Allometric equations to assess biomass, carbon and nitrogen content of black pine and red pine trees in southern Korea
iForest - Biogeosciences and Forestry, Volume 10, Issue 2, Pages 483-490 (2017)
doi: https://doi.org/10.3832/ifor2164-010
Published: Apr 12, 2017 - Copyright © 2017 SISEF
Research Articles
Abstract
A total of 74 Japanese black pine (Pinus thunbergii Parl.) and red pine (P. densiflora S. et Z.) trees were destructively sampled in southern Korea, which is severely affected by pine wilt disease (PWD). Species-specific allometric equations were developed to estimate the biomass, carbon (C) and nitrogen (N) content of the tree components (i.e., stem wood, stem bark, branches, needles and roots) based on the diameter at breast height (DBH) and stem diameter at 20 cm aboveground (D20). The C concentrations of the various tree components were not correlated with DBH (P > 0.05), except for the C concentration in the stem bark (r = -0.29, P < 0.05) of the black pine and the branches (r = 0.40, P < 0.05) of the red pine. However, the N concentrations in the stem wood (r = -0.53, P < 0.05), stem bark (r = -0.37, P < 0.05) and branches (r = -0.40, P < 0.05) of the black pine were negatively correlated with DBH. The mean C concentrations of the tree components were not significantly different between the black pine and red pine, except for the stem bark, whereas the mean N concentrations were significantly lower in the black pine than in the red pine, except for the stem bark. The allometric equations developed for the biomass, C and N content for all the tree components were significant (P < 0.05). The adjusted coefficient of determination (adj. R2) of the DBH allometric equations ranged from 0.66 to 0.97, while the coefficients for the D20 equations were between 0.66 and 0.95. Black pines consistently exhibited more biomass, C and N content in the tree components compared with the red pines with similar DBH or D20. These results suggest that the accuracy of estimates for biomass, C and N stocks in black pine and red pine forests could be improved by specific allometric equations for PWD-disturbed forests.
Keywords
Biomass Equations, Black Pine, Carbon Stocks, Nitrogen Stocks, Pine Wilt Disease, Red Pine
Introduction
Epidemic outbreaks of insects or pathogens in forest ecosystems in recent years have been unusually widespread and severe ([19], [9]) because global warming is permitting the invasion of tree populations that previously were inaccessible due to a short growing season. Trees infected or killed by biotic disturbances, such as insects or pathogens, no longer take up carbon (C) and nitrogen (N) and contribute to the loss of C and N stocks in the forest ecosystems. In addition, the export of dead trees may have a negative impact on the nutrient balance of the ecosystem and subsequently on soil chemical properties. Therefore, an accurate estimation of nutrient removal based on the biomass export is crucial to adjust forest management practices for disturbed forest sites.
Biomass, C and N content in forest ecosystems are commonly estimated by using allometric equations ([26], [6]). However, there is an inherent difficulty in using allometric equations to estimate multiple-scale biomass, C and N content of disturbed forests because most equations were developed using trees sampled from specific study sites or sites that represent small regions ([15], [25], [6], [24]). Although generalized allometric equations are available for biomass estimation of different tree components at regional landscape scales ([25]), the equations should be applied with caution to disturbed forests because pathogenic impacts can alter tree allometry.
Pine wilt disease (PWD), caused by the pine wood nematode (Bursaphelenchus xylophilus [Steiner et Buhrer] Nickle), has become a serious threat to pine ecosystems in many countries, including Korea ([19]), Japan ([22]), China ([7]) and several countries in Europe ([31]). PWD is considered the most destructive forest disease because the mortality rates of pine trees infected by PWD can approach 100% ([33]). Mortality in trees affected by PWD causes substantial losses of C and N stocks, with large influences on the C and N cycles in forest ecosystems. Analysis of the impacts of PWD outbreaks on C cycling have been performed on stand scales, and a previous stand-scale study in Korea documented large variability in PWD-affected C stocks ([13]). Numerous uncertainties limit estimates or predictions of the loss of C and N stocks in PWD-disturbed forests. Rapid, easily implemented methods are needed to quantify the degree of biomass, C and N loss on the local and regional scales in PWD-disturbed forests.
Japanese black pine (Pinus thunbergii Parl.) and red pine (P. densiflora S. et Z.) are the most dominant and important coniferous tree species that are severely disturbed by PWD throughout Korea ([14], [19], [13]). Although numerous biomass estimation models have been developed for red pine in Korea ([15], [23]), no studies are available to predict tree C and N content in disturbed forest ecosystems such as PWD-disturbed forests. This study aims to provide allometric equations to estimate the exact amount of biomass, C and N content within different tree components of black pine and red pine stands that are disturbed by PWD in southern Korea.
Materials and methods
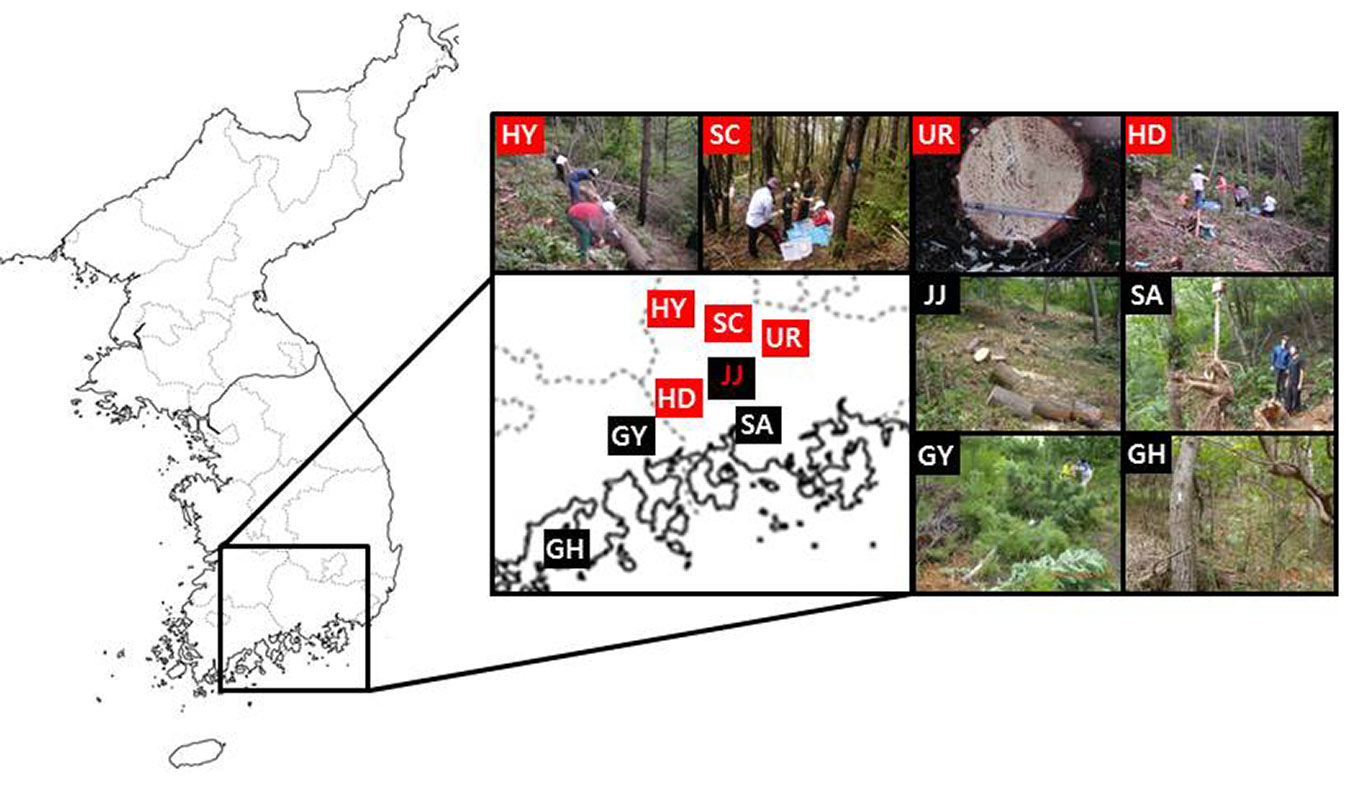
The study was conducted in natural black pine stands in four different regions (Goheung: GH1, GH2; Gwangyang: GY; Sacheon, SA; and Jinju: JJ1, JJ2, JJ3, JJ4) and natural red pine stands in five different regions (Hadong: HD; Hamyang: HY; Jinju: JJ5; Sancheong: SC; and Uiryeong, UR), located in the south-central part of Korea (Fig. 1). The study sites were severely PWD-disturbed regions in Korea since the outbreak of the disease in 1998 ([19], [13]). By 2014, 8.020.000 trees in forests had been killed by PWD throughout the country ([18]).
Fig. 1 - Location of the study site (black pine, GH: Goheung, GY: Gwangyang, SA: Sacheon, JJ: Jinju; red pine, HY: Hamyang, SC: Sancheong, UR: Uiryeong, HD: Hadong, JJ: Jinju).
The annual average precipitation and temperature in HD are higher than for other regions in the study area (Tab. S1 in Supplementary material). The soils in HD, HY, GH, GY and SC are well-drained, slightly wet, brown forest soils (mostly Inceptisols or Alfisol, USDA soil taxonomy) originating from granite with a loamy texture, while the soils in JJ, SA and UR are slightly dry, dark brown forest soils (Inceptisol) originating from sandstone with a silt loam texture. Because the black pine and red pine species are distributed in various environmental site conditions, the sampling for this study was planned to cover a broad range of site characteristics such as parent materials and stand age class, to provide a regional representation applicable at a landscape scale (Tab. S1 in Supplementary material).
The experimental design consisted of a 20 × 20 m plot within each site in the PWD-infected districts ([19]). The stand density for black pine was 2800 trees ha-1 at the GY site and ranged from 325 to 1000 trees ha-1 at the other sites. The stand density for red pine ranged from 925 to 2200 trees ha-1 (Tab. S1 in Supplementary material). The mean stand density was higher for red pine (1234 tree ha-1) than for black pine (875 tree ha-1 - Tab. S1). The mean tree age of red pine (36 years) was slightly lower than for black pine (43 years) (Tab. S2 in Supplementary material). The study sites were implemented for selective cutting of the infected and dead pine trees by PWD.
To develop allometric equations, a complete tree inventory was carried out for each stand to characterize the stem diameter distribution. Five or eight diameter classes based on the DBH ranges were established for each site, and sample trees were randomly chosen within each DBH class. However, it was preferable to select trees with a maximum and minimum DBH from each site. The trees were destructively sampled and separated into components (i.e., needles, branches and stems) in August of 2008 (red pine, 25 trees), August of 2009 (black pine, 25 trees) and August of 2012 (black pine, 24 trees). The roots of two or eight of the sampled trees within each site were excavated using a combined approach of hand pulling and machine excavation (Fig. 1). Coarse roots (diameter > 5 mm) were collected to determine the dry root biomass. Determination of the fresh biomass for all the tree components was conducted in the field using portable electronic balances. Sub-samples were taken from each tree component to determine the fresh-to-oven-dried biomass ratio. All the investigations were carried out in accordance with technical standards formulated by the Korea Forest Research Institute ([17]). The samples of each tree component were oven-dried at 85 °C for one week, and the dried samples were ground in a Wiley mill and passed through a 40-mesh stainless steel sieve. C and N concentrations from the ground materials were determined using an elemental analyzer (Thermo Fisher Scientific Flash 2000®, Milan, Italy). The C and N contents of the tree components (stem wood, stem bark, branches, needles and roots) of all the study trees were calculated to determine the relationships between C and N concentration and the biomass of each tree component.
The allometric equations developed for each of the tree components (stem wood, stem bark, branches, needles, roots) were of the following form (eqn. 1, eqn. 2):
where y is the biomass (kg), C or N content (kg) of the respective tree component, DBH is the diameter at breast height (cm) at 120 cm above the ground, and D20 is the stem diameter at 20 cm above the ground, and a and b are regression parameters. DBH was used as the primary variable to describe biomass and the C and N contents in both pine stands because DBH can be measured accurately as a variable for the prediction of tree biomass and C stocks ([2]). The other primary variable, D20, is the diameter at the 20 cm height of the tree stump, which is the easiest measurable variable after pine trees infected by PWD have been removed from the field. The accuracy of the allometric equations was evaluated by the adjusted coefficient of determination (adj. R2) and root mean square error (RMSE - [28]). Bias correction factors (CF) in the logarithmic transformation were calculated using the standard error of the estimate ([8]). Analyses of C and N concentrations were performed using the PROC GLM procedure of the software package SAS® ([27]), where tree DBH was classed as a covariate. Where significant differences occurred, the treatment means were compared using the post-hoc Tukey’s test at P < 0.05. The original data used to perform this analysis are given in Tab. S3 (Supplementary material).
Results
C and N concentrations among tree components
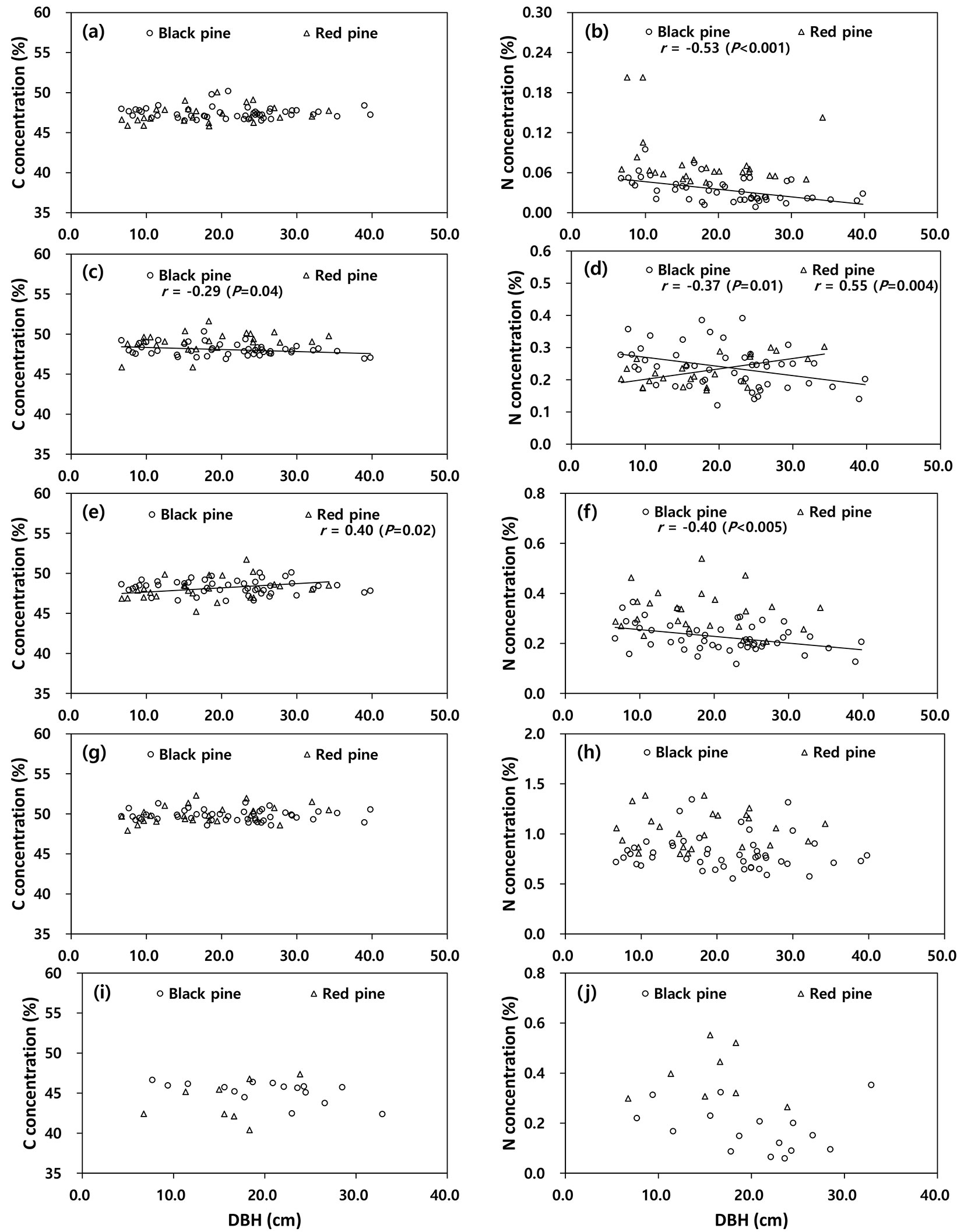
The C concentration among the tree components was not correlated with DBH (P > 0.05), except the C concentration in the stem bark of black pine (r = -0.29, P < 0.05) and the branches of red pine (r = 0.40, P = 0.04 - Fig. 2). The N concentrations in stem wood (r = -0.53, P = 0.02), stem bark (r = -0.37, P = 0.01) and branches (r = -0.40, P = 0.04) of black pine were negatively correlated with DBH, whereas the N concentration in stem bark of red pine (r = 0.55, P < 0.05) was positively correlated with DBH.
Fig. 2 - Correlation between carbon or nitrogen concentrations and diameter at breast height (DBH) among tree components (a-b: stem wood, c-d: stem bark, e-f: branches, g-h: needles, i-j: root) in black pine and red pine.
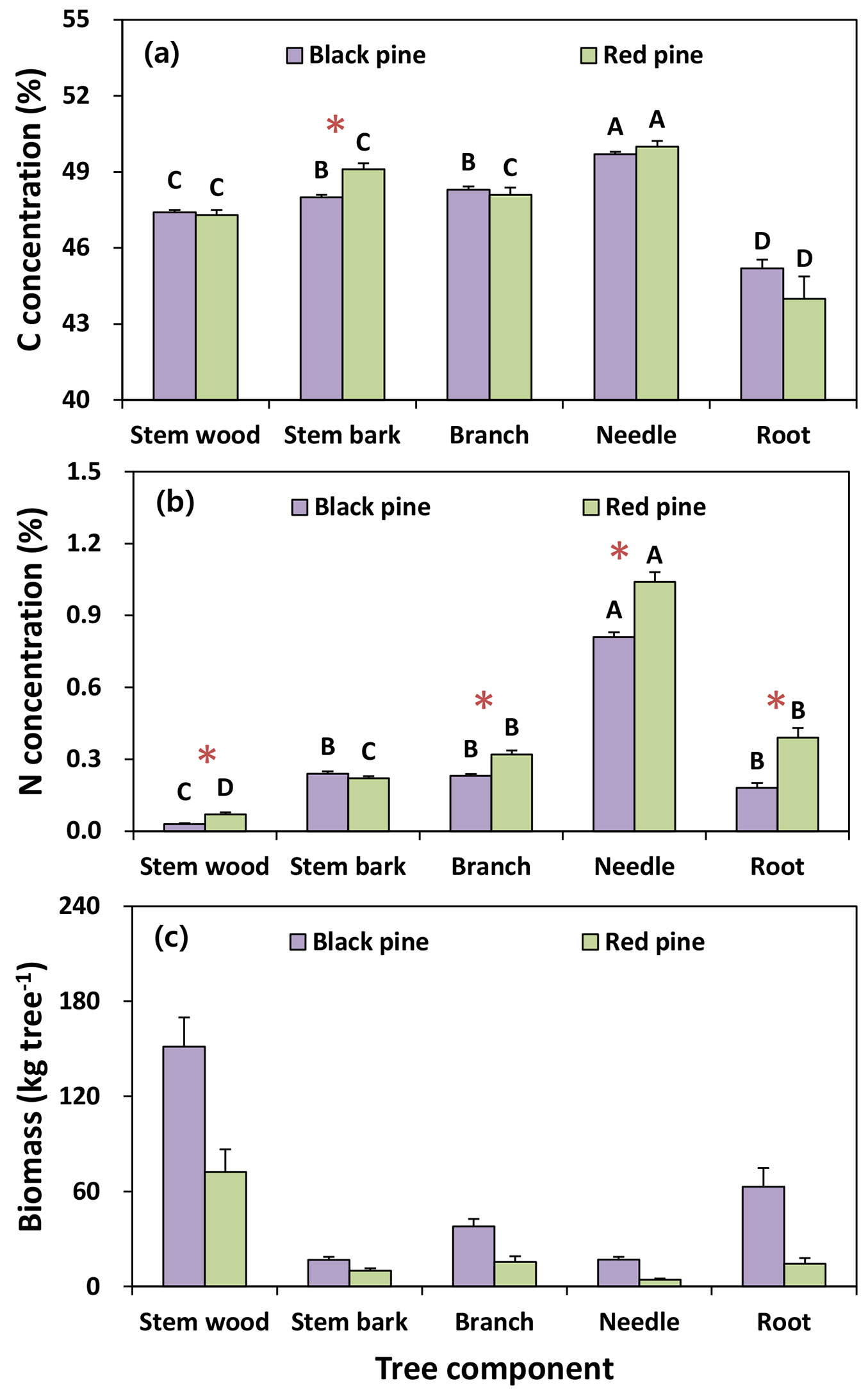
Mean C and N concentrations varied significantly among the tree components and tree species (Fig. 3). The mean values of C concentration in both pine species were significantly higher in needles (black pine: 49.7%; red pine: 50.0%) than in other woody components, such as stem bark (black pine: 48.0%; red pine: 49.1%), branches (black pine: 48.3%; red pine: 48.1%), stem wood (black pine: 47.4%; red pine: 47.3%) and roots (black pine: 45.2%; red pine: 44%). C concentrations in both pine species were not significantly different among the tree components, except for stem bark, which was significantly higher in the red pine (49.1%) than in the black pine (48.0%). Mean N concentrations in both pine species were significantly different among the tree components. The highest concentrations of N in both pine species were found in needles (black pine: 0.81%; red pine: 1.04%), whereas the lowest concentrations occurred in stem wood (black pine: 0.03%; red pine: 0.07%). In contrast to C concentration, N concentrations in stem bark were not significantly different between the two pine species, whereas the N concentrations in other tree components were significantly higher in the red pine than in the black pine (Fig. 3).
Fig. 3 - Mean values of biomass, carbon and nitrogen concentration for tree species and tree components in black pine (n = 49) and red pine (n = 25). Vertical bars represent standard error. Different letters among tree components in each tree species and asterisks between both pine species indicate a significant difference at P < 0.05.
Allometric equations in biomass, C and N content
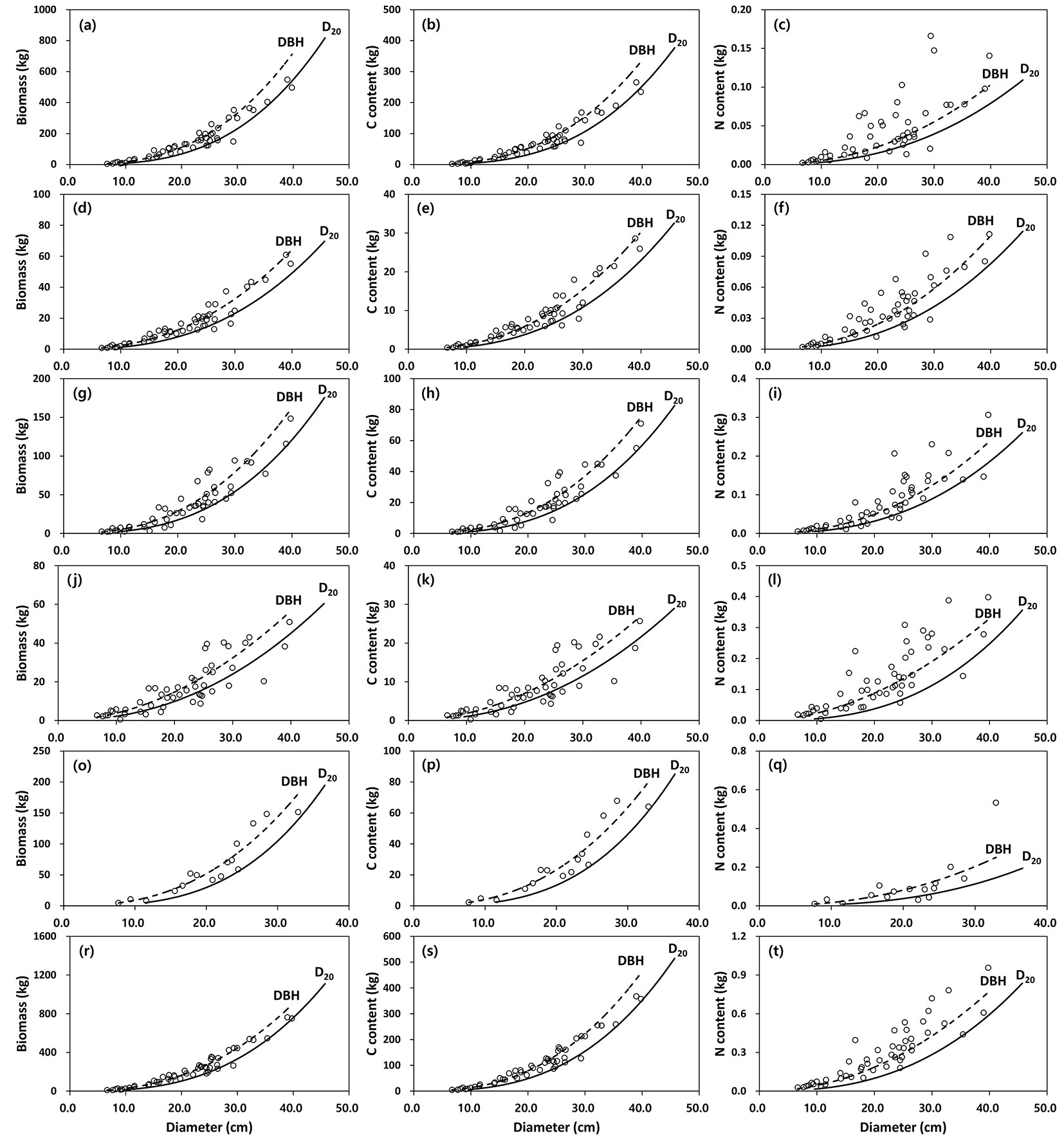
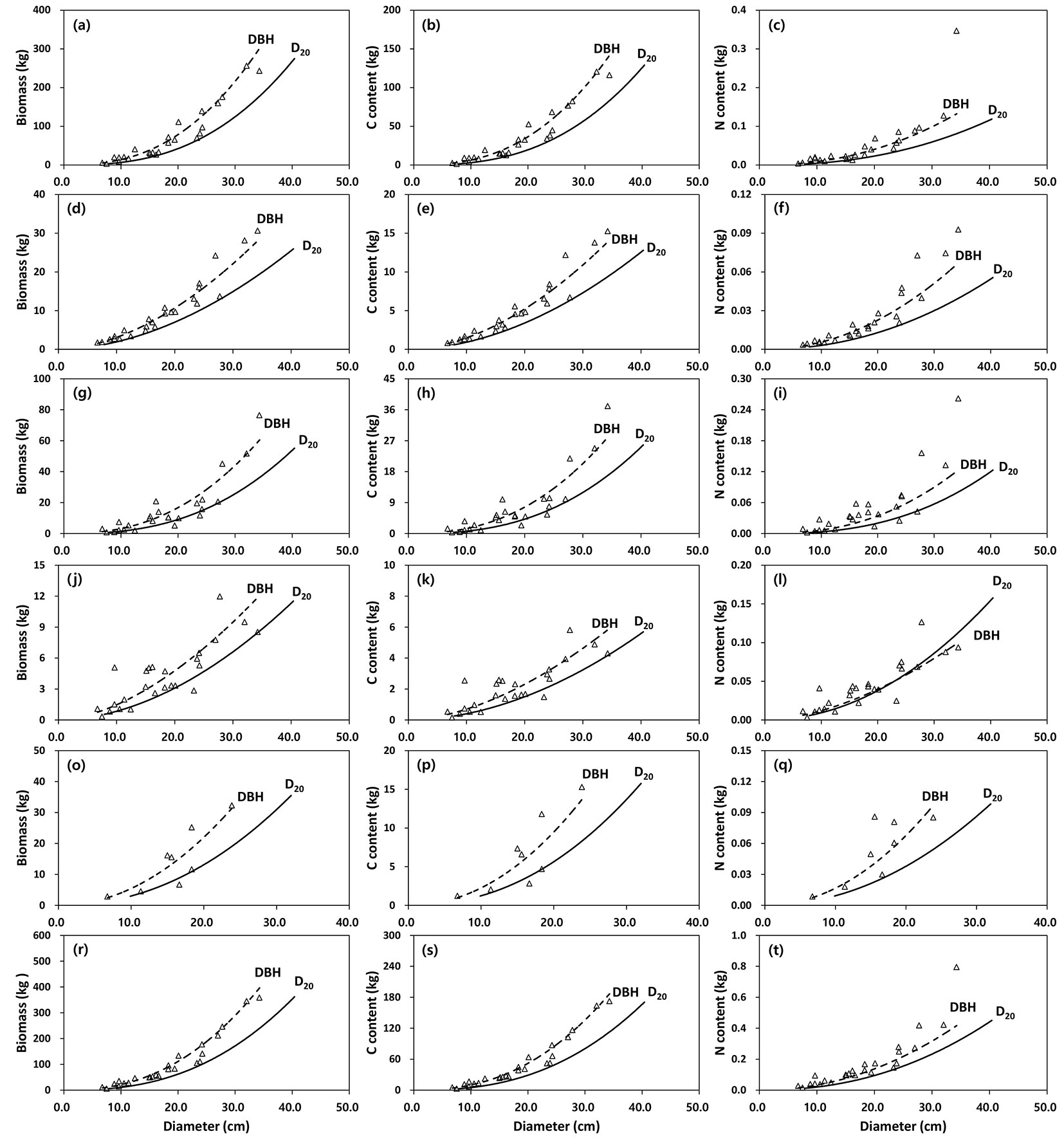
The relationships between biomass, C and N content for all the tree components and DBH or D20 are shown in Fig. 4 (black pine) and Fig. 5 (red pine). The allometric equations for all the tree components were significant (P < 0.05), with DBH or D20 accounting for 69-97% of the biomass, 70-97% of the C content and 66-91% of the variation in N content (Tab. 1, Tab. 2 and Tab. 3). In addition, the adjusted coefficient of determination (adj. R2) for the DBH allometric equations ranged from 0.66 to 0.97, while those for the D20 allometric equations were between 0.66 and 0.95. Based on the adj. R2 and RMSE, generally the best fits for biomass, C and N content were obtained for aboveground measurements (stem, branch, bark), whereas biomass, C and N content of needles or roots had the worst fit among all the tree components. The DBH allometric equations generally provided a better fit than D20 allometric equations on the basis of the values of adj. R2 and RMSE. The slopes (i.e., coefficient b) for biomass, C or N content in the tree components were generally lower in the DBH than in the D20 allometric equations (Tab. 1, Tab. 2 and Tab. 3).
Fig. 4 - Relationship between biomass (left column), carbon (central column) and nitrogen (right column) content of tree components (a-c: stem wood, d-f: stem bark, g-i: branches, j-l: needles, o-q: root, r-t: aboveground) between DBH and D20 in black pine (n=49). Observed (open circle) vs. predicted values (dashed line: DBH; solid line: D20) using allometric equations are displayed.
Fig. 5 - Relationship between biomass (left column), carbon (central column) and nitrogen (right column) content of tree components (a-c: stem wood, d-f: stem bark, g-i: branches, j-l: needles, o-q: root, r-t: aboveground) between DBH and D20 in red pine (n=25). Observed (open triangle) vs. predicted values (dashed line: DBH ; solid line: D20) using allometric equations are displayed.
Tab. 1 - Regression parameters for species-specific allometric equations of biomass estimation in tree components of black pine (n = 49) and red pine (n = 25). Allometric equation form is log10y = a + b log10(DBH or D20). The adj. R2 is the adjusted coefficient of determination. P-values represent the significance of the equations. (RMSE): root means squared error; (CF): correction factor.
| Variable | Tree species |
Tree component ( y) |
Coefficient | Adj. R2 | RMSE | P-value | CF | |
|---|---|---|---|---|---|---|---|---|
a
|
b
|
|||||||
| DBH | Black pine | Stem wood (kg) | -1.4410 | 2.6504 | 0.94 | 0.1272 | <0.0001 | 1.019 |
| Stem bark (kg) | -1.9608 | 2.3352 | 0.94 | 0.1124 | <0.0001 | 1.015 | ||
| Branch (kg) | -1.7242 | 2.4042 | 0.87 | 0.1823 | <0.0001 | 1.039 | ||
| Needle (kg) | -1.2966 | 1.8446 | 0.73 | 0.2186 | <0.0001 | 1.057 | ||
| Aboveground (kg) | -0.9897 | 2.4486 | 0.97 | 0.0856 | <0.0001 | 1.008 | ||
| Root (kg) | -1.5416 | 2.4869 | 0.94 | 0.2193 | <0.0001 | 1.012 | ||
| Red pine | Stem wood (kg) | -1.3140 | 2.4272 | 0.90 | 0.1547 | <0.0001 | 1.028 | |
| Stem bark (kg) | -1.3024 | 1.7847 | 0.95 | 0.0786 | <0.0001 | 1.007 | ||
| Branch (kg) | -1.7527 | 2.2283 | 0.78 | 0.2387 | <0.0001 | 1.068 | ||
| Needle (kg) | -1.4507 | 1.6100 | 0.69 | 0.2115 | <0.0001 | 1.053 | ||
| Aboveground (kg) | -0.8916 | 2.2289 | 0.94 | 0.1117 | <0.0001 | 1.014 | ||
| Root (kg) | -1.2218 | 1.9272 | 0.72 | 0.1940 | 0.005 | 1.044 | ||
| D20 | Black pine | Stem wood (kg) | -2.0111 | 2.9152 | 0.91 | 0.1594 | <0.0001 | 1.030 |
| Stem bark (kg) | -2.4595 | 2.5659 | 0.91 | 0.1422 | <0.0001 | 1.024 | ||
| Branch (kg) | -2.3117 | 2.6962 | 0.87 | 0.1794 | <0.0001 | 1.038 | ||
| Needle (kg) | -1.7701 | 2.0852 | 0.75 | 0.2122 | <0.0001 | 1.053 | ||
| Aboveground (kg) | -1.5361 | 2.7078 | 0.95 | 0.1130 | <0.0001 | 1.015 | ||
| Root (kg) | -2.5432 | 3.0676 | 0.93 | 0.1192 | <0.0001 | 1.016 | ||
| Red pine | Stem wood (kg) | -1.7845 | 2.5909 | 0.92 | 0.1481 | <0.0001 | 1.026 | |
| Stem bark (kg) | -1.5500 | 1.8304 | 0.88 | 0.1226 | <0.0001 | 1.017 | ||
| Branch (kg) | -2.2507 | 2.4288 | 0.82 | 0.2161 | <0.0001 | 1.055 | ||
| Needle (kg) | -1.8706 | 1.8004 | 0.77 | 0.1833 | <0.0001 | 1.039 | ||
| Aboveground (kg) | -1.3259 | 2.3809 | 0.95 | 0.1024 | <0.0001 | 1.012 | ||
| Root (kg) | -1.5667 | 2.0282 | 0.72 | 0.1921 | <0.005 | 1.043 | ||
Tab. 2 - Regression parameters of species-specific allometric equations of carbon content in tree components of black pine (n = 49) and red pine (n = 25). Allometric equation form is log10y = a + b log10(DBH or D20). The adj. R2 is the adjusted coefficient of determination. P-values represent the significance of the equations. (RMSE): root means squared error; (CF): correction factor.
| Variable | Tree species |
Tree component ( y) |
Coefficient | Adj. R2 | RMSE | P-value | CF | |
|---|---|---|---|---|---|---|---|---|
a
|
b
|
|||||||
| DBH | Black pine | Stem wood (kg) | -1.7618 | 2.6479 | 0.94 | 0.1267 | <0.0001 | 1.019 |
| Stem bark (kg) | -2.2675 | 2.3261 | 0.94 | 0.1124 | <0.0001 | 1.015 | ||
| Branch (kg) | -2.0378 | 2.4021 | 0.87 | 0.1824 | <0.0001 | 1.039 | ||
| Needle (kg) | -1.5977 | 1.8429 | 0.73 | 0.2185 | <0.0001 | 1.057 | ||
| Aboveground (kg) | -1.3014 | 2.4421 | 0.97 | 0.0846 | <0.0001 | 1.008 | ||
| Root (kg) | -1.8359 | 2.4472 | 0.94 | 0.1111 | <0.0001 | 1.014 | ||
| Red pine | Stem wood (kg) | -1.6616 | 2.4455 | 0.90 | 0.1557 | <0.0001 | 1.028 | |
| Stem bark (kg) | -1.6362 | 1.8048 | 0.95 | 0.0797 | <0.0001 | 1.007 | ||
| Branch (kg) | -2.0936 | 2.2473 | 0.77 | 0.2391 | <0.0001 | 1.067 | ||
| Needle (kg) | -1.7803 | 1.6330 | 0.69 | 0.2118 | <0.0001 | 1.053 | ||
| Aboveground (kg) | -1.2293 | 2.2427 | 0.94 | 0.1116 | <0.0001 | 1.014 | ||
| Root (kg) | -1.6365 | 1.9712 | 0.70 | 0.1940 | 0.006 | 1.052 | ||
| D20 | Black pine | Stem wood (kg) | -2.3335 | 2.9142 | 0.91 | 0.1580 | <0.0001 | 1.029 |
| Stem bark (kg) | -2.7648 | 2.5564 | 0.91 | 0.1418 | <0.0001 | 1.023 | ||
| Branch (kg) | -2.6253 | 2.6942 | 0.87 | 0.1794 | <0.0001 | 1.098 | ||
| Needle (kg) | -2.0714 | 2.0839 | 0.75 | 0.2120 | <0.0001 | 1.053 | ||
| Aboveground (kg) | -1.8485 | 2.7022 | 0.95 | 0.1109 | <0.0001 | 1.014 | ||
| Root (kg) | -2.8181 | 3.0162 | 0.93 | 0.1194 | <0.0001 | 1.017 | ||
| Red pine | Stem wood (kg) | -2.1329 | 2.6084 | 0.91 | 0.1502 | <0.0001 | 1.026 | |
| Stem bark (kg) | -1.888 | 1.8522 | 0.89 | 0.1235 | <0.0001 | 1.018 | ||
| Branch (kg) | -2.5957 | 2.4493 | 0.82 | 0.2140 | <0.0001 | 1.054 | ||
| Needle (kg) | -2.2052 | 1.8253 | 0.77 | 0.1830 | <0.0001 | 1.039 | ||
| Aboveground (kg) | -1.6642 | 2.3941 | 0.95 | 0.1034 | <0.0001 | 1.012 | ||
| Root (kg) | -1.9957 | 2.0842 | 0.71 | 0.2043 | <0.0001 | 1.049 | ||
Tab. 3 - Regression parameters of species-specific allometric equations of nitrogen content in tree components of black pine (n = 49) and red pine (n = 25). Allometric equation form is log10y = a + b log10(DBH or D20). The adj. R2 is the adjusted coefficient of determination. P-values represent the significance of the equations. (RMSE): root means squared error; (CF): correction factor.
| Variable | Tree species |
Tree component ( y) |
Coefficient | Adj. R2 | RMSE | P-value | CF | |
|---|---|---|---|---|---|---|---|---|
a
|
b
|
|||||||
| DBH | Black pine | Stem wood (kg) | -4.1655 | 2.0247 | 0.72 | 0.2444 | <0.0001 | 1.071 |
| Stem bark (kg) | -4.3109 | 2.1106 | 0.86 | 0.1645 | <0.0001 | 1.032 | ||
| Branch (kg) | -4.1078 | 2.1884 | 0.86 | 0.1748 | <0.0001 | 1.036 | ||
| Needle (kg) | -3.3399 | 1.7995 | 0.73 | 0.2172 | <0.0001 | 1.056 | ||
| Aboveground (kg) | -3.1703 | 1.9378 | 0.89 | 0.1309 | <0.0001 | 1.020 | ||
| Root (kg) | -3.7581 | 2.0185 | 0.66 | 0.2509 | <0.0001 | 1.075 | ||
| Red pine | Stem wood (kg) | -4.0840 | 2.1108 | 0.83 | 0.1878 | <0.0001 | 1.041 | |
| Stem bark (kg) | -4.1892 | 1.9772 | 0.91 | 0.1242 | <0.0001 | 1.018 | ||
| Branch (kg) | -4.2092 | 2.1941 | 0.73 | 0.2608 | <0.0001 | 1.081 | ||
| Needle (kg) | -3.4831 | 1.6452 | 0.73 | 0.1941 | <0.0001 | 1.044 | ||
| Aboveground (kg) | -3.2812 | 1.9287 | 0.90 | 0.1283 | <0.0001 | 1.019 | ||
| Root (kg) | -3.6781 | 1.9577 | 0.77 | 0.1711 | 0.003 | 1.034 | ||
| D20 | Black pine | Stem wood (kg) | -4.6370 | 2.2534 | 0.72 | 0.2479 | <0.0001 | 1.073 |
| Stem bark (kg) | -4.7760 | 2.3297 | 0.84 | 0.1781 | <0.0001 | 1.037 | ||
| Branch (kg) | -4.6553 | 2.4635 | 0.87 | 0.1681 | <0.0001 | 1.033 | ||
| Needle (kg) | -3.7952 | 2.0294 | 0.73 | 0.2125 | <0.001 | 1.053 | ||
| Aboveground (kg) | -3.6380 | 2.1688 | 0.89 | 0.1305 | <0.001 | 1.020 | ||
| Root (kg) | -4.6538 | 2.5506 | 0.69 | 0.2398 | <0.0001 | 1.068 | ||
| Red pine | Stem wood (kg) | -4.4445 | 2.2164 | 0.81 | 0.1990 | <0.0001 | 1.047 | |
| Stem bark (kg) | -4.4313 | 2.0035 | 0.82 | 0.1726 | <0.0001 | 1.035 | ||
| Branch (kg) | -4.7166 | 2.4044 | 0.79 | 0.2363 | <0.0001 | 1.066 | ||
| Needle (kg) | -3.9173 | 1.8436 | 0.83 | 0.1597 | <0.0001 | 1.030 | ||
| Aboveground (kg) | -3.6734 | 2.0727 | 0.92 | 0.1146 | <0.0001 | 1.015 | ||
| Root (kg) | -3.8574 | 1.9264 | 0.66 | 0.2097 | 0.009 | 1.052 | ||
Mean biomass and C content among tree components in the sampled trees were not significantly different between the observed values and the predicted values using the DBH or D20 allometric equations except for N content in needles (Tab. 4). However, biomass and C content in black pine were generally highest in the D20 equations, followed by the DBH equations and the observed values, while the values for red pine were highest in the DBH equations, followed by the D20 equations and the observed values. In contrast to mean biomass and C content in the tree components, mean N content was highest in the observed values, followed by the DBH equations and the D20 equations except for the N content in branches or needles in red pine. In the sampled trees black pine components consistently exhibited more biomass, C and N content than did red pine (Tab. 4).
Tab. 4 - Mean observed and predicted (DBH and D20) values (± standard error) using allometric equations in black pine (n = 49) and red pine (n = 25). Different letters among observed and predicted values indicate significant differences after Tukey’s test (P < 0.05).
| Biomass or nutrient | Tree species | Obs/Pred values | Tree component (kg tree-1) | |||||
|---|---|---|---|---|---|---|---|---|
| Stem wood | Stem bark | Branch | Needle | Above ground | Root | |||
| Biomass | Black pine | Obs. | 151.4 ± 18.4 a | 16.7 ± 2.0 a | 37.9 ± 4.7 a | 16.9 ± 1.8 a | 222.8 ± 26.2 a | 62.9 ± 11.9 a |
| DBH | 171.1 ± 23.1 a | 17.4 ± 2.1 a | 41.7 ± 5.2 a | 18.4 ± 1.8 a | 235.2 ± 29.3 a | 64.4 ± 11.8 a | ||
| D20 | 183.7 ± 26.7 a | 17.9 ± 2.3 a | 42.5 ± 5.7 a | 18.5 ± 2.0 a | 262.6 ± 36.7 a | 66.1 ± 12.7 a | ||
| Red pine | Obs. | 72.3 ± 14.3 a | 9.9 ± 1.6 a | 15.5 ± 3.6 a | 4.3 ± 0.6 a | 102.1 ± 19.5 a | 14.4 ± 3.6 a | |
| DBH | 79.9 ± 15.7 a | 9.9 ± 1.4 a | 16.7 ± 3.1 a | 4.4 ± 0.6 a | 114.5 ± 23.9 a | 14.8 ± 3.1 a | ||
| D20 | 77.6 ± 14.4 a | 9.8 ± 1.3 a | 16.1 ± 2.9 a | 4.3 ± 0.6 a | 107.8 ± 19.2 a | 15.2 ± 3.5 a | ||
| Carbon | Black pine | Obs. | 71.8 ± 8.8 a | 8.0 ± 0.9 a | 18.3 ± 2.3 a | 8.4 ± 0.9 a | 106.4 ± 12.5 a | 28.1 ± 12.7 a |
| DBH | 79.9 ± 10.8 a | 8.2 ± 1.0 a | 19.6 ± 2.5 a | 8.8 ± 0.9 a | 109.7 ± 14.0 a | 28.7 ± 5.5 a | ||
| D20 | 84.8 ± 12.3 a | 8.4 ± 1.0 a | 20.0 ± 2.7 a | 8.9 ± 1.0 a | 122.1 ± 17.0 a | 29.3 ± 5.5 a | ||
| Red pine | Obs. | 34.3 ± 6.8 a | 4.9 ± 0.8 a | 7.5 ± 1.7 a | 2.1 ± 0.3 a | 48.9 ± 9.3 a | 6.5 ± 1.8 a | |
| DBH | 37.2 ± 7.4 a | 4.8 ± 0.7 a | 7.7 ± 1.5 a | 2.1 ± 0.3 a | 53.0 ± 11.1 a | 6.3 ± 1.3 a | ||
| D20 | 36.2 ± 6.8 a | 4.8 ± 0.7 a | 7.5 ± 1.3 a | 2.1 ± 0.3 a | 50.6 ± 9.1 a | 6.5 ± 1.6 a | ||
| Nitrogen | Black pine | Obs. | 0.043 ± 0.005 a | 0.037 ± 0.004 a | 0.079 ± 0.010 a | 0.133 ± 0.014 a | 0.291 ± 0.031 a | 0.104 ± 0.031 a |
| DBH | 0.030 ± 0.003 a | 0.032 ± 0.004 a | 0.067 ± 0.010 a | 0.109 ± 0.011 ab | 0.270 ± 0.029 a | 0.096 ± 0.016 a | ||
| D20 | 0.031 ± 0.004 a | 0.032 ± 0.009 a | 0.069 ± 0.009 a | 0.088 ± 0.012 b | 0.220 ± 0.028 a | 0.074 ± 0.013 a | ||
| Red pine | Obs. | 0.052 ± 0.014 a | 0.025 ± 0.005 a | 0.049 ± 0.012 a | 0.044 ± 0.006 a | 0.170 ± 0.034 a | 0.053 ± 0.011 a | |
| DBH | 0.039 ± 0.007 a | 0.021 ± 0.003 a | 0.034 ± 0.006 a | 0.036 ± 0.005 a | 0.162 ± 0.030 a | 0.046 ± 0.011 a | ||
| D20 | 0.038 ± 0.006 a | 0.019 ± 0.003 a | 0.036 ± 0.006 a | 0.056 ± 0.008 a | 0.149 ± 0.024 a | 0.043 ± 0.010 a | ||
Discussion
C and N concentration among tree components
Increased DBH values might be expected to entail higher C concentrations because of increase in lignin concentration within lignified wood tissues ([4]), but C concentrations in stem wood and roots showed no patterns with increasing DBH in both pine species. Similarly, another study reported that the C concentration of stem wood was unrelated to the diameter growth rates in Sequoiadendron giganteum, though it was related to the year when the wood was formed ([20]). Additionally, the C concentration of needles did not display any trend relative to DBH. Generally, the C concentration of needles was not related to needle age or relative height of the crown in Pinus pinaster ([4]), but the needle C concentration in black pine was affected significantly by the sampling month during the growing season ([16]). In this study, needle biomass in both pine species was collected in the same month, early August during the growing season. However, the reasons for the negative correlation between stem bark C of black pine and DBH are not clear, but variations in bark C concentration with DBH could be attributed to decreased polyphenolic compounds of conifer bark in trees with increased DBH ([4]). Although the stem bark C concentration in black pine and branch C concentration in red pine were related linearly to growth factors such as DBH, the inter- and intra-specific variation of the C concentration in the tree species are determined by the interaction of genetic and environmental factors ([4], [30], [21]). The mean values of the C concentration can be directly used to estimate the C content of both pine species regardless of different site or DBH because of no or only marginally significant correlations between DBH and C concentrations for the tree components in both pine species.
There was a clear influence of DBH on N concentrations in stem wood, stem bark and branches, which were negatively correlated with increasing DBH in black pine. Negative relationships between N concentration and increasing DBH are commonly observed in tree stems ([1], [5]). This may be explained by an increased proportion of wood in trees with larger DBH ([1], [5]) because the wood may have some dead parts, lacking living cells. The results suggest that the N content of woody components (stem wood, stem bark and branches) in black pine should be estimated with caution because of the variation of N concentration across site and DBH range. In contrast to the black pine, the N concentration in stem bark of red pine was positively correlated with increasing DBH. The N concentration in stem bark is generally affected by N availability in different sites ([3]).
There were no species-specific differences in mean C concentrations between both pine species except for stem bark. Genetic differences in biomass allocation among tree species might result in different mean C concentrations of stem bark, which was significantly higher in the red pine than in the black pine. For example, the greater mean C concentration of bark tissues in red pine is likely due to a difference in bark thickness and branch biomass compared to black pine. In this study, the mean bark biomass of the sampled red pines was 9.9 kg tree-1, whereas that of black pines was 16.7 kg tree -1 (Fig. 3). This indicates that thin-barked species might be more sensitive to changes of DBH because cellulose and sugar in the inner bark lead to a C enrichment with increased DBH ([4], [21]).
Mean C concentrations among tree components were highest in needles, lowest in roots, and slightly lower in stem bark and branches than in stem wood. High C concentrations in needles could be non-structural C compounds such as sugar and starch ([11]). C concentrations in stem wood were 47.3% for red pine and 47.4% for black pine. These values were similar to a global mean value of 47.5 ± 0.5% for wood C concentration ([29]). A factor of 50% has commonly been used for the C content of tree biomass ([12], [10]), but the C concentration in tree components such as stem wood, stem bark, branches and roots were significantly lower than that in the needles (50%) in this study. This result indicates that use of a component-specific C concentration to estimate the tree C content in both pine tree species is more appropriate than a fixed C conversion factor of 50%.
The mean N concentrations of stem wood, branches, needles and roots in red pine were significantly greater than those of the respective components in black pine. The N concentrations in tree components were significantly affected by tree species, as the N concentration among tree components are controlled by factors such as the combined effects of the available N status of the soil, tree growth, and climate factors ([3]). A high N concentration in red pine is likely due to an increased uptake of available N at sites with better soil quality, as tree species with high N availability in mineral soil tend to produce high N concentration in tree components ([25], [16]). Needle N concentration, which is an indicator well correlated to soil N status ([16]), was significantly higher in the red pine than in the black pine. In addition, some dilution occurred because of the greater needle biomass in black pine compared with red pine. The lowest concentrations of N were found in stem wood, whereas the high concentrations occurred in needles, branches, and stem bark. These results indicate that indirect methods to estimate the N content in a forest stand based on the average N concentration in the entire forest ecosystem could induce larger errors because of the large variation of N concentration among the tree components.
Biomass, C and N content for allometric equations
The species-specific allometric equations to estimate biomass, C and N content for tree components in the black pine and red pine stands were highly significant (P < 0.001) for both DBH and D20. The allometric equations of aboveground biomass, C and N content provided better fits among the tree component-specific allometric equations, ranging from 81 to 96% of the observed variation. However, the low values of adj. R2 for biomass, C and N content of needles in both pine species could be due to the large scatter in data for tree biomass collected from different locations over a large spatial scale.
In this study the slope coefficient (b) of the DBH equation to estimate the aboveground biomass of red pine varied from 1.6100 for needles to 2.4869 for stem wood, which was comparable to the values of 1.5669 for needles and 2.4998 for stem wood over the whole country ([23]). This suggests there has been little impact of PWD infestation on allometry equations for needle or stem wood. The slope values of the allometric equations for biomass or C contents were generally higher in the black pine than in the red pine, but the N content was similar or slightly higher in the red pine than in the black pine. This indicated that the biomass and C content of the components of trees with similar DBH or D20 were higher in the black pine than the red pine. In addition, considerable differences were observed in biomass, C and N content for stem wood from trees with similar DBH or D20 in both pine species. These differences could be attributed to the difference of stem density in the two pine species (black pine: 0.50 Mg m-3; red pine: 0.42 Mg m-3).
Although the predicted values were generally overestimating biomass and C content or underestimating N content as compared to the observed values, those measures in both pine species were not significantly different between the observed and the predicted values, except for needle N content of black pine. This result indicates that the biomass, C and N content equations developed in this study can provide a means for estimating these values in other stands. However, the equations available for large-scale landscape, such as disturbed forests, should be applied with caution because such sites might have variation in tree biomass and its allocation to tree components ([32]).
Conclusion
Allometric equations for estimating biomass, C and N content of black pine and red pine stands in PWD-disturbed forests were developed based on DBH and D20. The accuracy of estimates for the C and N content in PWD-disturbed forests has been improved by the measurement of C and N concentrations per tree components in the allometric equations, because the C and N concentrations vary between different tree species and among tree components. This study emphasizes the importance of separating tree species and tree components to estimate the C and N content by tree biomass in the PWD-disturbed forest ecosystems.
Acknowledgements
CK performed the experiment and wrote the paper, SJ and KL provided constructive suggestions on the study, and BY designed the experiments. We thank one anonymous referee and the subject editor for helpful reviews of the manuscript. This study was carried out with the support of Southern Forest Resource Research Center, National Institute of Forest Science, Korea.
References
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Department of Forest Resources, Gyeongnam National University of Science and Technology, Jinju 52725 (Korea)
Su Young Jung
Kwang Soo Lee
Southern Forest Resource Research Center, National Institute of Forest Science, Jinju 52817 (Korea)
Corresponding author
Paper Info
Citation
Kim C, Yoo BO, Jung SY, Lee KS (2017). Allometric equations to assess biomass, carbon and nitrogen content of black pine and red pine trees in southern Korea. iForest 10: 483-490. - doi: 10.3832/ifor2164-010
Academic Editor
Mike Perks
Paper history
Received: Jul 06, 2016
Accepted: Feb 22, 2017
First online: Apr 12, 2017
Publication Date: Apr 30, 2017
Publication Time: 1.63 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2017
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 41242
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 34861
Abstract Page Views: 2389
PDF Downloads: 3126
Citation/Reference Downloads: 18
XML Downloads: 848
Web Metrics
Days since publication: 2565
Overall contacts: 41242
Avg. contacts per week: 112.55
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Feb 2023)
Total number of cites (since 2017): 10
Average cites per year: 1.43
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Estimation of fuel loads and carbon stocks of forest floor in endemic Dalmatian black pine forests
vol. 13, pp. 382-388 (online: 01 September 2020)
Research Articles
Allometric relationships for volume and biomass for stone pine (Pinus pinea L.) in Italian coastal stands
vol. 6, pp. 331-335 (online: 29 August 2013)
Research Articles
Allometric equations to estimate above-ground biomass of small-diameter mixed tree species in secondary tropical forests
vol. 13, pp. 165-174 (online: 02 May 2020)
Research Articles
Equations for estimating belowground biomass of Silver Birch, Oak and Scots Pine in Germany
vol. 12, pp. 166-172 (online: 15 March 2019)
Research Articles
Effects of black locust and black pine on extremely degraded sites 60 years after afforestation - a case study of the Grdelica Gorge (southeastern Serbia)
vol. 9, pp. 235-243 (online: 22 August 2015)
Research Articles
Amount and distribution of coarse woody debris in pine ecosystems of north-western Spain, Russia and the United States
vol. 7, pp. 53-60 (online: 28 October 2013)
Research Articles
Effects of tree species, stand age and land-use change on soil carbon and nitrogen stock rates in northwestern Turkey
vol. 9, pp. 165-170 (online: 18 June 2015)
Technical Notes
Effect of tree age on chemical compounds of ancient Anatolian black pine (Pinus nigra subsp. pallasiana) needles in Northwest Turkey
vol. 11, pp. 406-410 (online: 15 May 2018)
Research Articles
Analyzing regression models and multi-layer artificial neural network models for estimating taper and tree volume in Crimean pine forests
vol. 17, pp. 36-44 (online: 28 February 2024)
Research Articles
Scots pine’s capacity to adapt to climate change in hemi-boreal forests in relation to dominating tree increment and site condition
vol. 14, pp. 473-482 (online: 18 October 2021)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword