Forest litter as the mulch improving growth and ectomycorrhizal diversity of bare-root Scots pine (Pinus sylvestris) seedlings
iForest - Biogeosciences and Forestry, Volume 8, Issue 4, Pages 394-400 (2014)
doi: https://doi.org/10.3832/ifor1083-008
Published: Aug 20, 2014 - Copyright © 2014 SISEF
Research Articles
Abstract
In this paper, we report the influence of pine, oak and spruce forest litter on the growth and ectomycorrhizal (ECM) formation of Scots pine seedlings after the first growing season in a bare-root forest nursery. The mixture of collected forest litters and humus were used to obtain a 20-cm mulching layer on the prepared seedbeds. The concentrations of all nutrients and the C/N ratio of growth media were significantly higher in forest litter treatments than in negative control represented by mineral soil without litter. Addition of each forest litter type significantly enhanced pine seedling height and root-collar diameter compared to negative control. A significant positive influence on dry mass of stem, needles, roots and total dry mass of the seedling has been found only for pine litter. Based on molecular identification, seven ECM fungal taxa (Wilcoxina mikolae, Suillus luteus, Cenococcum geophilum, Meliniomyces bicolor, Laccaria laccata, unidentified Atheliaceae, unidentified Ascomycetes) were distinguished in the observed mycorrhizal communities. Each forest litter type significantly increased the total number of mycorrhizal tips and ECM fungal diversity compared to the control soil. However, results showed a lack of significant differences in species composition and relative abundance of ECM fungi between different litter types. Such result suggests that forest litter has not been a key source of inoculum for tested fungal species, as root systems of all pine seedlings from different litter types were dominated by a few nursery- adapted ECM fungi, probably originating from natural air-borne inoculum. Our data rather indicate that forest litter considerably improves environmental conditions for development of ECM fungi previously present in the nursery soil. Therefore, any of the forest litter types used in our studies may be able to promote planting stock quality on a small scale in the nursery phase.
Keywords
Introduction
Scots pine (Pinus sylvestris L.) is a broadly distributed forest tree species relevant in terms of planted areas and harvest yields ([40]). Scots pine grows only naturally in Lithuania ([13]) and represents approximately 37% of the forest area ([33]). Lithuania produces close to 80 million tree seedlings per year for reforestation and afforestation of abandoned agricultural lands, and approximately 20% of these are Scots pine ([32]). The entire seedling stock in Lithuania is produced in bare-root forest nurseries. The mass production of tree seedlings in bare-root nurseries drastically reduces the soil organic matter content without the possibility of its natural regeneration ([20]). The decline of organic matter content in nursery soil often leads to decreased microbiological activity and negatively influences the diversity of ectomycorrhizal (ECM) fungi ([29]). ECM fungi are a component of all forest trees and play a significant role in the uptake of soil nutrients and water, and inhibit the negative influence of soil borne pathogens and abiotic stresses ([46]). Deficiency of mycorrhizae or low species richness of ECM fungi on the root systems of tree seedlings in forest nurseries is one of the most common causes of poor seedling establishment and growth after transplanting ([2], [38], [15]). In contrast, high diversity of ECM fungi in bare-root seedlings production improves their growth and survival, and as a consequence the use of mineral fertilizers may be significantly reduced ([27]).
Forest litter is known as a rich source of soil organic matter and nutrients ([26], [19], [18]), and may significantly improve the physical characteristics of the soil ([5], [21]). The use of forest litter in nursery management practices may also revitalize the soil by the input of thousands of individual living organisms which decompose organic matter and facilitate seedling competition for nutrients ([41], [35]). The investigations performed by the Dunemann scheme indicated a better germination, growth and survival of conifer seedlings in spruce litter than in mineral soil ([23]). However, data regarding the effect of litter addition on ECM colonization and community structure are inconsistent. Both positive and negative effects of forest litter amendment have been observed in different experiments ([8], [11], [3]).
In our previous experiment, natural forest litter cover on the surface of the nursery bed soil considerably improved two years old Scots pine seedling survival and positively affected their ECM communities ([3]). However, in present forestry practice the use of two years old seedlings tends to be rare and the use of one-year-old seedlings, especially on dry sites, is becoming increasingly frequent ([6]). Nonetheless, information about the effectiveness of different forest litter types on one year old planting stock quality is limited to only laboratory studies with tree species other than Scots pine ([34]).
The aim of the present investigation was to evaluate how different forest litter types (pine, oak and spruce) affect growth and ECM formation of Scots pine seedlings after the first growing season in a bare-root forest nursery. We hypothesize that each of the forest litter type will differently affect: (i) the tested growth parameters of the seedlings; and (ii) quantitative and qualitative structure of ECM fungal community compared with nursery soil without litter amendments.
Materials and methods
Litter collection and seedling material
In March 2010, forest litter was collected in healthy, natural oak, pine and spruce stands of similar age, where natural seedling regeneration was abundant. The 90-years-old Quercus robur-dominated stand was classified as an aegopodiosa type with soil characterized as Eutric Planosols ([17]). To get a growth medium, litter (2 cm deep) on the top layer of the soil and humus horizon (12 cm deep), both taken from the same spot, was thoroughly mixed. The 80-years-old Scots pine-dominated stand was classified as a nemoral type with Vaccinium myrtillus in the understory and soil characterized as Albic Arenosols ([17]). The humus horizon was thin and spread to a uniform depth of 4 cm. The 70-years-old Norway spruce-dominated stand was classified as an oxalidosa type with soil characterized as Haplic Arenosols ([17]). Litter (3 cm deep) on the soil top layer was mixed with a 10-cm-deep humus horizon. The mixture of litter and humus has been stored in a forest as a heap and protected from direct sunlight with twigs of trees until further processing (ca. 3 weeks). From each growth medium, fine and coarse roots have been carefully picked up.
In April 2010, the mixture of collected forest litters and humus were transferred to the experiment site located in the bare-root nursery of the Vilnius University Botanical Garden (54° 43′ N, 25° 24′ E) and used as mulching layer (20 cm deep, approx. 40 kg m-2), allowing the largest part of the root system to grow up in the litter. A mixture of vermiculite and perlite (as chemically neutral substances) at a volume of 15% and 10%, respectively, was used to maintain the appropriate microclimate (moisture content, temperature and porosity) for plant growing in each growth medium (with and without litter amendments). The experimental design was divided into five complete blocks with four plots per oak litter, pine litter, spruce litter and control (mineral nursery soil), randomly allocated in each block. Each treatment variant was spaced approximately 1 m. The pine seeds for the trial originated from the local provenance of Labanoras (55° 16′ N, 25° 50′ E). The seeds of Scots pine required no stratification and a 3x3 cm sowing stencil was used. Two seeds were sown directly by hand into a single hole at the end of April 2010. Germinants were then thinned to one per hole. During the first growing season seedlings were manually weeded and irrigated, but not fertilized.
After the first year of growth under nursery conditions, 40 randomly selected seedlings were harvested from each block (50 seedlings per growth medium, 200 seedlings in total). The excavated seedlings with their surrounding soil were placed into labelled plastic bags and transported to the laboratory for subsequent mycorrhizal assessment and shoot height, root collar diameters and biomass measurement. Seedling biomass was determined after drying the plant material at 70 °C for 48 h.
Chemical analysis
For each plot, five soil cores were randomly sampled from the upper 15 cm of growth medium, where bare-root seedlings grew. Soil samples from growth medium were mixed, dried at 40 °C, and soil carbon content and the nutrient composition (N, P, K, Mg and Ca) were determined in four repetitions. A complete description of the growth medium analysis is presented in our earlier paper ([3]).
Mycorrhizal evaluation
One-year-old seedlings with surrounding soil were stored at -20 °C until processing. Frozen seedlings were processed in sets of ten to minimize the impact of thawing on fine root observations. The root systems of seedlings and single roots were extracted from the soil using a 0.5 mm mesh-sieve under tap water. The clean roots were cut into approximately 2-cm long fragments and placed in a Petri dish filled with water. Only roots with a diameter ≤ 2 mm were observed. The assessment with the aid of a stereomicroscope Zeiss Stemi 2000-C (Carl Zeiss, Germany; 10-60× magnification) was performed by counting all ECM root tips (85-249 root tips on average were sampled from each growth medium). Live roots (identified as swollen, without root hair or covered by fungal mantles) were considered ECM-colonized roots. Mycorrhizas were classified into morphotypes based on morphological characteristics (ramification system; colour, shape, texture and thickness of the mantle; presence and organization of the emanating hyphae; rhizomorphs; and other elements) according to Agerer ([1]) and database from our Laboratory of Mycorrhizal Research at the Institute of Dendrology. In doubtful cases (e.g., thin or non-evident mantle) we made a cross-section of potential mycorrhizas to determine the presence of a Hartig net and a mantle under the compound microscope Axio Imager A1 (Carl Zeiss, Germany) at 400-1000× magnification. The number of live mycorrhizas of each morphotype was recorded separately for each seedling. The relative abundance of each morphotype (number of root tips of each morphotype/total number of mycorrhizas) was calculated for each seedling.
The representative samples of three to four root tips for each morphotype from each growth medium were stored in a 2% cetyltrimethylammonium bromide buffer for molecular identification. No attempt was made to relate morphotypes between growth media until molecular analysis was complete; thus, each morphotype was treated separately in the molecular identification and pooled for abundance calculation only after the molecular analysis indicated that morphotypes were identical and belonging to the same taxa. The full methods used for molecular identification of mycorrhizas are reported in Pietras et al. ([39]). Fungal symbionts were identified using polymerase chain reaction (PCR) amplification of the internal transcribed spacer of rDNA (ITS rDNA) using the ITS-1F and ITS-4 primers ([49]). The selected samples with ECM root tips were then sequenced, and in total 98 sequences were obtained. Sequencing of the PCR products was performed with a CEQ 20000XL automatic sequencer using the same set of primers. Consensus sequences were constructed, with manual editing of ambiguous readings, and were compared to sequences published in the GenBank and UNITE databases ([28]) using the BLAST tool. Species-level identification of mycorrhizae was defined as sharing ≥ 98% of the ITS region sequence identity with the reference sequence. The best representative of each unique ITS sequence type was deposited in NCBI GenBank with the accession numbers KJ596425-KJ596431.
Statistical analysis
The chemical composition of soils, growth and biomass parameters, the amount of mycorrhizal tips and the mean fungal species richness per seedling were analysed by two-way analysis of variance, with the growth medium as a fixed factor and the block as the random factor. Normal distribution of data and homogeneity of variance were tested through the Shapiro-Wilk’s and Levene’s test, respectively. Post-hoc comparisons of means among the examined growth media were made using Tukey’s honestly significant difference (HSD) test (α = 0.05). As for relative abundances, no homogeneity of variance was found; therefore, differences in the relative abundance of morphotypes between growth media were tested using the Kruskal-Wallis and Mann-Whitney U tests. Prior to analysis, the relative abundance of mycorrhizal morphotypes was arcsine square root transformed. Computations were performed using the statistical software package Statistica 5.5 ([47]). Estimates of the true species richness (bootstrap and Chao 2) were calculated using the EstimateS program version 8.2.0 ([9]). Shannon’s diversity indices for the ECM assemblages of Scots pine seedlings were carried out using the PAST1.89 software ([22]) and were based on square root transformed data.
Multiple regressions were applied to assess the relationships between nutrient composition and the pH of growth media and the total number of mycorrhizal tips, species richness of ECM fungi, and growth parameters of the seedlings. Stepwise multiple regression with backward elimination was used after screening the potential independent variables (total number of mycorrhizal tips, species richness of ECM fungi) for significant autocorrelation. Dependent variables were log-transformed to improve their linearity. The total number of mycorrhizal tips and species richness of ECM fungi were log(n+1) transformed.
Results
The nutrient content and pH of growth media after one year of seedling growth are summarized in Tab. 1. Significant differences between forest litter types and control soil were found for all nutrient concentrations, C/N ratio and pHKCI of growth media. Concentrations of nutrients and the C/N ratio were highest in spruce litter. Mean values of soil total N, NH4+, NO3-, K, Ca, Mg, C and the C/N ratio were intermediate in oak and pine litters, and lowest in the control soil. The concentration of soil P was intermediate in the control soil and oak litter, and lowest in pine litter. The pHKCI was highest for the control soil, intermediate for pine and oak litters, and lowest for the spruce litter. The block effect was not significant (two-way ANOVA, P = 0.859).
Tab. 1 - Nutrient composition and pH of growth media at harvest of Pinus sylvestris L. seedlings after one year of growth in a bare-root nursery with mineral soil and growth media with pine litter, oak litter and spruce litter (values are mean ± SE, n=16). Within a row, values with different letters are significantly different after Tukey’s test (P < 0.05).
| Parameter | Growth medium | P>F | |||
|---|---|---|---|---|---|
| Pine litter | Oak litter | Spruce litter | Mineral nursery soil (control) |
||
| Ntotal (%) | 0.12 ± 0.01 c | 0.22 ± 0.01 b | 0.31 ± 0.02 a | 0.06 ± 0.01 d | 0.0001 |
| C (%) | 1.88 ± 0.01 c | 3.77 ± 0.03 b | 6.64 ± 0.06 a | 0.70 ± 0.02 d | 0.0001 |
| C/N | 16.00 ± 0.91 bc | 17.50 ± 0.57 b | 22.30 ± 1.23 a | 12.40 ± 1.41 c | 0.0001 |
| NH4-N (mg kg-1) | 0.28 ± 0.01 c | 0.86 ± 0.01 b | 1.77 ± 0.01 a | 0.12 ± 0.01 d | 0.0001 |
| N-NO3(mg kg-1) | 0.47 ± 0.01 c | 1.57 ± 0.01 b | 4.12 ± 0.03 a | 0.24 ± 0.02 d | 0.0001 |
| P (mg kg-1) | 1.28 ± 0.01 d | 1.79 ± 0.02 c | 10.30 ± 0.04 a | 2.08 ± 0.04 b | 0.0001 |
| K (mg kg-1) | 15.70 ± 0.08 c | 20.00 ± 0.13 b | 62.40 ± 0.37 a | 9.88 ± 0.07 d | 0.0001 |
| Ca (mg kg-1) | 10.70 ± 0.05 c | 14.70 ± 0.08 b | 39.30 ± 0.17 a | 6.62 ± 0.08 d | 0.0001 |
| Mg (mg kg-1) | 4.31 ± 0.05 c | 5.69 ± 0.05 b | 15.90 ± 0.08 a | 2.73 ± 0.03 d | 0.0001 |
| pHKCI | 4.81 ± 0.04 b | 4.65 ± 0.04 c | 3.93 ± 0.03 d | 5.66 ± 0.01 a | 0.0001 |
The statistical analysis of growth parameters revealed an overall significant differences among growth media (two-way ANOVA, F=6.477, P=0.0001), with the block having no significant influence (two-way ANOVA, F=0.388, P=0.819 - Tab. 2). The height and root-collar diameter of the seedlings from all litter types were significantly higher than seedlings from the control mineral soil. Only pine litter significantly increased standing biomass of seedlings (dry weight of total seedling, stem and needles; post-hoc Tukey’s HSD test - Tab. 2).
Tab. 2 - Growth parameters of Pinus sylvestris L. seedlings after the first growing season in a nursery with mineral soil and growth media with pine litter, oak litter and spruce litter (values are mean ± SE, n=50). Within a row, values with different letters are significantly different after Tukey’s test (P < 0.05).
| Parameter | Growth medium | P>F | |||
|---|---|---|---|---|---|
| Pine litter | Oak litter | Spruce litter | Mineral nursery soil (control) |
||
| Seedling height (cm) |
9.00 ± 0.39 a | 7.90 ± 0.46 a | 8.50 ± 0.36 a | 5.10 ± 0.30 b | 0.0001 |
| Root collar diameter (mm) | 1.70 ± 0.04 a | 1.60 ± 0.07 a | 1.60 ± 0.05 a | 1.40 ± 0.06 b | 0.0001 |
| Stem dry weight (g) |
0.09 ± 0.01 a | 0.07 ± 0.01 ab | 0.08 ± 0.01 ab | 0.04 ± 0.00 b | 0.001 |
| Needles dry weight (g) | 0.29 ± 0.04 a | 0.17 ± 0.03 b | 0.16 ± 0.02 b | 0.09 ± 0.01 b | 0.0001 |
| Roots dry weight (g) |
0.17 ± 0.03 a | 0.10 ± 0.02 b | 0.10 ± 0.01 b | 0.06 ± 0.00 b | 0.0001 |
| Total seedling dry weight (g) | 0.55 ± 0.07 a | 0.33 ± 0.05 b | 0.32 ± 0.03 b | 0.18 ± 0.02 b | 0.0001 |
| Ratio of above-/ belowground weight |
2.30 ± 0.09 | 2.40 ± 0.16 | 2.30 ± 0.15 | 2.20 ± 0.10 | 0.723 |
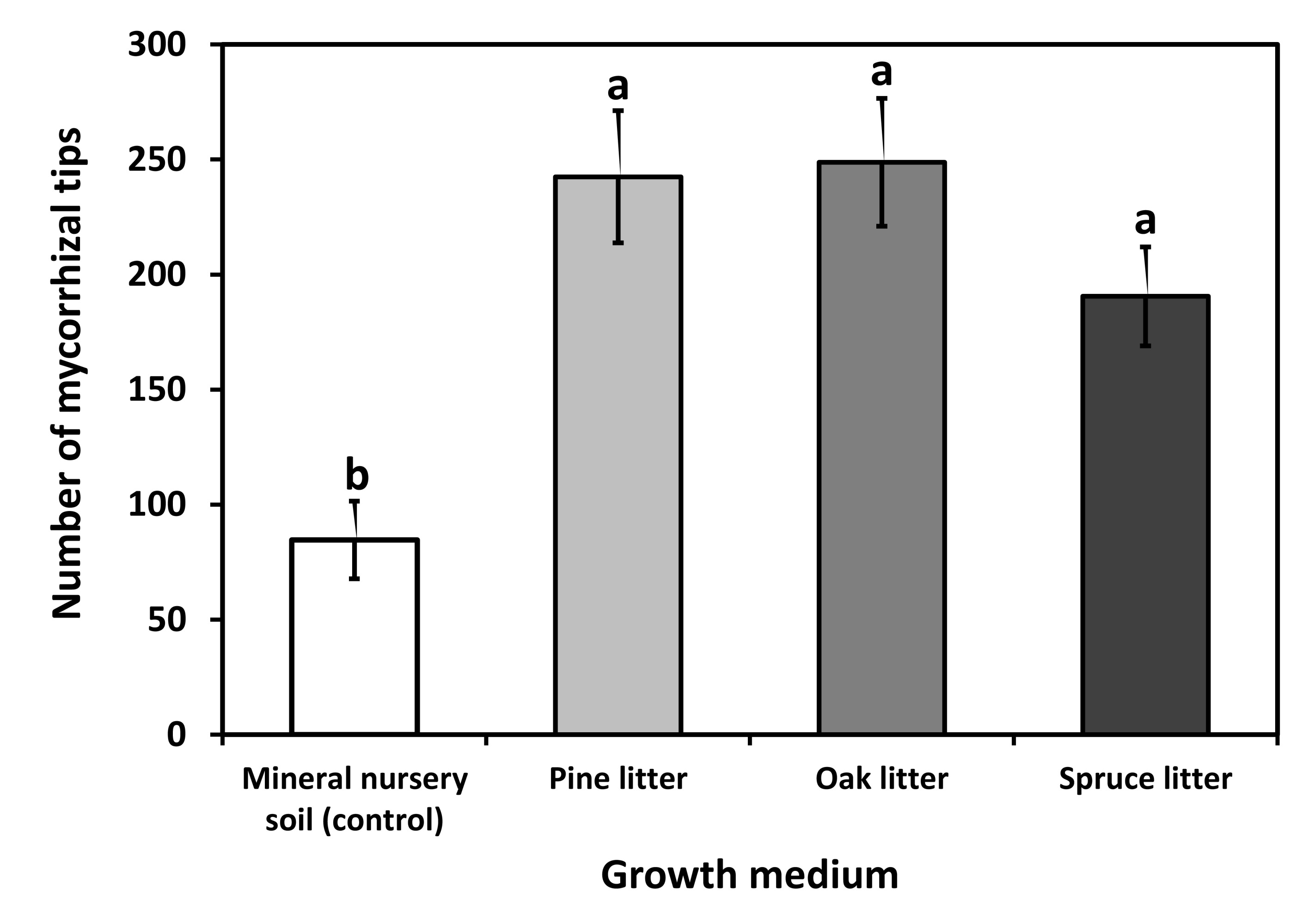
In all growth media, the rate of mycorrhizal colonization of Scots pine seedlings was high and neared 100%. The total number of mycorrhizal tips was significantly higher (two-way ANOVA, P=0.0003) for the litter variants than for the control (Fig. 1). The block effect was not significant (two-way ANOVA, P=0.713). On seedlings from all growth media, seven ECM fungal taxa (Wilcoxina mikolae, Suillus luteus, Cenococcum geophilum, Meliniomyces bicolor, Laccaria laccata, unidentified Atheliaceae, unidentified Ascomycetes) have been found: two taxa in the control mineral soil, seven in the pine and oak litter types and six in spruce litter type (Tab. 3). The mean fungal species richness per seedling ranged from 1.1 to 3.0, with significant differences among the litter types and the control soil (two-way ANOVA, P=0.001). The block effect was not significant (two-way ANOVA, P=0.534). According to the bootstrap species richness estimates, seedlings from oak litter type were estimated to be the most rich in ECM fungi (8.02 taxa), with control seedlings showing the lowest species richness estimates (2.35 taxa - Tab. 3).
Fig. 1 - Total number of mycorrhizal tips on Scots pine seedlings after the first growing season in a bare-root nursery with mineral soil and growth media with pine litter, oak litter and spruce litter. Each bar shows the mean for 50 replicates ± standard error (SE). Different letters indicate significant differences between growth media (P<0.05, Tukey’s test).
Tab. 3 - Molecular identification, relative abundance, observed total and mean species richness (± SE), and estimated species richness of ECM fungi on the roots of Pinus sylvestris L. seedlings after one year of growth in a bare-root nursery with mineral soil and growth media with pine litter, oak litter and spruce litter (values are mean ± SE, n=50). Within a row, values with different letters are significantly different (P<0.05; Mann-Whitney U test for relative abundance and Tukey’s test for mean species richness per seedling and Shannon diversity index).
| Group | Identifi-cation | Accession | Closest match |
Identity (%) |
Relative abundance (%) - Growth medium | |||
|---|---|---|---|---|---|---|---|---|
| Pine litter |
Oak litter | Spruce litter | Mineral nursery soil (control) |
|||||
| Ascomycota | Wilcoxina mikolae | KJ596425 | Wilcoxina mikolae (JQ310818) |
99 | 84.8 ± 3.6 | 87.1 ± 3.2 | 89.1 ± 2.9 | 96.3 ± 3.7 |
| Meliniomyces bicolor | KJ596426 | Meliniomyces bicolor (HM190124) |
98 | 8.5 ± 2.4 | 8.8 ± 2.4 | 5.9 ± 2.6 | - | |
|
Cenococcum
geophilum |
KJ596427 | Cenococcum geophilum (HM189727) | 99 | 1.9 ± 0.6 | 0.9 ± 0.7 | 2.2 ± 0.8 | - | |
| Ascomycetes | KJ596428 | unidentified Ascomycetes (JN172989) | 89 | 1.7 ± 1.2 | 0.5 ± 0.5 | 1.2 ± 1.0 | - | |
| Basidiomycota | Atheliaceae | KJ596429 | Amphinema sp. (JN943925) |
91 | 0.5 ± 0.6 | 2.4 ± 1.8 | - | 3.7 ± 3.7 |
|
Suillus
luteus |
KJ596430 | Suillus luteus (UDB000930) |
98 | 2.4 ± 1.7 | 0.3 ± 0.2 | 0.2 ± 0.2 | - | |
|
Laccaria
laccata |
KJ596431 | Laccaria laccata (UDB000106) |
98 | 0.1 ± 0.1 | 0.1 ± 0.1 | 1.5 ± 0.8 | - | |
| Statistics | Observed species richness | - | 7 | 7 | 6 | 2 | ||
| Mean species richness per seedling | - | 2.9 ± 0.4 a | 2.8 ± 0.4 a | 3.0 ± 0.3 a | 1.1 ± 0.1 b | |||
| Chao 2 | - | 8 | 7.67 | 6.5 | 2 | |||
| Bootstrap | - | 7.91 | 8.02 | 6.46 | 2.35 | |||
| Shannon diversity index (H′) | - | 0.47 ± 0.10 a | 0.39 ± 0.09 a | 0.37 ± 0.07 a | 0.07 ± 0.07 b | |||
W. mikolae was the most common taxon and dominated ECM communities on pine seedlings from all growth media. The mycorrhizas formed by M. bicolor, C. geophilum, unidentified Ascomycetes, S. luteus and L. laccata were present on pine seedlings grown in all forest litter types. Mycorrhizas formed by Atheliaceae sp. were detected on seedlings from the control soil and oak and pine litters. No significant differences in ECM abundance between the growth media were found (Tab. 3). The Shannon’s diversity indices for the ECM assemblages of Scots pine seedlings ranged from 0.07 to 0.47 and revealed significant differences between litter types and the control soil.
With regard to seedling height, results from the stepwise multiple regression analysis indicated significant components of beta for the total number of mycorrhizal tips and species richness of ECM fungi. The rest of the tested growth parameters (root collar diameter and stem, needles and roots dry weight) showed significant positive interactions with total number of mycorrhizal tips, but not with species richness of ECM fungi. When carbon, mineral content and pH of growing medium were entered into the stepwise multiple regression model, total nitrogen content explained a significant amount of variation both in the total number of mycorrhizal root tips and the ECM species richness. Carbon, nitrogen, potassium, calcium and magnesium had a positive impact on species richness of ECM fungi. Moreover, pH of the growth medium showed significant negative interactions with the species richness of ECM fungi (Tab. 4).
Tab. 4 - Results from the stepwise multiple regression analysis between the total number of mycorrhizal tips, species richness of ECM fungi, nutrient composition and the pH of growth media and growth parameters of Pinus sylvestris L. seedlings.
| Growth parameter, nutrient | Total number of mycorrhizal tips |
Species richness of ECM fungi |
||
|---|---|---|---|---|
| β | P | β | P | |
| Seedling height | 0.43 | 0.005 | 0.33 | 0.025 |
| Root collar diameter | 0.42 | 0.016 | 0.11 | 0.508 |
| Stem dry weight | 0.46 | 0.009 | 0.01 | 0.969 |
| Needles dry weight | 0.46 | 0.009 | 0.01 | 0.943 |
| Roots dry weight | 0.47 | 0.008 | -0.02 | 0.912 |
| C | 0.28 | 0.051 | 0.49 | 0.001 |
| Ntotal | 0.31 | 0.044 | 0.41 | 0.009 |
| P | -0.14 | 0.469 | 0.26 | 0.172 |
| K | 0.12 | 0.480 | 0.45 | 0.010 |
| Ca | 0.14 | 0.399 | 0.46 | 0.006 |
| Mg | 0.12 | 0.448 | 0.46 | 0.006 |
| pHKCI | -0.23 | 0.125 | -0.49 | 0.002 |
Discussion and conclusions
Bare-root forest nurseries can be considered as a simplified ecosystem with a disturbed cycle of organic matter. Long-term use of nursery soil and organic matter reduction may have important implications for the physical, biological and chemical properties of the nursery soil ([20]) and thus negatively result in growth responses of the seedlings. Therefore, mulching the nursery seedbeds with forest litter may be an attractive way of supplementing nursery soil with organic matter. The results of the present investigation provide evidence that forest litter amendment can significantly influence some growth parameters and ECM formation of Scots pine seedlings even after a single growing season in a bare-root forest nursery. Improved heights and stem diameters of the seedlings from all litter types in respect to the control trees (Tab. 2) supported our first hypothesis and was consistent with data reported by Bakuzis ([4]), suggesting that tree species seedlings rapidly develop a large root system and undergo a great height increment in undecomposed or partly decomposed forest litter. Forest floor as an appropriate rooting medium for planted tree seedlings has also been noted by other authors ([5], [21], [36], [48]). Also in our previous study performed on two-years-old Scots pine seedlings, height and survival of seedlings were considerably improved by natural pine and oak litter cover on the surface of the nursery bed soil ([3]). Although the foliar nutrient composition of the pine seedlings in our study was not examined, we presumed that the enhanced seedling growth and higher biomass parameters (in pine litter only) might be (at least partly) related to the nutritional effect of the tested forest litters. Forest litter with the organic horizon is an important reservoir of organic matter and mineral nutrients, especially nitrogen ([26]). Different litter types and the environmental conditions in the litter layer affect the decomposition rate and the microbial activity of litter, and thus regulate the rate of nutrient release from the organic matter ([16], [44], [10]). Consequently different litter types may selectively promote seedling growth responses ([8]), as found in our experiment. Moreover, each forest litter type (pine, oak and spruce) increased the total number of mycorrhizal tips and ECM fungal diversity. The uptake of nutrients from decomposing litter by ECM fungi associated with seedling roots could further promotes seedling growth ([8]). The presence of a high number of mycorrhizal tips and higher ECM fungal diversity in litter variants facilitate the access to an organic nutrient source and improve the seedling development, since mycorrhizal fungi often produce extracellular enzymes ([30]) capable of releasing nutrients from organic matter that are normally unavailable to plants. Baxter & Dighton ([7]) found a positive influence of increased species richness of ECM fungi on the growth and nutrient uptake of Pinus rigida seedlings after one growing season in field soils.
In the observed mycorrhizal communities, we distinguish seven ECM fungal taxa by morphotyping of 38 320 seedling root tips and sequencing of 98 mycorrhizas. Based on our sampling effort, the estimated numbers of species reach higher values by selected estimators for seedlings grown in litter variants than in the control soil. These values indicate that our estimated sampling completeness (percent of observed species) constitutes approximately 87% of estimated species richness. Similar sampling completeness is noted in many other studies as reported by Dickie ([14]). All examined mycorrhizas are ubiquitous and the identity of EM fungi composing these morphotypes was established by molecular methods in bare-root forest nurseries in a previous study ([42], [24], [3], [31]). Our results indicate that the widespread W. mikolae mycorrhizae at the initial stages of seedling growth is better adapted to nursery conditions than other ECM fungal taxa, such as M. bicolor, C. geophilum, Atheliaceae sp., S. luteus, L. laccata, and unidentified Ascomycetes. This is consistent with numerous studies suggesting that E-strain fungi are the most common symbionts not only in conifer nurseries and degraded sites, but also in northern boreal forests ([12], [37], [43], [31]).
Contrary to second hypothesis, different litter types used in our experiment did not differentiated neither species composition nor relative abundance of ECM fungi. In previous experiments with seedlings from different pine species, an effect of different forest litter amendment was rather observed on the relative proportions of mycorrhizal symbionts than on their species composition ([10], [25], [3]). Such results suggest that the forest litter has not been a source of inoculum for the identified fungal species, as root systems of pine seedlings from all litter types were colonized by a few nursery-adapted ECM fungi ([42], [24], [3], [31]), probably originating from a natural air-borne inoculum. Our data rather indicate that forest litter considerably improves environmental conditions for development of ECM fungi previously present in the nursery soil. We presume that forest litter amendment has constituted better microclimatic conditions for the maintenance of a high number of mycorrhizal tips and ECM fungal diversity in litter growth media than in the control without any supplement.
We conclude that the use of forest litter as a mulch improves the growth parameters and performance of ECM formation on Scots pine seedlings after the first growing season in a bare-root forest nursery. Therefore, any of the forest litter types used in our studies may be able to promote planting stock quality on a small scale in the nursery phase. It is likely that seedlings may benefit from litter growth media for their successful outplanting from nursery to dry field sites. Drought resistance of many ECM fungi is obviously of advantage for nursery seedlings following outplanting ([45]). Further experiments are in progress to examine growth performance and survival of such seedlings in the field.
Acknowledgements
We are grateful to Halina Narozna and Maria Wójkiewicz (Institute of Dendrology, Polish Academy of Sciences) for technical assistance. This research was funded by the EU Commission and Government of the Republic of Lithuania (Project No 1PM-PV-09-1-001461).
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Audrius Skridaila
Izolda Pašakinskiene
Botanical Garden of Vilnius University, 43 Kairenu Str., LT-10239 Vilnius (Lithuania)
Tomasz Leski
Institute of Dendrology, Polish Academy of Sciences, 5 Parkowa Str., 62-035 Kórnik (Poland)
Department of Sylviculture, Aleksandras Stulginskis University, 11 Studentu Str., Akademija, LT-53361 Kaunas district (Lithuania)
Corresponding author
Paper Info
Citation
Aučina A, Rudawska M, Leski T, Skridaila A, Pašakinskiene I, Riepšas E (2014). Forest litter as the mulch improving growth and ectomycorrhizal diversity of bare-root Scots pine (Pinus sylvestris) seedlings. iForest 8: 394-400. - doi: 10.3832/ifor1083-008
Academic Editor
Silvano Fares
Paper history
Received: Jul 24, 2013
Accepted: Apr 29, 2014
First online: Aug 20, 2014
Publication Date: Aug 02, 2015
Publication Time: 3.77 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2014
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 43979
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 36950
Abstract Page Views: 2127
PDF Downloads: 3783
Citation/Reference Downloads: 26
XML Downloads: 1093
Web Metrics
Days since publication: 3537
Overall contacts: 43979
Avg. contacts per week: 87.04
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Feb 2023)
Total number of cites (since 2015): 8
Average cites per year: 0.89
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Conservation of Betula oycoviensis, an endangered rare taxon, using vegetative propagation methods
vol. 13, pp. 107-113 (online: 23 March 2020)
Research Articles
Controlled-release fertilizers combined with Pseudomonas fluorescens rhizobacteria inoculum improve growth in Pinus halepensis seedlings
vol. 8, pp. 12-18 (online: 12 May 2014)
Research Articles
Substrates and nutrient addition rates affect morphology and physiology of Pinus leiophylla seedlings in the nursery stage
vol. 10, pp. 115-120 (online: 02 October 2016)
Short Communications
The effects of salicylic acid, oxalic acid and chitosan on damping-off control and growth in Scots pine in a forest nursery
vol. 13, pp. 441-446 (online: 24 September 2020)
Research Articles
Combined pre-hardening and fall fertilization facilitates N storage and field performance of Pinus tabulaeformis seedlings
vol. 9, pp. 483-489 (online: 07 January 2016)
Research Articles
The combined effects of Pseudomonas fluorescens CECT 844 and the black truffle co-inoculation on Pinus nigra seedlings
vol. 8, pp. 624-630 (online: 08 January 2015)
Research Articles
The effect of silver and copper nanoparticles on the growth and mycorrhizal colonisation of Scots pine (Pinus sylvestris L.) in a container nursery experiment
vol. 11, pp. 690-697 (online: 23 October 2018)
Research Articles
Using field and nursery treatments to establish Quercus suber seedlings in Mediterranean degraded shrubland
vol. 13, pp. 114-123 (online: 26 March 2020)
Research Articles
Linking nursery nutritional status and water availability post-planting under intense summer drought: the case of a South American Mediterranean tree species
vol. 9, pp. 758-765 (online: 03 June 2016)
Research Articles
Fertilisation of Quercus seedlings inoculated with Tuber melanosporum: effects on growth and mycorrhization of two host species and two inoculation methods
vol. 10, pp. 267-272 (online: 13 December 2016)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword