Latent infection of Biscogniauxia nummularia in Fagus sylvatica: a possible bioindicator of beech health conditions
iForest - Biogeosciences and Forestry, Volume 9, Issue 1, Pages 49-54 (2015)
doi: https://doi.org/10.3832/ifor1436-008
Published: Jun 18, 2015 - Copyright © 2015 SISEF
Research Articles
Abstract
Biscogniauxia nummularia is a xylariaceous fungus known as a common endophyte of European beech, living in plant tissues without development of symptoms, or even inducing strip-cankers and wood decay on trees stressed by drought. We studied the presence of the fungus in apparently healthy beech trees, growing in two different bioclimatic zones characterized by Continental and Mediterranean climates. Asymptomatic twigs were collected in each zone over the season and evaluated for the presence of B. nummularia infections using both cultural and qPCR methods. Results from qPCR indicated differences in the detection of B. nummularia among the seasons and between the study sites. In both sites the highest frequency of detection was in summer. B. nummularia was more frequently detected in the Mediterranean bioclimatic area, where drought is more common. These results suggest that B. nummularia may be a possible bioindicator of beech health stands.
Keywords
Fagus sylvatica, Latent Pathogen, Real Time PCR, Xylariaceae
Introduction
European beech (Fagus sylvatica L.) is a late successional species widely distributed in European temperate forests. Beech forests are usually managed as high stands so that centenary trees are common ([19]).
Along the Italian peninsula, beech forests grow at elevations ranging from 300 to 2000 m a.s.l. They are distributed in two phytoclimatic regions: Northern or Continental, and Central-Southern or Peninsular ([52]). In the Alps and northern Apennines, the climate is continental and the sub-alliances of forest associations are Abieti-Fagion, Eu-Fagion, Cephalanthero-Fagion and Luzulo-Fagion. In central-southern Italy, the climate is Mediterranean and the associations are are Aremonio-Fagion and Geranio versicoloris-Fagion ([50], [52], [18]). Beech forests located in the northern Apennines have greater affinity with the Alpine beech forests than with those in central Italy ([50], [52]). Because of its longevity, widespread distribution, and climate sensitivity, beech is a potential species for bio-monitoring programs on the status of European temperate forests ([3], [51], [44], [52]).
In a scenario of global climatic change, drought is expected to become one of the most limiting factors to beech forest sustainability, especially at lower latitudes ([66]). Monitoring of drought events and their impact on beech therefore are paramount needs in order to evaluate the latitudinal and altitudinal migration of the species and develop adaptation strategies. Immediate consequences of drought events on beech are extensive mortality of fine roots during peak drought ([35]), and an increase in host susceptibility to secondary pathogens ([56], [17]). Several pathogens attack beech trees opportunistically after extended drought, such as the root rot fungi Armillaria spp. and occasionally Heterobasidion annosum ([65], [6], [53], [9], [36]). Other opportunistic pathogens that normally colonize symptomless tissue may have an indirect effect on the survival of stressed trees. Among these pathogens, Biscogniauxia nummularia, a Xylariaceous fungus, has been found in stressed beech trees ([23], [48], [7], [22]). This fungus spends part of its life cycle as endophyte and may induce disease symptoms on its host under unfavorable environmental conditions. In prolonged summer drought, B. nummularia takes advantage of the altered host physiology, invades host tissue and causes elongated blackish bark lesions on trunk and branches, known as strip-cankers, and wood decay in mature trees ([28], [45]). In the related species B. mediterranea ([62]), outbreak of disease on oaks is likely to follow an increase in endophytic colonization. Therefore, monitoring of the endophytic stage of these fungi may provide an indication of drought-induced stress conditions of the host ([17]), i.e., the water deficit that can predispose the plant to disease ([5], [56]).
The detection of opportunistic pathogens within symptomless tissue is of primary importance to study disease progress and
particularly the transition from latent to active tissue colonization. This process may also account for seasonal disease outbreaks caused by opportunistic pathogens, especially when these diseases are related to host decline.
In the past, the occurrence of endophytic microorganisms in healthy trees and the seasonality of occurrence in woody plants was detected by isolation from tissues on growth media in order to obtain axenic fungal cultures ([25]). However, these traditional isolation methods probably underestimate the frequency of a target microorganism ([10]).
Over recent years, rapid and sensitive molecular methods, such as real-time quantitative PCR (qPCR), have been developed to identify and quantify bacteria ([46]) and fungi from woody plants, before symptoms occur in the host ([39], [40]). The effectiveness of this molecular method allows a target pathogen to be quantified directly in DNA extracted from plant tissue, reducing the time of diagnosis.
The aim of the work reported here was to investigate the potential of B. nummularia as bioindicator of beech health conditions in Italy, using a qPCR assay ([40]) to assess the occurrence of this fungus in asymptomatic twigs.
Material and methods
Study area
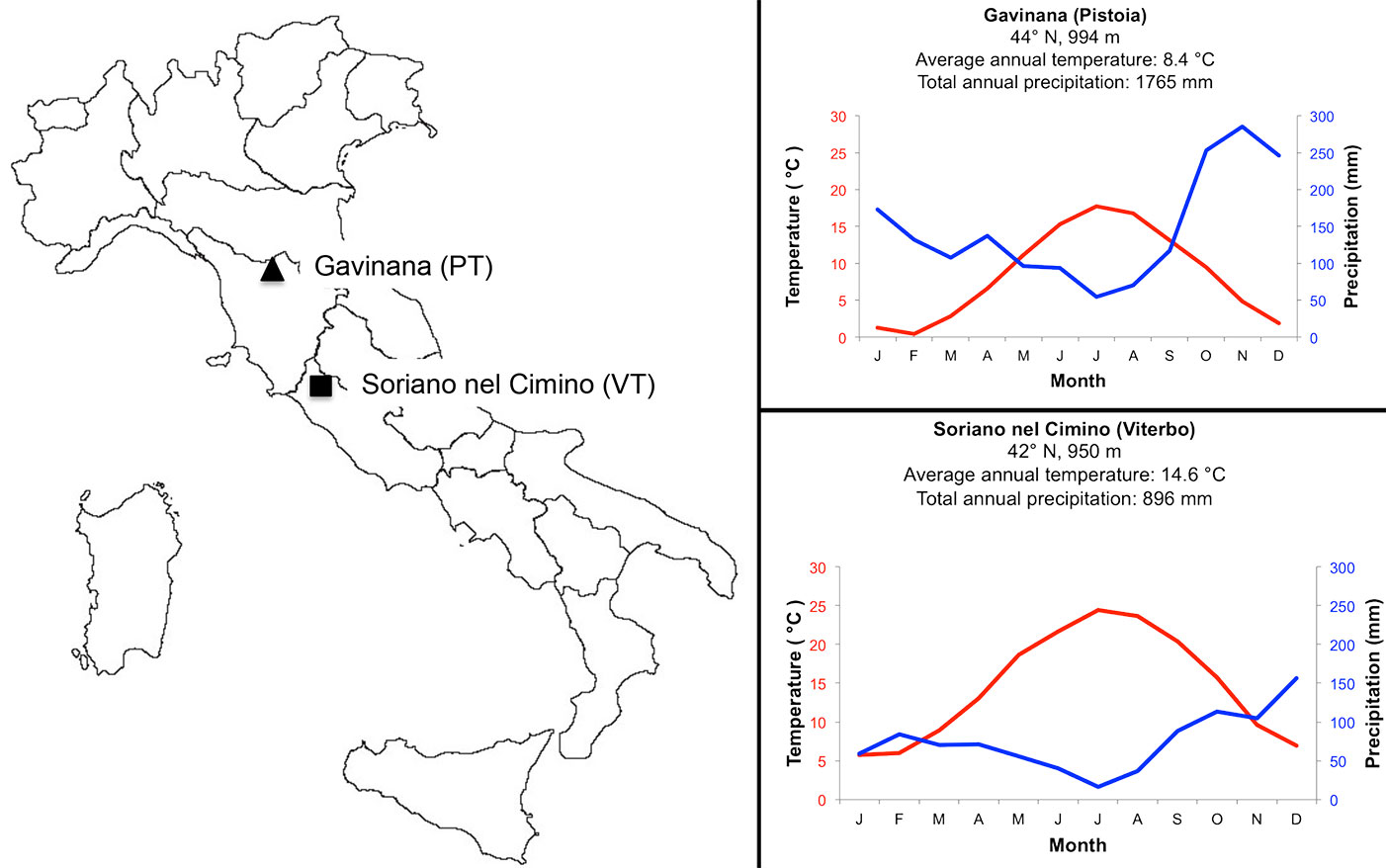
The survey was carried out between 2007 and 2008 comparing beech samples from two distinct phytoclimatic regions: one forest in the Northern Apennines (Gavinana - Pistoia) and the second forest in the central Apennines (Soriano nel Cimino, Viterbo, Italy - Fig. 1). The geographic and climatic characteristics of the sites are given in Tab. 1. Climatic data were provided by the AgroMeteo (⇒ http://agrometeo.arsia.tos cana.it) and the Banca Dati Agrometerologica Nazionale (⇒ http://cma.entecra.it) services for the Gavinana (Pistoia) and Soriano nel Cimino (Viterbo) site, respectively. The meteorological stations were at similar altitude and approximately 10 Km from the sampling sites. Sites were different in terms of climate conditions. In the period between 2003-2007 Gavinana had a shorter dry period (from May to September) in comparison with Soriano nel Cimino (from March to November - Fig. 1). During the sampling period (2007-2008), the annual precipitation was 1948 mm year-1 at Gavinana and 897 mm year-1 at Soriano nel Cimino. In the same period, highest values of precipitation generally occurred in November for Gavinana and October for Soriano nel Cimino. The average annual temperatures in 2007-2008 were 8.69 °C (Gavinana) and 14.8 °C (Soriano del Cimino).
Fig. 1 - (Left panel): location of beech-sampling areas along the Apennines Mountains; (right panel): Walter-Lieth diagram of the two sampling sites in the period 2003-2008.
Tab. 1 - Characteristics of beech sampling forests in the Apennine Mountains (Italy).
| Sampling site | Longitude N |
Latitude E |
Elevation (m a.s.l.) |
Phytoclimatic unit |
Xerothermic index (Xi) |
|---|---|---|---|---|---|
| Soriano nel Cimino (VT) | 42°24′ 36.9″ | 12°12′ 08.9″ | 950 | Aquifolio-Fagion | 38.5 |
| Mediterranean climate | |||||
| Gavinana (PT) | 44°04′ 07.9″ | 10°48′ 41.7″ | 994 | Luzulo-Fagion | 0 |
| Sub-continental climate |
For each sampling area a xerothermic index (Xi) was calculated using the following formula (eqn. 1):
where TM is the monthly mean of the maximum and minimum temperatures in °C and P is the monthly precipitation in mm ([24]). Xi was calculated on a historical data series from the last 10 years (1997-2007).
Previous studies showed the presence of Phytophthora cactorum and B. nummularia in the Soriano del Cimino forest ([64], [11]), while in Gavinana the stromata of B. nummularia were mainly associated with declining beech trees, especially on the edge of the forest ([21]).
Latent infections of B. nummularia
For each area two slopes (N-S) were chosen in the same growing conditions and 10 asymptomatic beech trees of the same age (five on a northern and five on a southern facing slope) were arbitrarily selected inside the forest. Site topographies were gently sloping (5%). No symptomatic beech trees were present close to those sampled. Each tree was numbered and its position registered with GPS for the subsequent sampling. From each tree, three arbitrarily selected apical portions of current-year shoots were collected from the lower part of the crown, with a total of 30 twigs per stand. Collections were made in autumn (25 October 2007), winter (13 February 2008), spring (3 June 2008) and summer (1 September 2008). A total of 240 twigs were collected.
Twig segments (30 mm length, 5-6 mm diameter) were surface sterilized in the lab with 75% ethanol (1 min), 3% NaClO (3 min) and 75% ethanol (30 s) and rinsed three times with sterile water ([37]). Each twig was split longitudinally into two parts containing both wood and bark tissue: one part was utilized for DNA extraction and the second for fungal culturing in agar media. The portion of twig used for fungal isolation was cut into 15 small fragments (2-3 mm each), placed on 1.5% PDA (potato dextrose agar - Difco, Milan, Italy) and incubated in darkness at 20°C for 10 days ([40]).
The portions of twigs used for DNA extraction (100 mg fresh weight) were transferred to 2-ml microfuge tubes and ground with a Mixer Mill 300® (Qiagen, Valencia, CA, USA) for 2 min (20 Hz). DNA was extracted from all samples using the DNeasy® Plant Minikit (Qiagen), as described previously ([40]). B. nummularia DNA was detected and quantified by real-time quantitative PCR (qPCR) using specific primers and the TaqMan™ probe ([40]). Quantitation of B. nummularia DNA was expressed as pg per mg fresh twig weight (pg fungal DNA/mg fw).
Data analysis
Frequency of occurrence (OF) of B. nummularia was calculated with the formula (eqn. 2):
where Ni is the number of twig segments in which B. nummularia was detected and Nt is the total number of segments tested.
Statistical analysis of data on the detection of B. nummularia among trees in each area, between sites, detection methods and seasons was calculated using the χ2 test. The D’Agostino-Pearson K2 test ([16]) was applied to test for departure from normality of data (α = 0.05). Data were also tested for homogeneity of variances using the Levene’s test, and equality of variances using plots of residuals and the Shapiro-Wilks test. Comparisons of differences in the amount of B. nummularia were performed after log transformation. The results were analysed using one-way analysis of variance (ANOVA - α = 0.05). Differences among means were tested by the post-hoc Tukey’s test (α = 0.05). An unpaired, two-tails test (t-test) was used to compare the sampling periods between sites. The temporal trend in B. nummularia abundance was analyzed by regression analysis using B. nummularia abundance (total pg fungal DNA) as dependent variable and the precipitation of the month before sampling (mm) as predictor. DNA data were log transformed to take into consideration the multiplicative effect of the independent variable (precipitation). All analyses were performed using the Graphpad Instat® software (San Diego, CA, USA).
Results
Detection of B. nummularia in symptomless beech trees
A total of 240 F. sylvatica twigs (120 for each forest) were processed to determine the presence of B. nummularia. The fungus was detected from samples using both cultural and molecular methods.
Frequency of occurrence of the fungus at each site was not significantly influenced by the tree aspect (χ2[1] = 2.09; P > 0.05), thus data from both sites were pooled. No significant differences (χ2[1] = 0.02; P > 0.05) were found in the annual frequency of occurrence of B. nummularia between the Soriano nel Cimino (OF = 25%) and Gavinana (OF = 23.3%) sites. No differences in isolation were observed between single trees. B. nummularia was significantly more frequently found using qPCR (72.5%) than the cultural method (24.1% - χ2[1] = 112.2; P < 0.001). Overall frequency of detection using qPCR was greater for samples from Soriano nel Cimino (90.8%) than for those collected in Gavinana (54.1% - χ2[1] = 40; P < 0.001). Due to the higher sensitivity of the molecular detection method, only data based on qPCR were used in further analyses.
Seasonality of B. nummularia occurrence
The percentage of qPCR-positive twig samples was significantly higher in the Soriano nel Cimino than in Gavinana site both in autumn (χ2[1] = 22.5; P < 0.001) and winter (χ2[1] = 28.7; P < 0.001). In contrast, spring detection of B. nummularia was higher in twigs collected at Gavinana (100%) than at Soriano nel Cimino (86.7% - χ2[1] = 4.28; P=0.04 - Tab. 2). As for summer, no significant differences were observed between the two sampling sites (χ2[1] = 1.4; P>0.05 - Tab. 2).
Tab. 2 - Results of Biscogniauxia nummularia detection by qPCR from beech trees growing in two different forests in central Italy. Sampling periods: Autumn 2007, Winter 2008, Spring 2008 and Summer 2008.
| Sampling period | Pres/Abs | qPCR detection of B. nummularia n. (%) of twigs inspected (out of 30 twigs) | Total | χ2 | P | |
|---|---|---|---|---|---|---|
| Gavinana (PT) |
Soriano nel Cimino (VT) |
|||||
| Autumn | Presence | 8 (26.7) | 29 (96.7) | 37 | 31.09 | <0.001 |
| Absence | 22 (73.3) | 1 (3.3) | 23 | |||
| Total | 30 (100) | 30 (100) | 60 | |||
| Winter | Presence | 10 (33.3) | 30 (100) | 40 | 30.00 | <0.001 |
| Absence | 20 (66.6) | 0 | 20 | |||
| Total | 30 (100) | 30 (100) | 60 | |||
| Spring | Presence | 30 (100) | 26 (86.7) | 56 | 4.28 | 0.04 |
| Absence | 0 | 4 (13.3) | 4 | |||
| Total | 30 (100) | 30 (100) | 60 | |||
| Summer | Presence | 17 (56.7) | 24 (80.0) | 41 | 1.4 | >0.05 |
| Absence | 13 (43.3) | 6 (20.0) | 19 | |||
| Total | 30 (100) | 30 (100) | 60 | |||
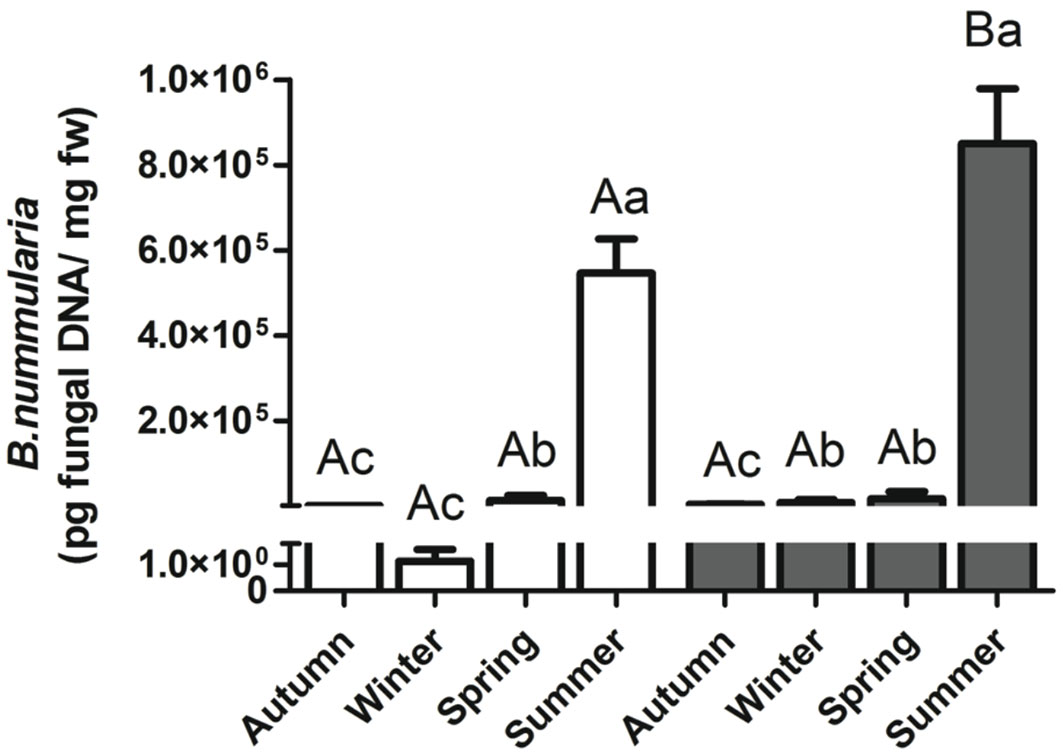
B. nummularia DNA in symptomless beech tissues was quantified by qPCR for each sample in both forests. Quantities ranged from 1.14E-02 to 2.26E+06 pg/mg of fresh weight beech tissue. Results from ANOVA showed significant differences in fungal DNA among sampling periods both in Soriano nel Cimino (F[1, 119] = 80.07; P < 0.001) and Gavinana (F[1, 119] = 319.9; P < 0.001). An increasing trend in the amount of fungal DNA was detected along the vegetative season, with a minimum in autumn (Soriano nel Cimino) or winter (Gavinana), and the maximum in summer at both sites (Fig. 2). The amount of B. nummularia DNA in healthy beech twigs was significantly higher in summer in Soriano nel Cimino (unpaired test, P=0.035) than in Gavinana (Fig. 2). No significant differences were detected for the other sampling periods (unpaired test, P > 0.05).
Fig. 2 - Quantification of Biscogniauxia nummularia DNA using qPCR in the northern (Gavinana, PT - white bars) and central Apennines (Soriano nel Cimino, VT - grey bars), in Autumn 2007, Winter 2008, Spring 2008 and Summer 2008. Different lowercase letters indicate significant differences among seasons (ANOVA, P < 0.05). Different capital letters indicate significant differences between two sites in the same season (Unpaired test, P < 0.05). Bars and whiskers represent the standard error and the mean value, respectively.
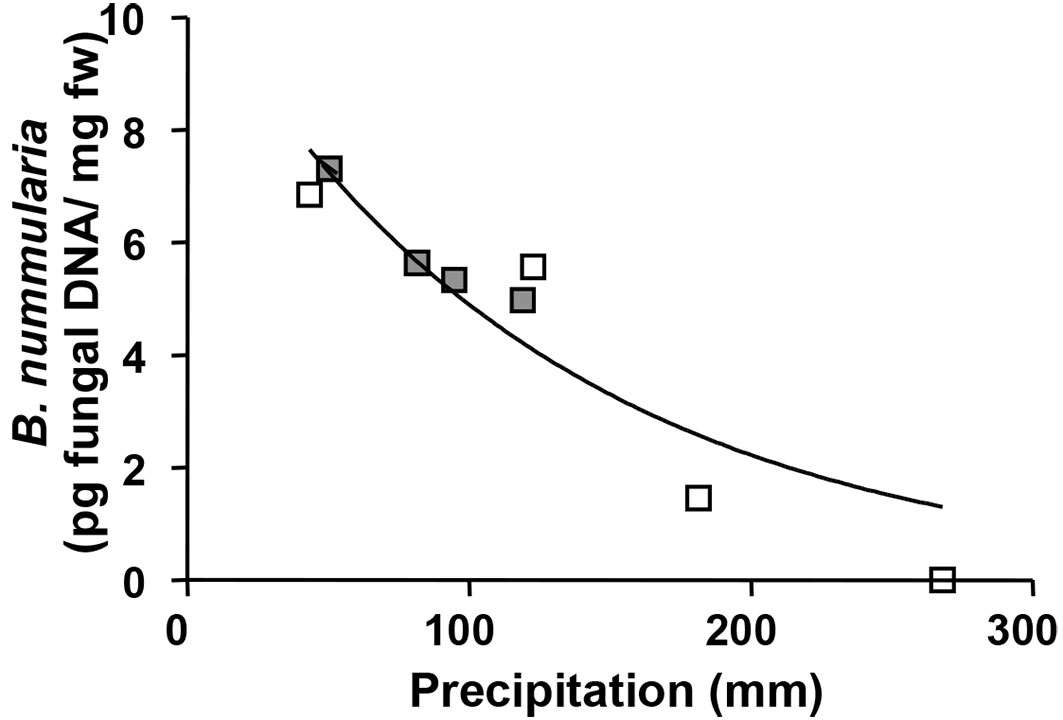
Regression analysis revealed that pathogen occurrence (in terms of DNA amount detected) was inversely proportional to the total precipitation of the month preceding the sampling (P<0.05, R2 = 0.86 - Fig. 3).
Fig. 3 - Amount of Biscogniauxia nummularia DNA detected in symptomless beech tissue by qPCR and inverse relationship with the precipitation (previous month of the sampling time) in Gavinana (white square) and Soriano nel Cimino sites (grey square).
Discussion
The results of this investigation confirmed that a specific qPCR assay could be used to detect B. nummularia DNA in the apical shoots of Fagus sylvatica, as already reported in a previous study ([40]). The qPCR technique has been successfully applied to quantify the fungal pathogen in a range between 1.14E-02 to 2.26E+06 pg/mg of fresh weight. The sensitivity of this technique has significantly improved in the last few years. Indeed, qPCR is currently a reliable method to detect even small amounts of latent pathogens in the trees, before visible symptoms occur in the host ([39], [42]).
Differential patterns of B. nummularia occurrence in symptomless beech trees in the two Apennines forests were likely due to their different climatic regimes, Mediterranean in Soriano nel Cimino (VT), and continental in Gavinana (PT). However, more pronounced differences in B. nummularia DNA amounts were observed in tissues at the end of the growing season in the site characterized by a warmer climate (Xi = 38.5), and with a longer arid period (Fig. 1). In this context, it can be hypothesized that water stress in beech stands may lead to an increase of the amounts of fungal inoculum within apparently healthy tissue, with possible changes in host susceptibility.
Interactions between water stress and the activity of forest pathogens have been recognized since long time ([47], [43], [30]). Both air moisture and soil drought have important roles in the epidemiology of tree diseases. Drought-induced tree diseases are often caused by secondary pathogens with endophytic abilities developing in secondary tissues (bark/wood) of stressed hosts ([34]). Such fungi may establish interactions with the tree ranging from mutualism to antagonism depending on host physiology and environmental conditions. Nevertheless, water stress can trigger the disease development by endophytic fungi in asymptomatic hosts. In this sense, the presence of and fluctuations in populations of pathogenic endophytes can reflect physical changes in the environment ([17]).
In this study fluctuations in the amounts of B. nummularia were also observed in symptomless beech twigs, with significant differences occurring among sampling periods in both sites. The amount of fungal DNA was significantly higher during the vegetative season (summer) in both sampled forests. From an ecological point of view, B. nummularia behaves in a similar way to B. mediterranea, which proliferates in asymptomatic tissues of Q. cerris during dry growing seasons ([62]).
The increase of quantities of B. nummularia within symptomless tissues may have a negative impact on beech health, possibly inducing the outbreak of disease, when climatic or host conditions change. Previous studies have also suggested that plant pathogenic fungi with endophytic behavior are sensitive to variations in host physiology driven by stress conditions ([35], [17]). In conifers, outbreaks of Diplodia sapinea have been attributed to several stress factors and experimental work has underlined the interaction with water stress in the Pinus - D. sapinea pathosystem ([59], [47]). In Fagaceae species of the Mediterranean area, B. mediterranea - the causal agent of charcoal disease of oak - can rapidly colonize the xylem and bark tissues of Q. cerris and Quercus ilex ssp. ballota subjected to water stress, inducing necrosis and canker formation and accelerating tree decline and death ([17], [33], [8]). Water stress favors the proliferation of the fungus in the endophytic phase; the pathogenic ability of B. mediterranea induces cankers and wood decay ([63], [14], [62]). Similarly, Bassett & Fenn ([2]) showed that asymptomatic seedlings of Quercus alba L. and Q. velutina Lam. naturally infected with B. atropunctata were rapidly colonized by the pathogen under water stress conditions.
The richness of fungal endophyte populations is strictly related to environmental parameters, such as temperature and rain ([26], [67]). Recently, it was shown that the composition of leaf-associated fungi on F. sylvatica is correlated primarily with the annual mean temperature ([15], [13]). In the present work, the detection B. nummularia DNA in symptomless tissue of beech showed a significant decreasing trend as the mean precipitation of the month preceding the sampling increases (R2= 0.86), i.e., the amount of latent infection increased with decreasing precipitation. A relationship between environmental parameters and fungal behavior has been observed in several studies. Gange et al. ([20]) showed that changing temperature and rainfall regimes cause different germination and growth rates of Auricularia auricula-judae. Straatsma et al. ([61]) observed a correlation between the time of fruitbody appearance and temperature in a Swiss forest plot. In the Diplodia-Austrian pine pathosystem variations in the quantities of D. sapinea DNA in healthy pine shoots was positively correlated with the yearly amount of solar radiation received by trees ([42]).
Variation in latent pathogens in apparently healthy plant tissues is important to consider when monitoring the health status of plants. Usually symptoms caused by biotic or abiotic factors represent successful bioindicators in assessing the health of forest trees ([57]). However, several studies have demonstrated the effectiveness of flora such as lichens as bioindicators of forest health ([38], [31], [41]). Fungi colonizing plant tissues also could be considered as potential bioindicators of environmental changes. For example, Helander ([27]) found a negative effect of air pollution on fungal endophytes in pine needles along a gradient from two factory complexes. Recently, Romeralo et al. ([55]) showed that the frequency of fungi inside pine needles was negatively correlated with air quality. Furthermore, fungal endophytes were also used as bioindicators of tree vitality. Several authors reported a negative correlation between needle fungal endophytes and P. abies vitality ([1], [58], [54]). Some fungal species may change their behavior towards a plant host after the alteration of environmental conditions ([58], [4]).
The ability of latent pathogens to quickly adapt to new environmental conditions suggests that these organisms could be used as bioindicators of forest health ([62], [55]), in that: (i) their presence can be easily detected; (ii) they are sufficiently sensitive to provide an early warning of change; (iii) they are widely applicable, and independent of sample size. Such characteristics match the criteria for their selection as good markers ([32]). In this study, B. nummularia was easily detected using a molecular method based on qPCR. In addition, the fungus occurred more frequent in the Mediterranean bioclimatic area, in which beech trees are more susceptible to drought stress, predisposing the host to fungal disease.
Conclusions
The identification of target species or functional groups for assessing the effects of climate on forests is particularly important in a scenario of global climatic change that affects the ecosystem functions and the survival of species ([60]). Italy and the Mediterranean basin in general are considered hot-spots for climatic change. Models for these areas predict extreme climatic events with prolonged drought periods and anomalous precipitation events that might seriously affect sustainability of several ecosystems ([12]). Forests are particularly sensitive to climate change, because the long lifespan of trees does not allow for a rapid adaptation response ([29]).
Tools for assessing the general health status of individual trees or forest community, as well as for forecasting the outbreak of complex decline syndromes, may increase in importance in the next future. During recent years, climate change has altered host distributions, with shifts towards higher altitudes ([29]). This phenomenon has been observed in several tree species, including European beech that shifted upwards at the highest altitudes, displacing heath lands and grasslands and a few conifer species ([49]). Such shifts could potentially favor the spread of pathogens into new bioclimatic zones causing additional damage. Considering that beech is a common forest species along the Italian peninsula and is particularly sensitive to climatic conditions, the interactions of beech with B. nummularia (or potentially other Xylariaceous endophytes) might be used as a tool in monitoring the health status of mountain forests. The seasonal variation in fungal quantities within healthy beech tissues suggest that B. nummularia should be considered a possible bioindicator of beech health condition.
Acknowledgments
This paper is dedicated to the memory of Prof. Francesco Moriondo (1926-2014).
This work was supported by DIGESFAM (“DIfesa e GEStione FAggete Mediterranee” - Ministero delle Politiche Agricole e Forestali, Italy) and BACCARA (“Biodiversity And Climate Change, A Risk Analysis” - European Community’s Seventh Framework Program FP7/2007-2013) under the grant agreement n° 226299) projects. The authors are grateful to Prof. Stephen Woodward (University of Aberdeen, UK) for a critical review of the manuscript. We also appreciate the helpful comments and suggestions from the anonymous reviewers.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Online | Gscholar
Online | Gscholar
Authors’ Info
Authors’ Affiliation
National Research Council - Institute for Sustainable Plant Protection, (CNR-IPSP), Via Madonna del Piano 10, I- 50019 - Sesto Fiorentino, Firenze (Italy)
Matteo Feducci
Department of Agrifood Production and Environmental Sciences, University of Florence (DISPAA). Piazzale delle Cascine, 28, I-50144, Firenze (Italy)
Barbara Ceccarelli
Anna Maria Vettraino
Department for Innovation in Biological, Agro-food and Forest Systems, University of Tuscia (DIBAF), Via San Camillo De Lellis, I- 01100, Viterbo (Italy)
Corresponding author
Paper Info
Citation
Luchi N, Capretti P, Feducci M, Vannini A, Ceccarelli B, Vettraino AM (2015). Latent infection of Biscogniauxia nummularia in Fagus sylvatica: a possible bioindicator of beech health conditions. iForest 9: 49-54. - doi: 10.3832/ifor1436-008
Academic Editor
Elena Paoletti
Paper history
Received: Aug 29, 2014
Accepted: Mar 05, 2015
First online: Jun 18, 2015
Publication Date: Feb 21, 2016
Publication Time: 3.50 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2015
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 54380
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 44065
Abstract Page Views: 4445
PDF Downloads: 4204
Citation/Reference Downloads: 72
XML Downloads: 1594
Web Metrics
Days since publication: 3870
Overall contacts: 54380
Avg. contacts per week: 98.36
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2016): 20
Average cites per year: 2.00
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Real-Time PCR for Ceratocystis platani detection: in-depth validation to assess the diagnostic potential and include additional technical options
vol. 11, pp. 499-509 (online: 18 July 2018)
Research Articles
Variation in resistance to the rust fungus Melampsora larici-populina Kleb. in Populus nigra L. in the Czech Republic
vol. 9, pp. 146-153 (online: 26 October 2015)
Research Articles
Identification and molecular characterization of LTR and LINE retrotransposable elements in Fagus sylvatica L.
vol. 2, pp. 119-126 (online: 10 June 2009)
Research Articles
Patterns of genetic diversity in European beech (Fagus sylvatica L.) at the eastern margins of its distribution range
vol. 10, pp. 916-922 (online: 10 December 2017)
Research Articles
Kretzschmaria deusta, a limiting factor for survival and safety of veteran beech trees in Trentino (Alps, Northern Italy)
vol. 14, pp. 576-581 (online: 18 December 2021)
Review Papers
Dutch elm disease and elm bark beetles: a century of association
vol. 8, pp. 126-134 (online: 07 August 2014)
Research Articles
Suitability of Fagus orientalis Lipsky at marginal Fagus sylvatica L. forest sites in Southern Germany
vol. 15, pp. 417-423 (online: 19 October 2022)
Research Articles
Effects of abiotic stress on gene transcription in European beech: ozone affects ethylene biosynthesis in saplings of Fagus sylvatica L.
vol. 2, pp. 114-118 (online: 10 June 2009)
Research Articles
Hearing nature’s heartbeat: towards large-scale real-time forest monitoring network in Italy
vol. 18, pp. 202-211 (online: 09 August 2025)
Research Articles
Application of fungicides and urea for control of ash dieback
vol. 8, pp. 165-171 (online: 13 August 2014)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword