrared Gas Analyzer (IRGA) and a conifer chamber, in the middle (August 20th, 2012) and the end (October 3rd) of the dry season, in outdoors environmental conditions. During measurements (12:00 to 16:00 h), the average PPFD ranged from 150 to 201 µmol m-2 s-1. Average leaf chamber CO2 concentration and temperature ranged from 393 to 400 ppm and from 30 to 35.6 °C, respectively. After gas exchange measurements, the projected needle area was estimated with an image analyzer, and the gas exchange variables were referred to the measured needle area.
Spectral signatures were recorded from all 24 plants with an ASD FieldSpec 3® (Analytical Spectral Devices Inc., Boulder, CO, USA) field spectroradiometer. Measurements were always made in clear sky conditions, on June 25th and July 2nd, 2012, at midday, operating in the spectral range between 350 and 2500 nm with an average spectral resolution of 3 nm FWHM (Full-Width-Half-Maximum) and a sampling interval of 1.4 nm across the 350-1050 nm spectral range. The FWHM and the sampling interval for the 1051-2500 nm spectral range are 30 and 2 nm, respectively. A fibre optic cable, providing a field of view edge of 25°, was connected to the spectroradiometer to collect reflectance spectra in nadir direction. Three signatures were recorded per plant from their tops, and the average was used to estimate the spectral response. Before and between every series of three plant reflectance measurements, three white references were taken for calibration, by measuring the reflectance of a 1×1 m polystyrene panel, covered with a mixture of barium sulphate powder and white paint, as white standard. The ViewSpec Pro® (ASD) software was used for pre-processing reflectance spectra and then reflectance indices were derived. The reflectance responses of all 24 pines used in this experiment were tested for the species and the irrigation treatment. Reflectance in the range from 350 to 2500 nm wavelength was measured, and spectral indices were calculated to track changes in pigment concentration as a function of the water stress condition: green (531 nm), yellow (570 nm), red (670 nm), different Near Infra-Red (NIR) reflectances: NIR1 (760 nm), NIR2 (895 nm), NIR3 (900 nm), NIR4 (970 nm), NIR5 (1090 nm), NIR6 (1190 nm), two Short Wave Infra-Red: SWIR1 (1650 nm), SWIR2 (2217 nm); Normalized Difference Vegetation Index: (NDVI - [32]), Physiological (or Photochemical) Reflectance Index (PRI - [8]), Normalized Difference Infrared Index (NDII - [11]); different indices based on reflectance ratios: SWIR1/SWIR2, simple ratio (SR) = NIR5/Red ([31]), SWIR1/Red, SWIR2/Red (see Tab. S2 in Supplementary material), and the Global Environmental Monitoring Index (GEMI - [28]), the latter being calculated according to the formula (eqn. 3):
where η is (eqn. 4):
Micrometeorological variables were recorded with a Davis Vantage Pro2™ (Davis Instruments, CA, USA) meteorological station every five minutes, during all the experiment duration. Temperature (°C), relative air humidity (rH, %), vapour pressure deficit (VPD), solar radiation (W m-2), wind speed (m s-1), barometric pressure (hPa), rain rate (mm h-1), soil moisture (kPa) at 5 and 15 cm depth and soil temperature (°C) at 5 and 15 cm depth were averaged per hour. Rainfall (mm) and reference evapo-transpiration (ETo, mm) were accumulated per hour. Reference evapo-transpiration was estimated according to the modified Penman’s equation as implemented by the California Irrigation Management Information System, CIMIS, including Net Radiation calculation ([34]).
Statistics
Tree species, leaf-level variables, irrigation treatments and dates of measurement were compared through multifactor Analysis of Variance (ANOVA). Water potential, sap flow, leaf-level gas exchange variables and spectral indices were also tested through correlation analysis. Significant variables were estimated with regression models. The statistical packages Statgraphics Centurion XVI® v. 16.1.17 (StatPoint Technologies, Warrenton, VA, USA) and STATISTICA v. 7.1 (StatSoft Inc., Tulsa, OK, USA) were used. Several reflectances and spectral indices were compared through ANOVA: green (531 nm), yellow (570 nm), red (670 nm), different Near Infra-Red (NIR) wavelengths: NIR1 (760 nm), NIR5 (1090 nm), NIR6 (1190 nm), two Short Wave Infra-Red: SWIR1 (1650 nm), SWIR2 (2217 nm); Normalized Difference Vegetation Index NDVI5, Photochemical Reflectance Index (PRI), Normalized Difference Infrared Index (NDII), Global Environmental Monitoring Index (GEMI); and different ratios: SWIR1/SWIR2, NIR5/Red, SWIR1/Red, SWIR2/Red.
Results
The leaf area (Al) per plant was compared before the beginning of the experiment between the two sets of plants which were going to be subjected to the different irrigation treatments by ANOVA, to test possible differences in size which could bias the results. However, no statistically significant differences were found among plants subjected to the irrigation treatments (p = 0.999), among the three species (p = 0.460) or the interaction between species and treatment (p = 0.467). Therefore, we assumed that differences in the results of the experiments carried out cannot be attributed to differences in leaf area among plants (see Tab. S1 in Supplementary material). On the other hand, stone pine plants had a significantly larger (p ≤ 0.0001) cross sectional area (As) than the other two species, due to the intrinsic anatomical traits of stone pine. As a consequence, significant differences in the leaf area to cross-sectional sapwood area ratio (Al:As) were found (p = 0.026). The Al:As ratio of stone pine was significantly lower than in Scots pine but it was not significantly different from that of Aleppo pine (Tab. S1 in Supplementary material).
Observed climatic conditions over the study period (temperatures, precipitation, and air relative humidity) were compared with average conditions from the 1982-2010 period (Tab. S3 in Supplementary material), indicating that May, June and August were warmer and dryer in the study period than the average, while July and September were close to the average.
Water potential
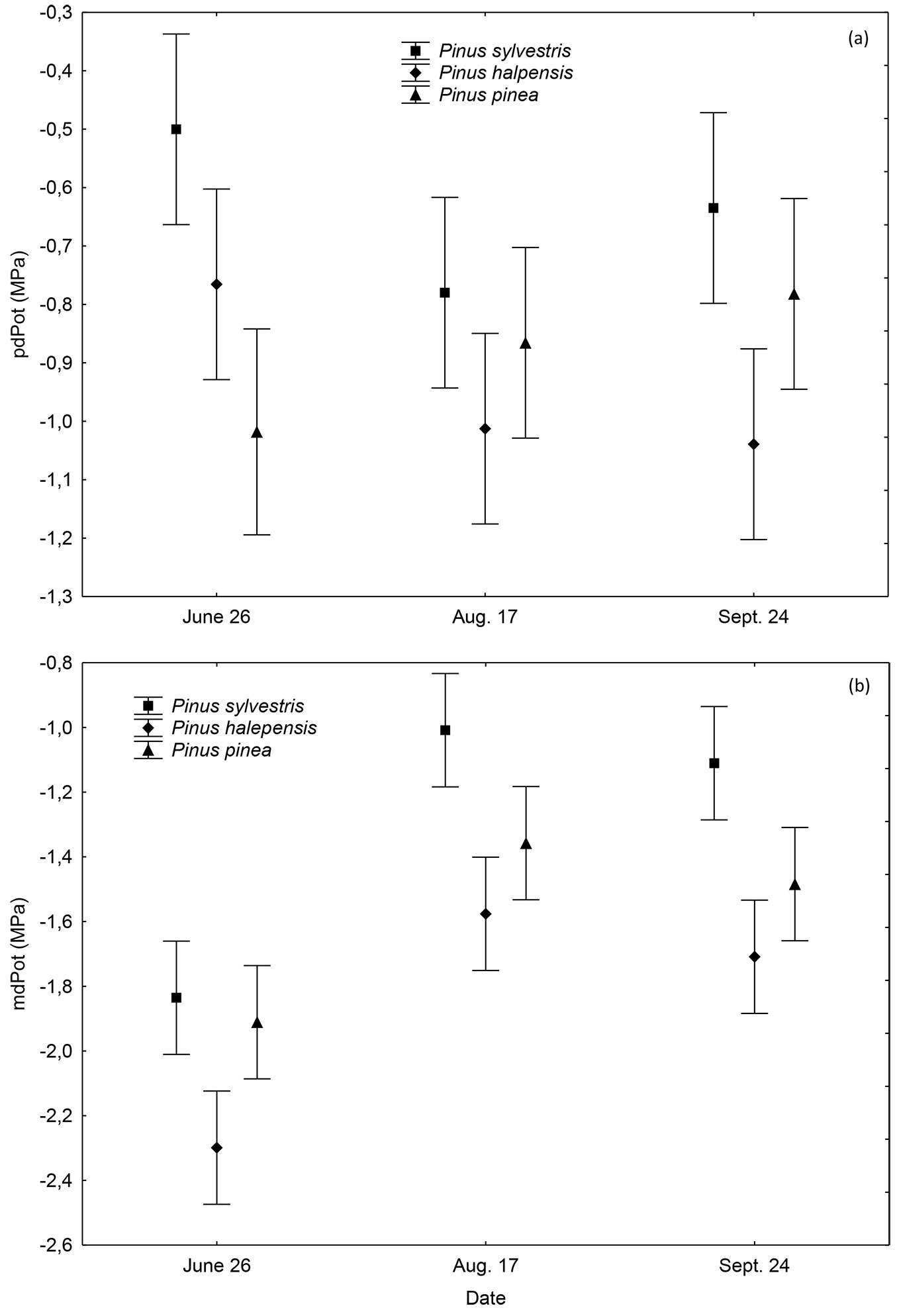
Significant differences in pre-dawn water potential were found for the interaction between species and date of measurement (p = 0.020). Pinus sylvestris showed the highest pre-dawn water potential as compared to P. halepensis, while Pinus pinea was lowest in June, and intermediate in August and September. In this period, the pre-dawn water potential was relatively stable (Fig. 1a).
Fig. 1 - (a) Mean pre-dawn leaf water potential (pdPot, MPa) and (b) Midday water potential (mdPot, MPa) in Pinus sylvestris, P. halepensis and P. pinea. Plants were subjected to two irrigation regimes, high irrigation (7 mm day-1) and low irrigation (3.5 mm day-1) from June to September. Vertical bars denote 95% confidence intervals.
On the other hand, midday water potential had statistically significant differences between species (p ≤ 0.001) and between dates of measurement (p ≤ 0.001 - Fig. 1b), but not between the irrigation treatments nor for the interactions. Pinus sylvestris and P. pinea plants had the highest midday water potential and P. halepensis the lowest, without differences between treatments. Midday water potential was lowest on June 26th, especially for Pinus halepensis (-2.3 MPa) and recovered as the season advanced, in parallel with the decreasing temperature and ETo and the increasing rH (Fig. S1 in Supplementary material). Pinus pinea had an initial water potential (-1.91 MPa) closer to Pinus sylvestris (-1.84 MPa), but did not recover to the same high levels in August and September (Fig. 1b).
Sap flow measurements
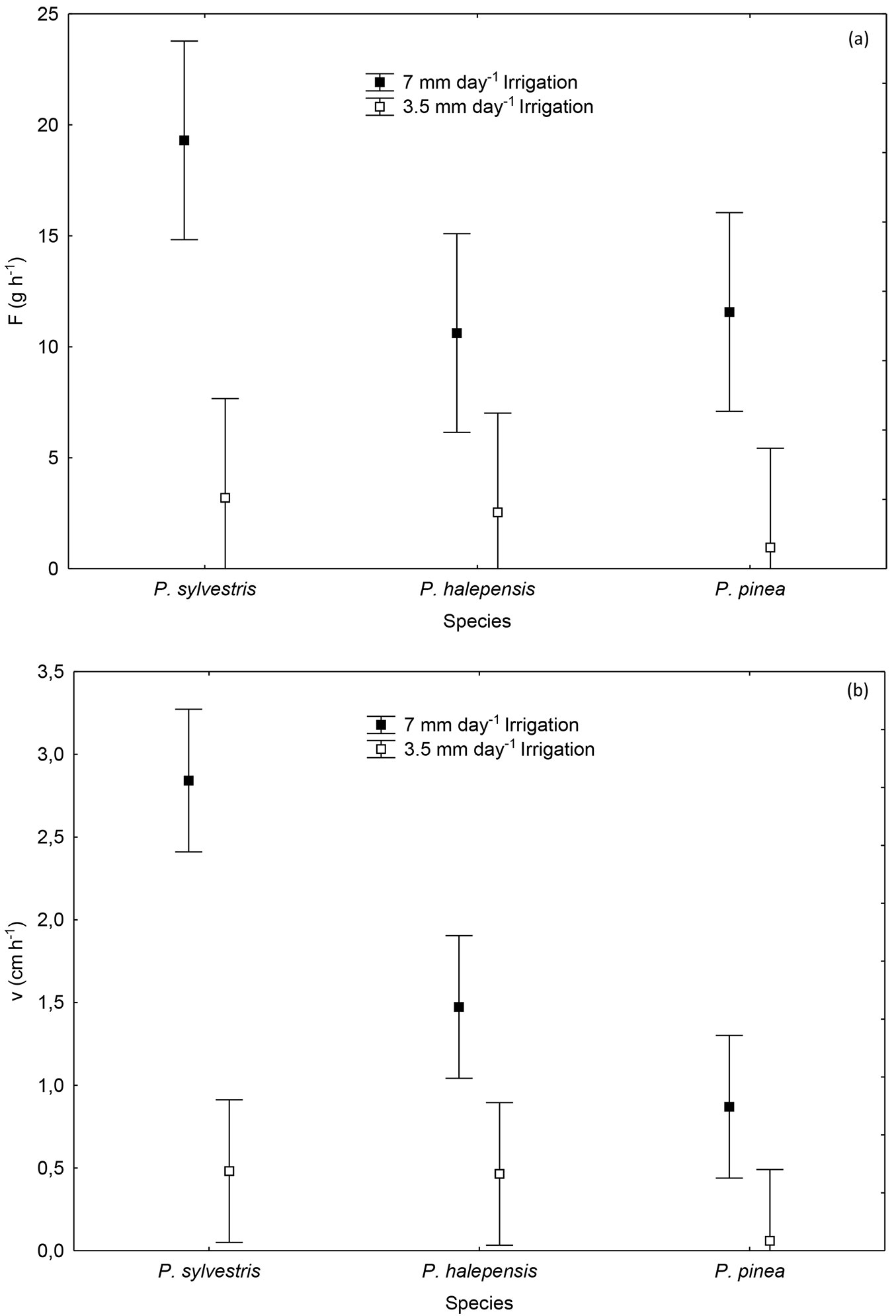
Mean sap flow rate of the whole season was only significantly different among irrigation treatments (p ≤ 0.001 - Fig. 2a) but not among species or for the interaction. On the other hand, mean sap-flow velocity showed significant differences for the irrigation treatment (p ≤ 0.001), species (p ≤ 0.001) and the interaction (p = 0.003 - Fig. 2b). As expected, sap flow rate and mean sap-flow velocity were higher in the more watered plants. Pinus sylvestris had higher mean sap flow rate than the other two species with more irrigation, but the rate was reduced to approximately the same level as the other two pine species when less water was available (Fig. 2a). On the other hand, correlation analysis did not show significant relationships between water potential and sap flow either.
Fig. 2 - (a) Mean sap flow rate (F, g h-1) and (b) mean sap-flow velocity (v, cm h-1) in Pinus sylvestris, P. halepensis and P. pinea plants subjected to two irrigation regimes: high irrigation (7 mm day-1) and low irrigation (3.5 mm day-1). Vertical bars represent 95% confidence intervals.
Leaf-level gas exchange variables
ANOVA of leaf-level gas exchange variables revealed significant differences in net assimilation rate (An) among species (p = 0.044) and dates of measurement (p ≤ 0.001) but not for the interactions. During the adverse period of August, net assimilation was lower for all species, but as temperatures descended, P. sylvestris showed a better capacity of recovery by a higher assimilation rate than the other pines.
Transpiration rate (E, mmol m-2 s-1) was significantly lower in August than in October (p ≤ 0.001) and for the interaction between species and irrigation treatment (p = 0.035 - Fig. S2a in Supplementary material). As for net assimilation, transpiration showed a depression in August, while in October Pinus sylvestris recovered with a higher E rate than the other species at the low irrigation treatment (Fig. S2a in Supplementary material). Stomatal conductance also showed significant differences among the dates of measurement (p ≤ 0.001) and for the interaction between species and irrigation treatment (p = 0.009 - Fig. S2b in Supplementary material). Stomatal conductance followed the same pattern of An and E, as it was higher in October than in August. On the other hand, neither water use efficiency (WUE) nor intrinsic water use efficiency (IWUE) were statistically significant for any of the factors tested.
Correlations among water potential, sap flow and gas exchange variables
Tab. 1 - Correlation coefficients between leaf-level gas exchange variables (An: net assimilation; E: transpiration rate; gs: stomatal conductance; Ci: internal CO2 concentration; VPG: vapor pressure gradient; IWUE: intrinsic water use efficiency), midday water potential (mdPot) and daily sap flow rate (Sap Flow) during August and September. Values of the second row for each variable are the significance values (prob, in italic). (* ): p ≤ 0.05; (**): p ≤ 0.01; (***): p ≤ 0.001.
| Parameter | E | gs | Ci | mdPot | VPG | IWUE | Sap Flow |
|---|---|---|---|---|---|---|---|
| An | 0.798 *** | 0.848 *** | 0.072 | 0.122 | -0.633 *** | 0.268 | 0.266 |
| prob | 0.000 | 0.000 | 0.640 | 0.432 | 0.000 | 0.079 | 0.081 |
| E | - | 0.897 *** | 0.138 | 0.113 | -0.574 *** | -0.089 | 0.209 |
| prob | - | 0.000 | 0.373 | 0.463 | 0.000 | 0.566 | 0.174 |
| gs | - | - | 0.328 * | 0.039 | -0.765 *** | -0.158 | 0.306 * |
| prob | - | - | 0.030 | 0.802 | 0.000 | 0.305 | 0.043 |
| Ci | - | - | - | -0.100 | -0.410 ** | -0.515 *** | 0.211 |
| prob | - | - | - | 0.517 | 0.006 | 0.000 | 0.169 |
| mdPot | - | - | - | - | 0.062 | -0.037 | 0.447 ** |
| prob | - | - | - | - | 0.691 | 0.814 | 0.002 |
Correlation analysis among leaf-level gas exchange variables, water potentials and sap flow rates revealed a general good correlations between pairs of gas exchange variables, between water potentials, or between sap flow rates, but not between variables of different sets. Only An, E and gs showed good correlations among them and with VPG, and a weak correlation was found between midday water potential and sap flow (Tab. 1). Remarkably, E did not show correlation with sap flow measurements. The best regression model for predicting gs using VPG as predictor variable was the square root-Y reciprocal-X model (p ≤ 0.000, r = 0.84, R2 = 71.03% - eqn. 5):
Sap flow correlations with meteorological variables
Tab. 2 - Linear correlation coefficients between meteorological variables and mean sap flow per species (Pinus sylvestris, P. halepensis, P. pinea) and irrigation treatment (high irrigation: 7 mm day-1; low irrigation: 3.5 mm day-1). (T): air temperature; (rH): relative humidity; (VPD): vapour pressure deficit; (Rad): solar radiation; (ETo): reference evapo-transpiration; (sylv-HighIrr): sap flow of Pinus sylvestris with high irrigation; (halep-HighIrr): sap flow of P. halepensis with high irrigation; (pinea-HighIrr): sap flow of P. pinea with high irrigation; (Sylv-LowIrr): sap flow of Pinus sylvestris with low irrigation; (halep-LowIrr): sap flow of P. halepensis with low irrigation; (pinea-LowIrr): sap flow of P. pinea with low irrigation. Values of the second row for each variable are the significance values (prob, in italic). (* ): p ≤ 0.05; (**): p ≤ 0.01; (***): p ≤ 0.001.
| Parameter | rH | VPD | Rad | ETo | sylv- HighIrr |
halep- HighIrr |
pinea- HighIrr |
sylv- LowIrr |
halep- LowIrr |
pinea- LowIrr |
|---|---|---|---|---|---|---|---|---|---|---|
| T | -0.8197 * | 0.9527 * | 0.5529 * | 0.6842 * | 0.4874 | 0.3889 | 0.4085 | 0.1712 | 0.2635 | -0.0739 |
| prob | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0001 |
| VPD | - | - | 0.5446 * | 0.7119 * | 0.4493 | 0.3315 | 0.3553 | 0.0868 | 0.1999 | -0.1176 |
| prob | - | - | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 |
| Rad | - | - | - | 0.9015 * | 0.8286 * | 0.7250 * | 0.8068 * | 0.5383 * | 0.6831 * | 0.1305 |
| prob | - | - | - | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 |
| ETo | - | - | - | - | 0.7248 * | 0.5937 * | 0.6136 * | 0.3627 | 0.5232 * | -0.0311 |
| prob | - | - | - | - | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0965 |
Tab. 3 - Mean sap flow rate (F, g h-1) regression models, with solar radiation (W m-2) as the independent variable, in Pinus sylvestris, Pinus halepensis and Pinus pinea subjected to high irrigation (7 mm day-1) or low irrigation (3.5 mm day-1). All of them fitted to a double square root model, except for P. halepensis with low irrigation, which followed a square root-Y model. Model parameters: b, slope; a, y-intercept.
| Species | Irrigation | Regression Model | r | adjusted R2 | p-value |
|---|---|---|---|---|---|
| P. sylvestris | 7 mm day-1 | eqn. 6; a = 0.55; b = 0.239 | 0.89 | 78.5% | 0.000 |
| P. halepensis | 7 mm day-1 | eqn. 6; a = 0.43; b = 0.165 | 0.79 | 62.32% | 0.000 |
| P. pinea | 7 mm day-1 | eqn. 6; a = 0.43; b = 0.182 | 0.86 | 74.09% | 0.000 |
| P. sylvestris | 3.5 mm day-1 | eqn. 6; a = 0.44; b = 0.07 | 0.63 | 39.46% | 0.000 |
| P. halepensis | 3.5 mm day-1 | eqn. 7; a = 0.38; b = 0.003 | 0.70 | 49.14% | 0.000 |
| P. pinea | 3.5 mm day-1 | eqn. 6; a = 0.54; b = 0.01 | 0.15 | 2.31% | 0.000 |
Mean sap flow of each species and irrigation treatment were tested for correlations with meteorological data recorded during the experiment duration. Solar radiation had the strongest correlation with mean sap flow, with differences among species (Tab. 2). The high irrigation treatment had better correlation than the low irrigation treatment. Also, ETo was moderately correlated with mean sap flow of the well-watered pines, but the correlation coefficients decreased for the less irrigated plants. Correlations of VPD with sap flow were weak (Tab. 2). As expected, some meteorological variables were strongly correlated: mean temperature (T) and relative humidity (rH), T and ETo, VPD and ETo. However, no good correlations were observed between them or between precipitation and sap flow, except for the mentioned correlation of ETo with sap flow (Tab. 2). Significant regression models predicted sap flow responses with solar radiation as the independent variable for all species and treatments (Tab. 3). In all cases, the best fit was a double square root model (eqn. 6), except for Pinus halepensis subjected to the low irrigation treatment, which followed a quadratic model (eqn. 7):
where a and b are the model parameters (Tab. 3).
Seasonal effect on leaf-level gas exchange and sap flow variables
Again, An, E and gs were highly correlated (r = 0.89 to 0.99) while the correlation with the internal CO2 concentration Ci was negative at the end of the growing season (Tab. 4). Sap flow rate was weakly associated with An. A positive correlation of Ci in October with VPG of the same day (r = 0.61) and a negative one with the sap flow rate of September (r = -0.63) was recorded (Tab. 4). A similar pattern has been observed for the other gas exchange variables: An, E and especially gs show no correlation with VPG in August, but are negatively correlated with VPG in October (Tab. 4).
Tab. 4 - Correlation coefficients between leaf-level gas exchange variables (An: net assimilation; E: transpiration rate; gs: stomatal conductance; Ci: internal CO2 concentration; VPG: vapour pressure gradient), and daily sap flow rate (Sap Flow), during September (Sep) and October (Oct). Values of the second row for each variable are the significance values (prob, in italic). (* ): p ≤ 0.05; (**): p ≤ 0.01; (***): p ≤ 0.001.
| Parameters | E (Oct) |
gs (Oct) |
Ci (Oct) |
VPG (Oct) |
Sap Flow (Sep) |
|---|---|---|---|---|---|
| An (Oct) | 0.891 *** | 0.898 *** | -0.769 *** | -0.509 * | 0.486 * |
| prob | 0.000 | 0.000 | 0.000 | 0.015 | 0.022 |
| E (Oct) | - | 0.995 *** | -0.561 ** | -0.563 ** | 0.303 |
| prob | - | 0.000 | 0.007 | 0.006 | 0.170 |
| gs (Oct) | - | - | -0.616 ** | -0.611 ** | 0.360 |
| prob | - | - | 0.002 | 0.003 | 0.100 |
| Ci (Oct) | - | - | - | 0.614 ** | -0.633 ** |
| prob | - | - | - | 0.002 | 0.002 |
Analysis of spectral indices
The spectral reflectance responses of all 24 pines used in this experiment were tested for the species and the irrigation treatment (Fig. S3 in Supplementary material). The spectrum of the well-watered pines (high irrigation, 7 mm day-1) was characterized by a low reflectance (< 0.12) in the visible region (VIS: 400-700 nm), while in the less irrigated pines (3.5 mm day-1) this reflectance was slightly higher. Reflectance in the near infrared wavelengths (NIR: 700-1350 nm) was relatively higher (≥ 0.3). Gaps between the NIR and SWIR bands are spectral regions affected by the atmospheric moisture and these were omitted.
For green reflectance (531 nm - not shown) and yellow reflectance (570 nm - Fig. S4a in Supplementary material), significant differences were found in both wavelengths among species (p ≤ 0.000) and irrigation doses (p = 0.033 for 531 nm and p = 0.022 for 570 nm), but not for the interaction (p ≥ 0.50). Aleppo pine had a significantly higher green reflectance than the other pines, with slightly greater values for the lower watering. Similar results were obtained for red reflectance (670 nm), where significant differences were also found among species (p ≤ 0.000) but not for the irrigation doses (p = 0.06) nor for the interaction (p = 0.56). Again, Aleppo pine had a significantly higher green reflectance than the other pines, without significant differences between irrigation treatments (Fig. S4b in Supplementary material).
For NIR reflectances, no statistical differences were found among species and water dose, except at 1190 nm wavelength, for which both the factor species (p = 0.004) and irrigation (p = 0.03) were significant. Again, P. halepensis showed greater reflectance with low irrigation than the other species (Fig. S4c in Supplementary material).
For SWIR1 (1650 nm) and SWIR2 (2217 nm), Aleppo pine had significantly higher reflectance (p = 0.0012 and p = 0.005, respectively) with no differences between Stone and Scots pine. On the other hand, Aleppo pine had a significantly lower NDVI5 than the other two species (p = 0.003).
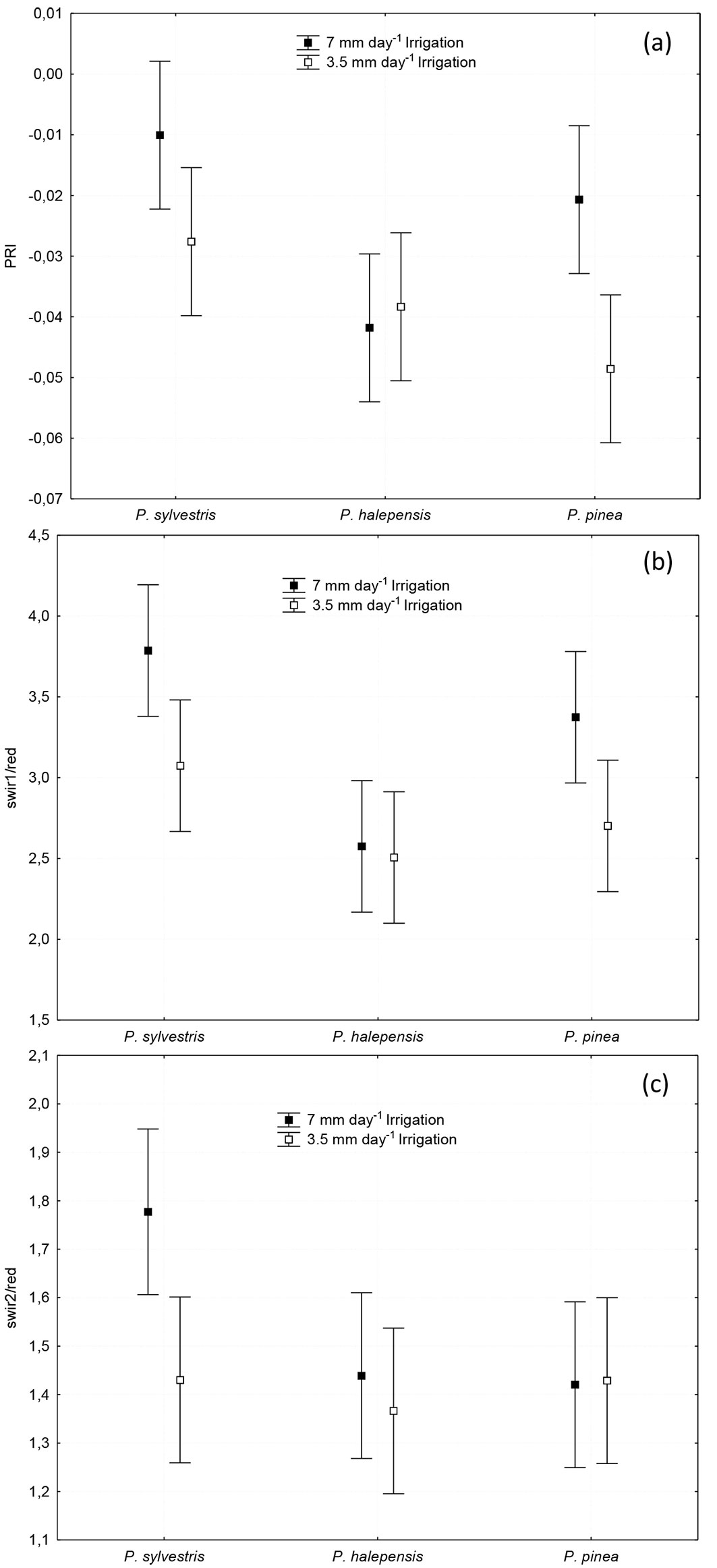
The ANOVA of PRI showed significant differences between species (p = 0.005), water dose (p = 0.009) and the interaction of both factors (p = 0.042). In this case, Scots pine had a higher PRI than the other two species and the higher the water availability, the higher the PRI (Fig. 3a).
Fig. 3 - (a): Photochemical Reflectance Index (PRI); (b): Short Wave Infra-Red (reflectance at wavelength 1650 nm; SWIR1) to Red (670 nm) ratio (SWIR1/Red); and (c): Short Wave Infra-Red (reflectance at wavelength 2217 nm; SWIR2) to Red (670 nm) ratio (SWIR2/Red), of Pinus sylvestris, P. halepensis and P. pinea subjected to high (7 mm day-1) and low (3.5 mm day-1) irrigation treatments. Vertical bars represent 95% Least Significant Difference (LSD) intervals.
No statistical differences were found for the SWIR1/SWIR2 ratio between species, irrigation treatment nor the interaction (p = 0.23). On the other hand, The NDII of Aleppo pine (0.475) was significantly lower (p = 0.0365) than that of Scots pine (NDII = 0.599) and Stone pine (NDII = 0.633). Also, the NIR5/Red ratio index was significant for the species (p = 0.025), with higher values for both Stone pine (NIR5/Red = 7.219) and Scots pine (NIR5/Red = 6.849) than for Aleppo pine (NIR5/Red = 4.089).
The SWIR1/Red ratio was significant for the species (p ≤ 0.001) and for the water dose (p = 0.007), but not for the interaction (p = 0.20 - Fig. 3b). The SWIR2/Red ratio was significant for the species (p = 0.046) but not for the water dose (p = 0.054) or for the interaction (p = 0.10). Only this index has discriminated Scots pine from Stone pine with the high watering dose (Fig. 3c). On the other hand, GEMI was not significant for any factor or for their interaction (p = 0.253).
The multiple correlation analysis among reflectances, reflectance indices, leaf-level gas exchange variables, water potentials and sap flow-derived variables, showed significant correlation indices (Tab. 5). Green reflectance (531 nm) showed moderate associations with midday water potential (r = -0.573) of June 26th, being slightly higher with the pre-dawn (r = -0.659) and midday water potential (r = -0.62) of September. This effect could be explained by the cumulative action of drought, which was more severe at the end of the summer. Yellow reflectance (570 nm) also showed a moderate association with the midday water potential of June (r = -0.59 - Tab. 5), with the September water potential at dawn (r = -0.69) and at midday (r = -0.66). Slightly better correlations with pre-dawn water potential of September were found for red (reflectance at 670 nm, r = -0.72) SWIR2 (r = -0.71) and for NDVI5 (r = 0.73). For midday water potential in September, correlations were -0.67 for red (670 nm), -0.65 for SWIR2 and 0.645 for NDVI5 (Tab. 5).
Tab. 5 - Correlation indices and their significance level (prob, in italic) among reflectances, reflectance indices, leaf-level gas exchange variables, water potentials and sap flow-derived variables: mdPot, midday water potential; pdPot, pre-dawn water potential; Sap Flow rate; An, net assimilation rate. Variables with only non-significant correlations have been omitted for clarity. (*): p ≤ 0.05; (**): p ≤ 0.01; (***): p ≤ 0.001.
| Parameter | mdPot (Jun) |
Sap Flow rate |
Monthly Flow (Jun) |
mdPot (Aug) |
Monthly Flow (Aug) |
pdPot (Sep) |
mdPot (Sep) |
Monthly Flow (Sep) |
|---|---|---|---|---|---|---|---|---|
| green 531 nm | -0.573 ** | -0.280 | -0.283 | -0.538 ** | -0.353 | -0.659 ** | -0.623 ** | -0.237 |
| prob | 0.003 | 0.186 | 0.180 | 0.007 | 0.090 | 0.001 | 0.001 | 0.264 |
| yellow 570 nm | -0.587 ** | -0.324 | -0.329 | -0.570 ** | -0.400 | -0.694 *** | -0.666 *** | -0.282 |
| prob | 0.003 | 0.122 | 0.116 | 0.004 | 0.053 | 0.000 | 0.000 | 0.182 |
| red 670 nm | -0.523 ** | -0.327 | -0.343 | -0.550 ** | -0.396 | -0.721 *** | -0.667 *** | -0.270 |
| prob | 0.009 | 0.119 | 0.101 | 0.005 | 0.055 | 0.000 | 0.000 | 0.203 |
| SWIR2 2217 nm | -0.536 ** | -0.210 | -0.218 | -0.562 ** | -0.294 | -0.714 *** | -0.646 ** | -0.158 |
| prob | 0.007 | 0.326 | 0.306 | 0.004 | 0.163 | 0.000 | 0.001 | 0.460 |
| NDVI5 | 0.493 * | 0.294 | 0.319 | 0.513 * | 0.339 | 0.735 *** | 0.645 ** | 0.235 |
| prob | 0.015 | 0.163 | 0.129 | 0.011 | 0.105 | 0.000 | 0.001 | 0.270 |
| PRI | 0.394 | 0.623 ** | 0.642 ** | 0.542 ** | 0.672 *** | 0.673 *** | 0.723 *** | 0.592 ** |
| prob | 0.057 | 0.001 | 0.001 | 0.006 | 0.000 | 0.000 | 0.000 | 0.002 |
| NDII | 0.445 * | 0.157 | 0.175 | 0.458 * | 0.214 | 0.666 *** | 0.571 ** | 0.104 |
| prob | 0.029 | 0.465 | 0.415 | 0.025 | 0.315 | 0.000 | 0.004 | 0.630 |
| swir1/red | 0.380 | 0.583 ** | 0.603 ** | 0.434 * | 0.604 ** | 0.645 ** | 0.643 ** | 0.541 ** |
| prob | 0.067 | 0.003 | 0.002 | 0.034 | 0.002 | 0.001 | 0.001 | 0.006 |
| An (Oct) | 0.345 | 0.444 * | 0.447 * | 0.475 * | 0.442 * | 0.517 * | 0.672 *** | 0.428 * |
| prob | 0.098 | 0.030 | 0.029 | 0.019 | 0.031 | 0.010 | 0.000 | 0.037 |
PRI showed moderately good correlations with the total mean sap flow rate (r = 0.62), the monthly sap flow in June (r = 0.64), the sap flow in August (r = 0.67), and also with the pre-dawn (r = 0.67) and midday (r = 0.72) water potentials of September (Tab. 5). The SWIR1/red ratio did not provide higher correlations than PRI for the same variables. On the other hand, no good correlations were found for SWIR2/ red, nor in general for other variables, such as E, gs, WUE, IWUE and PPFD (data were omitted from Tab. 5 for the sake of clarity and simplification).
Multivariate analysis of spectral signatures
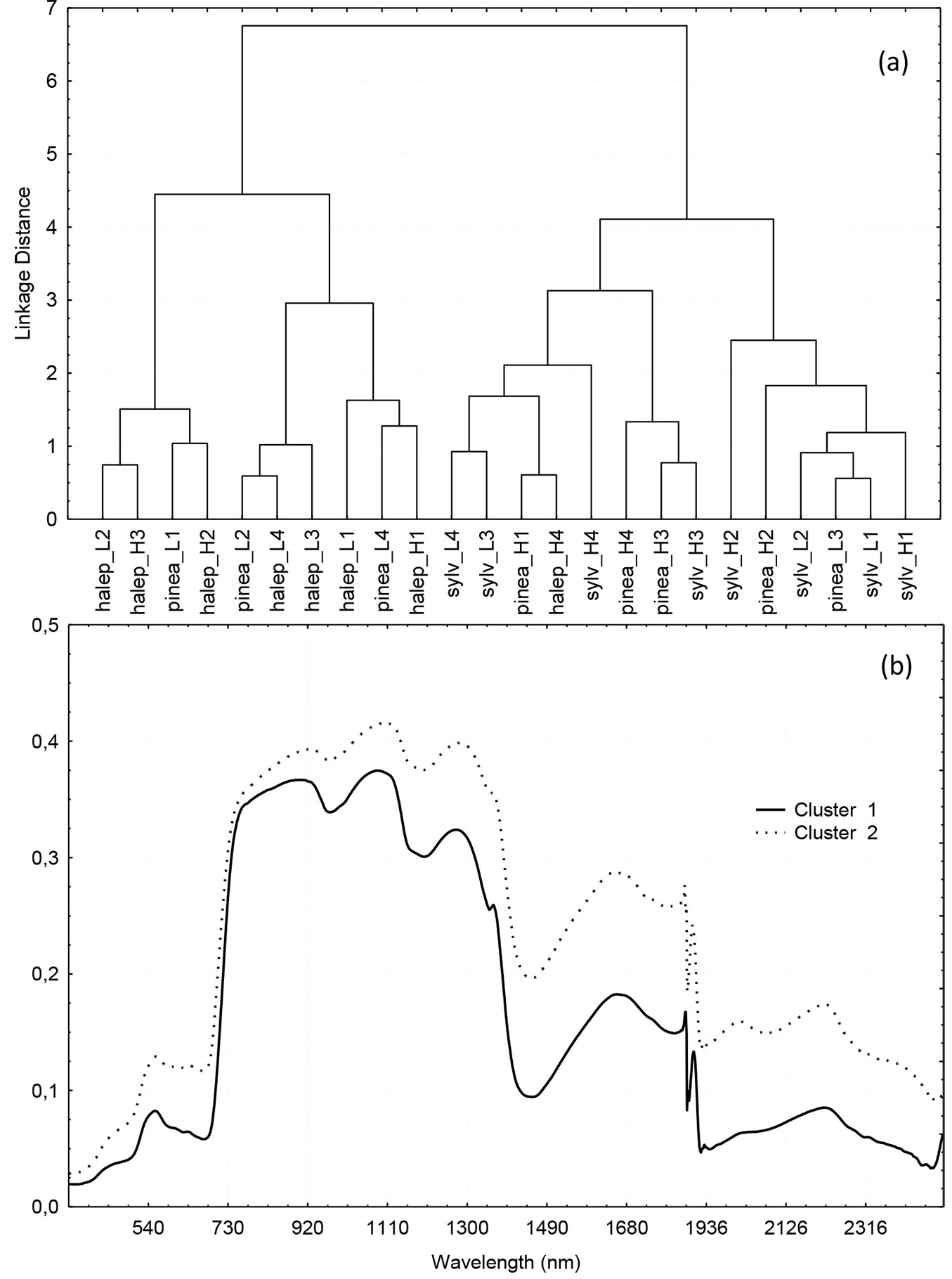
Spectral signatures of all 24 plants were grouped by cluster analysis. Different linkage rules were compared: single linkage, complete linkage, unweighted and weighted pair-group average, unweighted pair-group centroid, weighted pair-group centroid, median and Ward’s method. The best results were provided by the complete linkage rule, which discriminated two groups: one integrated by Aleppo pine plants (except for Aleppo pine plant H4) and Stone pine plants of the low irrigation treatment (except for Stone pine plant L3), and the other one formed by Scots pine plants, Stone pine plants of the high irrigation treatment and the two exceptions mentioned (Fig. 4a). Interestingly, there was no clear discrimination among Aleppo pine plants of either watering regime in the first group or among Scots pine plants of either irrigation treatments in the second group.
Fig. 4 - (a): Cluster analysis of the spectral signatures of Pinus sylvestris (sylv), P. halepensis (halep) and P. pinea (pinea) plants subjected to high (7 mm day-1) and low (3.5 mm day-1) irrigation; (b): Plot of means for clusters grouped by k-means (k=2). Cluster 1: P. sylvestris + P. pinea H + P. pinea L3 + P. halepensis H4. Cluster 2: P. halepensis + P. pinea L - (P. pinea L3 + P. halepensis H4). Plants labeled with an H received high irrigation (7 mm day-1), and plants with label L received low irrigation (3.5 mm day-1).
Similar results were obtained when spectral signatures were grouped by k-means (Fig. 4b). The average spectral signature of the cluster composed of Scots pine and highly watered Stone pine had a lower reflectance than the Aleppo pine and less watered Stone pine, for the spectral range of the study (from 350 to 2500 nm). This pattern difference was highest in the SWIR range, though it is also apparent in the visible green and red, and in the NIR wavelengths (Fig. 4b).
Discussion
Ecophysiological responses of young plants of three Mediterranean pine species (Pinus sylvestris, P. pinea and P. halepensis) to low water availability have been investigated. We observed that the midday water potential was lower on June 26th than on August 17th and on September 24th (Fig. 1b). This rise of the midday water potential that occurred between June and August suggests that pines closed stomata when aridity reaches a threshold. Similar results were obtained by Oliveras et al. ([26]) in adult individuals of P. halepensis, which also showed a more pronounced decrease in midday water potential as compared with P. pinea, while pre-dawn water potentials were not significantly different. These results were also in agreement with those reported for minimum needle water potential measured in severe drought field conditions, -2.6 MPa for P. halepensis, ca. -2.35 for P. pinea, and ca. -2.25 MPa for P. sylvestris ([20]). Also, the water potentials for a 50% loss of xylem conductivity were, respectively, -3.1 MPa, -3.65 MPa and -3.57 MPa ([20]). The low overall variation of minimum water potential as a general rule in pine species implies that structural and physiological adjustments take place to maintain water potentials within relatively narrow limits, in agreement with an isohydric behavior. These results suggest a homeostatic behavior in pines to avoid extremely negative xylem water potentials and to prevent embolism by closing stomata and/or reducing leaf area ([20]). Our results corroborate those considerations, with similar water potential values, though not so excessively low, indicating a moderate water stress in the low irrigated plants (Fig. 1b).
Pinus sylvestris had a higher mean sap flow rate than the other two species with more irrigation but the rate was reduced to approximately the same level as the other two pine species when less water was available. This behavior, again, may be explained either by a superior stomatal conductance or by a more efficient water transport capacity. Indeed, it has been reported that Pinus sylvestris shows a higher wood-specific hydraulic conductivity than P. halepensis and P. pinea ([20]). Our observations partly confirmed that P. sylvestris plants had higher Al:As ratio, which is related to hydraulic conductivity, than P. pinea, but not significantly different from that of P. halepensis. On the other hand, Martínez-Vilalta & Piñol ([19]) found no differences among three pine species (Pinus sylvestris, P. nigra Arn. and P. pinaster Ait.) and their populations of NE Spain in maximum water transport capacity or vulnerability to embolism, corroborating that this homogeneity in hydraulic capacity is quite general in pines ([20]). In Pinus canariensis Sweet, transpiration estimated by sap flow responded to canopy conductance, which was found to decline significantly with increasing evaporative demand and thus significantly reduced tree water loss, and this decrease was more pronounced during the soil drought ([2]). These considerations suggest that other mechanisms, i.e., stomatal regulation, may play a major role in adaptation and acclimatization to drought stress in pine species ([13]).
During the adverse period of August, net assimilation was lower for all species, but as temperatures decreased, Pinus sylvestris showed a better capacity of recovery by a higher assimilation rate than the other pines. In contrast, in an experiment with Pinus halepensis, Klein et al. ([15]) found slow rates of CO2 emission in trees under the highest drought intensity, i.e., ≈ 0.2 µmol m-2 s-1 at midday and 0.4 µmol m-2 s-1 in the late evening in the leaves of the less irrigated plants. Those plants later died, indicating that the CO2 uptake threshold had been surpassed and carbohydrate reserves were depleted ([15]). Similarly in P. sylvestris, CO2 depletion due to leaf mass reduction together with the tight stomatal closure in extremely dry conditions may lead to plant death, in spite of hydraulic adjustments that may allow short-term survival, for long-term resilience in response to drought is jeopardized ([29]). In our case, even in the driest period (August), average An was 0.33 µmol m-2 s-1 for P. pinea, 1.24 for P. sylvestris, and 1.49 for P. halepensis, indicating a non-extreme water limitation. In agreement with our results, Flexas et al. ([7]) reported positive An at a lower leaf water potential in P. halepensis than in P. pinea. Stomatal conductance followed the same pattern of An and E, as it was higher in October than in August. The interaction results indicate that P. halepensis regulates stomata better than the other pines tested, limiting conductance with water shortage in greater measure than Pinus pinea and P. sylvestris. Leaf stomatal conductance also decreased in response to drought, in P. halepensis near the Negev desert ([15]). These authors also observed that gs fell as midday leaf water potential declined and at -2.8 MPa conductance approached zero as a result of 99% stomatal closure. This tight regulation in P. halepensis is a characteristic of isohydric, water saving plant species, while P. pinea follows an intermediate pattern and P. sylvestris shows a tendency closer to the isohydric, water spending pattern, as indicated by the higher transpiration rates maintained by P. sylvestris with low water availability. On the other hand, this lower stomatal sensitivity of Pinus sylvestris may be in benefit of an increase in growth capacity. An, E and gs showed good correlations among them, and of gs with VPG (r = -0.76 - Tab. 1). The model response of gs to VPG described by Eamus & Shanahan ([5]) resembles our model in eqn. 5. Similar patterns of decreasing stomatal conductance with increasing VPG, while the correlation of net assimilation and transpiration with VPG are not so strong, have been described in Pseudotsuga menziesii (Mirb.) Franco ([22]), supporting the hypothesis that stomata respond to the water vapor demand favoring CO2 uptake with respect to water loss.
Similar results of the relationship between solar radiation and sap flow were obtained in Pinus sylvestris ([9]) and also in Pinus cembra L. of the Austrian Alps timberline, although VPG was the variable that best correlated with sap flow density ([39]). A strong association was also found between sap flow-estimated transpiration and potential or reference evapo-transpiration in a Pinus halepensis stand with high soil water content, but this relationship was weaker with low water availability ([37]). These results resemble our finding that sap flow correlated with ETo in the well irrigated plants, but the correlation coefficients were lower for all three species in the low irrigation treatment. Indeed, transpiration (estimated by sap flow) depends on both water availability and evaporative demand (ETo), and when water is a limiting factor, transpiration cannot keep pace with the ETo increase, as a probable consequence of stomatal closure.
Several authors have described pines, especially P. halepensis, as very sensitive to water deficit, showing a strong stomatal control under drought conditions ([1]), in agreement with our observations. This adaptation mechanism of P. halepensis by means of extremely conservative water use (water-saver), contribute in this species to high drought resistance. On the other hand, Pinus halepensis is highly vulnerable to embolism, with a narrow safety margin between stomatal closure and xylem dysfunction as small as 0.3 MPa ([15]). It has been hypothesized that P. halepensis has narrow tracheids and low hydraulic conductance to adjust to low water availability ([15]). This is probably why we observed lower sap flow and stomatal conductance rates than in the other species. Also, it has been reported that P. halepensis leaves are adapted to optimize stomatal conductance for avoiding carbon starvation as much as possible, resulting in leaf water potential approaching the danger zone for xylem collapse. That failure was usually avoided due to rapid stomatal responses. Therefore, P. halepensis is more efficient than most tree species in terms of using the carrying capacity of the xylem ([15]).
We also tested the relationships of leaf spectral reflectance with whole-plant transpiration, water relations and leaf-level gas exchange variables, finding that PRI was a better predictor of sap flow rate, i.e., whole plant transpiration, and of water potential than other spectral indices (Tab. 5). Although the PRI index was first proposed as a good indicator of photosynthetic activity and dissipation of excess energy not absorbed in the photochemical process ([8]), we did not observe a statistical relationship of PRI with net assimilation. Inter-specific differences in xanthophyll content may affect the role of PRI as indicator in drought stress conditions ([6]). Nevertheless, we found a significant relationship between PRI and the water status associated to the irrigation treatment, in agreement with the findings of Ripullone et al. ([30]). Similar results were obtained in Olea europaea, where trees subjected to irrigation treatment had a higher PRI as compared to control rain-fed olive trees ([18]). Also significant differences in PRI value have been observed when comparing several species, indicating that PRI is species-specific ([30]). In Quercus coccifera it was concluded that, contrary to early reports, PRI is a sensible, indirect, non-destructive water stress indicator, even in plants experiencing intense drought ([27]), confirming our finding. Our results also are in agreement with those observed in Pinus sylvestris and P. nigra afforestations, where PRI showed a moderately good association with water potential and stomatal conductance ([12]), and in Olea europaea trees, which also showed a linear relationship between PRI and sap flux density ([18]), though the association was stronger in our case (Tab. 5). A good correlation between PRI and pre-dawn water potential has been described in the short term, in a pool of forest species, i.e., Arbutus unedo, several Quercus species, Castanea sativa, Fagus sylvatica and Populus x euroamericana ([30]).
Characterization of ecosystem functioning and plant ecophysiology by means of associated spectral indices has been a major objective of remote sensing ([8]). We identified simple spectral indices, such as PRI, NDVI, NDII, SR, SWIR/red and specific reflectances, for the discrimination of three dominant Pinus species present in many temperate and semi-arid Mediterranean ecosystems. We also found relationships between those spectral indices and ecophysiological features of the mentioned species, as tools opening possibilities for large-scale environmental studies. Previous studies reported the use of NIR and SWIR-based spectral indices associated with leaf water potential in Pinus contorta and P. coulteri ([3]). Close results were obtained in Olea europaea trees, also showing a good relationship between NDVI and water potential, for which R2 = 0.668 was reported ([18]), similar to our results (Tab. 5). In contrast, Dzikiti et al. ([4]) did not find significant correlations between water potential and various combinations of spectral reflectances, but the use of the first order spectral derivatives improved their results in orange trees.
Conclusions
In conclusion, ecophysiological responses of young plants of three Mediterranean pine species (Pinus sylvestris, P. pinea and P. halepensis) to water availability have been investigated. Two irrigation treatments were compared: a high water dose (7 mm day-1) and a low dose (3.5 mm day-1). Significant differences have been recorded in water potential, sap flow and leaf-level gas exchange variables and in their interactions. Our results revealed different responses to low water availability among the three pine species studied. All species showed the isohydric, drought-avoiding strategy which is common in the genus Pinus, maintaining relatively high water potentials. Nevertheless, P. halepensis showed a strong water-saving pattern, having a tight stomatal closure with drought, even at the expense of affecting CO2 uptake. On the other hand, stomatal regulation is less strict in Pinus sylvestris, closer to a water-spending behavior. Pinus pinea shows an intermediate behavior between the other two species. We may conclude that Pinus sylvestris may be endangered in a climate change context of more aridity in Mediterranean mountains. The search for inter- and intra-population variability, to explore more drought-tolerant individuals, more adapted ecotypes or other species should be considered in reforestation strategies. Alternatively, thinning may improve drought recovery and should be considered in forest management to improve drought tolerance in endangered forest species.
The spectral reflectance indices tested in this study may be valuable for fast, nonintrusive detection of water stress. We propose remotely sensed reflectance indices, in particular PRI, as promising predictive tools of the impact of drought on photosynthetic activity, water status and whole-plant transpiration.
Future applications of these techniques could be forest cover classifications in areas where Pinus sylvestris, P. pinea and P. halepensis are sympatric and need to be discriminated among them. We could discriminate the spectral signatures of P. sylvestris and P. halepensis by cluster analysis, and conclude that both species may be distinguished by hyperspectral sensors. On the other hand, P. pinea is intermediate between the other two species, and the hydric status of this pine can mimic the other two spectral patterns. With a high hydric availability, P. pinea spectral signature cannot be differentiated from that of P. sylvestris, and with less water, can be confused with P. halepensis. Presumably, in forested areas where P. halepensis and P. pinea are sympatric, the best moment for an adequate discrimination of the areas occupied by both species by hyperspectral imagery would be during the moist season. The findings of this experiment may help to quantify the impacts of early and mid-summer water deficit on Mediterranean pines and evaluate their potential responses to future climate regimes.
Acknowledgements
This work is the result of collaboration in the Moncloa Campus of International Excellence. EM carried out gas exchange measurements, MR and JM carried out the spectro-radiometry measurements, JZ was in charge of the nursery and laboratory measurements, EMF and JM performed the statistical analysis, AGG, BP and AGA conceived the study and JM helped to draft the manuscript. This work was supported by the Spanish Ministry of Industry, Project FIT330221200610 and Ministry of Education and Science, Project AGL-2010-17505. We thank Prof. Alicia Palacios and Victor Cicuendez for their scientific advice. We also thank Prof. J.C. Robredo for providing the meteorological data.
Conflict of interest
The authors declare no conflict of interest.
References
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Authors’ Info
Authors’ Affiliation
Javier Zazo
Eugenio Martínez-Falero
Antonio García-Abril
Technical University of Madrid, College of Forestry and Natural Environment, Ciudad Universitaria, Moncloa Campus of International Excellence, 28040 Madrid (Spain)
Beatriz Pintos
Eugenio Moreda
Complutense University of Madrid, Dept. of Plant Physiology, Ciudad Universitaria, Moncloa Campus of International Excellence, 28040 Madrid (Spain)
Autonomous University of Madrid, Madrid (Spain)
Corresponding author
Paper Info
Citation
Manzanera JA, Gómez-Garay A, Pintos B, Rodríguez-Rastrero M, Moreda E, Zazo J, Martínez-Falero E, García-Abril A (2016). Sap flow, leaf-level gas exchange and spectral responses to drought in Pinus sylvestris, Pinus pinea and Pinus halepensis. iForest 10: 204-214. - doi: 10.3832/ifor1748-009
Academic Editor
Tamir Klein
Paper history
Received: Jun 24, 2015
Accepted: Jul 16, 2016
First online: Nov 01, 2016
Publication Date: Feb 28, 2017
Publication Time: 3.60 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2016
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 53282
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 44041
Abstract Page Views: 3746
PDF Downloads: 4096
Citation/Reference Downloads: 59
XML Downloads: 1340
Web Metrics
Days since publication: 3358
Overall contacts: 53282
Avg. contacts per week: 111.07
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2017): 10
Average cites per year: 1.11
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Growing season water balance of an inner alpine Scots pine (Pinus sylvestris L.) forest
vol. 11, pp. 469-475 (online: 02 July 2018)
Review Papers
Should the silviculture of Aleppo pine (Pinus halepensis Mill.) stands in northern Africa be oriented towards wood or seed and cone production? Diagnosis and current potentiality
vol. 12, pp. 297-305 (online: 27 May 2019)
Research Articles
Allometric equations to assess biomass, carbon and nitrogen content of black pine and red pine trees in southern Korea
vol. 10, pp. 483-490 (online: 12 April 2017)
Review Papers
Climate change impacts on spatial distribution, tree-ring growth, and water use of stone pine (Pinus pinea L.) forests in the Mediterranean region and silvicultural practices to limit those impacts
vol. 14, pp. 104-112 (online: 01 March 2021)
Research Articles
Coping with spring frost-effects on polyamine metabolism of Scots pine seedlings
vol. 10, pp. 227-236 (online: 27 January 2017)
Research Articles
Developing stand transpiration model relating canopy conductance to stand sapwood area in a Korean pine plantation
vol. 14, pp. 186-194 (online: 14 April 2021)
Research Articles
Amount and distribution of coarse woody debris in pine ecosystems of north-western Spain, Russia and the United States
vol. 7, pp. 53-60 (online: 28 October 2013)
Research Articles
The growth dynamics of East European Scots pine (Pinus sylvestris L.) populations - a Lithuanian field trial
vol. 17, pp. 59-68 (online: 06 March 2024)
Review Papers
Impacts of climate change on the establishment, distribution, growth and mortality of Swiss stone pine (Pinus cembra L.)
vol. 3, pp. 82-85 (online: 15 July 2010)
Review Papers
Biomass, radial growth and regeneration capacity of Aleppo pine, and its possible use as rootstock in arid and degraded areas
vol. 15, pp. 213-219 (online: 16 June 2022)
iForest Database Search
Search By Author
- JA Manzanera
- A Gómez-Garay
- B Pintos
- M Rodríguez-Rastrero
- E Moreda
- J Zazo
- E Martínez-Falero
- A García-Abril
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
- JA Manzanera
- A Gómez-Garay
- B Pintos
- M Rodríguez-Rastrero
- E Moreda
- J Zazo
- E Martínez-Falero
- A García-Abril
Search By Keywords
PubMed Search
Search By Author
- JA Manzanera
- A Gómez-Garay
- B Pintos
- M Rodríguez-Rastrero
- E Moreda
- J Zazo
- E Martínez-Falero
- A García-Abril
Search By Keyword