Hydrological properties of litter layers in mixed forests in Mt. Qinling, China
iForest - Biogeosciences and Forestry, Volume 11, Issue 2, Pages 243-250 (2018)
doi: https://doi.org/10.3832/ifor2535-011
Published: Mar 12, 2018 - Copyright © 2018 SISEF
Research Articles
Abstract
The litter layer is an important component of forest ecosystems, although little is known about the differences in litter characteristics and hydrological properties of unmanaged, overgrown broadleaved and coniferous mixed forests in the subtropical and temperate zones. This study was carried out in a naturally generated broadleaved and coniferous mixed forest at the Qinling National Forest Ecosystem Research Station, Shaanxi Province, China. We quantified the litter thickness, mass, and its hydrological properties in evergreen pine (Pinus tabulaeformis) stands and deciduous oak (Quercus aliena var. acuteserrata) stands through in situ surveys and laboratory immersion experiments. The thickness of the litter layer for P. tabulaeformis averaged 9.3 ± 2.8 cm, and it was not significantly different from that detected in the Q. aliena stand (8.3 ± 3.6 cm). The total mass of the litter layer for P. tabulaeformis, 27.94 ± 9.81 Mg ha-1, was significantly higher than the total mass of 16.04 ± 3.60 Mg ha-1 found for Q. aliena. The mass of the semi-decomposed, fermented litter (OF) layer was significantly higher than that of the non-decomposed litter (OL) layer, irrespective of species. The rate of water absorption by dry litter was the highest at the onset of the immersion experiment and decreased exponentially with time. The water-holding capacity (Wm) and water-interception capacity (Wi) of the OF layer were higher than the OL layer for both forest stands. The Wm and Wi for the P. tabulaeformis stand were higher than those for the Q. aliena stand at our study sites. The higher Wm and Wi for P. tabulaeformis may be more effective in ameliorating the splash impact from high-intensity storms to improve water quality, while relatively lower Wm and Wi for Q. aliena may be considered favorable for augmenting water yield. Forest resource managers should consider those differences along with the other components of the water budget when making management decisions.
Keywords
Water Conservation Forest, Hydrological Properties, Forest Succession
Introduction
Many natural forests and plantations have become overgrown during the past few decades, and this is a global phenomenon ([24], [40], [17], [32]). The implication of overgrown forests on water resource management in humid regions has been under-appreciated in locations where water scarcity is less pressing compared with semiarid and arid regions ([7], [9]). The litter layer is an important component of forest ecosystems and its importance is often underestimated in water resource management ([30], [19], [15]). More recent studies have added evidence of a substantial impact of litter on the water budget in semiarid shrubland and woodland with a substantial litter layer ([6], [3], [2], [1]). However, the litter production dynamics, litter layer structure and hydrological properties of secondary forests in humid forest ecosystems, as well as their relevance in water conservation management have not been widely studied. With the increasing acreage of secondary forests and overgrown plantations in subtropical and temperate forest systems, the quantity and quality of ecosystem services provided by those forest stands deserve further studies.
Forest litter, a sponge layer separating precipitation from mineral soil, mediates rainfall splash impact and reduces, delays or often eliminates overland flow and soil erosion from the forest floor ([8], [26]). As a result, the amount of litter and its hydrological properties in a forested catchment could affect not only the water quality but also the water yield of the catchment. A recent study on Japanese red pine (Pinus densiflora) and konara oak (Quercus serrata) mixed forest in Japan ([9]) demonstrated that the litter layer plays an important role in the hydrological cycle of humid ecosystems, and litter removal resulted in the increase of peak flow, particularly during relatively large flood events. However, we know little about the differences in litter characteristics and hydrological properties between broadleaved species and coniferous species in the mixed forests of the subtropical and temperate zones. Such information is particularly useful from the perspective of managing water conservation forests for water yield and water quality ([29], [16]).
The Qinling Mountains are the highest mountain range in central and eastern China, with an east-west length of 1600 km and a north-south width ranging from tens to two-three hundred kilometers. They divide the Yangtze River system to the south and the Yellow River system to the north, and are located in the central and southern part of Shaanxi Province between the Wei River and the Han River. The forest area encompasses gullies, ravines, and streams, and contains abundant water resources reaching a total amount of 22.2 billion m3 ([35]). The southern slope of the Qinling Mountains has a water resource amount of approximately 18.2 billion m3 and is an important recharge source for the Danjiangkou Reservoir, the headwater source of the central route of China’s South to North Water Diversion Project (SNWDP - [35]). The average annual streamflow into the reservoir was reported to have substantially decreased in past decades ([23]).
The southern slope of the Qinling Mountains is co-dominated by P. tabulaeformis and Quercus aliena var. acuteserrata (Q. aliena, hereafter) between 1300 and 1800 m a.s.l. of elevation. A 30-year-old study quantified the litter characteristics of young and middle-aged P. tabulaeformis and Q. aliena forests ([18]). Currently, most of those forests have become overgrown due to lack of management. A recent study revealed that the streamflow in this region decreased during the same period as a result of the reduced precipitation, though 10-15% of such decrease was attributed to non-climate related causes, including vegetation ([23]). Nonetheless, there have been no studies on the characteristics and hydrological properties of the litter layer of these forests, especially on possible differences between P. tabulaeformis and Q. aliena (the dominant species in the area) stands in favoring water resource management.
In this study, we quantified the thickness, mass, and hydrological properties of the litter layer in representative Pinus tabulaeformis and Quercus aliena stands through in situ site surveys and laboratory immersion experiments, with the aim of providing data support and theoretical guidance to water resource management and conservation in the Qinling Mountains.
Materials and methods
Study site
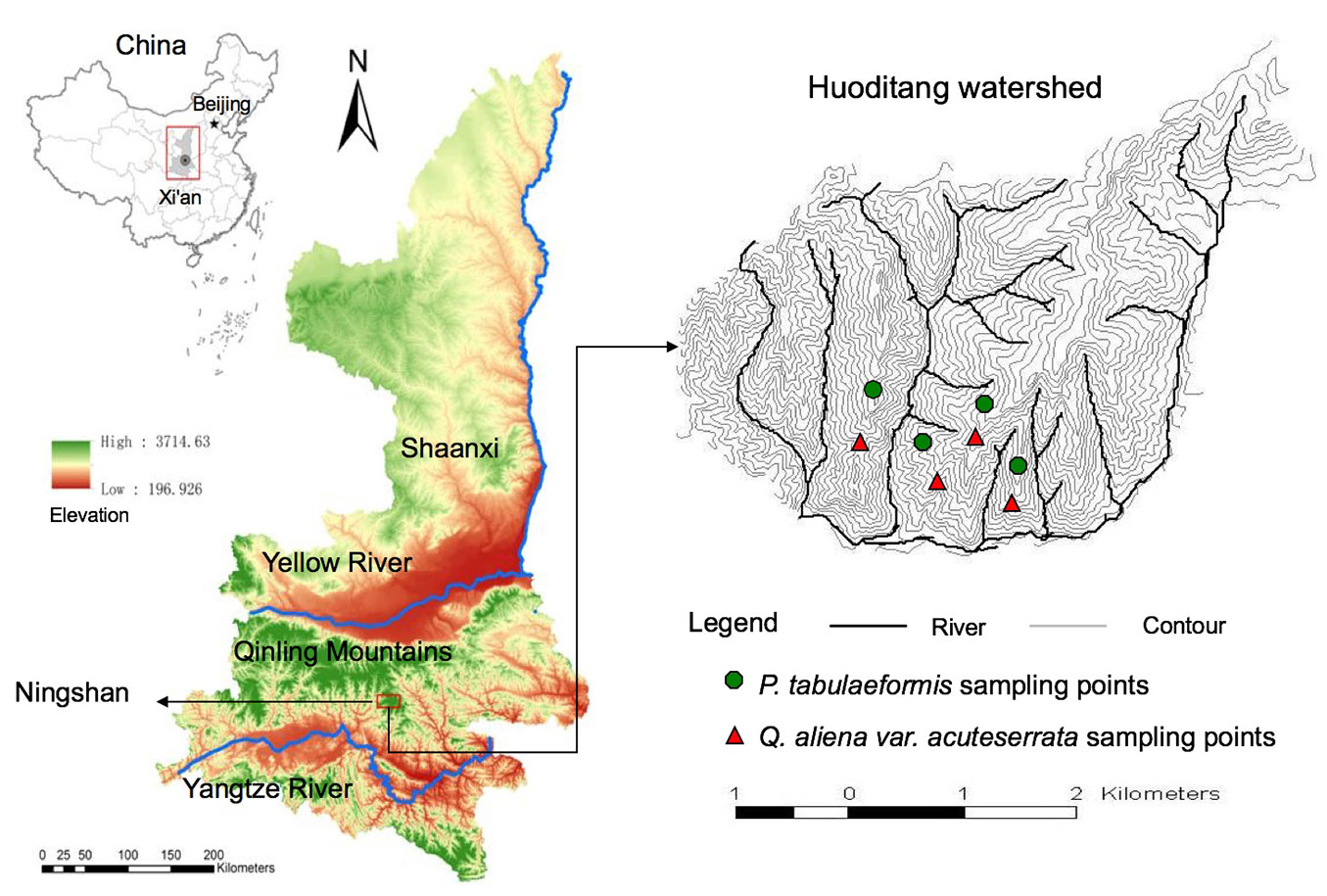
The study was carried out in the Huoditang Watershed at the Qinling National Forest Ecosystem Research Station at Ningshan County, Shaanxi Province, China (33° 18′ N, 108° 20′ E - Fig. 1). The station is located on the southern slope of the Qinling Mountains, falling within the transition zone of the warm-temperate deciduous broadleaved forest region and the subtropical evergreen broadleaved forest region ([27]). The elevation ranges from 800 to 2474 m a.s.l., declining from east to west. The topography is diverse with mainly southwestward slopes ranging from 20° to 50°. The average annual temperature ranges from 8 to 10 °C The average annual precipitation ranges from 900 to 1200 mm, with rainfall mostly occurring from May to October and snowfall occurring from November to the beginning of April. The average annual evaporation reaches 800 to 950 mm ([5]). The soil at this site belongs to mountain brown soil, and the bedrock mainly consists of granite, gneiss, metamorphic sandstone and schist ([39]). The study site is primarily covered by naturally regenerated, secondary forests. Distinct zoning in forest types exists along an elevation gradient, with the deciduous broad-leaved forest distributed at the bottom, coniferous and broad-leaved mixed forest in the middle, and subalpine coniferous forest on the top. In this study, we focused on the P. tabulaeformis stands and Q. aliena stands forming the coniferous and broad-leaved mixed forest at the altitudes ranging from 1300 to 1800 m a.s.l. These stands were primarily naturally regenerated after forest harvesting during the 1960s and the 1970s. For the P. tabulaeformis stand, the associated tree species were Pinus armandi, Toxicodendron vernicifluum, Carya cathayensis Sarg, and Sorbus folgneri. The shrub layer included Viburnum dilatatum, Stachyurus chinensis, Rosa omeiensis, Lonicera hispida Pall; and the herbaceous layer contained Carex lanceolata and Stellaria media. For the Q. aliena stand, the associated tree species included P. tabulaeformis, Acer davidii, Carpinus cordata, Quercus aliena, P. armandi, Acer maximowiczii, and Toxicodendron vernicifluum; the shrub layer was dominated by Rosa multiflora, Lespedeza bicolor, Abelia dielsii, Spiraea fritschiana, Euonymus alatus, and Corylus heterophylla; and the herbaceous layer included Thalictrum aquilegifolium, Anemone tomentosa, Dryopteris chinensis, and Carex duriuscula sp. stenophylloides.
Fig. 1 - Geographic location of study site and spatial distribution of sampling points in the Huoditang Watershed, located in the Qinling National Forest Ecosystem Research Station (southern Qinling Mountains, central China, 161 km south of Xian City).
Litter collection
We selected four P. tabulaeformis sites and four Q. aliena sites that were representative of the overall stand age and growth condition, located in areas away from the road and relatively free of human disturbance. At each site, a section or an entire surface of a slope was selected following the protocol of “Observation Methodology for Long-term Forest Ecosystem Research” of the National Standards of the People’s Republic of China (GB/T 33027-2016). The basic site and stand information are presented in Tab. 1. For each site, upper, middle, and lower positions were selected. Two 0.5 × 0.5 m quadrats were randomly selected for each slope position for litter sampling. Intact litter layers were collected and the un-decomposed litter (OL layer) and semi-decomposed litter (OF layer) were packed separately into plastic litter bags following the method described in the aforementioned protocol. A total of 96 litter bags (2 species × 4 sites × 3 slope positions × 2 random quadrats × 2 litter layers) were collected. Eight points around each quadrat were randomly selected to measure the average thickness of both OL and OF layers representing that quadrat. Litter bags were sealed and brought back to the laboratory to determine the fresh mass. After fresh mass determination, litter was left to air dry in the lab. Dry mass was determined by drying the air-dry samples in an oven at 85 0C for 12 hours. Collection of litter samples was conducted after all deciduous trees dropped their leaves in November and December of 2013.
Tab. 1 - Basic characteristics of the sampling sites.
| Characteristics | Variable | Vegetation type | |
|---|---|---|---|
| P. tabulaeformis | Q. aliena var. acuteserrata |
||
| Topography | Elevation (m) | 1542-1774 | 1423-1752 |
| Slope aspect | S, SW | S, SW | |
| Slope (°) | 25-35 | 32-40 | |
| Stands characteristics | Height (m) | 23.35 ± 3.04 | 17.33 ± 1.82 |
| DBH (cm) | 23.62 ± 1.94 | 25.36 ± 2.30 | |
| Age (yrs) | 51-65 | 51-65 | |
| Canopy density (%) | 0.85-0.90 | 0.80-0.85 | |
Laboratory analyses
One sub-sample from each air-dried litter bag was used to compare the litter composition by separating the litter into leaves, needles, branches, cones and herbaceous components for both P. tabulaeformis and Q. aliena stands. Then, one sub-sample from the air-dried litter bags for each site and slope position (n = 12 per species and litter horizon) was packed into porous nylon bags of known mass and weighed to calculate the corresponding gross dry litter mass (m0) of each sample. The nylon bags were immersed in a water bath for a pre-determined time and then taken out at fixed time intervals and re-weighed to determine the gross wet mass of litter (mi) after full drainage. The water-holding ratio of litter (Ri, kg kg-1) was calculated as the ratio between the total water held in the litter tissue and the dry litter mass using the following formula (eqn. 1):
where Ri is the water-holding capacity of litter at the immersion time i (i = 0.5, 1, 2, 4, 6, 8, 10, 12 and 24 h), m0 is the dry litter mass and mi is the litter mass at the immersion time i after free drainage.
The litter water-holding capacity (Rm) was defined as the highest ratio achieved when the increase of wet mass of litter was negligible with increased immersion time. In this study, the water-holding ratio rarely increased after litter was immersed in water for 24 h, thus Rm was numerically equivalent to the R24.
The total water retention capacity of the litter layer (Wm, Mg ha-1) in a given stand was calculated on the unit area basis using the following formula (eqn. 2):
where M is the unit litter mass (Mg ha-1).
Assuming the water-holding ratio of litter under ambient conditions is R0, the maximum water interception capacity of litter (Wi, Mg ha-1) was calculated as (eqn. 3):
where R0 is the water-holding capacity of the litter under ambient conditions, m1 is the fresh litter mass and m0 is the dry litter mass (eqn. 4):
Data analysis and statistics
We treated tree species (P. tabulaeformis and Q. aliena) and the litter layers (OL and OF) as fixed factors, the site (four representatives for each species) as a random factor, and the slope position (lower, middle and upper) was nested under each site. Linear mixed-effects models were fitted to explore the effects of these factors and their interactions on the litter thickness and mass of the OL layer and OF layer using the “lmerTest” package in R (version 3.4.2). We also fitted models with the total thickness and total mass of the OL layer and the OF layer combined. To check the model assumptions, we applied the normality test as well as the test of equal variance of the residuals for each model. A logarithm transformation was applied to the total thickness of litter since the original data failed the normality test. For the hydrological properties determined in the laboratory, a Student’s t-test (α=0.05) was performed.
Results
Litter composition, thickness and mass
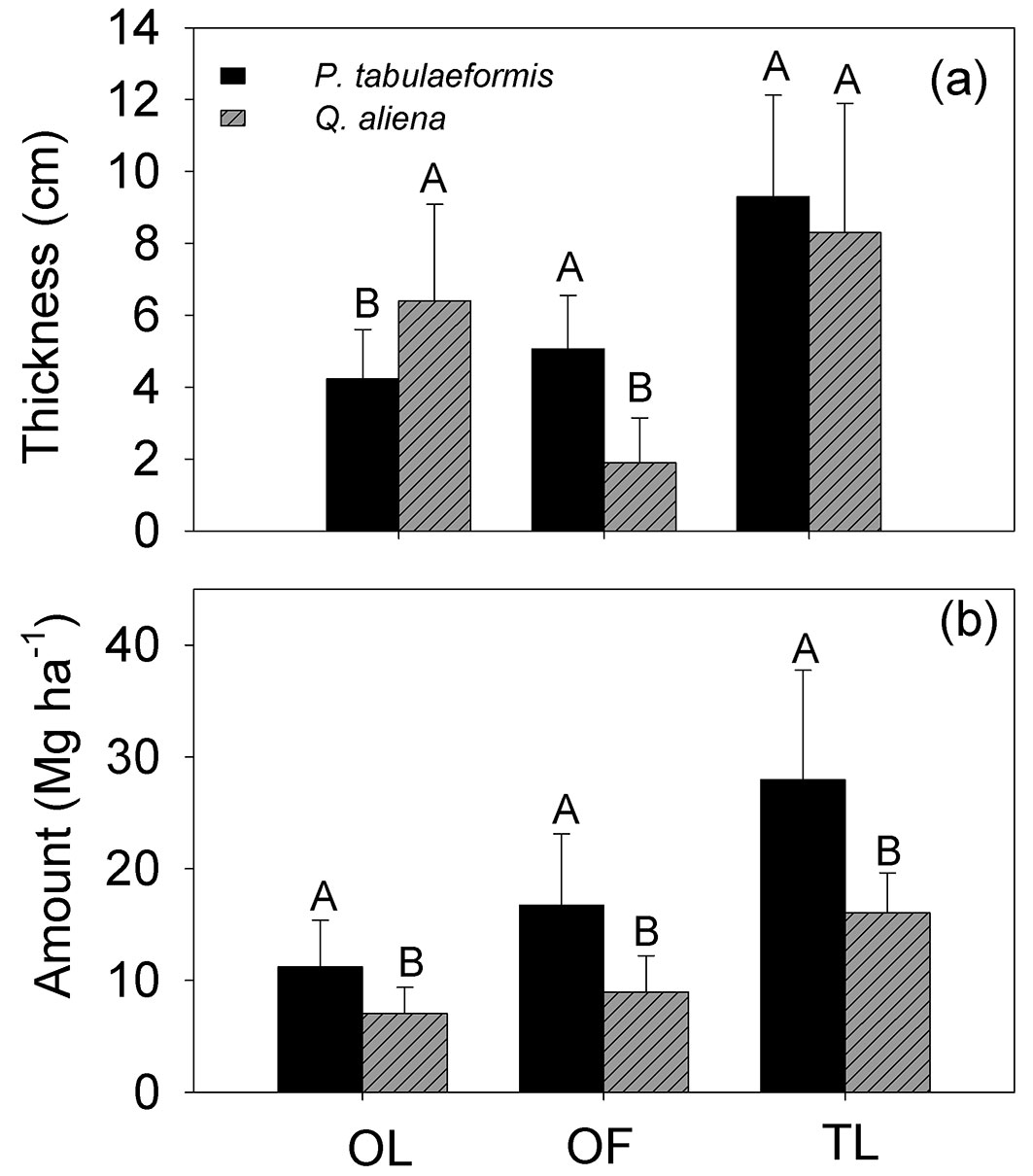
The thickness differed significantly between the OL layer and the OF layer (p = 0.001) and this difference depended on the tree species (p<0.001). There was a significant difference in litter mass between tree species and litter layer (p<0.001). The total thickness of both OL and OF layers was not significantly different between tree species (p = 0.3178), but different tree species did significantly differ in total mass of either OL and OF layers (p < 0.001 - Tab. 2). The composition of the litter layer and the defoliation period of P. tabulaeformis and Q. aliena are presented in Tab. 3. The collected litter is mainly composed of leaves or needles. The needles of P. tabulaeformis fall both during early spring and later autumn, while the leaves of Q. aliena fall mostly during the autumn. The thickness of the OF of P. tabulaeformis stand was 5.1 ± 1.5 cm (mean ± standard deviation), slightly larger than the 4.2 ± 1.4 cm thickness of its OL. The OF of Q. aliena was 1.9 ± 1.2 cm, significantly lower than the 6.4 ± 2.7 cm thickness of its OL (p = 0.001). The combined depth of the OF and OL (total litter mass, TLL) of P. tabulaeformis was 9.3 ± 2.8 cm, not significantly different from the 8.3 ± 3.6 cm depth of Q. aliena (Fig. 2a). The mass of the P. tabulaeformis litter layer was 27.94 ± 9.81 Mg ha-1, significantly higher than the 16.04 ± 3.60 Mg ha-1 mass of the Q. aliena litter layer (p = 0.001 - Fig. 2b). The mass of the OF layer was higher than the mass of the OL layer in both stand types (Fig. 2b). The mass of the OF layer was 16.74 ± 6.36 Mg ha-1 for P. tabulaeformis, accounting for 59.85 ± 8.46% of the total litter mass, and 8.98 ± 3.23 Mg ha-1 for Q. aliena, accounting for 55.49 ± 14.43% of the total litter mass.
Tab. 2 - F-statistics from factorial ANOVA to assess tree species (P. tabulaeformis and Q. aliena var. acuteserrata), litter layer (OL and OF) and slope position (lower, middle and upper) effects on the litter thickness, mass, total litter thickness and total litter mass. (df): degrees of freedom; (SS): sum of squares; (MS): mean square; (ns): p > 0.05; (*): p < 0.05; (**): p < 0.01; (***): p < 0.001
| Variable | Factor | df | SS | MS | F-value | P-value |
|---|---|---|---|---|---|---|
| Litter thickness | Species | 1, 42 | 2.8 | 2.8 | 0.367 | 0.3671 ns |
| Litter layer | 1, 42 | 41.44 | 41.44 | 0.001 | 0.001 ** | |
| Slope position | 2, 42 | 0.34 | 0.34 | 0.951 | 0.9512 ns | |
| Species × Litter layer | 1, 42 | 85.86 | 85.86 | 0 | <0.0001 *** | |
| Litter mass | Species | 1, 39 | 9.24 | 9.24 | 36.64 | <0.0001 *** |
| Litter layer | 1, 39 | 3.44 | 3.44 | 13.63 | 0.0006 *** | |
| Slope position | 2, 7.7 | 1.14 | 0.57 | 2.27 | 0.1669 ns | |
| Species × Litter layer | 1, 39 | 0.47 | 0.47 | 1.89 | 0.1767 ns | |
| Log (Total litter thickness) | Species | 1, 20 | 0.164 | 0.164 | 1.04 | 0.3178 ns |
| Slope position | 2, 20 | 0.013 | 0.006 | 0.04 | 0.9591 ns | |
| Total mass | Species | 1, 17 | 851.09 | 851.09 | 27.32 | <0.0001 *** |
| Slope position | 2, 6.43 | 119.91 | 59.96 | 1.92 | 0.221 ns |
Tab. 3 - Composition of the litter layer and defoliation period of P. tabulaeformis and Q. aliena var. acuteserrata.
| Characteristics | Vegetation type | ||
|---|---|---|---|
| P. tabulaeformis | Q. aliena var. acuteserrata |
||
| Woody litter | Leaf (%) | 86.1 | 93.4 |
| Branch (%) | 10.8 | 4.6 | |
| Cone, seed scale, others (%) | 2.7 | 1.5 | |
| Shrub and herb litter (%) | 0.4 | 0.5 | |
| Defoliation period | Mar. to Apr., Nov. to Dec. |
Sep. to Nov. | |
Fig. 2 - Comparison of the mean thickness and masses of the non-decomposed litter layer (OL), the semi-decomposed litter layer (OF), and the total litter (TL) of P. tabulaeformis and Q. aliena var. acuteserrata. The vertical bar represent the standard error (n = 12). For each category, different capital letters indicate a significant difference in the mean value between the two species.
Water-holding capacity of litter
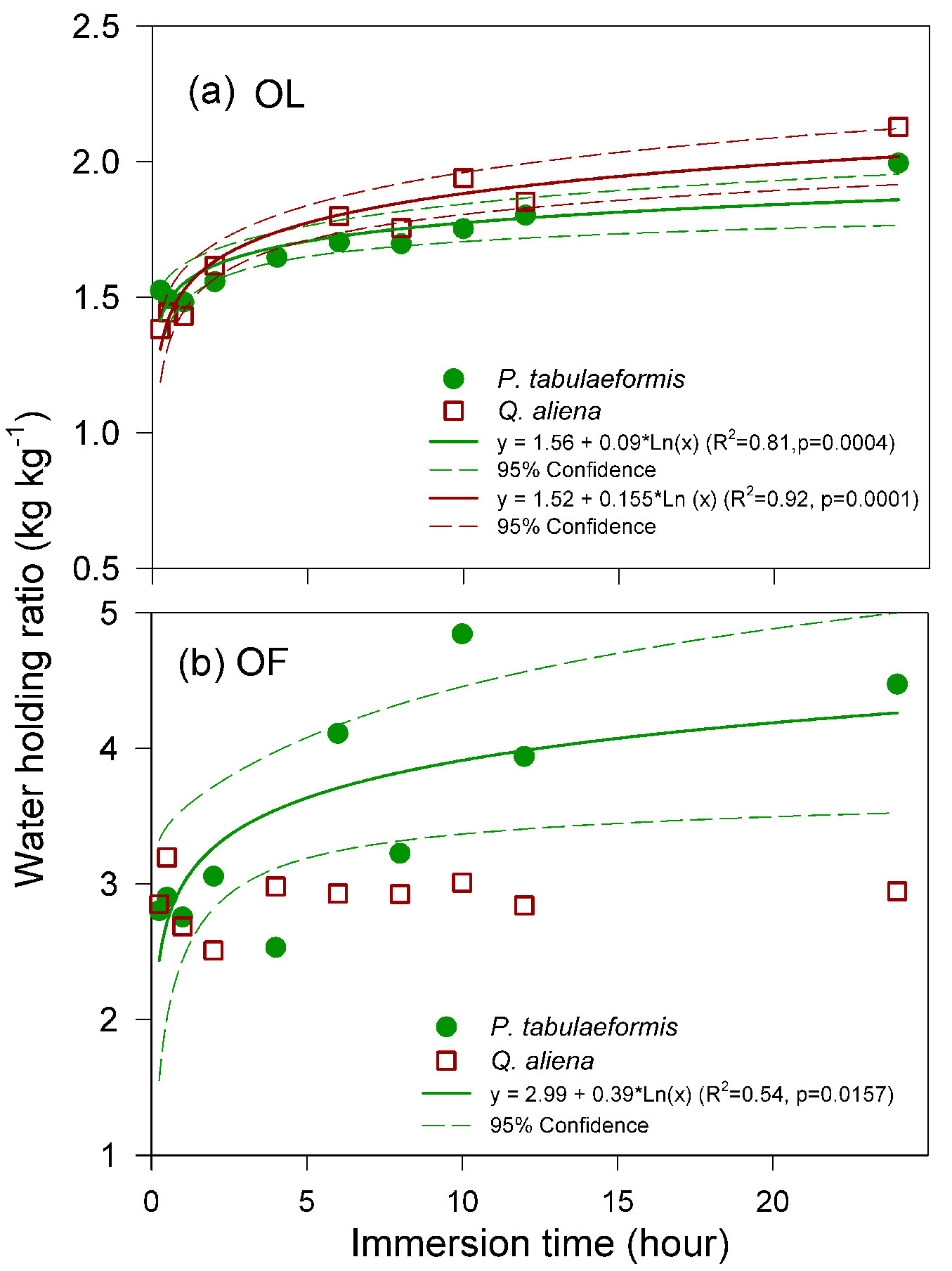
After one hour of water immersion, the water-holding ratio of OL and OF reached 74.30% and 61.61% of their holding capacity in P. tabulaeformis, respectively, while it was 67.26% and 91.26% in Q. aliena, respectively. The relationship between the water-holding ratio of OL and the time immersion in the water was fitted to a logistic model (Fig. 3a). This relationship was similar between the two types of litter. The water-holding ratio was relatively higher for OF compared to the OL at the same immersion time. However, the water-holding ratio of the OF of Q. aliena did not increase substantially with an increase in the time of water immersion (Fig. 3b) and was mostly lower than the OF of P. tabulaeformis.
Fig. 3 - Relationships between the water-holding ratio and the immersion time of (a) non-decomposed litter (OL) and (b) semi-decomposed litter (OF) collected from P. tabulaeformis stands and Q. aliena var. acuteserrata stands. Each point is the average value of 12 replicates.
Litter water-absorption rate
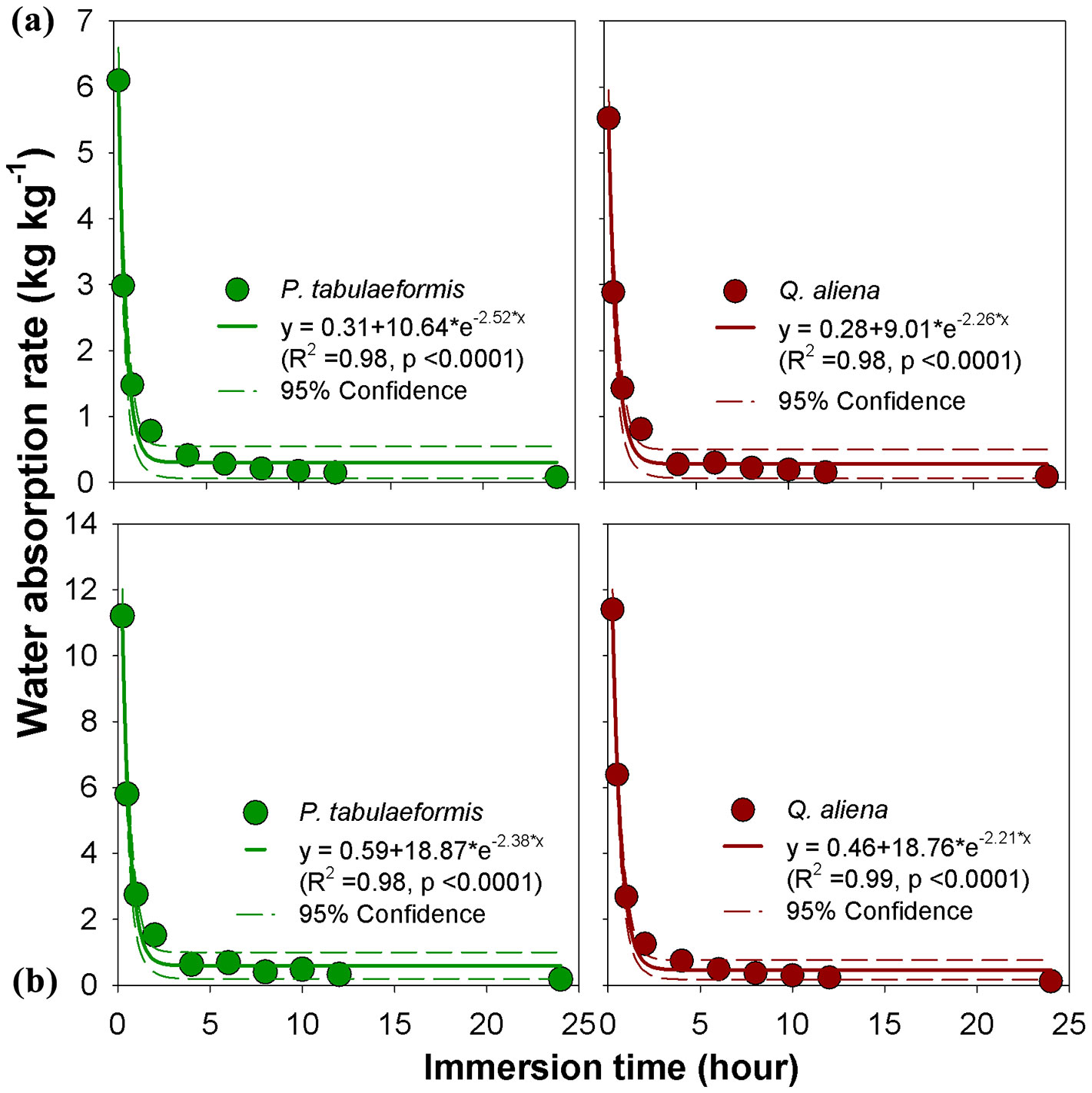
The water-absorption rates of undecomposed and semi-decomposed litter layer were highest at the beginning of the experiment and declined quickly in the first 4 hours. The rate slowed down during the time interval of 4-8 h, and was nearly unchanged after 8 h (Fig. 4). The water-absorption rate of the OL was 2.99 kg kg-1 h-1 and 2.89 kg kg-1 h-1 for P. tabulaeformis and Q. aliena, respectively, at the first 0.5 h (Fig. 4a). The water-absorption rate of the OF was 5.80 kg kg-1 h-1 and 6.39 kg kg-1 h-1 for P. tabulaeformis and Q. aliena, respectively (Fig. 4b). There were no observable differences in water-absorption rates between P. tabulaeformis and Q. aliena at the same time of water immersion.
Fig. 4 - Litter water-absorption rate of (a) non-decomposed litter (OL) and (b) semi-decomposed litter (OF) of P. tabulaeformis and Q. aliena var. acuteserrata. Each point is the average value of 12 replicates.
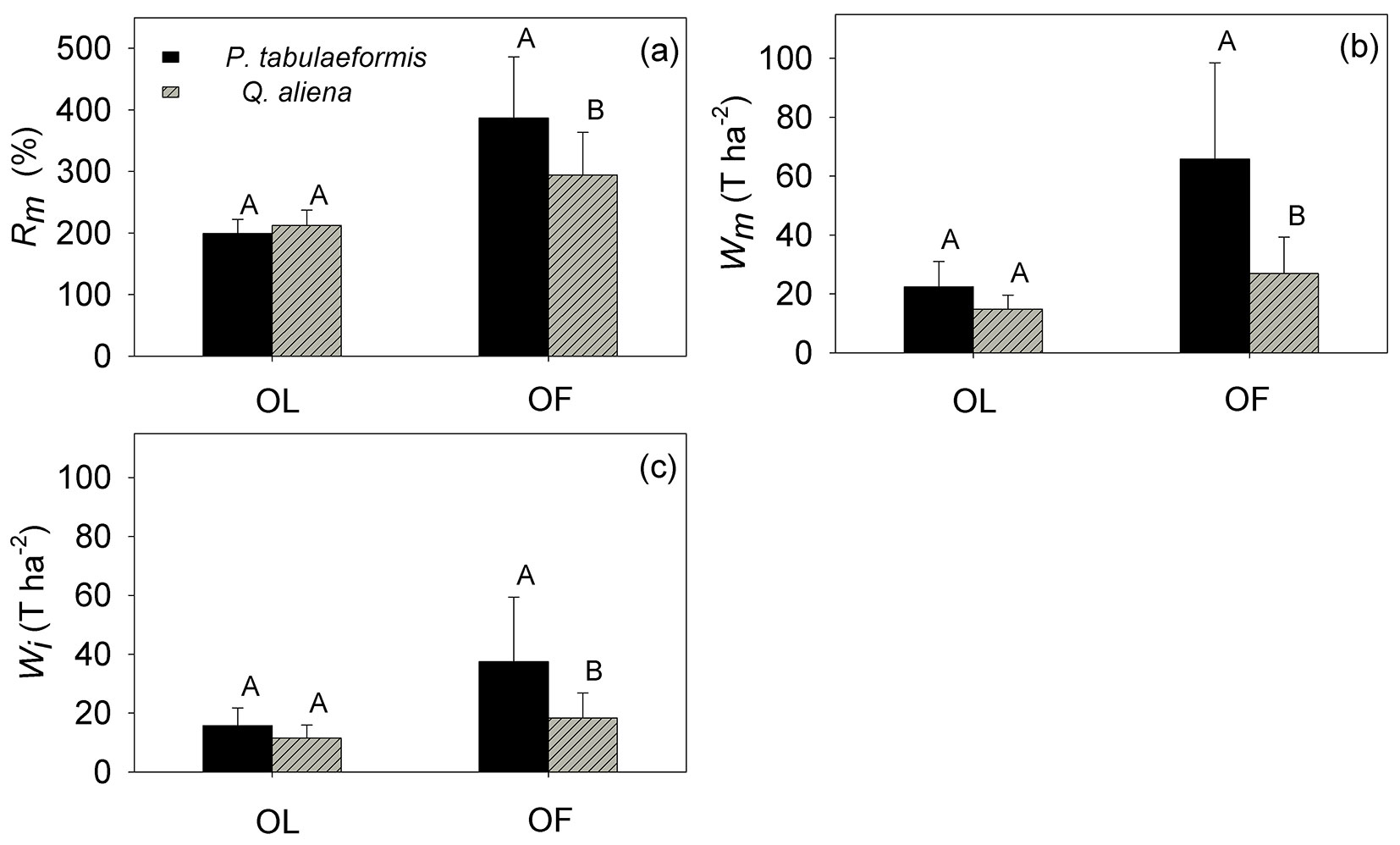
Rm, Wm and Wi
The litter water-holding capacity (Rm) of the OL was 199.45 ± 22.83% for P. tabulaeformis, not significantly different from the value observed for Q. aliena (212.76 ± 24.64%) The Rm of the OF for P. tabulaeformis was 386.80 ± 99.33%, significantly higher than 294.53 ± 69.21% for Q. aliena (p <0.05 - Fig. 5a).
Fig. 5 - (a) Maximum water-holding capacity (Rm, %), (b) maximum water retention capacity (Wm, T ha-2), and (c) maximum water interception capacity (Wi, T ha-2) of the non-decomposed litter layer (OL) and the semi-decomposed litter layer (OF) of P. tabulaeformis and Q. aliena var. acuteserrata. The vertical bar represents the standard error (n = 12). For each category, different capital letters indicate significant differences in the mean value between the two species.
The total water retention capacity (Wm) of the OL for P. tabulaeformis was 22.35 ± 8.68 Mg ha-1, which was not significantly different from 14.82 ± 4.78 Mg ha-1 for Q. aliena. The Wm of the OF was 65.87 ± 32.66 Mg ha-1 for P. tabulaeformis, significantly higher than 26.91 ± 12.38 Mg ha-1 observed for Q. aliena (Fig. 5b). The Wm of the entire litter layer for P. tabulaeformis was 88.22 ± 38.24 Mg ha-1, which was nearly two times the value of 41.74 ± 12.63 Mg ha-1 Wm for Q. aliena (Fig. 5b).
The maximum water interception capacity (Wi) of the OL was 15.82 ± 5.96 Mg ha-1 for P. tabulaeformis, which was not significantly different from 11.52 ± 4.40 Mg ha-1 for Q. aliena. The Wi of the OF was 37.53 ± 21.86 Mg ha-1 for P. tabulaeformis, which was significantly higher than the Wi detected for the OF layer in Q. aliena (18.39 ± 8.50 Mg ha-1 - Fig. 5c).
Discussion
P. tabulaeformis is an evergreen pine tree and its needles last for three to four years ([25]). The litter fall is concentrated between March and April and between November and December, with the litter fall from these two periods accounting for 50% of the annual litter fall. The needles account for more than 85% of the total litter. Contrastingly, the litter fall of the broadleaved Q. aliena is concentrated between September and November, with few leaves, dead branches and cones falling at other times ([13]). In this study, the P. tabulaeformis stands were at the near-mature stage and dead branches, cones, seeds, and flakes made up approximately 3% of the whole litter. These litter components are slower to decompose as compared with needles and leaves, and result in the accumulation of a thick litter layer and a relatively large litter mass ([31], [14]). We found that he percentage of undergrowth shrub and herbaceous litter was relatively small, only 0.4% and 0.5% in P. tabulaeformis and Q. aliena stands, respectively (Tab. 3). The litter of Q. aliena contains a high carbohydrate content and low Klason lignin content, and decomposes more easily than that of P. tabulaeformis. The leaf litter decomposition half-life is 1.26 years for Q. aliena, much shorter than the 2.28 years reported for P. tabulaeformis ([13], [12]).
The litter layer is reported to substantially reduce the evaporation from mineral soil ([22], [34]). Therefore, a thick litter layer or the conservation of the litter layer may favor moisture retention in shallow soil. On the other hand, the precipitation intercepted by the litter layer will mostly evaporate back into the atmosphere, and this loss can be substantial in a hot and dry climate ([1]). The net effect of litter on forest surface water flux deserves further study in the studied climate conditions.
Gong et al. ([11]) reported a rapid rise in the water content and a fast increase in the litter water-holding ratio at the onset of the water-immersion experiment in the laboratory. This is related to the low matrix water potential associated with drier litter. Our study showed that the absorption rate decreases exponentially when the litter becomes moist (Fig. 4). The litter in arid and semiarid ecosystems often has a low moisture content, whereby exhibits a great potential to retain the occurring rainfall. The semi-decomposed litter layer (OF) was able to quickly reach a high water-holding ratio of approximately 3 within an hour, which was higher than the non-decomposed litter layer (OL) at its holding capacity ([33]). This may be explained by the fact that the OF has a higher degree of fragmentation, a larger surface area per mass unit and is more conducive to water entrance ([21]). This characteristic is important in retaining water from high-intensity rainfall.
In this study, the OL of P. tabulaeformis stands showed a lower Rm than that of Q. aliena stands, which is consistent with previous reports ([28]). This difference in hydrological properties is likely attributed to the difference in physical and chemical properties of the needles and leaves, since the non-decomposed needles of P. tabulaeformis have thicker cuticles and a higher oil content than the leaves of Q. aliena ([10]). Additionally, our results showed that the value of Rm for non-decomposed coniferous tree species was lower than that observed for broadleaved tree species in general ([38]). In contrast, the Rm for the OF of P. tabulaeformis was nearly twice as great as the Rm for the semi-decomposed litter of Q. aliena. The OF of Q. aliena reaches its water-holding capacity quickly while it takes hours for the OF of P. tabulaeformis to reach its holding capacity. Further studies are needed to throw light on the biological and hydrological mechanisms underlying the above evidence, as well as to better understand whether such differences are either species-specific or more general between conifers and broadleaves.
While the Rm defines the effectiveness of unit mass litter in retaining water, ultimately the water retention capacity of the litter layer (Wm) depends on the litter mass ([38], [20]). Our study showed that the water-retention capacity of P. tabulaeformis is nearly two-fold that of Q. aliena as a result of greater mass and more effective OF. However, Zhang et al. ([38]) reported a smaller water-retention capacity of the semi-decomposed OF layer as compared to the non-decomposed OL layer, and Li et al. ([20]) showed a higher water retention capacity in broadleaved than in coniferous forest litter of co-occurring stands in other forest ecosystems. A comprehensive study is needed to systematically investigate litter characteristics and their hydrological properties across different climate regimes and forest ecosystems, also aiming to assess the latent heat transfer, the microclimate for microbes, and the carbon flux from forest litter.
The water interception capacity (Wi) of the litter is a hydrological property which sets the upper limit of precipitation loss to the litter layer for any given event. This value varies depending on the antecedent litter water content, which varies substantially under a hot and dry climate ([1]). A simple conversion of Wi obtained in this study suggests that the litter layer can theoretically intercept up to 53.35 mm of throughfall in P. tabulaeformis stands and up to 29.91 mm in Q. aliena stands (Fig. 5c). This provides a substantial capacity to minimize the splash erosion from high-intensity storms. Chen et al. ([4]) reported that surface runoff rarely occurs until the rainfall is above 50 mm for the P. tabulaeformis forest in the Miyun District of Beijing. There is a little surface runoff in the forested area of this study; however, it is largely unknown whether Wi will be reached in situ for a given storm. Recent development in the measurement of in situ litter water content may provide more realistic estimates of the rainfall interception by the litter ([34], [1]). Nevertheless, the Wi value may give a useful indication of the maximum litter interception and a combination of both approaches may improve our estimation of water flux in the forest ground, which is important not only for the hydrological budget but also for an improved understanding of energy partitioning and microbe-driven carbon flux in forest ecosystems.
The P. tabulaeformis and Q. aliena mixed forest analyzed in this study is at a late successional stage for this climate zone, and is the result of forest harvesting carried out in the 1960s and the 1970s ([36]). From the perspective of water resource management, our results suggest that P. tabulaeformis stands have a dense litter layer suitable to intercept rainfall and buffer storm impact. The increase in litter mass along with the development of an overgrown canopy of P. tabulaeformis may be one of the aforementioned non-climate causes and can be partially responsible for the decreasing trend in the streamflow ([23]). P. tabulaeformis is an endemic species in northern China and will eventually out-compete Q. aliena at the mid-elevations of the Qinling Mountains ([37]).
The precipitation in the Qinling Mountains area is characterized by uneven seasonal distribution and large annual variation. The majority of rainfall is concentrated from May to October, accounting for more than 85% of the annual precipitation with frequent occurrence of heavy rains, torrential rains, and other extreme weather events. Moreover, the mountainous topographic features of this area are prone to geological disasters, including floods, mudslides and landslides ([5]). From the management perspective, a thick and dense litter layer may favor the delay and decrease in the formation of surface runoff, reducing the occurrence of geological disasters. Strategic removal of some unhealthy Q. aliena trees may promote P. tabulaeformis growth, thus improving litter layer characteristics. Conversely, in areas with low precipitation, such as the Loess Plateau, the rocky mountain area of North China and other areas, the thin litter layer of forest or savanna dominated by Q. aliena may be considered favorable to rainfall infiltration and potential streamflow enhancement. Additionally, the quantity and quality of litter affects the morphology and properties of soils as well as the nutrition of plants and soil heterotrophic organisms ([15]).
The overgrown secondary forests on the Qinling Mountains are the result of the lack of management in the past, similar to the vast cypress and red cedar plantations in Japan ([17], [32]). The impact of overgrown plantations on the hydrologic budget and water quality requires further studies to sustain the multiple ecosystem services provided by forests ([32], [16]).
Conclusions
The overgrowth of the mixed forests in the Qinling Mountains causes uncertainty on their current water-conservation function. We studied the structure and hydrologic properties of the litter layer in stands of the two dominant species, P. tabulaeformis and Q. aliena, finding significant differences between them. P. tabulaeformis stands have a much thicker semi-decomposed layer with a higher water-holding capacity than Q. aliena and is likely more effective in reducing overland flow and surface erosion. The selective removal of Q. aliena trees in overgrown mixed forests may promote the growth of P. tabulaeformis and improve water quality. However, the impact of Q. aliena removal on the total evapotranspiration of the forest is unknown. Further studies should aim at directly quantifying the impact of different scenarios of tree removal and their impact on water balance and water yield using an experimental watershed approach.
Acknowledgments
This study was supported by the Fundamental Research Funds for the Central Universities, Special Fund Programs of Basic Scientific Research of Northwest A&F University (2452015043), the Natural Science Basic Research Program of Shaanxi Province of China (2014JM2-3029), Ph.D. research start-up foundation of Northwest A&F University (Z109021505), National Natural Science Funds of China (Grant No. 31600580, 31670646). The Qinling National Forest Ecosystem Research Station provided all the background information and field equipment. This work was also supported by CFERN & Beijing Techno Solutions Award Funds on excellent academic achievements. We thank Joseph Dale for language editing of the manuscript.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Tianjian Cao
Tian Gao
College of Forestry, Northwest A&F University, Yangling 712100 (China)
Qinling National Forest Ecosystem Research Station, Ningshan 711600 (China)
Chris B Zou
Ecohydrology Research Institute, The University of Tokyo Forests, Graduate School of Agricultural and Life Sciences, The University of Tokyo, Seto, Aichi 489-0031 (Japan)
Department of Statistics, Oklahoma State University, Stillwater 74078 (USA)
Tianjin Key Laboratory of Water Resources and Environment, Tianjin Normal University, Tianjin 300387 (China)
Department of Natural Resource Ecology and Management, Oklahoma State University, Stillwater 74078 (United States)
Corresponding author
Paper Info
Citation
Chen S, Cao T, Tanaka N, Gao T, Zhu L, Zou CB (2018). Hydrological properties of litter layers in mixed forests in Mt. Qinling, China. iForest 11: 243-250. - doi: 10.3832/ifor2535-011
Academic Editor
Giorgio Alberti
Paper history
Received: Jun 26, 2017
Accepted: Dec 27, 2017
First online: Mar 12, 2018
Publication Date: Apr 30, 2018
Publication Time: 2.50 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2018
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 38902
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 32939
Abstract Page Views: 1946
PDF Downloads: 3230
Citation/Reference Downloads: 18
XML Downloads: 769
Web Metrics
Days since publication: 2230
Overall contacts: 38902
Avg. contacts per week: 122.11
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Feb 2023)
Total number of cites (since 2018): 6
Average cites per year: 1.00
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Soil water deficit as a tool to measure water stress and inform silvicultural management in the Cape Forest Regions, South Africa
vol. 13, pp. 473-481 (online: 01 November 2020)
Research Articles
Soil fauna communities and microbial activities response to litter and soil properties under degraded and restored forests of Hyrcania
vol. 14, pp. 490-498 (online: 11 November 2021)
Research Articles
Hydrological simulation of a small forested catchment under different land use and forest management
vol. 13, pp. 301-308 (online: 10 July 2020)
Research Articles
Evaluation of hydrological and erosive effects at the basin scale in relation to the severity of forest fires
vol. 12, pp. 427-434 (online: 01 September 2019)
Research Articles
Effect of size and surrounding forest vegetation on chemical properties of soil in forest gaps
vol. 8, pp. 67-72 (online: 04 June 2014)
Research Articles
Influences of forest gaps on soil physico-chemical and biological properties in an oriental beech (Fagus orientalis L.) stand of Hyrcanian forest, north of Iran
vol. 13, pp. 124-129 (online: 07 April 2020)
Research Articles
Soil stoichiometry modulates effects of shrub encroachment on soil carbon concentration and stock in a subalpine grassland
vol. 13, pp. 65-72 (online: 07 February 2020)
Research Articles
Spatial heterogeneity of soil respiration in a seasonal rainforest with complex terrain
vol. 6, pp. 65-72 (online: 07 February 2013)
Research Articles
Wood-soil interactions in soil bioengineering slope stabilization works
vol. 2, pp. 187-191 (online: 15 October 2009)
Research Articles
Soil respiration along an altitudinal gradient in a subalpine secondary forest in China
vol. 8, pp. 526-532 (online: 01 December 2014)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword