Effect of soil-applied lead on mineral contents and biomass in Acer cappadocicum, Fraxinus excelsior and Platycladus orientalis seedlings
iForest - Biogeosciences and Forestry, Volume 10, Issue 4, Pages 722-728 (2017)
doi: https://doi.org/10.3832/ifor2251-010
Published: Jul 27, 2017 - Copyright © 2017 SISEF
Research Articles
Abstract
Phytoremediation is an effective and affordable approach to extract or remove lead from contaminated soil. An understanding of the physiological responses of different species subjected to heavy metal contamination is necessary before considering their use for environmental clean-up. The objective of this study was to assess the effect of lead (Pb) on growth and nutrient uptake in three forest species native to Iran: Cappadocian maple (Acer cappadocicum), European ash (Fraxinus excelsior) and Oriental aborvitae (Platycladus orientalis). The capability of lead uptake in different organs was studied in one-year-old potted seedlings grown in contaminated soils with Pb concentration ranging from 100 to 500 mg kg-1 for six months in a nursery. Several phytoextraction parameters such as translocation factor (TF), tolerance index (TI) and bioconcentration factor (BCF) were assessed to investigate the phytoremediation potential of these species. Increasing Pb application in the soil caused a gradual decrease in dry weight of leaf and shoot of all species, while the dry weight of root remains unaffected. However, such inhibition was less marked in the conifer (P. orientalis) compared to the two broad-leaf species. Phosphorus uptake of all species slightly declined in contaminated soils. Contrastingly, Pb application did not hinder nitrogen and potassium uptake in seedlings. Atomic absorption thermo electron analysis of Pb-treated plants showed an increasing Pb accumulation in all plant compartments, although the result was more evident in the tissues of P. orientalis. This species also showed the highest values for TF, TI and BCF, indicating this conifer species as a potential candidate for phytoremediation of lead-polluted soils in Iran.
Keywords
Phytoremediation, Seedling Stage, Growth, Nutrient Uptake, Lead Accumulation, Cappadocian Maple, European Ash, Oriental Arborvitae
Introduction
The increase of heavy metals in soil is one of the major environmental problems that threatens agriculture, horticulture, natural forests, plantations and finally the food chain in Iran ([38]). Lead pollution in Iranian soils has increased because of increased disposal of municipal, industrial solid as well as liquid wastes. Vehicle exhausts and mining industry also contribute towards environmental pollution ([38]). Lead is a heavy metal that can cause disorder of plant metabolism, but some plant species can absorb large amounts of Pb through roots and accumulate the element in their tissues, with no negative effect on their biology ([15], [16]). Identifying an economical, convenient and effective solution for removing or decreasing the harmful elements in polluted soils is necessary.
Phytoremediation technology is a bioremediation strategy that uses plants as filters for accumulating, immobilizing and transforming contaminants to a less harmful form. It generally uses tolerant plants to extract heavy metals from the contaminated soil, and accumulate them in plant tissues ([28]).
Although some herbaceous plants have demonstrated good resistance to heavy metals, tree species with their bigger roots may reach deeper polluted soils, making them more suitable for phytoremediation ([1]).
Previous studies ([31]) reported that some cationic channels such as calcium channels are involved in lead uptake in plant tissues. After Pb entry and accumulation in plant tissues, photosynthesis can decrease due to a lower carboxylase activity and negative effects on the metabolites of the carbon reduction cycle ([33]). As a non-essential element for plants, lead inhibits not only photosynthesis, changing enzyme activities, hormonal status and respiration, but also the metabolism and mineral nutrient balance, and finally results in decrease of plant growth and production ([25], [32], [13]). Impairment of nutrient elements leads to decreasing of plant production, and it has been reported that heavy metals, like Pb, can significantly influence the uptake and translocation of some nutrients in plants ([9], [18], [35]). Thus, the imbalance of nutrients and decreasing of plant biomass might be a convenient and reliable symptom of heavy metal toxicity in plants. On the other hand, lead accumulation in plant tissues increases with an increase in the exogenous lead level ([19]). Therefore, plant species with high accumulation ability in their organs are good candidates for heavy metal remediation.
Fewer fast-growing tree species, especially the whole family of Salicaceae, have been considered for metal remediation ([8], [22], [34]). Despite the importance of lead contamination in Iran, it remains unclear as to which native tree species can be used in phytoremediation of polluted soils. Indeed, the phytoremediation potential of many tree species native to Iran has not been investigated hitherto.
The present study focuses on the assessment of selected tree species in lead remediation from contaminated soils by surveying their biomass production and nutrient uptakes under lead pollution. Cappadocian Maple (Acer cappadocicum), European ash (Fraxinus excelsior) and Oriental arborvitae (Platycladus orientalis) are tree species native to Iran with potential interest in phytoremediation. We investigated the effect of lead exposure on mineral content (N, P, and K) as well as the percentage of carbon and Pb amount in different plant tissues, and their consequences on biomass production in one-year-old seedlings of the above-mentioned species.
Material and methods
Nursery and plant materials
The experiment was conducted in the Khoshkedaran nursery, located at the west of Mazandaran province, Iran, approximately 4 km from Nashtarood city (36° 42′ 42″ N and 51° 02′ 36.9″ E). The climate in the region is temperate humid, characterized by an annual average temperature of 16.7 °C and average annual precipitation of 1233.27 mm. A total of 180, one-year-old seedlings of Acer cappadocicum, Fraxinus excelsior and Platycladus orientalis were planted in individual pots. The pots with the 3-kg capacity were filled with sandy-clay-loamy soil (46.2: 26.4: 24.5), bearing 39.5 % moisture, pH 7.4 and EC 0.26 ds m-1. Percentage of organic carbon, organic matter and total nitrogen (N) in the soil was 1.35, 2.322 and 0.11%, respectively. Besides, the amount of absorbable phosphorus (P) and potassium (K) was 18 and 146 mg kg-1. The Pb content of the untreated soil in the nursery was found to be 12.7 mg kg-1.
Lead contamination treatments
Soil samples were first contaminated with six different concentrations of lead (0, 100, 200, 300, 400 and 500 mg kg-1), which was used in the form of Pb(NO3)2. All the pots were labeled to distinguish between treatments and then randomly arranged to avoid possible bias due to their location within the nursery. The treated seedlings grew under natural environment during the growth season (six months). The experiment was carried out in 10 replicates for each lead contamination treatment. Overall, 60 seedlings were used for each species. All treated seedlings were irrigated based on 100% field capacity once every three days.
Measurement of growth and biomass
At the end of the experiment, all seedlings were harvested and the leaf, stem and root were separated. To obtain the dry weight of each plant part, samples were oven dried at 80 °C until constant weight was achieved. Finally, the dry weight of each seedling was recorded.
Measurement of Pb in root, stem and leaf
Lead analysis in plant tissues was performed by atomic absorption thermo electron (ICE™ 3000 Series AAS, ThermoFischer Scientific, Waltham, MA, USA) according to a modified manufacturer’s method. For the sample preparation, 0.5 g of dried ground plant tissue was digested by 3.5 ml of H2SO4 and ashed in the furnace for 30 minutes at 250 °C. Finally, the ash was dissolved in HCl and lead content was determined.
Measurement of nutrients in root, stem and leaf
Ground plant samples were oven dried at 65 °C and then digested with a mixture of H2SO4, H2O2 and lithium sulfate for the determination of N, P and K ([3]). The concentration of the nutrients in the digest was measured by an atomic absorption spectrophotometer (ICE™ 3000 Series AAS). Micro-Kjeldahl method ([27]) was used for the determination of nitrogen. Phosphorus was determined by vanadomolybdo phosphoric yellow color method in the nitric acid system ([27]) and potassium was measured by a flame photometer ([17]).
Phytoextraction parameters
Phytoextraction parameters such as translocation factor (TF), tolerance index (TI) and bioconcentration factor (BCF) were examined ([36], [40], [23], [37]). The TF is the capacity of a plant to transfer metal from its roots to shoots. TI is based on the dry weight of the plant, whereas BCF is an index commonly used to determine the potential of a plant for remediation of heavy metals from contaminated soils to its root ([4]). In the current study, the TI, TF ([37]) and BCF ([4]) values for lead were calculated as follows (eqn. 1, eqn. 2, eqn. 3):
where DWPb and DWc are the dry weights of the seedlings grown in contaminated soil and control, respectively, and Cshoot, Croot and Csoil are the concentrations of lead in shoots, roots and soil, respectively.
Statistical analysis
The experiment was performed in a completely randomized design. Biomass data was analyzed by one-way ANOVA, while data of nutrient elements and Pb adsorption were analyzed by two-way ANOVA. Shapiro-Wilk’s and Levene’s test were applied to test for normality and variance homogeneity across treatments, respectively. All statistical analyses were carried out using SPSS® ver. 16.0 (IBM, Armonk, NY, USA). Differences among treatments were tested using the Duncan’s test with α=0.05.
Results
Seedling mortality
Seedling mortality was not observed after the application of lead in the soil. Symptoms of leaf senescence were less evident in A. cappadocicum, whereas other species had sufficient vitality.
Dry biomass
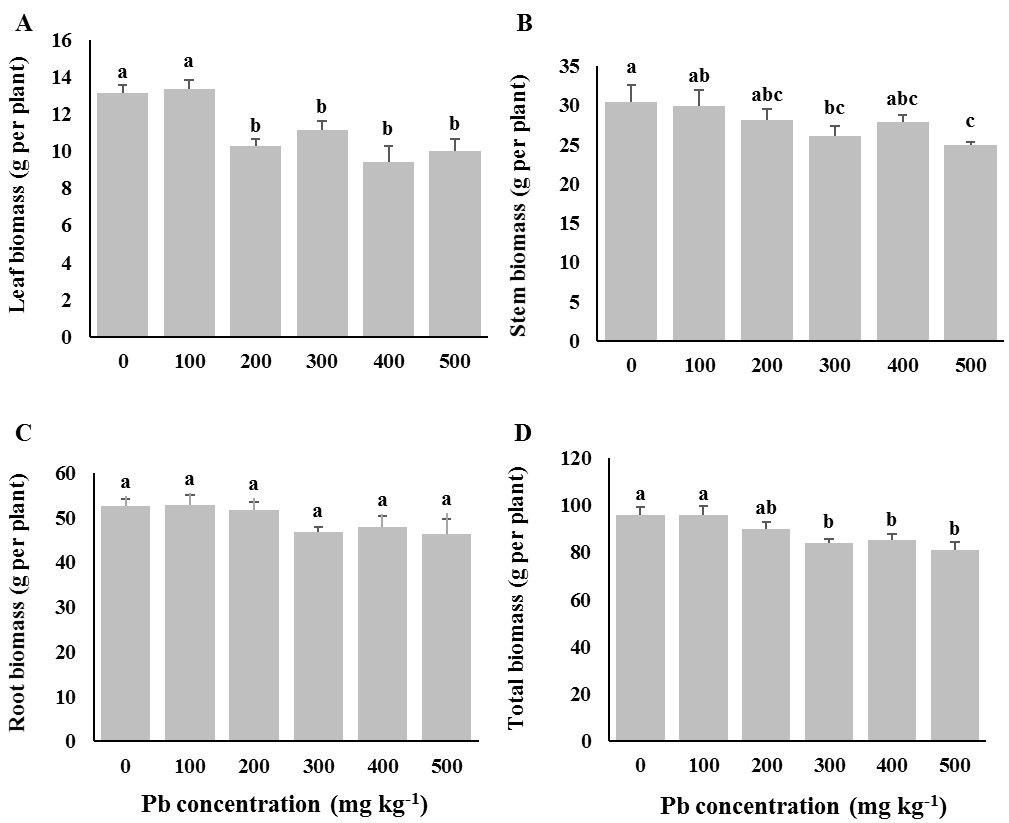
Lead contamination in the soil showed a negative effect on dry biomass in all the treated seedlings of A. cappadocicum, F. excelsior, P. orientalis (Tab. 1), but the level of adverse effects was different for each species and their parts (Fig. 1, Fig. 2 and Fig. 3). A. cappadocicum seedlings exposed to moderate and high concentrations of Pb (from 200 to 500 mg kg-1) showed a significant decrease in leaf and stem biomass, whereas the dry weight of roots did not show any considerable difference across Pb treatments (Fig. 1). The total biomass of the species was significantly affected by soil Pb at concentration ≥ 200 mg kg-1.
Tab. 1 - One-way ANOVA of plant biomass of the three species grown in lead contaminated soil. Only F values are presented. (*): p<0.05; (**): p<0.01; (ns): not significant.
| Biomass | P. orientalis | F. excelsior | A. cappadocicum |
|---|---|---|---|
| Leaf | 17.3** | 4.319** | 8.652** |
| Stem | 1.307 ns | 2.916* | 3.24* |
| Root | 0.428 ns | 9.209* | 1.667 ns |
| Total | 5.04** | 10.159* | 4.882** |
Fig. 1 - Dry biomass in root, shoot, and leaf of Cappadocian Maple (Acer cappadocicum) seedlings grown at different Pb concentrations in the soil. Control seedlings (“0” treatment) received 12 mg kg-1 Pb from untreated soils in the nursery. Different letters indicate significant differences (P< 0.05) among Pb treatments after Duncan’s tests.
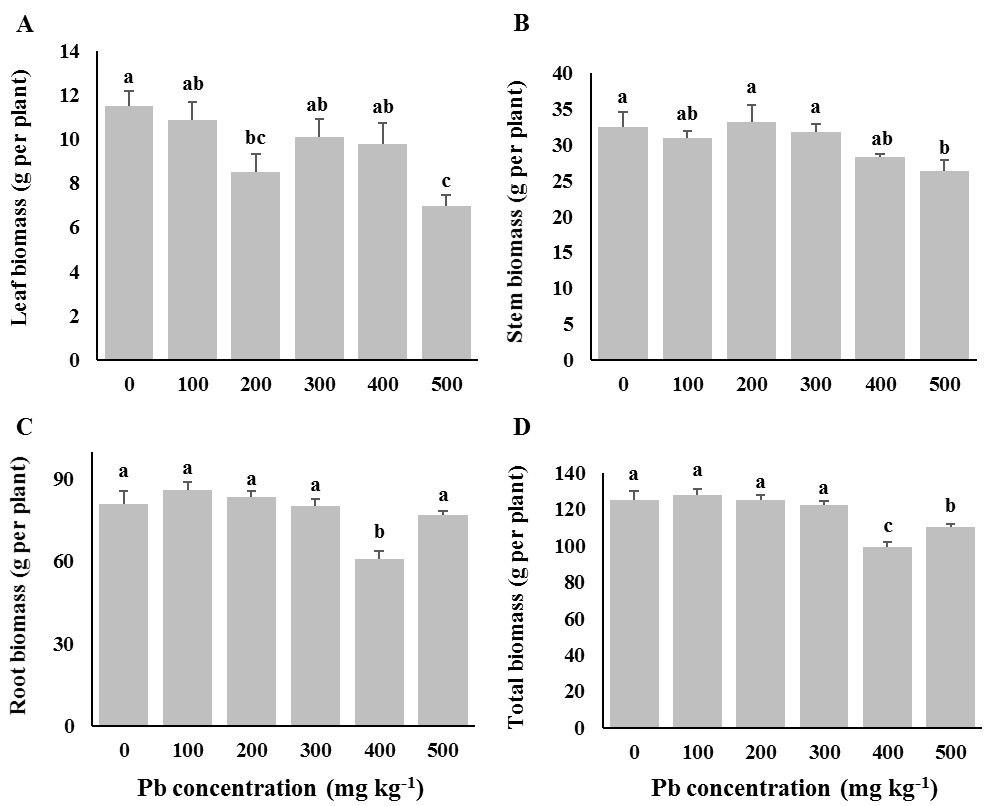
Fig. 2 - Dry biomass in root, shoot, and leaf of European ash (Fraxinus excelsior) seedlings grown at different Pb concentrations in the soil. Control seedlings (“0” treatment) received 12 mg kg-1 Pb from untreated soils in the nursery. Different letters indicate significant differences (P< 0.05) among Pb treatments after Duncan’s tests.
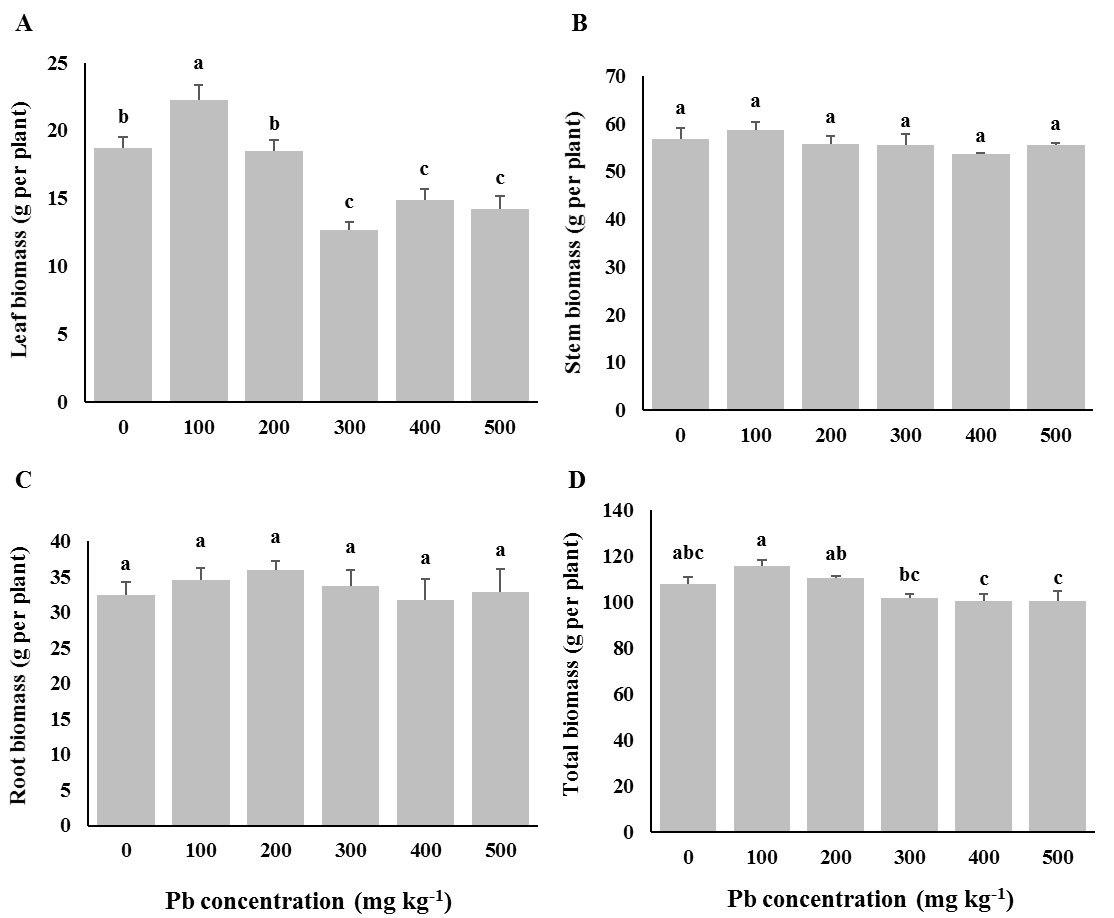
Fig. 3 - Dry biomass in root, shoot, and leaf of Oriental arborvitae (Platycladus orientalis) seedlings grown at different Pb concentrations in the soil. Control seedlings (“0” treatment) received 12 mg kg-1 Pb from untreated soils in the nursery. Different letters indicate significant differences (P< 0.05) among Pb treatments after Duncan’s tests.
Exposure of F. excelsior seedlings to 500 mg kg-1 of soil Pb concentration declined the dry biomass of shoots and leaves (Fig. 2), while no significant changes were detected for low and moderate concentrations of Pb. Root biomass of seedlings of this species was significantly affected at 400 mg kg-1 of soil Pb concentration, while root biomass at 500 mg kg-1 of soil Pb concentration was not significantly different from that observed in the control. As expected, the minimum total biomass in this species was observed for seedlings subjected to 400 and 500 mg kg-1 Pb in the soil.
An inhibitory effect of Pb on leaf biomass of P. orientalis seedlings was observed, compared to control seedlings, while the effect of Pb contamination on stem and root was not significant. On the other hand, the total dry biomass of the species did not show considerable modifications in response to increasing Pb concentrations (Fig. 3).
Lead uptake and nutrient element content
The results of two-way ANOVA indicated that Pb uptake and N, P and K content in seedlings of all species were significantly different among Pb treatments and different plant organs (Tab. 2). Also, the interaction effect of Pb treatment and plant organ was significant for all species and element contents, except for K in Platycladus orientalis.
Tab. 2 - Two-way ANOVA of nutrient content and Pb uptake in plant organs of the three species subjected soil lead contamination. Only F values are presented. (**): p<0.01.
| Species | Element | Plant organ × Pb treatment |
Pb treatment | Plant organ |
|---|---|---|---|---|
| Acer cappadocicum | N | 7.28** | 13.56** | 11.152** |
| P | 8.92** | 7.25** | 308.7** | |
| K | 3.39** | 5.38** | 640.8** | |
| Pb | 31.48** | 123.12** | 438.59** | |
| Fraxinus excelsior | N | 5.00** | 7.97** | 1949.3** |
| P | 12.67** | 26.64** | 501.08** | |
| K | 6.22** | 17.2** | 873.2** | |
| Pb | 45.3** | 104.08** | 794.73** | |
| Platycladus orientalis | N | 2.04** | 8.92** | 699.1** |
| P | 5.21** | 23.27** | 334.38** | |
| K | 0.73ns | 2.69* | 19.21** | |
| Pb | 111.83** | 329.71** | 1467.73** |
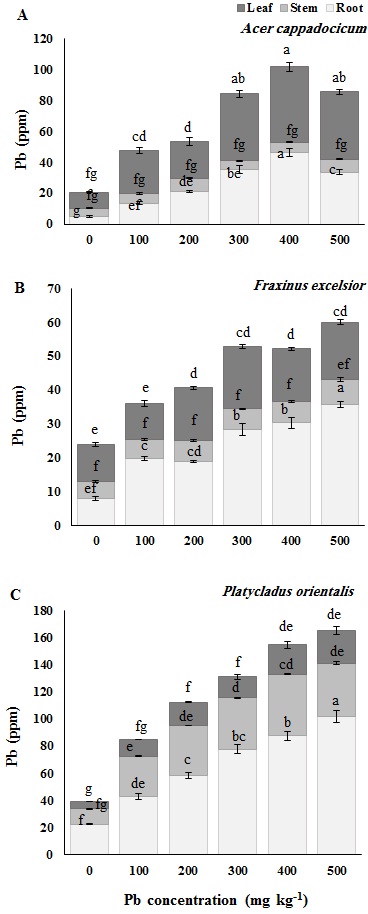
Concentrations of lead in leaf, stem and root of seedlings of each species across the different Pb treatments are shown in Fig. 4. The results showed that the Pb concentration in controls were lowest in Cappadocian maple (A. cappadocicum) and highest in Oriental arborvitae (P. orientalis). The results of Duncan’s test clearly indicated that the application of lead to soil significantly increased the concentration of this element in the roots, shoots and leaves, as compared with the relative control, in all the studied species. Total lead concentrations reached 60 mg kg1 dry matter in F. excelsior seedlings and 120 mg kg1 dry matter in P. orientalis at 500 mg kg-1 Pb in the soil, while the highest amount of lead for A. cappadocicum seedlings was recorded at soil Pb concentration of 400 mg kg-1, reaching 100 mg kg1 dry matter. The lowest content of lead was recorded in stems of A. cappadocicum and F. excelsior and in the leaves of P. orientalis. It has also been observed that only P. orientalis could accumulate a high amount of Pb in the stem.
Fig. 4 - Pb concentration (mg kg-1 or ppm) in different tissues of Cappadocian Maple (Acer cappadocicum - A), European ash (Fraxinus excelsior - B) and Oriental arborvitae (Platycladus orientalis - C) seedlings at the end of growing season. Control seedlings (“0” treatment) received 12 mg kg-1 Pb from untreated soils in the nursery. Different letters indicate significant differences (P< 0.05) among treatments after Duncan’s test.
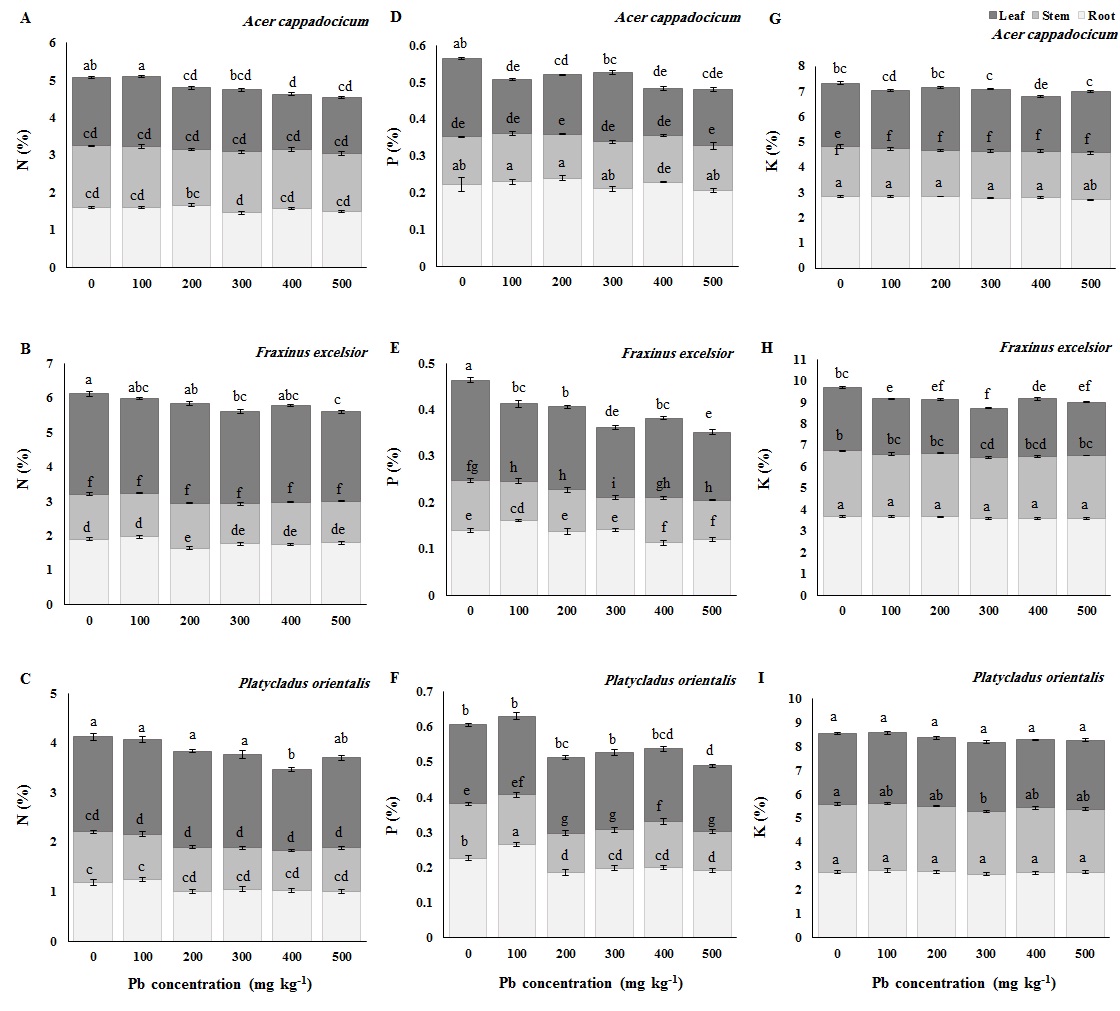
Comparison of macroelements (N, P and K) mean content in different plant organs among Pb treatments is represented in Fig. 5. The results of Duncan’s test revealed a negligible difference among treatments in the amount of nitrogen (N) in various tissues of all the three species (Fig. 5A-B-C). In contrast, total phosphorus (P) content significantly declined with increasing lead supply in all the species (Fig. 5D-E-F). The decrease in P concentration in the different organs of P. orientalis was more prominent than in the other species, whereas the K content was stable in various tissues of the three species across Pb treatments (Fig. 5G-H-I).
Fig. 5 - Macro-element content (nitrogen: A, B, C; phosphorus: D,E,F; potassium: G, H, I) in leaves, stem and roots of Cappadocian Maple (Acer cappadocicum - top), European ash (Fraxinus excelsior - middle) and Oriental arborvitae (Platycladus orientalis - bottom) seedlings grown at different Pb concentrations in the soil (0, 100, 200, 300, 400, 500 mg kg-1). Control seedlings (“0” treatment) received 12 mg kg-1 Pb from untreated soils in the nursery. Different letters indicate significant differences (P< 0.05) among treatments after Duncan’s test.
Phytoextraction parameters
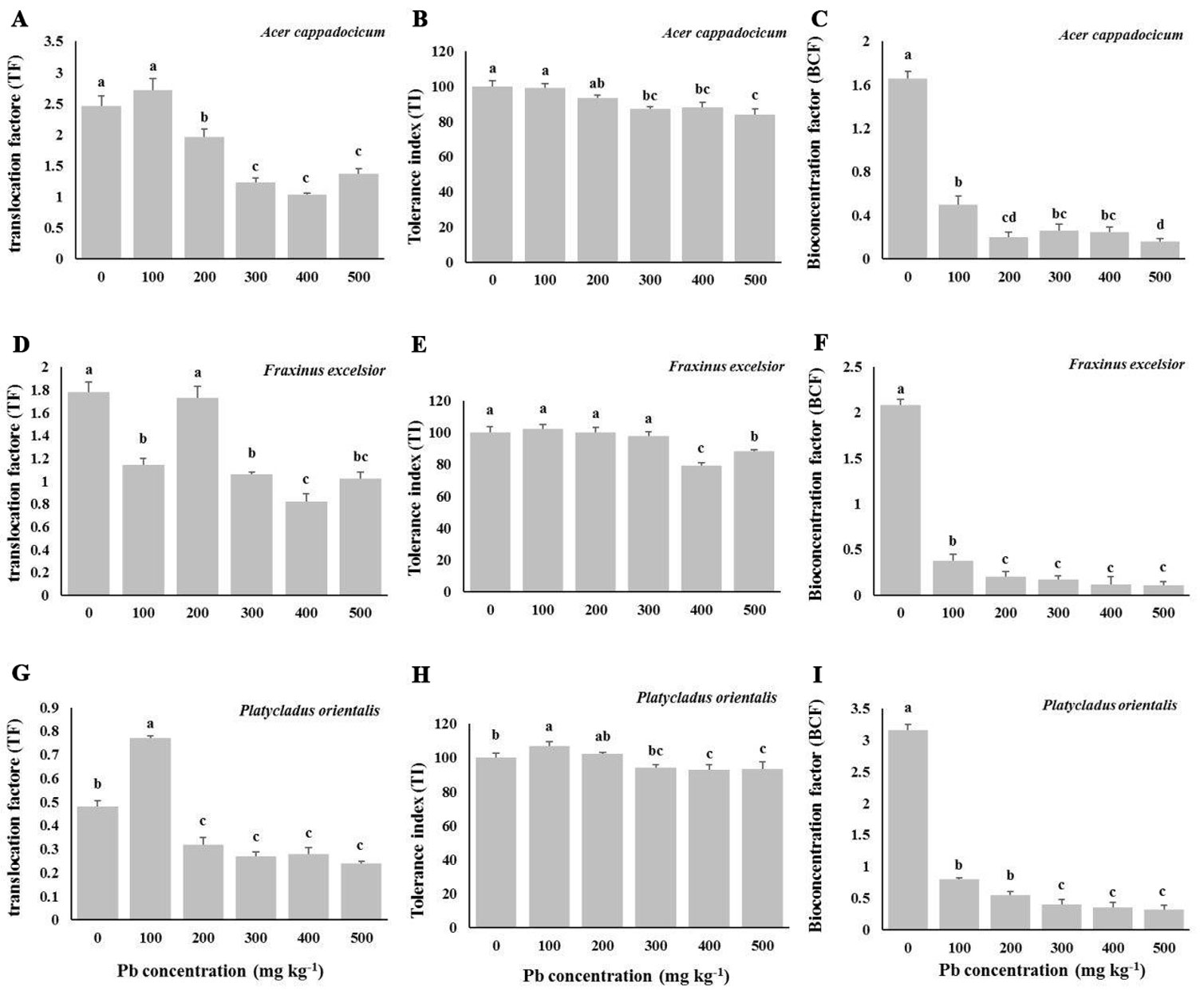
Lead contamination in the soil significantly affected the Translocation Factor (TF), Tolerance Index (TI) and Bio-Concentration Factor (BCF - Tab. 3). Lead contamination decreased the seedling TF in all the species (Fig. 6). Interestingly, low level of contamination (100 mg kg-1 Pb treatment) resulted in a considerable improvement of TI in the seedlings of P. orientalis. TI values ranged from 80 to 100 in all the treatments (Fig. 6B-E-H). The lowest values of TI were recorded in the 400 and 500 mg kg-1 Pb treatments in all the species, with the minimum value (TI=80) recorded for F. excelsior at 400 mg kg-1 Pb. Maximum TI was found in the control and sometimes in the low-level contamination treatments, which showed no significant difference with their respective controls at 5% level.
Tab. 3 - One-way ANOVA of phytoextraction parameters of the three studied species under different lead contamination levels. Only F values are presented. (**): p<0.01.
| Parameter | Platycladus orientalis | Fraxinus excelsior | Acer cappadocicum |
|---|---|---|---|
| Translocation factor (TF) | 17.017** | 29.39** | 32.529** |
| Tolerance index (TI) | 5.04** | 10.159** | 6.103** |
| Bioconcentration factor (BF) | 127.16** | 280.37** | 151.64** |
Fig. 6 - Effect of lead contamination in the soil on Translocation Factor (TF - A, D, G), Tolerance Index (TI - B, E, H) and Bioconcentration Factor (BCF - C, F, I) in seedlings of the three studied species (Acer cappadocicum - top; Fraxinus excelsior - middle; Platycladus orientalis - bottom). Control seedlings (“0” treatment) received 12 mg kg-1 Pb from untreated soils in the nursery. Different letters indicate significant differences (P< 0.05) among treatments after Duncan’s test.
The highest mean value of BCF was estimated for the control seedlings, while the lowest at 500 mg kg-1 Pb (Fig. 6C-F-I). Increasing the Pb amount in soil significantly decreased BCF across all the studied species. The highest value of BCF (3.16) was observed in control seedlings of P. orientalis.
Discussion
Growth disorders and biomass reductions are commonly observed in plants subjected to high levels of metal contamination ([24]). The decline in plant growth occurs due to the adverse effect of heavy metals on cell division and may be due to their influence on DNA, RNA or protein metabolism ([5]). In this study, total dry biomass of seedlings declined by 14, 8 and 5% for A. cappadocicum, F. excelsior and P. orientalis, respectively, when exposed to lead at high concentration (500 mg kg-1 Pb). Malar et al. ([20]) reported that plant biomass is a good indicator of plant growth performance in presence of heavy metals. Accordingly, we observed an adverse effect of lead on seedling growth, which became more evident at the higher concentrations of lead in the soil. Among the studied species, growth inhibition was lower in P. orientalis compared to the other two species, while seedling mortality was not observed in any species. Roots of studied tree species seemed to be more tolerant to lead pollution than shoots and leaves. Indeed, we observed that the dry biomass of seedling roots was not significantly affected by increasing Pb concentrations in the soil, whereas leaf biomass was more sensitive to the lead contamination.
Lamhamdi et al. ([19]) reported that the decrease of plant dry weight can be essentially attributed to a deficiency of some nutrient elements, especially N, P and K, resulting from the inhibition of their uptake under Pb exposure. In this study, lead contamination significantly affected N, P and K adsorption, but the contamination could not remarkably inhibit the uptake of essential elements, so much as P content in P. orientalis seedlings varied across treatments by a negligible amount. Contrastingly, Akinci et al. ([2]) and Päivöke ([25]) observed a negative correlation between P uptake and lead concentration in the soil.
Our results clearly showed that a remarkable deficiency of N, P and K did not occur when seedlings were subjected to lead contamination, though a slight reduction in plant biomass was observed, particularly for broad-leaved species (A. cappadocicum and F. excelsior). Lead contamination results in phytotoxicity primarily by inactivating enzymes and causing physiological changes ([26], [29]). Therefore, we conclude that the presence of the heavy metal Pb in soil can have some inhibitory effects on physiological and biochemical parameters of the three studied species. Lead can create physical barriers and disturb the physiology of root absorption, thereby inhibiting the uptake of many essential ions ([12], [7], [11]). Pb toxicity disrupts the uptake of other macroelements (Ca, Mg and Na) or microelements (Fe, Cu, and Zn), which are essential for plant growth ([30], [6]).
The observed ability of Pb bioaccumulation suggests the existence of an efficient hyper-accumulation mechanism in the studied species, which allows for removal of the heavy metal from contaminated soils ([20]). Our findings clearly showed the ability of P. orientalis in phytoremediation of highly-contaminated soil. Furthermore, the amount of Pb accumulated by P. orientalis was more than two times as compared to A. cappadocicum and F. excelsior.
The Translocation Factor (TF) reflects the capacity of a plant to transfer metal from its roots to shoots ([10]). We found that TF declined for all species in response to increasing levels of Pb concentration in soil. Interestingly, TF significantly increased in P. orientalis when seedlings were subjected to 100 mg kg-1 Pb. The Tolerance Index (TI) is assumed to equal 100 when there is no influence of the treatment on growth, whereas it takes values higher or lower than 100 in the case of positive or negative effects of the treatment, respectively ([39]). We observed that TI values slightly decrease in all the species with increasing soil Pb contamination, the lowest TI values being for F. excelsior at 400 and 500 mg kg-1 Pb. On the other side, TI for P. orientalis was close to 100 even when the plant was subjected to a high concentration of the heavy metal.
Bioconcentration factor (BCF) is defined as the ratio of heavy metal concentration in plant organs to that in the soil ([21]). In this study, BCF varied across species and Pb concentrations in the soil, with values < 1 in all treatments except in control. BCF decreased with increasing Pb concentration in the soil, which suggests a restriction in soil-root transfer at higher Pb concentrations in the soil ([14]). Although BCF was < 1 in all the three studied species, P. orientalis was more successful in the extraction of lead from the soil.
Conclusion
Our findings indicate that lead treatments even at high concentrations do not induce a significant disturbance in N, P and K uptake by plants. Moreover, we observed negative effects of lead contamination on plant production in all the three species.
The lack of mortality and the lack of a significant reduction in growth suggest that the studied species are promising in phytoremediation of lead-polluted soils, although Oriental arborvitae (Platycladus orientalis) appears to be far more resistant to lead contamination. Indeed, this conifer showed a larger Pb uptake and accumulation in tissues, while nutrient uptake, growth and biomass production did not change considerably. Further, seedlings of P. orientalis showed higher TI values compared to the two broadleaf species, and a higher lead translocation from root to stem, according to TF and BCF. Finally, the two broadleaves showed a lower lead accumulation in their stems as compared to the conifer species.
Therefore, it is concluded that Platycladus orientalis exhibits more relative resistance to lead contamination and is more suitable for phytoremediation of lead-polluted soils as compared to Acer cappadocicum and Fraxinus excelsior. Further studies are needed to explore its response to more severe contaminations under prolonged exposure time.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Authors’ Info
Authors’ Affiliation
Mohammad Reza Pourmajidian
Seyed Mohammad Hodjati
Asghar Fallah
Department of Forestry, Faculty of Natural Resources, Sari Agriculture and Natural Resource University, Sari (Iran)
Department of Biotechnology, Gurucharan College, Silchar (India)
Corresponding author
Paper Info
Citation
Abbasi H, Pourmajidian MR, Hodjati SM, Fallah A, Nath S (2017). Effect of soil-applied lead on mineral contents and biomass in Acer cappadocicum, Fraxinus excelsior and Platycladus orientalis seedlings. iForest 10: 722-728. - doi: 10.3832/ifor2251-010
Academic Editor
Claudia Cocozza
Paper history
Received: Oct 15, 2016
Accepted: May 04, 2017
First online: Jul 27, 2017
Publication Date: Aug 31, 2017
Publication Time: 2.80 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2017
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 38666
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 32616
Abstract Page Views: 1752
PDF Downloads: 3420
Citation/Reference Downloads: 17
XML Downloads: 861
Web Metrics
Days since publication: 2462
Overall contacts: 38666
Avg. contacts per week: 109.94
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Feb 2023)
Total number of cites (since 2017): 9
Average cites per year: 1.29
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Heavy metal accumulation characteristics of Nepalese alder (Alnus nepalensis) growing in a lead-zinc spoil heap, Yunnan, south-western China
vol. 7, pp. 204-208 (online: 27 February 2014)
Review Papers
A review of the performance of woody and herbaceous ornamental plants for phytoremediation in urban areas
vol. 13, pp. 139-151 (online: 14 April 2020)
Research Articles
Effect of cadmium (Cd) and lead (Pb) soil contamination on the development of Hymenoscyphus fraxineus on Fraxinus excelsior and F. angustifolia seedlings
vol. 16, pp. 307-313 (online: 09 November 2023)
Research Articles
Analysis of biometric, physiological, and biochemical traits to evaluate the cadmium phytoremediation ability of eucalypt plants under hydroponics
vol. 10, pp. 416-421 (online: 24 March 2017)
Review Papers
Monitoring the effects of air pollution on forest condition in Europe: is crown defoliation an adequate indicator?
vol. 3, pp. 86-88 (online: 15 July 2010)
Research Articles
Mapping Cadmium distribution in roots of Salicaceae through scanning electron microscopy with x-ray microanalysis
vol. 4, pp. 113-120 (online: 01 June 2011)
Research Articles
Nutrient uptake, allocation and biochemical changes in two Chinese fir cuttings under heterogeneous phosphorus supply
vol. 11, pp. 411-417 (online: 05 June 2018)
Review Papers
Heavy metals and woody plants - biotechnologies for phytoremediation
vol. 4, pp. 7-15 (online: 27 January 2011)
Research Articles
Preliminary results of the tolerance to inorganic contaminants and phytoextraction potential of twelve ornamental shrub species tested on an experimental contaminated site
vol. 11, pp. 442-448 (online: 18 June 2018)
Research Articles
A comparison between stomatal ozone uptake and AOT40 of deciduous trees in Japan
vol. 4, pp. 128-135 (online: 01 June 2011)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword