Assessing the habitat conservation status by soil parameters and plant ecoindicators
iForest - Biogeosciences and Forestry, Volume 7, Issue 3, Pages 170-177 (2014)
doi: https://doi.org/10.3832/ifor0963-007
Published: Feb 14, 2014 - Copyright © 2014 SISEF
Research Articles
Abstract
The aim of this study is to evaluate the conservation status of a Natural Reserve through an integrated approach analysing simultaneously soils, lithotypes, land forms, edaphic parameters and plant species. In focusing the relationships between soil and vegetation, plant ecoindicators, expressed by i) the Ellenberg bioindication model and by ii) the Hemeroby Index, and soil measured parameters were utilized.Vegetation and soil data have been collected simultaneously through thirty vegetation relevés and soil profiles. Cluster analysis, performed on matrix 12 variables/ 30 relevés allowed a division into two main clusters, each one divided into sub-clusters, distinguished by floristic composition and soil characteristics. The clusters were markedly discriminated by soil Available Water Capacity (AWC). Canonical Correspondence Analysis (CCA), performed on variables and species matrices, allowed to discriminate two main habitats: i) a core habitat represented by patches of temperate forest correlated to soil cycles of water and nutrients; ii) an ecotonal habitat shaped by mixed evergreen and thermophilous deciduous oak forest depending on light, temperature and human disturbance.
Keywords
Mediterranean Forest Vegetation, Soil, AWC, Ellenberg’s Indicators, Hemeroby Index, CCA
Introduction
The assessment of habitat conservation status, especially in protected areas, is nowadays one of the main tasks of local, national and international offices attending to nature conservation and management. In this context, recent studies in applied ecology focused on methods based on ecoindicators for habitat survey and monitoring. The usefulness of ecoindicators is well known ([6]), in particular for the early detection of trends in ecological factors such as climate, soils and disturbances ([18], [32]).
The main challenge in applied ecology is the identification of the key factors involved in the response of species and communities to disturbances and in their ecological structure ([19], [4]). Focusing on the relationship between soil and vegetation, static factors such as soil pH and texture, soil carbon and nitrogen stock, exchangeable bases, etc. can be easily measured by soil profiles and laboratory analyses. Instead, dynamic factors like mineralization rate, water availability and annual average temperature, etc. are harder to be obtained without long-term research efforts ([62]). Nonetheless, the above factors may be easily assessed by the use of plant ecoindicators, such as Ellenberg’s indicators ([14], [68], [12]) and Hemeroby Index for the anthropic disturbance evaluation ([37]). Using the above indexes, the detection of changes in species and communities along spatio-temporal gradients and at different scale levels have been successfully carried out ([1], [38], [33]).
Ellenberg’s indicator values (EIVs) express through a numerical value the average reali-zed niches along seven fundamental gradients (light, temperature, continentality, soil moisture, soil pH, nutrients, salinity). EIVs summarize in scales with nine degrees (up to twelve for soil moisture indicator) the large amount of ecological information on plants and plant communities associated with environmental measurements of edaphic and climatic parameters. Limitation and strengths of the Ellenberg’s approach have been long debated (e.g., [16]), though a number of studies showed a good agreement between indicators and environmental variables (e.g., [21], [61], [63], [64], [35]). EIVs represent a first model of bioindication applied for the first time to the flora of Germany ([14]), and then extended to Netherlands ([74]), Norway ([75]), Sweden ([12]), Estonia ([56], [57]), Poland ([59]), Great Britain ([31]), northeastern France ([71]) and Italy ([7], [53], [20]). EIVs have been shown to successfully describe the ecological patterns of plant communities and to be related to important functional traits ([61], [54], [67]). They are mainly used for environmental monitoring ([15]), and in ecological studies for the interpretation of ordinations in terms of known gradients ([29]). EIVs have been largely applied in botanical studies ([73], [12]) and more recently in ecological investigations ([70], [26], [34]).
Closely related to EIVs is the hemeroby index (H) which is related to the degree of past and present human impacts on ecosystems according to a ten-point scale ([73], [37], [17]). Direct estimation of disturbance and human impact is usually difficult. However, hemeroby index has been successfully applied in studies based on changes in the composition of communities and species in order to assess the response of vegetation to disturbance ([18]).
The ecosystemic approach based on ecological indicators described above is particularly useful in the assessment of the conservation status of forest habitats that are increasingly threatened by human activities and urban sprawl, like in the surrounding of large cities. The aim of this study is to evaluate the conservation status of a natural reserve in the surroundings of Rome (Italy) through an integrated approach, analysing simultaneously soils, lithotypes, landforms, edaphic parameters and plant species.
Materials and methods
Description of the study area
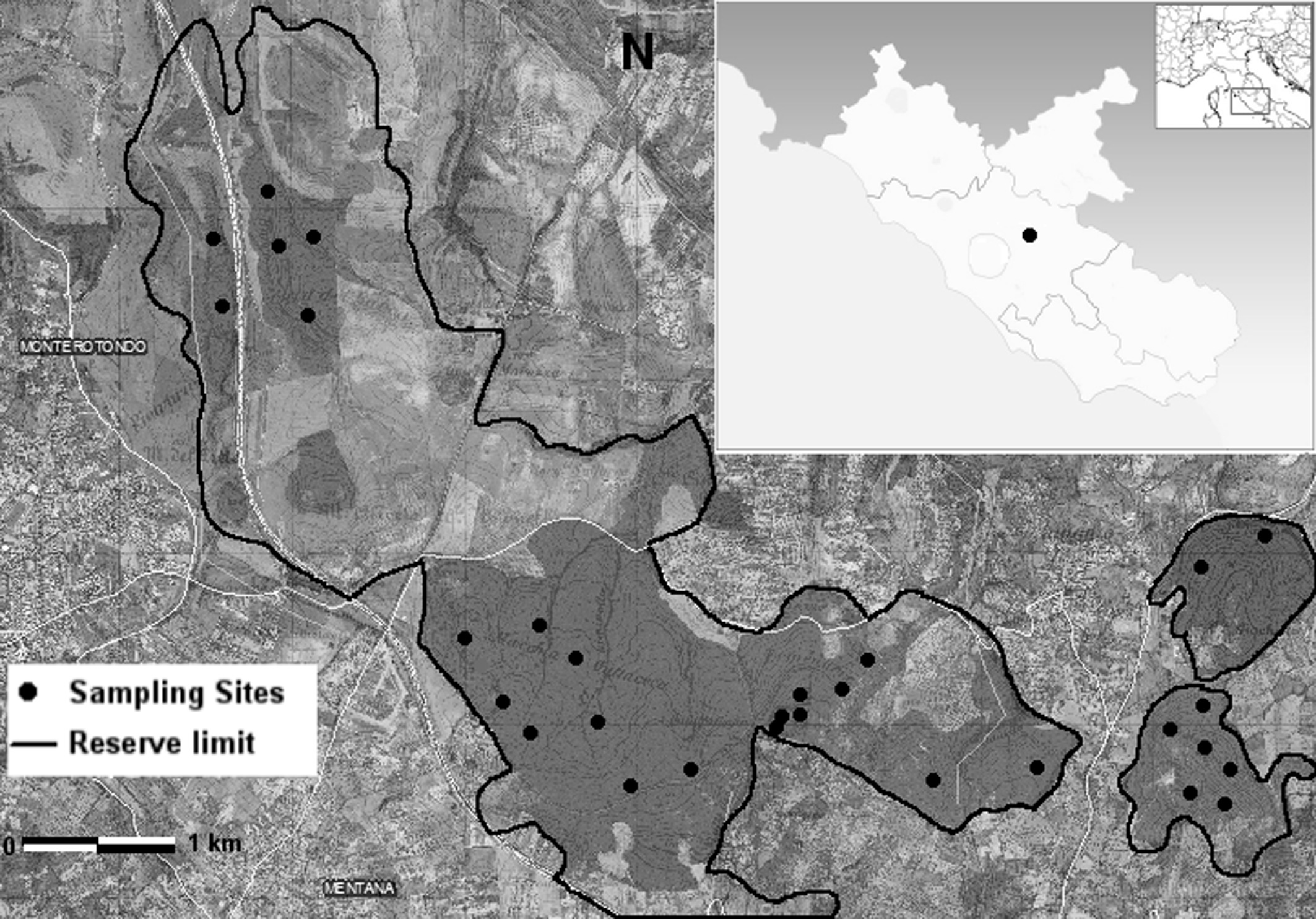
The “Macchia di Gattaceca e Macchia del Barco” natural reserve, established in 1997, is located in the Province of Rome, between the Tiber Valley and the Lucretili Mountains (UTM N 42° 05’, E 12° 50’ - Fig. 1). The protected area covers about 1000 hectares of forested hills, with altitude ranging between 78 and 241 m a.s.l. The area was subjected to a long-term historical disturbance, due to heavy cutting and burning, as well as to grazing, which has been recently restricted to some edge zones only.
From the geological point of view, the area is mainly made up of Mesozoic limestone formations of the Tiber Ridge (Soratte Mt - Cornicolani Mts), referring to the Umbro- Sabina succession. Since Pliocene, marine ingression/regression led to the deposition of yellow sand, calcareous conglomerates, clayey sand and yellowish clay, until the final emergence of the whole area in the Pleistocene ([42]). Pleistocenic pyroclastic products from the Sabatino and Laziale volcanic districts were then deposited ([2]). Limestones in the area show intense fracturing and karst phenomena with numerous sink-holes, including Pozzo del Merro, the deepest explored sink-hole in the world (-392 m a.s.l. - [23]). Geological heterogeneity in the area is one of the causes of its geomorphological diversity, dolines, ditches, plains, slopes make the environmental mosaic.
The most widespread vegetation is a deciduous mixed oak forest, dominated by Quercus cerris and Quercus frainetto, co-dominants in some sites. Quercus robur is present in the diches, while Quercus pubescens and Quercus ilex shrublands are present on sunny calcareous slopes ([13], [66]).
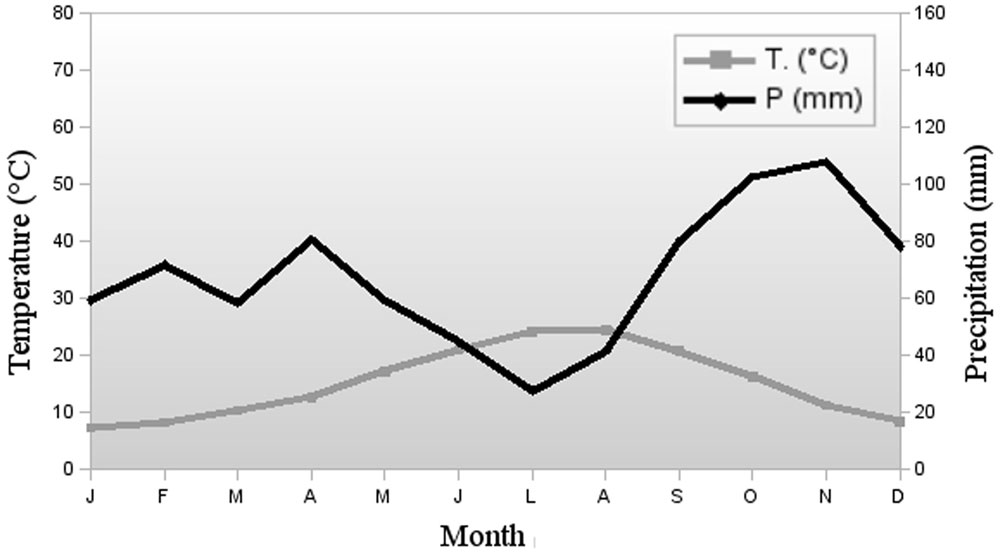
Climate is moderately Mediterranean with about 2 months drought in summer (July and August); average yearly temperature is 15.2 °C and annual rainfall 813 mm, with two peaks in October-November and April (Fig. 2). As for the pedoclimate ([72]), temperature regime resulted to be Thermic. The soil moisture regime is Xeric for most of the studied soils. Only soils on plio-pleistocenic sediments with the highest available water capacity have Udic moisture regime.
Fig. 2 - Climogram of the meteorological Guidonia- Montecelio station (89 m a.s.l.) nearby to the study area. Monthy means for the period 1971-2000.
Field data collecting and analysing
Thirty sampling sites, each of 100 m2, were chosen using randomized-systematic method ([24], [55]). Samplings were distributed over the whole study area within the following geomorphological units:
- Unit A: ridges (6 samplings),
- Unit B: mountain slopes (6 samplings),
- Unit C: plain surfaces (6 samplings),
- Unit D: ditches, subdivided into D1-ditches bottom (4 samplings), D2-ditches gentle slopes (4 samplings) and D3-ditches steep slopes (4 samplings).
In each site phytosociological relevés and soil profiles were carried out simultaneously.
Vegetation
Phytosociological relevés were carried out using Braun-Blanquet method ([5], [76]). All vascular plant species were determined in each site and their relative coverage recorded on a percentage basis. Nomenclature of species followed Pignatti ([52]) revisited with MedCheckList ([28]). Overall, a floristic matrix of 130 species x 30 relevés was obtained.
Ecoindicators
Ellenberg’s Indicator Values (EIVs - [14], [15]) and Hemeroby Index ([37]) were applied in this study. The full list of the indicators applied was as follows: (1) L: light; (2) T: temperature; (4) F: soil moisture; (5) R: soil reaction; (6) N: soil nitrogen; (7) hemeroby. Since salinity (S) is used only for saline soils and continentality (K) has a meaning only on a geographical scale, these two indicators were excluded from the analysis.
All the above indexes were weihted on species coverage.
Pedological analysis
In each sampling site soil profiles were obtained and described following the guidelines for Soil Survey by Costantini ([9]). Seventy seven samples were collected and analysed with standard methods ([43]) for: (i) texture; (ii) pH in a 1:2.5 soil/ water suspension; (iii) total carbonates; (iv) organic carbon and organic matter; (v): exchangeable acidity; (vi): exchangeable bases; (vii): cation exchange capacity; (viii) total nitrogen; (v) available phosphorus. Moreover, three additional parameters were calculated: available water capacity (AWC), base saturation and carbon/nitrogen ratio (C/N). Available water capacity (AWC, mm H2O cm-1 soil depth) was estimated by the Salter & Williams ([60]) equation based on textural composition and percentage of organic matter (eqn. 1):
where CS is the percentage of the coarse sand, S is the percetage of silt and CO is the percentage of organic carbon in the sample.
Although all soil horizons were sampled and analysed separately, statistical analysis was applied on weighted averages, calculated by multiplying the value of each parameter by the horizon thickness, summing these values and dividing them by the total depth of the profile ([10], [22], [3]).
Soils were classified to the subgroup level according to the Soil Taxonomy ([72]).
Statistical treatment
For each one of the 30 sites studied, values were obtained for 12 variables: 6 ecoindicators and 6 soil parameters (Tab. S1 in Appendix 1). All variables were normalized and standardized (since having different scales and units) by subtracting the variable mean and dividing by their standard deviation ([55]).
The following multivariate statistical analyses were applied on standardized variables: (i) cluster analysis (CA) on the variables/ relevés matrix (12x30); (ii) Canonical Correspondence Analysis (CCA) on the whole data set (variables/relevés 12x30 and species/relevés 130x30 matrices).
All statistical analyses were performed by the software R ([58]) using the package “vegan” for community ecology ([48]), “cluster” for cluster analysis ([41]) and “ecodist” for distance calculation ([27]).
To recognize the main gradients in the dataset, normalized variable scores were calculated from their weights on CCA axes. To test the significance of species/environmental factors correlation, two-ways ANOVA with permutation was performed on the CCA model, variables and axes ([39]). Using the R package “anova.cca” the number of permutations is controlled by targeted “critical” P value and accepted Type II or rejection error (β); permutations were performed until the P value obtained differs from the targeted α at risk level given by β.
Analysis of variance (one-way ANOVA) was performed on clusters obtained by CA to test for possible differences in soil parameters and ecoindicator means.
Results
Cluster Analysis
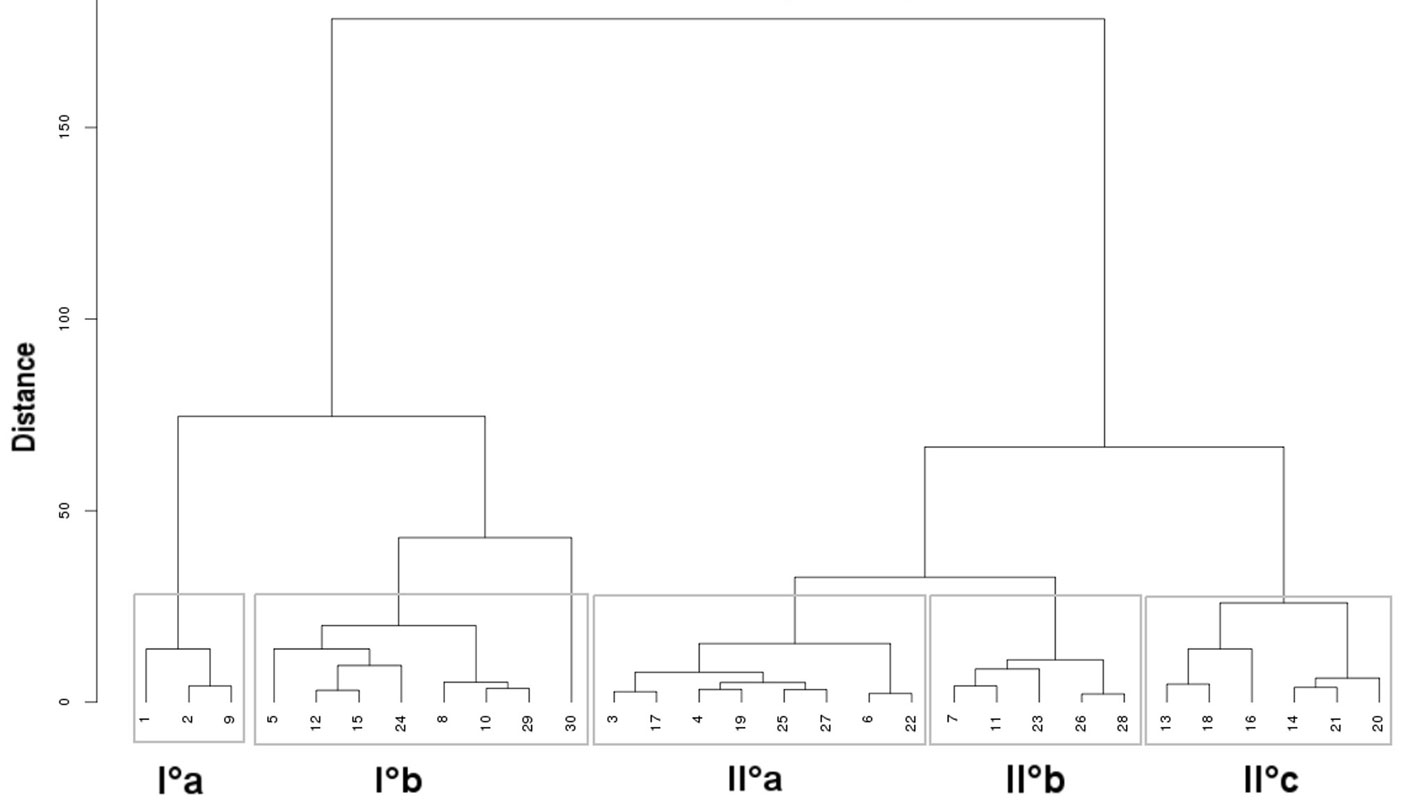
Results of the cluster analysis carried out on the variables/relevés matrix revealed the existence of two main groups of relevés (Fig. 3), each one divided into sub-clusters, distinguished by floristic (Table S2 in Appendix 1), ecological (Ellenberg’s indicators) and edaphic characteristics. Overall, 5 clusters were identified (I°a, I°b; II°a, II°b, II°c - see Tab. 1). Vegetation, soil types, bedrocks and geomorphological units of the sampled areas are summarized in Tab. 2. Following is a brief description of the two main clusters and the five subclusters identified.
Fig. 3 - Dendrogram of the relevés from the cluster analysis carried out on the matrix of 12 variables (6 ecoindicators and 6 soil parameters) x 30 sites. Two main clusters are outlined, each one further distinguished into sub-clusters.
Tab. 1 - Mean values (± standard deviations) of soil parameters and ecoindicators in the 5 groups identified by cluster analysis.
| Parameters | I°a | I°b | II°a | II°b | II°c |
|---|---|---|---|---|---|
| pH | 7.8 ± 0.5 | 7.2 ± 0.5 | 6.4 ± 0.7 | 7.6 ± 0.8 | 6.8 ± 1 |
| CaCO3 (%) | 0.3 ± 0.5 | 1.4 ± 4 | 0.1 ± 0.1 | 3.4 ± 4.4 | 0.1 ± 0.2 |
| Org (%) | 2.8 ± 0.4 | 4.3 ± 1.3 | 2.5 ± 0.8 | 3.3 ± 0.1 | 1.4 ± 0.5 |
| N (%) | 0.2 ± 0 | 0.5 ± 0.2 | 0.3 ± 0.1 | 0.3 ± 0 | 0.2 ± 0.1 |
| C/N | 11.5 ± 2.1 | 8.9 ± 1.4 | 8.9 ± 1.5 | 10.3 ± 0.8 | 8.6 ± 1.7 |
| AWC (mm) | 137 ± 6.9 | 98 ± 14 | 219 ± 5.1 | 240 ± 6.3 | 195 ± 9.7 |
| H | 2.7 ± 0.4 | 2.6 ± 0.6 | 2.5 ± 0.4 | 2.3 ± 0.1 | 2.3 ± 0.2 |
| L | 3.7 ± 0.5 | 4.3 ± 1 | 3.4 ± 0.3 | 3.5 ± 0.4 | 3.3 ± 0.1 |
| T | 7 ± 0.6 | 7 ± 0.6 | 6.4 ± 0.3 | 6.6 ± 0.3 | 6.4 ± 0.3 |
| F | 5.3 ± 0.6 | 5.3 ± 0.7 | 5.9 ± 0.2 | 5.8 ± 0.4 | 5.9 ± 0.2 |
| R | 7.2 ± 0.3 | 7.4 ± 0.1 | 7.3 ± 0.2 | 7.4 ± 0 | 7.4 ± 0.1 |
| N | 5 ± 1.2 | 5.4 ± 0.8 | 6.1 ± 0.4 | 5.9 ± 0.4 | 6.2 ± 0.3 |
Tab. 2 - Integrated table of geomorphological units, bedrocks, soil types and vegetation following sub-clusters.
| Subcluster | Bedrocks | Land Forms | Dominat soil types | Dominat tree species |
|---|---|---|---|---|
| I°a | Calcareous rocks |
A-ridge | TYPIC HAPLOXEREPTS: fine, thermic, superactive. Shallow soils, with low water availability (< 140 mm). Clay. Neutral to weakly alkaline slightly calcareous. Very high E.C.C.(40-50 cmol/kg) and very high Base Saturation (80-90%) |
Quercus ilex Quercus pubescens Quercus cerris |
| I°b | Calcareous rocks |
B-mountain slope |
LITHIC HAPLOXEROLLS: fine, thermic, superactive. Shallow soils, stony, with rather low water availability (< 110 mm). Clay loam- clay. Weakly alkaline. Slightly calcareous or acalcareous. Very high E.C.C. (50-70 cmol/kg) and very high Base Saturation (>90%). |
Quercus ilex Quercus pubescens Quercus cerris |
| II°a | Tuffs | D2-ditches gentle slopes C-plain surfaces |
TYPIC HAPLOXEREPTS, MOLLIC/TYPIC HAPLOXERALFS: fine, thermic, active. Shallow to moderately deep soils, with moderate to high water availability (210 to 220 mm). Weakly acid. Silty clay to clay. High E.C.C. and high Base Saturation (60-75%). |
Quercus cerris Quercus frainetto |
| II°b | Plio-pleistocenic marine sediments and calca- reous rocks |
D2-ditches gentle slopes D1-ditches bottom |
ENTIC HAPLUDOLLS: fine silty, thermic, superactive. Very deep soils, with very high water availability (230-250 mm) and udic moisture regime. Silt loam. Moderately alkaline, weakly to moderately calcareous (2-14% CaCO3) . Very high E.C.C.(60-70 cmol/kg) and very high Base Saturation (100%). |
Quercus cerris Quercus frainetto Quercus robur |
| II°c | Calcareous rocks | D1-ditches bottom C-plain surfaces |
ULTIC HAPLOXERALFS and TYPIC EUTRUDEPT: fine, thermic, active. Deep soils with very high water availability (>200 mm). Silty clay - clay. Moderately to weakly acid. High E.C.C.(40 to >50 cmol/kg) , high Base Saturation (60-70%). |
Quercus cerris Quercus frainetto Quercus robur |
I° cluster: Thermophilous submediterranean /mediterranean woodlands and shrublands with Quercus cerris along with Quercus ilex and Quercus pubescens (locally dominant) on calcareous rocks, lithic soils. Geomorphological units are ridges (A) and mountain tops (B).
- I°a sub-cluster: sclerophyllous shrublands dominated by Quercus ilex, with Viburnum tinus on the ridges.
- I°b sub-cluster: open woodlands and shrublands of Quercus pubescens and Quercus ilex along with Mediterranean species (such as Pistacia terebinthus, Phyllirea latifolia and Cercis siliquastrum) on moderately to strongly steep slopes.
II° cluster: Mesophilous and hygrophilous woodlands dominated by Quercus cerris and Quercus frainetto with the occurrence of Quercus robur on deep and very deep soils on plio-pleistocenic marine sediments (slopes of ditches and gorges), tuffs (gentle slopes) and calcareous rocks (depressions and gentle slopes). This vegetation type is the most common in the study area. Geomorphological units are plain surfaces (C) and ditches (D).
- II°a sub-cluster: mesophilous woodlands with Quercus robur on tuffs.
- II°b sub-cluster: hygrophilous woodlands with Quercus robur, Acer obtusatum, Ulmus minor, Sambucus nigra and Corylus avellana on marine sediments and calcareous bedrocks and soils with the highest AWC values (Tab. 1).
- II°c sub-cluster: more open woodlands characterized by large cover of Prunus spinosa and exclusive occurrence of ruderal and edge species, such as Onopordum illyricum, Bellis perennis, Dactylis glomerata, Holcus lanatus on calcareous rocks.
Analysis of variance
Significant differences among the five sub-clusters identified were detected for the variable AWC after one-way ANOVA (df=4; SS=92959.3; F=252.2, P<0.0001). To test the significance of differences among subclusters’ means, Fisher LSD test was carried out (P<0.05). Results showed that sub-cluster I°a and I°b had means significantly lower than the mean of the other three sub-clusters (II°a, II°b, II°c). Sub-cluster I°b showed that means for ecoindicator L (DF=4, SS=4.44; F=3.39, P<0.024), organic carbon (DF=4, SS=31.49; F=11.14, P<0.0001) and nitrogen (DF=4, SS=0.42; F=5.29, P<0.003) were significantly higher than the means of sub-clusters II°a, II°b, II°c. Furthermore, sub-cluster I°a showed a N-indicator mean significantly lower (DF= 4, SS=4.98; F=3.38, P<0.024) than the mean of sub-clusters II°a, II°b and II°c.
CCA
Results of the CCA analysis on the matrix of 30 relevés, 130 species and 12 variables confirmed the differences between the two main clusters previously identified (Tab. 3, Fig. 4a and Fig. 4b) . The first CCA axis accounted for 66% of the total variance and was highly correlated (P<0.01) with the ecoindicators for N (r = 0.73) and T (r = -0.64); it was also correlated (P<0.05) with F (r = 0.52) and H (r = -0.41) indicators, as well as with the ratio C/N (r = - 0.42).
Tab. 3 - CCA Outputs. (1) Partitioning of mean squared contingency coefficient; (2) Eigenvalues and their contribution to the mean squared contingency coefficient (first three axes); (3) Biplot scores for constrained variables. (*): p<0.05; (**) p<0.01; (***): p<0.001.
| Output 1 | Inertia | Proportion | Rank |
|---|---|---|---|
| Total | 3.768 | 1 | - |
| Constrained | 2.505 | 0.665 | 13 |
| Unconstrained | 1.263 | 0.335 | 17 |
| Output 2 | CCA1 | CCA2 | CCA3 |
| Eigenvalue | 0.655 | 0.415 | 0.295 |
| Proportion Explained | 0.174 | 0.11 | 0.078 |
| Cumulative Proportion | 0.174 | 0.284 | 0.362 |
| Output 3 | CCA1 | CCA2 | CCA3 |
| pH | -0.089 | 0.106 | -0.362 |
| CaCO3 % | 0.003 | 0.132 | -0.335 |
| Org C% | -0.199 | 0.626** | -0.131 |
| N % | -0.088 | 0.762* | -0.05 |
| C/N | -0.415* | -0.234 | -0.052 |
| AWC (mm) | 0.289 | -0.457** | 0.074 |
| H | -0.409* | 0.348 | -0.27 |
| L | -0.184 | 0.912** | -0.156 |
| T | -0.641** | 0.635 | 0.03 |
| F | 0.522* | -0.756** | 0.04 |
| R | 0.181 | 0.181 | -0.802* |
| N | 0.733** | -0.519 | 0.041 |
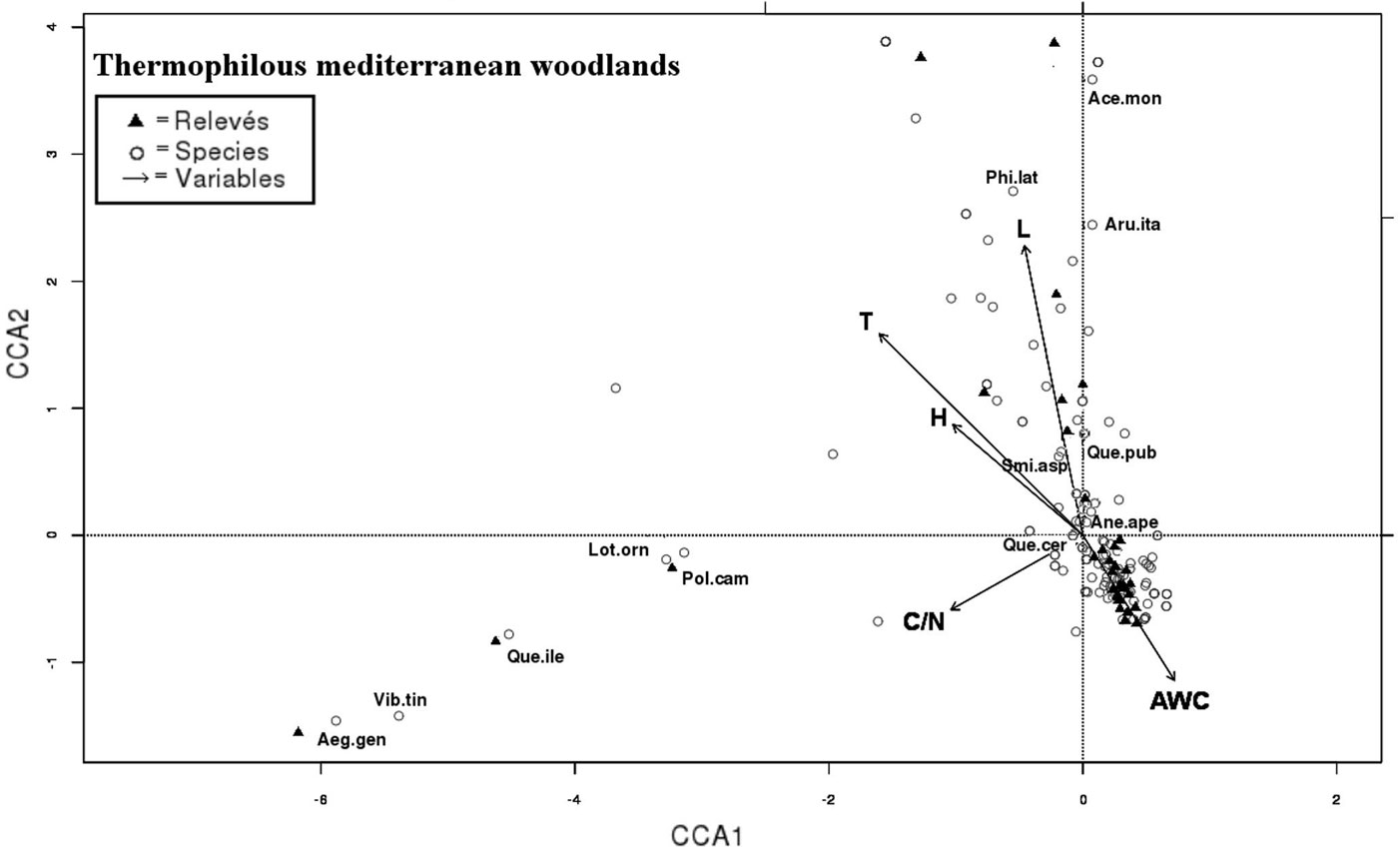
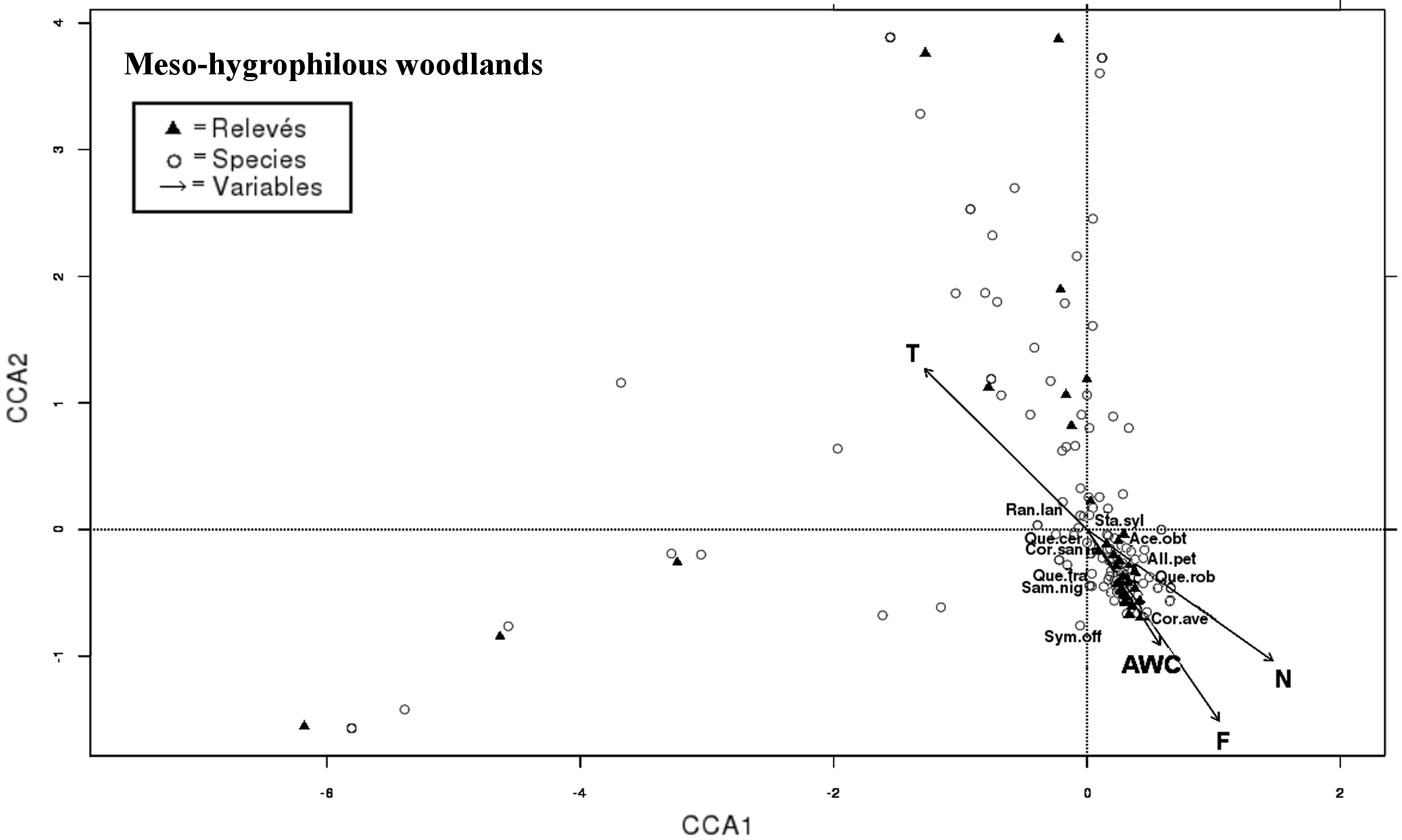
Fig. 4 - CCA triplot according to axes 1 and 2: species, relevés and variables of thermophilous Mediterranean woodlands (cluster I°) are projected into two branches on the bottom left (sub-cluster I°a) and on the top left (sub-cluster I°b). Most representing species are indicated with labels (see Tab. S2 in Appendix 1 for species abbreviations) and most correlated soil parameters are displayed.
The second CCA axis accounted for 42% of the total variance and showed a highly significant (P<0.01) correlation with L (r = 0.91) and F (r = -0.76) ecoindicators; it was also correlated (p<0.01) with organic carbon (r = 0.63) and with measured nitrogen (r =0.76). The third axis accounted for 29% of the variance in the dataset and showed a significant correlation (p<0.05) with the R indicator (r = -0.80).
A small group of species exclusive of cluster I° was projected onto two branches of CCA plane (Fig. 4): on the bottom left of the scattergram, species with large abundance or exclusive presence in the sub-cluster I°a (like Quercus ilex, Viburnum tinus, Aegilops geniculata, Lotus ornithopodioides and Polypodium cambricum) are displayed; on the right top of the same plot, species largely abundant in the sub-cluster I°b may be found, such as Acer monspessulanum, Cercis siliquastrum, Quercus pubescens, Smilax aspera, Arum italicum.
In the central sector of CCA (Fig. 5), the cloud of species projected in the direction of AWC, F and N indicators belongs to the II° cluster and corresponds to meso-hygrophilous woodlands dominated by Quercus cerris with the exclusive occurrence of Quercus frainetto and Quercus robur. All the species belonging to cluster II° (like Sambucus nigra, Corylus avellana, Acer obtusatum, Cornus, sanguinea, Symphytum officinale, Ranunculus lanuginosus and Stachis sylvatica) exhibit a low requirement for T and high for N and F indicators (Tab. 1).
Fig. 5 - CCA triplot according to axes 1 and 2: species, relevés and variables of meso-hygrophilous woodlands (cluster II°) are projected in the centre of axes plane. Most representing species are indicated by labels (see Tab. S1 in Appendix 1 for species abbreviations) and most correlated variables are displayed.
All the species of the two branches of cluster I° exhibit a high requirement for T (range 7-9) and low for N (range 2-6) and F indicators.
The two main forest types respectively represented by thermophilous (cluster I°) and meso-hygrophilous (cluster II°) communities were clearly separated along CCA axis 1, while the two sub-clusters of cluster I° were separated along CCA axis 2.
Discussion
The results from the canonical correspondence analysis carried out may be easily interpreted based on the ecoindicator values used in this study. Species of the meso-hygrophilous woodlands (cluster II°) were distributed along the gradients of water and nutrients, as revealed by their F and N indicator values, as well as by the measured soil AWC parameter. In fact, water and nutrients represent the key factors responsible for survival of forest patches dominated by Quercus frainetto and Quercus robur on deep soils with the highest water availability ([30]). In the investigated area the above communities occur on plain surfaces, as well as on the steep slopes of the ditches, on plio-pleistocenic marine sediments and on tuffs (Tab. 2).
Shrublands and open woodlands of Quercus pubescens and Quercus cerris (subcluster I°b) were distributed along the ma-ximum variation of the T, L and H indicators (Fig. 4). T and H indicators were significantly correlated with CCA axis 1, while L indicator with CCA axis 2 (Tab. 3). Soils in the above communities are lithic and shallow, with low AWC values (Tab. 2). The above evidence indicate that light, temperature and disturbance are the key factors shaping these communities, mostly occurring on calcareous bedrocks.
As clearly revealed by the CCA triplot in Fig. 4, a small group of species dominated by Quercus ilex and Viburnum tinus are related with the highest C/N ratio values in the dataset. The above result can partly be explained by the slower decomposition rate of the sclerophyllous leaves leading to a consistent accumulation of organic carbon in these communities ([25], [78]).
Through the correlation of CCA axes with species and variables (Fig. 4, Fig. 5, Tab. 3) it was possible to identify two different types of habitats, corresponding to the clusters I° and II°:
- a core habitat (cluster II°) represented by patches of temperate forest with higher diversity in species as well as in landforms and lithotypes, supported by high water and nutrients availability ([50]);
- an ecotonal habitat (cluster I°) characterized by mixed evergreen and thermophilous deciduous oak forest depending on light, temperature and human disturbance, on soils with the lowest AWC values (Tab. 1).
The above habitats were distributed along three multi-composite gradients identified by CCA: (i) soil moisture and nutrients (F, N indicators, AWC); (ii) light and temperature (L, T indicators); and (iii) disturbance (H indicator). Soil AWC was the main factor summarizing the complexity of the vegetation gradients investigated, as well as the species and communities distribution in the two habitats. Indeed, large differences in soil AWC were detected among species assemblages characterized by high environmental heterogeneity of landforms, substrates and soils ([67], [49], [44]). AWC also showed marked differences among community structures (Tab. 1): from more mature and complex communities with closed canopy of the core habitat (cluster II°) to younger ones with tendency towards open woodlands of the ecotonal habitat (cluster I°).
Among the soil parameters measured in this study, AWC was the most efficient for the description of vegetation changes, being an aggregate set of more than one variable, such as organic matter, sand, silt and soil depth, and therefore summarizing soil characteristics at the community scale ([77]). Among EIVs, light (L) and nutrients (N) were the main factors responsible for structuring communities and determining species assemblage: higher N values were associated to more mature and complex meso-hygrophilous communities, while higher L values were linked to younger thermophilous woodlands.
In the Mediterranean environments anthropic disturbance affects mainly the meso-hygrophilous vegetation in respect to the more resilient thermo-xerophilous communities ([40], [32]). For this reason, the persistence of the meso-hygrophilous vegetation of the core habitat within an area historically affected by human activities is particularly valuable. The conservation of these patches of humid vegetation was favored not only by the peculiar geomorphology of some sites (ditches) in the Reserve, but also by water and nutrients availability (Fig. 5). For the above reasons, conservation activities aimed at preservation of the core habitat should be adopted, including the realization of ecological corridors connecting the different patches of habitat ([11], [46], [65]), and the conservation of soil nutrients and water regime to prevent soil fertility losses and erosion ([26]).
Our study highlights the importance of protecting the more vulnerable core habitat, keeping a light and controlled sheep grazing only on the edges of the thermophilous woodlands which are historically adapted to disturbance ([45]). The protection must be on a local scale, where the key ecological factors emerged from the multiple set of indicators and parameters ([47], [36], [8]).
Conclusion
Multi-dimensional data analysis may help in the identification of key factors underlying the ecosystem complexity ([38]). In this study, the combined use of soil parameters and plant ecoindicators allowed to detect differences and similarities among the investigated communities at a fine scale.
Our results showed the ability of light (L) and soil nutrients (N) indicators and of soil measured parameter AWC to detect differences in ecological requirements of species and communities ([67], [69]). AWC resulted a good synthetic soil parameter able to detect this diversity of gradients, species and communities, confirming the results of previous researches in the Mediterranean environments ([67], [51]).
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Department of Forest Environment and Resources, University of Tuscia, v. S. Camillo de Lellis snc, I-01100 Viterbo (Italy)
IBAF-CNR, v. Salaria km 29.3, I-00016 Monterotondo Scalo (RM - Italy)
Giuseppina Dowgiallo
Anna Testi
Department of Environmental Biology, Botanical Garden, “La Sapienza” University of Rome, l.go Cristina di Svezia 24, I-00165 Rome (Italy)
Corresponding author
Paper Info
Citation
Sicuriello F, De Nicola C, Dowgiallo G, Testi A (2014). Assessing the habitat conservation status by soil parameters and plant ecoindicators. iForest 7: 170-177. - doi: 10.3832/ifor0963-007
Academic Editor
Raffaele Lafortezza
Paper history
Received: Feb 01, 2013
Accepted: Dec 03, 2013
First online: Feb 14, 2014
Publication Date: Jun 02, 2014
Publication Time: 2.43 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2014
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 55891
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 45877
Abstract Page Views: 3052
PDF Downloads: 5241
Citation/Reference Downloads: 17
XML Downloads: 1704
Web Metrics
Days since publication: 4475
Overall contacts: 55891
Avg. contacts per week: 87.43
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2014): 8
Average cites per year: 0.67
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Towards a functional phytosociology: the functional ecology of woody diagnostic species and their vegetation classes in Northern Italy
vol. 14, pp. 522-530 (online: 22 November 2021)
Research Articles
Classification and mapping of Spanish Mediterranean mixed forests
vol. 12, pp. 480-487 (online: 14 October 2019)
Commentaries & Perspectives
The role of plant sociology in the study and management of European forest ecosystems
vol. 6, pp. 55-58 (online: 21 January 2013)
Research Articles
The cork oak in the Mountains of Palermo (Italy): ecological insights from the south-eastern edge of its distribution range
vol. 13, pp. 336-344 (online: 07 August 2020)
Research Articles
Drought effects on the floristic differentiation of Greek fir forests in the mountains of central Greece
vol. 8, pp. 786-797 (online: 08 April 2015)
Technical Reports
Diversity pattern of vegetation in and around proposed Kotlibhel hydroelectric project along the Alaknanda River in Garhwal Himalaya (India)
vol. 4, pp. 38-43 (online: 27 January 2011)
Research Articles
Growing at the forest edges: how natural regeneration develops under fragmentation
vol. 15, pp. 248-255 (online: 19 July 2022)
Research Articles
Typology and synecology of aspen woodlands in the central-southern Apennines (Italy): new findings and synthesis
vol. 13, pp. 202-208 (online: 19 May 2020)
Research Articles
A state-and-transition approach to alpine grasslands under abandonment
vol. 3, pp. 44-51 (online: 02 March 2010)
Research Articles
Influences of mature Pinus nigra plantations on the floristic-vegetational composition along an altitudinal gradient in the central Apennines, Italy
vol. 13, pp. 279-285 (online: 03 July 2020)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword