Does degradation from selective logging and illegal activities differently impact forest resources? A case study in Ghana
iForest - Biogeosciences and Forestry, Volume 9, Issue 3, Pages 354-362 (2016)
doi: https://doi.org/10.3832/ifor1779-008
Published: Jan 29, 2016 - Copyright © 2016 SISEF
Research Articles
Abstract
Degradation, a reduction of the ecosystem’s capacity to supply goods and services, is widespread in tropical forests and mainly caused by human disturbance. To maintain the full range of forest ecosystem services and support the development of effective conservation policies, we must understand the overall impact of degradation on different forest resources. This research investigates the response to disturbance of forest structure using several indicators: soil carbon content, arboreal richness and biodiversity, functional composition (guild and wood density), and productivity. We drew upon large field and remote sensing datasets from different forest types in Ghana, characterized by varied protection status, to investigate impacts of selective logging, and of illegal land use and resources extraction, which are the main disturbance causes in West Africa. Results indicate that functional composition and the overall number of species are less affected by degradation, while forest structure, soil carbon content and species abundance are seriously impacted, with resources distribution reflecting the protection level of the areas. Remote sensing analysis showed an increase in productivity in the last three decades, with higher resiliency to change in drier forest types, and stronger productivity correlation with solar radiation in the short dry season. The study region is affected by growing anthropogenic pressure on natural resources and by an increased climate variability: possible interactions of disturbance with climate are also discussed, together with the urgency to reduce degradation in order to preserve the full range of ecosystem functions.
Keywords
Tropical Forest, Remote Sensing, Degradation, Logging, Guild, Africa
Introduction
Forest degradation is defined as a reduction of the ecosystem’s capacity to supply goods and services ([23]). The West African forest belt is a biodiversity hotspot and a provider of fundamental ecosystem services, now degraded and reduced to a patchwork of forest fragments ([12]). Selective logging is possibly the primary cause of degradation in African forests with recognized impacts on carbon stocks ([11], [38]) and fine root production ([44]). In Ghana, concessions interest more than half of the reserves ([37]). Impacts on resources other than carbon, such as tree species richness, diversity, functional composition, and productivity are less clear: they are difficult to quantify and vary with disturbance type, intensity and the area under analysis. With respect to biodiversity, varied research results were reported for disturbed forests in Ghana ([2], [65]), and differentiated according to forest types ([7]). For functional composition, a drought-driven shift toward drier forests has been reported by Fauset et al. ([24]); while for net primary productivity, both climate-driven global increase ([58]) and intact African forests biomass increase ([47]) have been reported. Selective logging is also known to decrease productivity through larger trees removal ([54]).
Our research investigates the effects of selective logging and illegal land use and resources extraction (here defined as “illegal activities”) on forest resources, analyzing: forest structure and biomass; soil carbon content, tree richness and biodiversity, functional composition (guild and wood density); and productivity. With recent field data from two national parks and two forest reserves, we investigated whether the considered disturbance drivers differently affected forest resources, and the effectiveness of protection measures. With historical field data from the two reserves, we analyzed temporal changes in basal area and guild composition. Thirty years of remote sensing data allowed to study productivity trends in ten forests, mostly disturbed reserves, of southwestern Ghana and their relationships with precipitation, temperature and cloud cover.
We tested the following four hypotheses: (i) impacts on forests’ mean structure parameters, mean soil carbon (C) content, tree species’ richness and diversity, and guilds and wood density, were stronger in the less protected areas; (ii) a significant increase in basal area occurred in the reserve (Bia) where logging activities stopped about two decades ago, while its significant decrease occurred in the reserve affected by continuous illegal activities; (iii) a change in guilds composition in the direction of recovery occurred in the logged reserve, while a change toward further degradation occurred in the illegally disturbed one; (iv) the productivity in the ten forests increased over the last 30 years.
Methodology
We used 96 recent (2012-2013) and 94 historical (1987 and 1991) field plots. In the recent dataset we collected species’ name, diameter at breast height (DBH), and height (H) information in four areas in a ~150 km range on a south to north gradient (blue squares in Fig. S1 - Appendix 1): (i) the wet evergreen Ankasa National Park (NP), where logging was scarce and confined to the southern range; (ii) the moist evergreen Dadieso Forest Reserve (FR), surrounded by and including communities and cocoa farms which cause frequent illegal activities, such as timber extraction, hunting, gaps opening for cultivation, with presence of swampy zones; (iii) the moist evergreen Bia Resource Reserve (RR), selectively logged in 1980-90 and possibly also few years after; (iv) the moist semi-deciduous Bia National Park, where there are no logging records but fire, drought and elephants cause natural disturbance. Historical basal area and species records were collected by the Ghana Forestry Commission in one hectare plots: 59 in the Bia RR and 35 in Dadieso FR (dataset described in [34]). We collected orthophotos over the recent plots (10 cm spatial resolution) in March 2012 with an aerial survey: we screened for signs of disturbance (logging trails, large gaps, and irregular canopy height), also reviewing information gathered from rangers and villagers to check the scarce disturbance official records. This was done by visual interpretation of images and also by interaction with the local community.
Forest structure and guild data
In the recent plots, DBH, H, and species records were collected for trees having DBH>10 cm. For the 1600 m2 plots, 400 m2 subplots were defined for sampling also trees with 10-20 cm DBH (Tab. S1 - Appendix 1). Height was collected using a laser hypsometer. In total, we sampled 11.79 ha and 1819 trees for DBH>20 cm and 8.51 ha and 1988 trees for DBH in 10-20 cm range (Tab. S2 - Appendix 1 ). In historical plots all trees above 30 cm DBH were sampled, and trees in the 10-30 cm DBH range were collected in subplots of 0.2 ha.
Ordination scores for tree species (from [29], [30]) weighted by stem abundance were used to obtain scores for plots ([36]). This approach was used by Hawthorne ([35]) to define a wet to dry gradient across Ghana. We ordinated plot data using the Nonmetric Multidimensional Scaling (NMS) statistical method using the PCORD ver. 6 software ([53]).
From field records we calculated: wood density (WD, Global Wood Density Database - [16]); basal area (BA); above ground biomass (AGB, [15]); and tree guilds according to the Hawthorne ([34], [35], [36]) classification, which identifies pioneer (P), non-pioneer light-demanding (NPLD), shade-bearer (SB), and swamp-resistant (SR) species. Mean parameters difference among areas were tested in recent data using the Mann-Whitney (MW) U-test and the Kolmogorov-Smirnov (KS) test to additionally detect differences in the shape of distributions ([42]). We preferred non-parametric statistics to account for non-normal distributions and outliers, represented by large old trees. The two DBH classes (10-20 cm and over 20 cm) were tested separately at 95% confidence level, using the normal approximation to compute the p-value, as samples are large in statistical terms. Distributions of H, DBH, and WD were estimated with a kernel density method based on the Gaussian kernel and using the Silverman’s rule for bandwidth selection ([69]). For AGB and BA, which are density dependent, we reported the normalized cumulative values per hectare. The following pairs were tested: Ankasa vs. Bia NPs, with limited disturbance occurrence; Bia NP vs. Bia RR, with different disturbance levels; and Dadieso FR vs. the other three areas. To detect changes between recent and historical data, the Mann-Whitney U-test was used for BA and the paired z-test for guild composition.
Forest soil data
Soil information was derived from eight 50×50 m plots, two per forest area, located in close proximity to recent field plots. The soil field data used here are part of a larger soil data collection realized in the framework of the FP7 ERC Africa GHG project. As soil sensors were also used for project needs, it was decided to realize the soil survey just outside of the plots, to avoid problems to sensors and instruments that might be caused by the repeated passages of the tree survey team. Forest and soil plots were set up in homogeneous areas of small extent. In each plot, 10 samples of the organic horizon were randomly collected using a 40×40 cm frame. In the mineral soil, 10 samples were collected in the topsoil (0-30 cm depth) and 10 in the subsoil (30-100 cm depth), using a cylinder (diameter = 5 cm; height = 5 cm) to determine the bulk density ([5]). Samples were oven-dried (60 °C) to a constant weight and mineral soil was sieved at 2 mm to remove rock fragments; they were then ground in a ball mill to reach the highest homogenization possible and measured for total carbon via dry combustion (ThermoFinnigan Flash EA112® CHN, Gemini BV, Apeldoorn, Netherlands). To calculate the amount of carbon (C) each sample was corrected for the amount of rock fragments, and the soil organic carbon (SOC) stock calculated considering the bulk density, the C concentration and the depth of each layer ([8]). Significant differences (α = 0.001) between C stocks of different soil layers were tested within each site and among sites by one-way analysis of variance and Tukey’s test ([70]).
Species richness and biodiversity
To avoid area-effects on results, richness and biodiversity indices were computed using recent data from subplots of same area, equal to 500 m2 in Ankasa NP and 400 m2 in all the other sites (Tab. S3 - Appendix 1). We adopted the Margalef’s richness index because it is less sensitive to sampling efforts variations ([52]). Considering the high number of rare species, expressed as singletons, doubletons, and unique species ([51]), we adopted the Chao 1 index ([13]) as a diversity measure, useful when datasets are skewed toward the low-abundance classes ([14]). We also reported the Coleman’s rarefaction curve ([18]) and the Shannon’s evenness ([51]) for comparison purposes; however, both measures are sensitive to species abundance and may not be optimal for forests with many rare species ([27]). Chao 1 index results were used to estimate the minimum sampling area needed to detect most of the species in each site (A), using the area-species curve equation (eqn. 1):
where c and z are parameters derived from the curves and S is the Chao 1 index.
Remote sensing and meteorological data
The third generation of the GIMMS3g (Global Inventory Modelling and Mapping Studies) NDVI dataset has 15-day temporal frequency, 1/12th of a degree spatial resolution and continuity from July 1981 to December 2011 ([74]). We extracted NDVI values for 7 reserves and 3 national parks in southwestern Ghana (red stars in Fig. S1 - Appendix 1), using pixels with >75% of their area inside the following reserves/parks: Anhwiaso FR, Ankasa NP, Atewa FR, Bia RR, Dadieso FR, Kakum NP, Krokosua NP, Subri FR, Subuma FR, and Tano FR. From the Climate Research Unit (CRU) 3.2 dataset ([33]) we extracted monthly rainfall, temperature, and cloud cover for the same period (1981-2011). We calculated monthly, seasonal and yearly basic statistics (mean, minimum, maximum, standard deviation) for NDVI time series, applied a 12-month moving-window average to visualize trends and oscillations, and detected anomalies subtracting the 30-year averaged monthly values from the observations of that month, and then applying a 12-month moving-window average ([9]). The observed differences in trends among sites were evaluated considering also their distance from the center of the wet evergreen Ghana ecological zone ([22]). We calculated Spearman’s correlation among NDVI and precipitation, cloud cover and temperature; and trends in CRU 3.2 and NDVI time series using the Seasonal Modified Kendall test ([41]) suited to treat data with seasonal cycles, and covariance evaluating the trend magnitude with slope estimation.
Results
Ordination of trees and disturbance evidences
The ordination scores attributed to plots corresponded to eight forest subcategories (Tab. S4 - Appendix 1) from Hall & Swaine ([29], [30]). NMS results showed that our samples represent three different forest types found in the region along a primary latitudinal gradient: wet evergreen (Ankasa), moist evergreen (Dadieso) and moist semi-deciduous (Bia Reserve and NP); a secondary gradient is present, according to soil moisture content or soil drainage (Fig. 1). The orthophotos screening showed varied signs of disturbance. Ankasa showed no sign with closed and uniform canopy. In Bia NP, only uneven canopy height was detected in two plots. Logging scars, fallen trees, gaps and different canopy heights were observed in nine Bia RR plots. Even more signs were found in Dadieso (14 plots), where rangers, villagers and workers from timber companies reported frequent illegal activities (pers. comm. from March 2013), but no quantitative record was found.
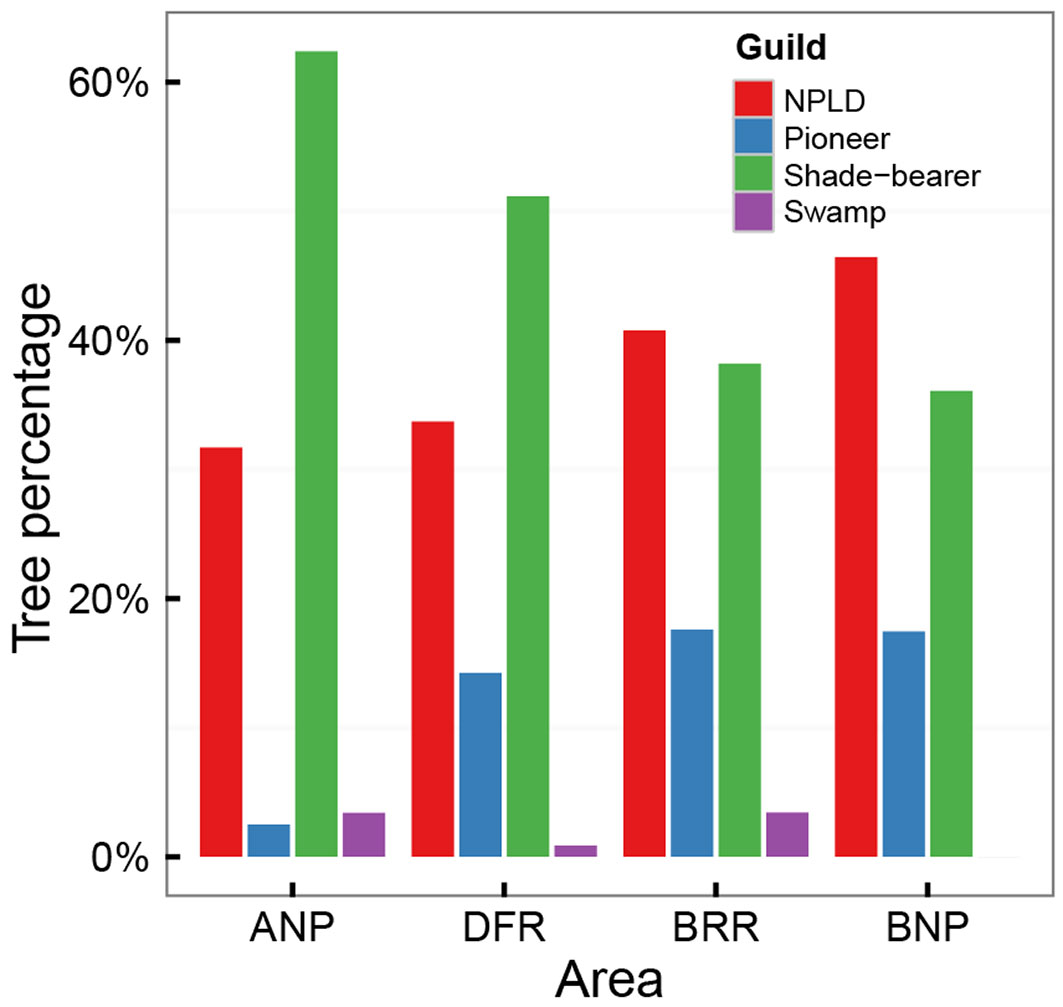
Fig. 1 - Percentage of guilds in recent data. For trees with diameter at breast height (DBH) > 20 cm. (ANP): Ankasa National Park; (DFR): Dadieso Forest Reserve; (BRR): Bia Resource Reserve; (BNP): Bia National Park.
Height, DBH, above ground biomass, and basal area
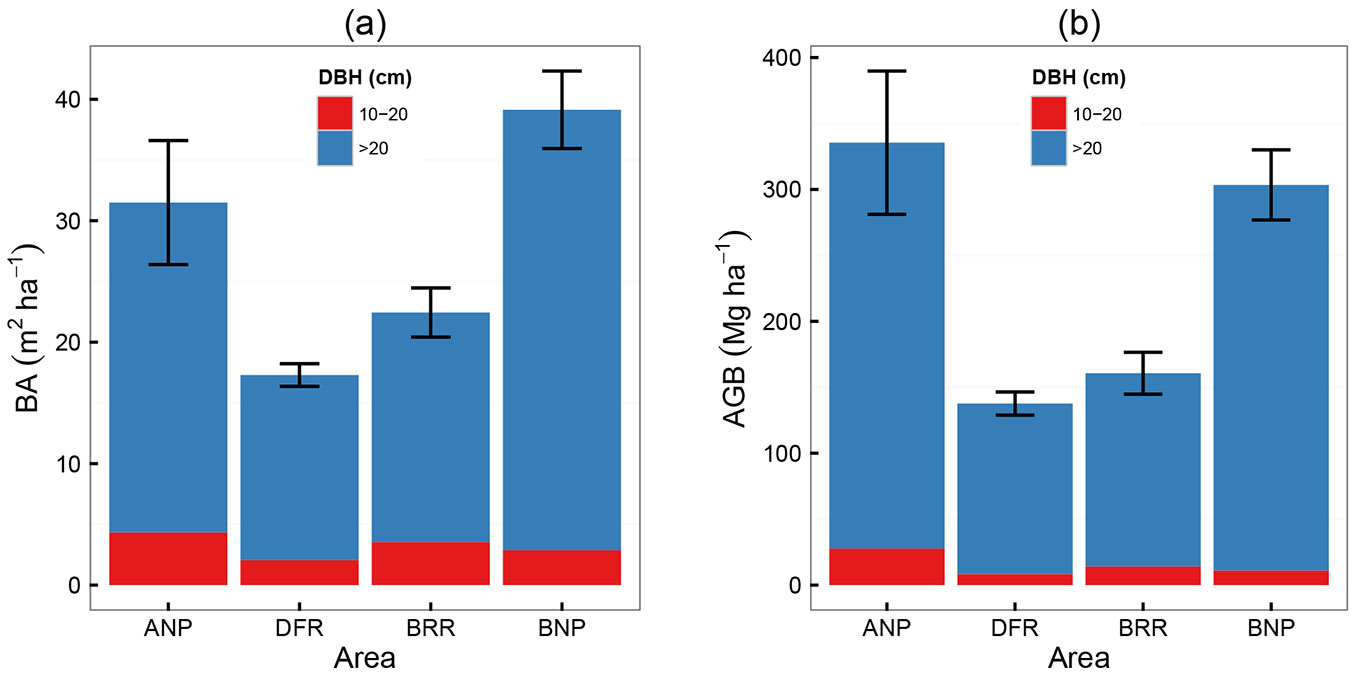
With recent data we performed descriptive statistics for H and DBH (Tab. S5 - Appendix 1), AGB and BA normalized by area (Fig. 2, Tab. S6 - Appendix 1), and paired comparison tests at 95% confidence level (Tab. 1). For mean H, Ankasa resulted to be the tallest forest, followed by Bia NP, and then Dadieso and Bia Reserves; differences were significant between all pairs for trees with DBH>20 cm, whereas for those within the DBH range 10-20 cm significant differences were found only between Ankasa and both Bia NP and Dadieso reserve. Regarding average AGB, Ankasa showed the highest value, followed by Bia NP, and Bia and Dadieso reserves; pairwise differences were always significant for both DBH ranges, except between the two Bia areas for 10-20 cm DBH trees.
Fig. 2 - Recent above ground biomass (AGB) and basal area (BA) values. (ANP): Ankasa National Park; (DFR): Dadieso Forest Reserve; (BRR): Bia Resource Reserve; (BNP): Bia National Park.
Tab. 1 - Results of statistical pairwise comparison tests, 2012-2013 data. (ANP): Ankasa National Park; (DFR): Dadieso Forest Reserve; (BRR): Bia Resource Reserve; (BNP): Bia National Park. (H): height; (DBH): diameter at breast height; (WD): wood density; (BA): basal area; (AGB): above ground biomass; (MW): Mann-Whitney test; (KS): Kolmogorov-Smirnov test; (*): p< 0.05; (**): p < 0.01; (***): p< 0.001.
| Comparison | Parameter | DBH 10-20cm | DBH > 20cm | ||||||
|---|---|---|---|---|---|---|---|---|---|
| MW stat | p-value | KS stat | p-value | MW stat | p-value | KS stat | p-value | ||
| BNP vs. BRR | H | -0.88 | - | 0.10 | - | -4.44 | *** | 0.20 | *** |
| DBH | 0.84 | - | 0.06 | - | -4.23 | *** | 0.17 | *** | |
| WD | -1.03 | - | 0.11 | - | 1.15 | - | 0.11 | * | |
| BA | 0.84 | - | 0.06 | - | -4.23 | *** | 0.17 | ** | |
| AGB | -0.19 | - | 0.03 | - | -4.62 | *** | 0.20 | *** | |
| BNP vs. ANP | H | -14.30 | *** | 0.50 | *** | -9.34 | *** | 0.28 | *** |
| DBH | 0.63 | - | 0.05 | - | 1.90 | * | 0.11 | ** | |
| WD | -8.29 | *** | 0.36 | *** | -12.62 | *** | 0.44 | *** | |
| BA | 0.63 | - | 0.05 | - | 1.90 | * | 0.11 | ** | |
| AGB | -9.73 | *** | 0.30 | *** | -3.95 | *** | 0.17 | *** | |
| DFR vs. BNP | H | -0.27 | - | 0.08 | - | 2.22 | ** | 0.14 | ** |
| DBH | -2.63 | ** | 0.14 | * | 4.56 | *** | 0.17 | *** | |
| WD | -2.56 | * | 0.21 | *** | -3.17 | *** | 0.21 | *** | |
| BA | -2.63 | ** | 0.14 | * | 4.56 | *** | 0.17 | *** | |
| AGB | -2.43 | * | 0.15 | * | 3.36 | *** | 0.15 | *** | |
| DFR vs. BRR | H | 0.55 | - | 0.10 | - | -2.75 | *** | 0.15 | *** |
| DBH | -3.52 | *** | 0.16 | ** | -0.33 | - | 0.05 | - | |
| WD | -1.21 | - | 0.14 | ** | -1.91 | * | 0.12 | * | |
| BA | -3.52 | *** | 0.16 | ** | -0.33 | - | 0.05 | - | |
| AGB | -2.25 | ** | 0.13 | * | -1.99 | ** | 0.11 | * | |
| DFR vs. ANP | H | -15.33 | *** | 0.49 | *** | -12.65 | *** | 0.42 | *** |
| DBH | 4.29 | *** | 0.14 | *** | -3.90 | *** | 0.16 | *** | |
| WD | -5.04 | ** | 0.19 | *** | -8.17 | *** | 0.30 | *** | |
| BA | 4.29 | *** | 0.14 | *** | -3.90 | *** | 0.16 | *** | |
| AGB | -6.75 | *** | 0.22 | *** | -9.01 | *** | 0.27 | *** | |
Mean BA and mean DBH resulted higher in Bia NP, followed by Ankasa, Bia and Dadieso reserves; distributions were significantly different between Dadieso reserve and both Ankasa and Bia NPs for trees with DBH>20 cm, and between Dadieso and the other three areas for trees in the 10-20 cm DBH range. The total number of trees per ha were: Ankasa 408; Dadieso 229; Bia reserve 368; Bia NP 352.
The analysis of these structural parameters confirmed our first hypothesis (i) of higher impacts on mean forest structure values in the less protected areas where selective logging or illegal disturbance occurred.
Wood density and guild composition
Mean WD analysis showed for both DBH ranges the highest in Ankasa and the lowest in Bia NP; differences in distributions were significant between Ankasa and both Bia NP and Dadieso, and between Dadieso and Bia NP.
Percentages of trees per guild for DBH >20 cm showed that, from Ankasa to Bia NP, pioneer and non-pioneer light-demanding species increased, while shade-bearer species decreased (Fig. 1, Tab. S7 - Appendix 1); for the 10-20 cm DBH range the pattern was similar. Swamp species were too scarce for evaluation.
The results from WD and guild composition analysis are in disagreement with the first hypothesis (i) of stronger impacts in the less protected reserves. Instead, WD and guild composition followed the latitudinal gradient.
Temporal changes in basal area and guild composition
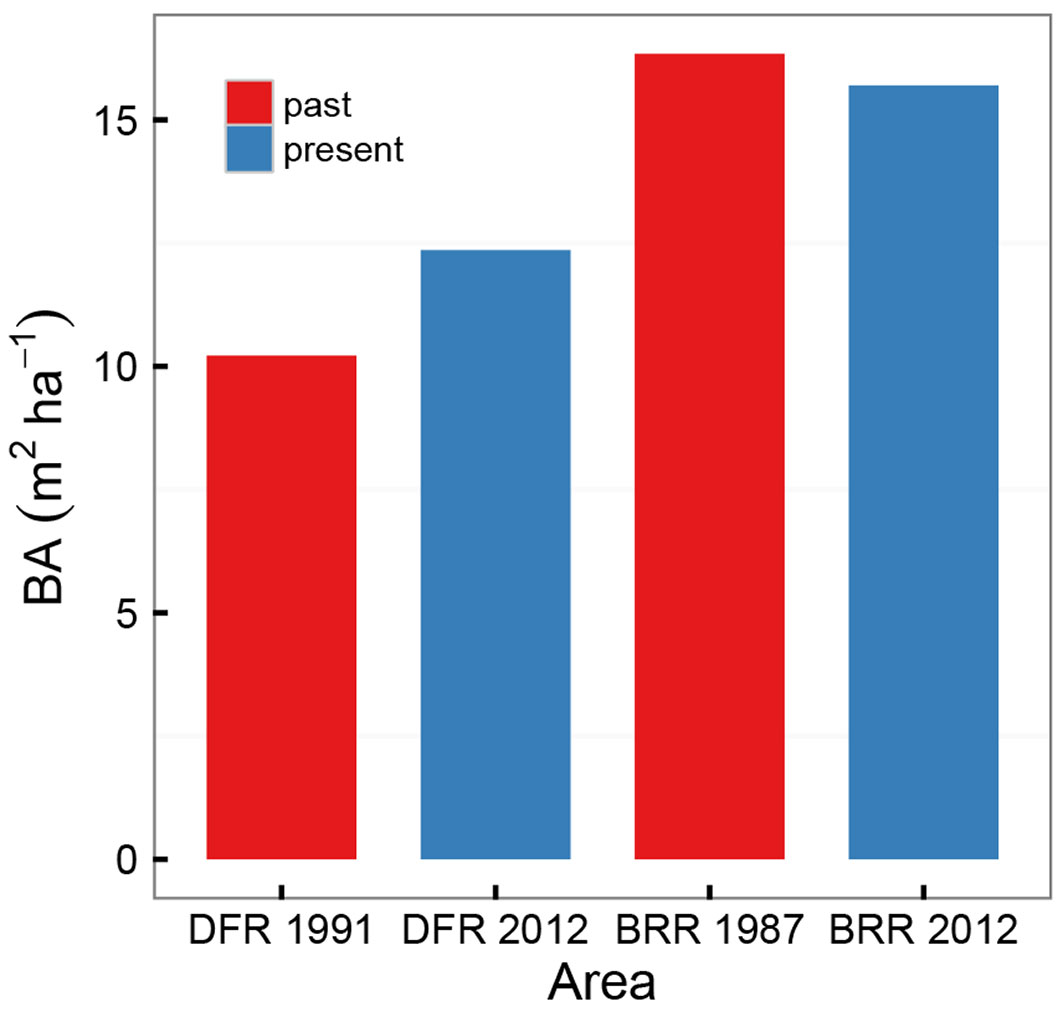
The BA from historical data totaled 15.96 m2 ha-1 in Dadieso and 23.82 m2 ha-1 in Bia reserves (Tab. S8 - Appendix 1). The BA change over time, tested for trees with DBH>30 cm to account for differences in sampling thresholds in the surveys, was not significant for any of the areas (Fig. 3). This is in contrast to what was defined in the second hypothesis (ii), namely a recovery in BA for Bia NP, where logging activities ceased two decades ago, and a decrease for Dadieso, still affected by illegal activities. For Bia Reserve, the difference in BA from the neighboring Bia NP is more than 50%, revealing very degraded conditions; the even lower BA in Dadieso indicates a highly degraded forest.
Fig. 3 - Changes in basal area (BA), from past to present values, for trees with DBH > 30 cm. (DFR): Dadieso Forest Reserve; (BRR): Bia Resource Reserve.
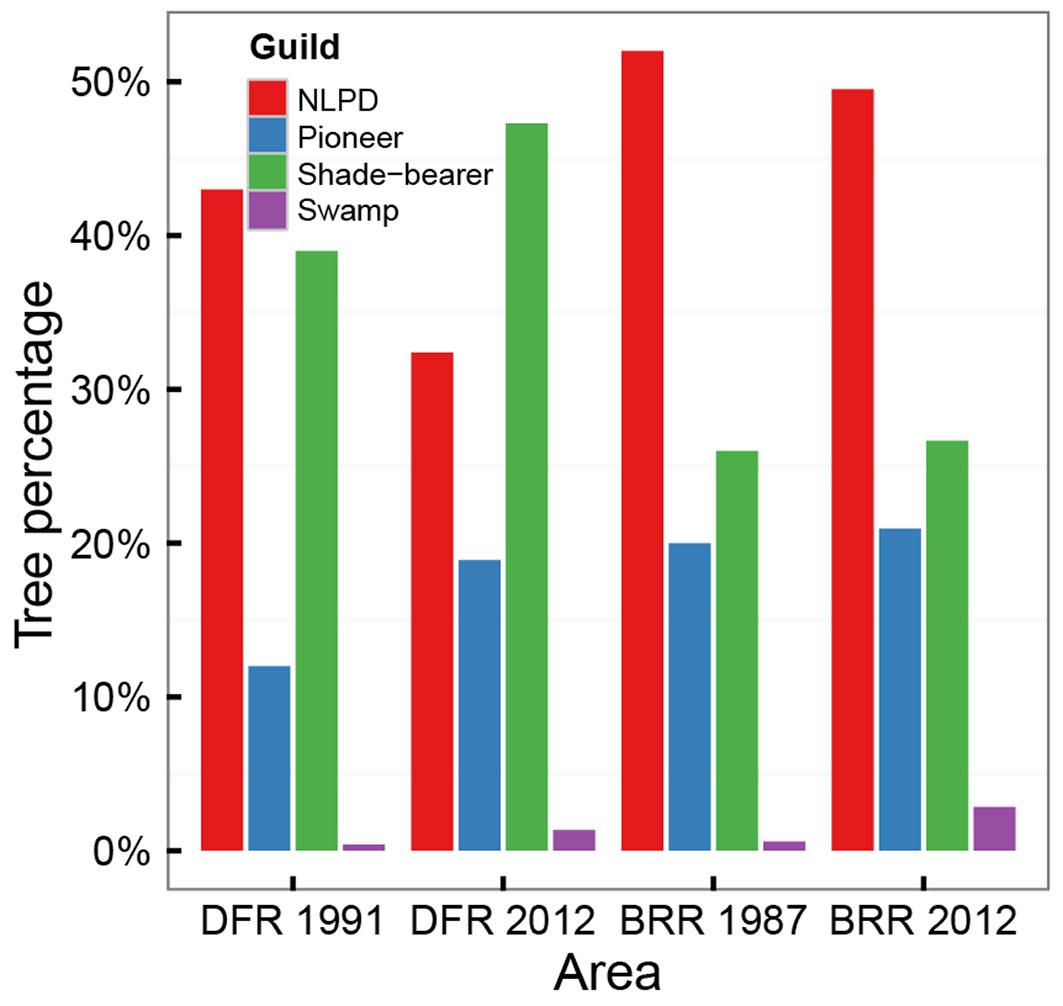
Historical guild data (Tab. S8 - Appendix 1) showed for that time a prevalence in Dadieso (trees > 30 cm DBH) of non-pioneer light-demanding species, followed by shade-bearer, pioneer and swamp species; in the 10-30 cm DBH range, the first two guilds were instead inverted in order. In Bia, for both DBH classes, the prevalent guild was non-pioneer light-demanding, followed by shade bearer, pioneer, and swamp-resistant.
Guild change over time, tested for trees with DBH>30 cm (Fig. 4), was not significant for any guild in Bia, contrary to the shift toward recovery predicted by our third hypothesis (iii). For Dadieso, a significant increase in pioneer and shade-bearer species was found, with significant decrease of non-pioneer light-demanding species; swamp-resistant trees were too limited for testing. The further Dadieso degradation predicted by our third hypothesis (iii) is in contrast with the increase of shadow-bearer species.
Fig. 4 - Changes in guild composition, from past to present values, for trees with DBH > 30 cm. (DFR): Dadieso Forest Reserve; (BRR): Bia Resource Reserve.
Overall, Bia recovery has not occurred after two decades, with unaltered BA and guilds composition. In Dadieso BA conditions are unchanged, while guild data only partially support our initial hypothesis.
Species richness and biodiversity
The Margalef index (Tab. 2) indicated an increasing species richness moving southward from Bia to Ankasa NPs. This result contradicts our first hypothesis (i) regarding a stronger impact in less protected areas. The richness distribution follows the latitudinal gradient as found for WD and guilds composition. The Chao 1 index (Tab. 2) was higher in NPs, and confirms our first hypothesis. For minimum sampling area, we obtained 3.02 and 3.27 ha respectively for Ankasa and Bia NPs, while for Bia and Dadieso Reserves 1.28 and 3.06 ha, respectively. We reported Shannon’s evenness in Tab. 2 and Coleman’s rarefaction curve in Fig. S2 - Appendix 1.
Tab. 2 - Biodiversity indices calculated per area. (ANP): Ankasa National Park; (DFR): Dadieso Forest Reserve; (BRR): Bia Resource Reserve; (BNP): Bia National Park.
| Biodiversity indices (DBH>10 cm) |
ANP | DFR | BRR | BNP |
|---|---|---|---|---|
| Area (m2) | 17000 | 18400 | 8800 | 10000 |
| Number of species | 147 | 120 | 94 | 84 |
| # of individuals | 841 | 402 | 290 | 335 |
| Singletons | 54 | 48 | 39 | 37 |
| Dubletons | 22 | 22 | 17 | 7 |
| Uniques | 57 | 55 | 43 | 37 |
| Margalef’s richness | 21.68 | 19.85 | 16.40 | 14.28 |
| Chao 1 ± st.dev. | 213.19 ± 24.29 | 172.23 ± 20.09 | 138.58 ± 19.11 | 181.49 ± 49.83 |
| Shannon’s Evenness | 0.85 | 0.91 | 0.91 | 0.88 |
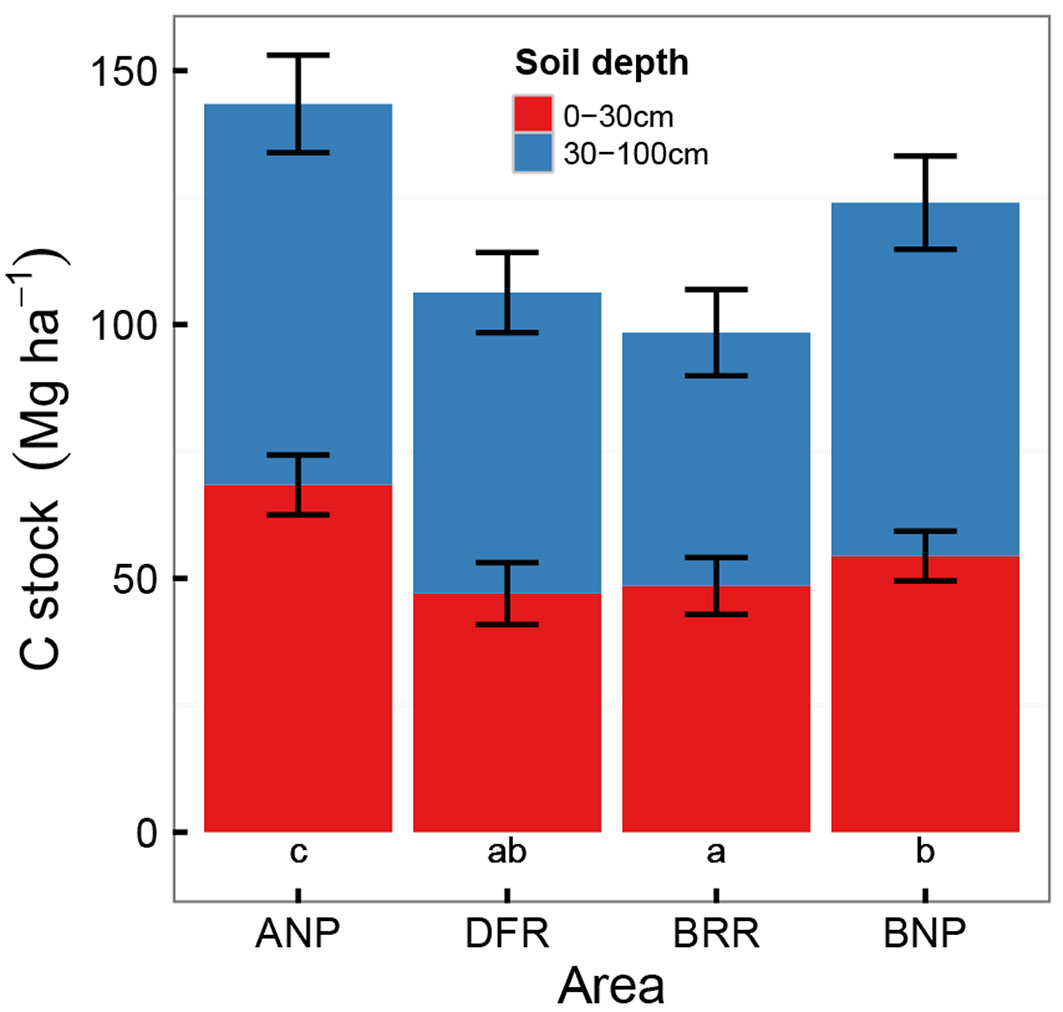
Soil carbon content
Fig. 5 illustrates the SOC results. In the topsoil, the C concentration in the Bia reserve was significantly lower (p<0.001) than in Ankasa and Bia NPs, with no difference between the two parks. In Dadieso the C concentration was only significantly lower than in Ankasa. Compared to the topsoil which stores roughly half of the total SOC stock stored down to 1 meter, the C concentrations in the subsoil considerably decreased: no detectable differences were observed among the sites. The significantly higher C stocks found in Ankasa and Bia NPs support the first hypothesis (I) of stronger impacts in less protected sites.
Fig. 5 - Carbon stock in the topsoil (0-30 cm) and subsoil (30-100 cm). Different letters indicate significant differences for the 0-100 cm depth. (ANP): Ankasa National Park; (DFR): Dadieso Forest Reserve; (BRR): Bia Resource Reserve; (BNP): Bia National Park.
Remote sensing and meteorological data
NDVI trends over 30 years were analyzed. Monthly averaged NDVI values in the ten forested areas remarked the two distinct productive seasons (Fig. S3 - Appendix 1). Oscillations in 30-year NDVI temporal series showed consistency among the areas, but different magnitude (Fig. S4 - Appendix 1). The Seasonal Modified Kendall tests (Tab. 3) indicated a significant NDVI increase for all the areas, in accordance to our fourth hypothesis (iv) of a forest productivity increase along the last 30 years. Also precipitation (with the exception of Atewa FR, an upland rainforest) and temperature significantly increased, while cloud cover data showed a significant positive trend only for areas located in the most northwest range (Tano, Dadieso and Bia Reserves, and Krokosua NP). Positive and negative NDVI fluctuations are more marked in areas closer to the wet evergreen zone.
Tab. 3 - Seasonal Modified Kendall tests for NDVI and climate data from ten forested areas in Ghana. Z-test values, p-values and slopes are displayed. (FR): Forest Reserve; (NP): National Park; (RR): Resource Reserve. (NDVI): Normalized Difference Vegetation Index; (-): non significant; (*): p < 0.05; (**): p < 0.01; (***): p<0.001.
| Variable | Site | Z | p-value | slope | Variable | Site | Z | p-value | slope |
|---|---|---|---|---|---|---|---|---|---|
| NDVI | Ankasa NP | 3.166 | *** | 0.003 | Precipitation | Ankasa NP | 1.775 | * | 4.789 |
| Subri FR | 2.560 | ** | 0.002 | Subri FR | 2.424 | ** | 6.767 | ||
| Tano FR | 3.331 | *** | 0.003 | Tano FR | 2.079 | ** | 5.500 | ||
| Kakum NP | 2.515 | ** | 0.003 | Kakum NP | 2.268 | ** | 6.070 | ||
| Subuma FR | 3.966 | *** | 0.004 | Subuma FR | 2.247 | ** | 5.527 | ||
| Dadieso FR | 2.269 | * | 0.002 | Dadieso FR | 1.839 | * | 4.373 | ||
| Anwhiaso FR | 2.149 | * | 0.002 | Anwhiaso FR | 2.247 | * | 5.257 | ||
| Krokosua NP | 3.071 | *** | 0.002 | Krokosua NP | 2.223 | * | 4.091 | ||
| Bia RR | 2.054 | * | 0.002 | Bia RR | 2.176 | * | 3.572 | ||
| Atewa FR | 1.902 | * | 0.001 | Atewa FR | 0.778 | - | 1.928 | ||
| Temperature | Ankasa NP | 2.515 | ** | 0.015 | Cloud cover | Ankasa NP | 1.302 | - | - |
| Subri FR | 2.795 | ** | 0.016 | Subri FR | 1.174 | - | - | ||
| Tano FR | 2.286 | * | 0.014 | Tano FR | 1.739 | * | 0.261 | ||
| Kakum NP | 2.969 | *** | 0.017 | Kakum NP | 0.681 | - | 0.167 | ||
| Subuma FR | 2.412 | ** | 0.008 | Subuma FR | 1.227 | - | 0.250 | ||
| Dadieso FR | 2.321 | ** | 0.014 | Dadieso FR | 1.771 | * | 0.294 | ||
| Anwhiaso FR | 2.412 | ** | 0.013 | Anwhiaso FR | 1.227 | - | 0.250 | ||
| Krokosua NP | 2.234 | * | 0.013 | Krokosua NP | 1.834 | * | 0.366 | ||
| Bia RR | 2.331 | ** | 0.014 | Bia RR | 1.952 | * | 0.364 | ||
| Atewa FR | 2.545 | ** | 0.014 | Atewa FR | 0.417 | - | 0.048 |
Spearman’s ρ correlation was tested between NDVI time series and climate parameters (Tab. 4), with higher ρ values found using 12 months of precipitation, 5 of cloud cover (June-October), and 3 of temperature (July-September). For all but one area (Tano FR) the highest correlation was found with cloud cover, with ρ values ranging from -0.43 to -0.60, followed by precipitation (ρ from 0.25 to 0.49) and temperature (ρ from 0.19 to 0.33).
Tab. 4 - Spearman’s correlation values between NDVI time series and monthly climate data for ten forested areas in Ghana. (NDVI): Normalized Difference Vegetation Index.
| Site | Spearman’s correlation (ρ) | ||
|---|---|---|---|
| Cloud cover | Precipitation | Temperature | |
| Ankasa NP | -0.51 | 0.40 | 0.28 |
| Subri FR | -0.44 | 0.25 | 0.33 |
| Tano FR | -0.44 | 0.48 | 0.31 |
| Kakum NP | -0.43 | 0.32 | 0.24 |
| Subuma FR | -0.54 | 0.49 | 0.28 |
| Dadieso FR | -0.52 | 0.48 | 0.31 |
| Anhwiaso FR | -0.55 | 0.43 | 0.25 |
| Krokosua NP | -0.57 | 0.42 | 0.28 |
| Bia FR | -0.60 | 0.40 | 0.19 |
| Atewa FR | -0.47 | 0.40 | 0.22 |
Discussion
Degradation impacts and protection status
The C stocks and BA of Bia and Dadieso reserves were reduced by 50% or more compared to the Ankasa and Bia NP; significant reduction was also observed for structural parameters (H, DBH). The illegal activities reported for the Dadieso reserve had impacts similar or even worse than selective logging in Bia. Even if we could collect only unofficial records, Hansen et al. ([32]) evaluated that in Ghana the informal lumber sector, driven primarily by domestic consumption, represents 80% of the market, and is six times the annual allowable cut. A balance between forest regrowth and persistent illegal activities, possibly coupled with natural disturbance, may explain the lack of BA change along time observed for Dadieso.
The complete absence of recovery of BA in the Bia reserve might be explained if severe logging occurred after historical data collection, possibly coupled with climate natural disturbance, which had been reported for the drier Ghana tropical range by Owusu & Waylen ([60]). In this case multiple stresses may have limited until now the ability of the forest to recover. Recovery is linked to the frequency of past disturbance events as well as their intensity and type, with some research estimating hundreds to a thousand years for a complete recovery of fragmented forests ([48]). Recovery from selective logging might be much longer than that estimated through growth models, as previously observed in Ghana by Hawthorne et al. ([38]), as forest biomass can continue to decline after logging ceases due to delayed effects on tree mortality ([6]). The BA change observed in the Bia reserve poses a warning for the sustainability of current selective logging schemes.
Bia NP had a significantly higher mean DBH and BA with respect to Ankasa NP; tree density was higher in Ankasa, but larger stems were recorded in Bia. Moist forests can have more favorable water conditions, more nutrients and lower light competition compared with drier forests, as observed by Hall & Swaine ([30]) in Ghana, thus explaining the presence of such very large trees in Bia.
With respect to SOC, the two NPs showed a C stock in the 0-100 cm depth similar or higher than the average reported for a series of Oxisols from Western and Central Africa by Batjes ([3]) and Henry et al. ([39]). In contrast, results from the two reserves showed SOC stocks significantly lower than these values. Logging, even if selective, promotes SOC decomposition by creating gaps in the canopy cover and reducing the inputs of organic C to soil ([72], [49]). All logging activities leave behind large amounts of slash which decomposes, and, combined with an increased mortality of residual trees, increases ecosystem heterotrophic respiration and CO2 emissions for years following harvest ([6], [43]). The topsoil, where most of the tropical forests C is stored, is the layer more prone to changes following disturbance, as observed by Chiti et al. ([17]) in Western Ghana. Our results are not consistent with what reported by Asase et al. ([2]) for both Bia areas, as our Bia NP values are much higher. This discrepancy may be due to the low number of samples (4) collected in the aforementioned study.
For species diversity, results from the Chao 1 index indicated that the two NPs have much higher species diversity than the reserves. This is agreement with the results of a meta-analysis by Gibson et al. ([26]), who showed that forest degradation has an overwhelmingly detrimental effect on tropical biodiversity; however, the authors reported that selecting logging causes smaller impact compared to other disturbance types. The area needed to sample diversity was higher in the NPs, with the about 3 ha value being far from the 1-1.5 ha commonly suggested as an adequate sampling size ([1], [46]); such data could represent an indication for planning field diversity studies in this region.
According to our study, NPs were more effective in conserving forest structure, soil C content, and biodiversity. The evenness component of species diversity is especially important for assessing the effects of degradation, as higher richness can be observed in disturbed areas as a result of an increased number of pioneer species. A reduced species evenness caused by disturbance represents alone an indication of biodiversity loss. This result adds to the existing evidence that selective logging has severe impacts in African forests ([4], [31], [38]). However, it is evident that this protection cannot be granted to all the forest fragments of the region, while on the edges of NPs, such as Ankasa, economic problems are causing an increase in anthropogenic pressure ([19]). In order to avoid further depletion of forest resources within and around protected areas, viable management solutions, such as the European Union FLEGT (Forest Law Enforcement, Governance and Trade) facility should be strengthened ([66]), as well as smart and cost-effective restoration approaches ([67]).
Forest variables less influenced by disturbance
In this study arboreal richness distribution followed the latitudinal gradient. According to the Intermediate Disturbance Hypothesis, higher richness values are found in moderately disturbed areas ([55]). Bongers et al. ([7]) found in Ghana that richness is less sensitive to disturbance in wet than in dry forests. Norris et al. ([59]) in a West African review study observed that, although in some instances overall species richness increased in disturbed sites, the richness of endemic species tended to decline. Species richness is also known to increase with rainfall amount in this region ([30]).
Functional composition, namely WD and guilds, was also distributed according to the latitudinal gradient; the percentage of pioneer species, an indicator of disturbance, was not higher in the more degraded areas.
These results indicate that both richness and functional composition are shaped by climate parameters and less affected by the disturbance types we considered.
Regional forest productivity and climate variable trends
Previous studies showed the ability of GIMMS3g to capture productivity dynamics ([71], [73]). Our result of NDVI increasing trends is in agreement with the increase in carbon storage observed for African intact tropical forests ([47]), as well as with the increase in net primary production recorded at a global scale ([58]). As most (7) of the ten sites we considered were reserves affected by multiple disturbances, our results supports the view that productivity can be enhanced by fast growing species that are abundant in disturbed sites ([10]). The magnitude of oscillations in NDVI decreased with increasing distance from the wetter tropical forest type center (Fig. S4 - Appendix 1), congruent with observations indicating that drier forests are characterized by higher resiliency to changes and have 50-75% of the productivity of wetter forest types ([21], [57]).
The increasing trend in precipitation and temperature resulting from 30 years of CRU 3.2 dataset analysis is opposite of the precipitation decrease observed by Fauset et al. ([24]) and Owusu & Waylen ([60]). We explain this difference considering that the CRU datasets up to version 3.10.01 were affected by a bug in precipitation data (⇒ http://www.cru.uea.ac.uk/cru/data/h rg/). Moreover, we examined a different period (1981-2011) with respect to the aforementioned studies, and the area we analyzed is much toward south than that of Fauset et al. ([24]). Finally, the CRU cells considered in this study are not so close to stations used in Owusu & Waylen ([60]).
Previous studies reported different results about which climate variables mostly influence forest productivity ([20], [28], [62]). However, it is known that light availability plays a key role in limiting photosynthesis ([56]). Seasonal productivity changes have been already observed in the dry tropics, as a result of climate change, but usually are not investigated as often or intensely as long-term changes ([25]). The strongest negative correlation of NDVI with cloud cover during the short dry season, that was higher than that with precipitation, stresses the importance of examining changes in seasonal patterns beside long-term changes, especially in “bimodal” seasonal forests.
Forest resources conservation in a changing climate
In mostly undisturbed forests of Ghana, Fauset et al. ([24]) found that prolonged drought led to a consequent change in functional composition toward drought-resistant species, with increase in pioneers and decrease in shadow-bearers. These shifts are similar to those caused by selective logging, which also increases pioneers and decreases shadow-bearers through gap openings. Unless there is certainty of absence of disturbance (including illegal activities), it is difficult to understand what contributes more to a guild change. Our historical data were collected in two disturbed areas, but the lack of guild change found in the Bia reserve and the increase in shadow-bearer species observed in Dadieso do not support a functional composition change in our study sites. Bongers et al. ([7]) found that in Ghana the response in guild composition to disturbance intensity changed according to forest type, being significant only in drier forests. It is possible that the forests located in the zone we focused are more resilient to functional changes. However, climate change is a concerning issue in West Africa, with predictions still uncertain, especially for precipitation ([61]). If considering climate alone, deciduous and evergreen forests might expand in the future, but human impacts will play a critical role in the future of forest resources ([40]). New technologies such as laser scanning for forests ([63], [64], [68]) will enable a better monitoring of changes to support decision and policies. Our results call for additional functional change studies focused on the possible varied responses of different forest types.
Conclusions
Overall, the impacts observed in reserves are alarming, especially considering that most of the West African forests are under this low protection status. Even the NPs of this region might be threatened by growing anthropogenic pressure, and lack of resources and buffer zones ([45]). Community-based resource management projects have been implemented in Ghana, but several issues hamper their full effectiveness ([19]). It is urgent to strengthen these approaches and properly value the range of services provided by forests, including agro-forestry systems that could generate C credits ([59]). Selectively logged and degraded forests remain critical for the conservation of biodiversity and C storage potential, once adequately protected ([50]). Forest productivity increase is an opportunity for recovery that is very important to seize. This translates into the need to reconsider the sustainability of selective logging schemes, to improve controls in forests, to strengthen viable forest management alternatives, providing more incentives for conservation through international programs.
Acknowledgments
We acknowledge the EC FP7 ERC grant Africa GHG #247349. The funding source was not involved in any part of the manuscript preparation. We are grateful to the Ghana Forestry Commission staff, to Justice Mensah, and to all the field collaborators. We thank Prof. Ranga Mynani and Dr. Zeichuan Zhu for providing the GIMMS3g dataset. G.V.L. thanks the EU for supporting the BACI project funded by the EU’s 692 Horizon 2020 Research and Innovation Programme under grant agreement 6401 76. None of the authors has any conflict of interest.
References
Gscholar
Online | Gscholar
CrossRef | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
CrossRef | Gscholar
Authors’ Info
Authors’ Affiliation
Tommaso Chiti
Arianna Di Paola
Roberto Cazzolla Gatti
Sergio Noce
Elisa Grieco
Riccardo Valentini
Impacts of Agriculture, Forests and Ecosystem Services Division, Euro-Mediterranean Center on Climate Change (IAFES-CMCC), v. Pacinotti 5, I-01100 Viterbo (Italy)
Department of Plant Sciences, University of Oxford, South Parks Road, Oxford OX1 3RB (United Kingdom)
Tommaso Chiti
Sergio Marconi
Riccardo Valentini
Department for Innovation in Biological, Agro-Food and Forest Systems (DIBAF), University of Tuscia, I-01100 Viterbo (Italy)
Interdepartmental Research Center of Geomatics (CIRGEO), Land, Environment and Agriculture and Forestry Department (TESAF), University of Padova, I-35020 Legnaro, Padua (Italy)
Corresponding author
Paper Info
Citation
Vaglio Laurin G, Hawthorne WD, Chiti T, Di Paola A, Cazzolla Gatti R, Marconi S, Noce S, Grieco E, Pirotti F, Valentini R (2016). Does degradation from selective logging and illegal activities differently impact forest resources? A case study in Ghana. iForest 9: 354-362. - doi: 10.3832/ifor1779-008
Academic Editor
Matteo Garbarino
Paper history
Received: Jul 26, 2015
Accepted: Nov 06, 2015
First online: Jan 29, 2016
Publication Date: Jun 01, 2016
Publication Time: 2.80 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2016
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 42942
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 35417
Abstract Page Views: 2496
PDF Downloads: 3967
Citation/Reference Downloads: 74
XML Downloads: 988
Web Metrics
Days since publication: 3000
Overall contacts: 42942
Avg. contacts per week: 100.20
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Feb 2023)
Total number of cites (since 2016): 26
Average cites per year: 3.25
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Review Papers
Remote sensing of selective logging in tropical forests: current state and future directions
vol. 13, pp. 286-300 (online: 10 July 2020)
Review Papers
Remote sensing-supported vegetation parameters for regional climate models: a brief review
vol. 3, pp. 98-101 (online: 15 July 2010)
Review Papers
Accuracy of determining specific parameters of the urban forest using remote sensing
vol. 12, pp. 498-510 (online: 02 December 2019)
Research Articles
Estimates of selective logging impacts in tropical forest canopy cover using RapidEye imagery and field data
vol. 9, pp. 461-468 (online: 11 January 2016)
Technical Reports
Detecting tree water deficit by very low altitude remote sensing
vol. 10, pp. 215-219 (online: 11 February 2017)
Technical Reports
Remote sensing of american maple in alluvial forests: a case study in an island complex of the Loire valley (France)
vol. 13, pp. 409-416 (online: 16 September 2020)
Research Articles
Assessment of land sensitivity to degradation using MEDALUS model - a case study of Grdelica Gorge and Vranjska Valley (southeastern Serbia)
vol. 15, pp. 163-170 (online: 07 May 2022)
Research Articles
Afforestation monitoring through automatic analysis of 36-years Landsat Best Available Composites
vol. 15, pp. 220-228 (online: 12 July 2022)
Research Articles
Fine root production and distribution in the tropical rainforests of south-western Cameroon: effects of soil type and selective logging
vol. 3, pp. 130-136 (online: 27 September 2010)
Research Articles
Assessing water quality by remote sensing in small lakes: the case study of Monticchio lakes in southern Italy
vol. 2, pp. 154-161 (online: 30 July 2009)
iForest Database Search
Search By Author
- G Vaglio Laurin
- WD Hawthorne
- T Chiti
- A Di Paola
- R Cazzolla Gatti
- S Marconi
- S Noce
- E Grieco
- F Pirotti
- R Valentini
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
- G Vaglio Laurin
- WD Hawthorne
- T Chiti
- A Di Paola
- R Cazzolla Gatti
- S Marconi
- S Noce
- E Grieco
- F Pirotti
- R Valentini
Search By Keywords
PubMed Search
Search By Author
- G Vaglio Laurin
- WD Hawthorne
- T Chiti
- A Di Paola
- R Cazzolla Gatti
- S Marconi
- S Noce
- E Grieco
- F Pirotti
- R Valentini
Search By Keyword