Soil chemical and physical status in semideciduous Atlantic Forest fragments affected by atmospheric deposition in central-eastern São Paulo State, Brazil
iForest - Biogeosciences and Forestry, Volume 8, Issue 6, Pages 798-808 (2015)
doi: https://doi.org/10.3832/ifor1258-007
Published: Apr 22, 2015 - Copyright © 2015 SISEF
Research Articles
Collection/Special Issue: IUFRO 7.01.00 - Ilhéus (Brazil, 2013) & Beijing (China, 2014)
Forest Response to Climate Change and Air Pollution
Guest Editors: Paoletti E, Kozovitz A, Feng Z, Bytnerowicz A
Abstract
The expansion of agricultural, urban and industrial areas in the São Paulo State (SE Brazil) led to the fragmentation of the original semideciduous Atlantic Forest into small, patchy forest remnants. Anthropogenic activities produce a variety of pollutants affecting many ecological processes in these remaining forest fragments through soil acidification and fertilization. In this study, we investigated the soil chemical and physical status of six forest remnants (Paulínia, Holambra, Americana, Jaguariúna, Campinas and Cosmópolis) differently affected by industrial, rural and urban pollution in central-eastern São Paulo in order to determine the soil potential to buffer the inputs of pollutants. Soil samples from 0-10, 10-20 and 20-40 cm depths were collected in the dry and the wet season and the following variables were analyzed: soil texture, pH in CaCl2 solution, exchangeable cations and exchange capacity, organic carbon, total nitrogen, extractable sulfur, phosphorus and heavy metals. Distinct buffering capacities were observed in industrial and in rural and urban areas, primarily due to the natural characteristics of the soils, such as soil texture, acidification and organic matter. The forest soils affected by atmospheric deposition from the industrial complex (Paulínia and Americana) were more sandy and acidic (pH = 3.6) than those near rural and urban sources (pH = 4.5). The optimal chemical conditions (high contents of organic matter, exchangeable bases, nitrogen, phosphorus and sulfur) were found in the clay soils of forest remnants located in Campinas and Jaguariúna, which were more affected by rural or urban pollution than by industrial emissions. Such clay soils provide the highest buffering capacity against environmental impacts in the study region.
Keywords
Tropical Soils, Atlantic Forest, Urban, Rural and Industrial Pollution, Soil Acidification, Buffering Capacity
Introduction
The Brazilian Atlantic biome is recognized as one of the hotspots for the conservation of biodiversity ([18]), though it is severely affected by diverse human interferences, particularly in the State of São Paulo, southeastern Brazil ([10]). The semideciduous Atlantic Forest is highly fragmented in central-eastern São Paulo because of the expansion of agriculture and urban and industrial growth ([47]). Besides the reduction of forest cover, human activities bring about a variety of pollutants from combustion of fuels, waste disposal, long-term sewage sludge and fertilizer application and other sources, which may be toxic to the plant community and modify the chemical status of soil, depending on its original chemical and physical conditions ([48], [60], [58], [51], [37]). Soil nutrients are among the main factors that regulate plant growth and play an important role in the sustainable use of soils; however, their excess due to pollution from human sources may damage the soils and affect the soil-plant relationships. Air pollution may also disrupt other nutrient cycling processes in natural ecosystems, such as the decomposition of litter ([9]).
Most anthropogenic pollutants deposited in forest ecosystems accumulate in the soil surface layers ([56]), where pollutants are typically immobilized for long periods ([21], [66]). However, these accumulations are considered a “chemical time bomb” ([29], [23]), because pollutants will eventually be leached into waterways ([42]).
Increased deposition of sulfur and nitrogen compounds (SO2, NOx and NH3) in natural communities induces soil acidification ([13], [1], [20]). Soil acidification depletes the basic nutrient cations, causes a decrease of pH, lowers the quality of humus, and accelerates the mobilization of aluminum ([4], [43]). The depletion of basic cations from the forest floor alters the mineral nutrition of trees, modifies tree growth patterns ([33], [26], [59]) and affects the distribution of roots ([28]). As a consequence, biodiversity, vegetation productivity and dynamics of the soil carbon pool are affected ([3]).
Altogether, such negative effects contribute to the forest decline observed in the Atlantic Rain Forest on the coastal mountain range named Serra do Mar (region of Cubatão, São Paulo State, Brazil). This forest is affected by air pollution from an industrial complex that caused destabilization of the land surface, disturbance of soil processes and landslides ([34], [40], [41]). Furthermore, the large metropolitan regions of the São Paulo State (SE Brazil) are responsible for the intense fragmentation of the semideciduous Atlantic Forest. This is observed in the Metropolitan Campinas Region (MCR) located in the central eastern region of the State (Fig. 1), which is composed of 19 municipalities with different types of land use (industrial, agricultural and urban areas). In addition to forest fragmentation, the expansion of agricultural, urban and industrial areas in the MCR has led to increased pollutant emissions, such as sulfur and nitrogen dioxides ([5]) that acidify the rain ([65]). Unless the natural characteristics of the forest fragments increase the resilience against pollution impacts, the atmospheric contamination and acid rain may endanger the local soil and may affect many of the ecological processes in these remaining forest patches.
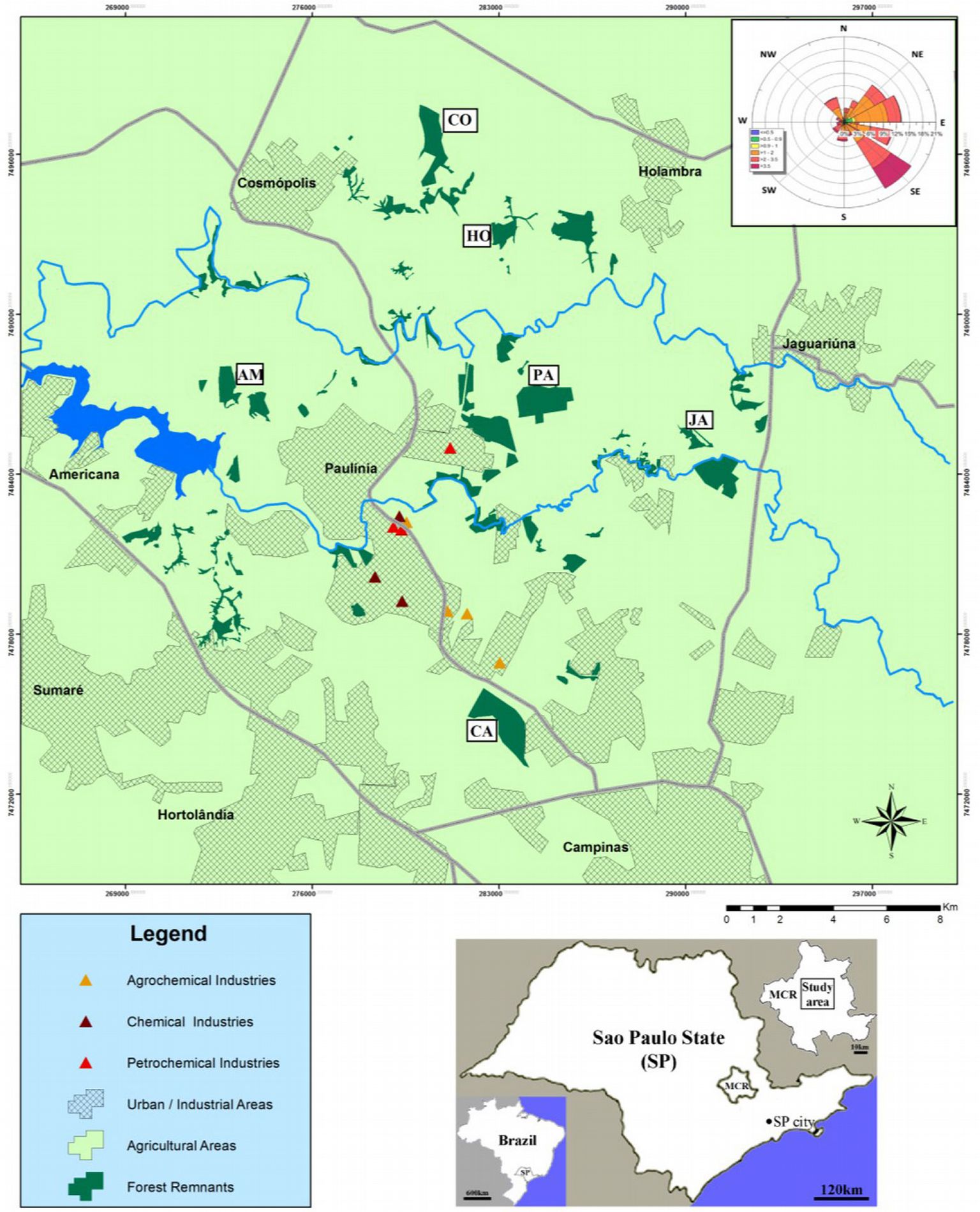
Fig. 1 - Locations of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil. (PA): Paulínia; (HO): Holambra; (AM): Americana; (JA): Jaguariúna; (CA): Campinas; (CO): Cosmópolis. The wind rose in the upper right corner summarizes the annual wind velocities (shown by distinct colors in m s-1), frequencies (%) and directions (distributed in the different azimuth degrees).
Because the soil is usually the most important buffer of forest ecosystems against such impacts, we analyzed the soils of the leftover forest fragments to determine their potential to buffer the pollutant inputs. We investigated the soil chemical and physical status in six forest remnants differently affected by industrial, rural and urban pollution in municipalities in central-eastern São Paulo. We assumed that wet and dry deposition of air pollutants in the MCR changed the soil chemical status in the semideciduous Atlantic Forest remnants, particularly those located downwind from the industrial area. Additionally, we assumed that soils with high buffering potential against atmospheric deposition in the MCR had the optimal chemical conditions. This study is the first of a series of ecological surveys aimed at assessing the impact of the complex mixture of air pollutants on semideciduous Atlantic Forests in the MCR, and planning the conservation of their high biodiversity.
Material and Methods
Study area
Oxisol soils and gentle sloping topography characterize the Metropolitan Campinas Region (MCR - [31], [52]). The predominant climate in the MCR is classified as B1rB’4a according to the Thornthwaite’s typology, or Aw (tropical with dry winter) according to Koeppen’s classification ([54]). The MCR is characterized by a hot and rainy season between October and March and a dry season between April and September. The average temperature ranges from 23.2 to 24.9 °C, and the rainfall is 1142 mm in the rainy season, while uuring the dry season the average temperature ranges from 18.5 to 23.0 °C, and the rainfall is 283 mm. The predominant wind direction is southeast (see the wind rose included in Fig. 1).
The MCR is the second most important economic center of the São Paulo State and is located 100 km from the São Paulo metropolitan region. The largest Brazilian oil refinery is located in Paulínia, which refines 20% of the crude oil in Brazil. Moreover, the industrial complex includes chemical, pharmaceutical and fertilizer industries ([64]). The high vehicle traffic associated with industrial and extensive agriculture activities remarkably contributes to increase the atmospheric pollution of the region ([44]).
Six semideciduous forest remnants were selected at different distances from the industrial areas of Paulínía, Holambra, Americana, Jaguariúna, Campinas and Cosmópolis municipalities (Fig. 1, Tab. 1). The largest forest remnant was located in Campinas (235 ha), followed by those in Paulínia (180 ha) and Cosmópolis (120 ha). Tree density varied from 400 ind ha-1 in Jaguariúna to 706 ind ha-1 in Cosmópolis, and the basal area ranged from 17.2 m2 ha-1 in Paulínia to 29.7 m2 ha-1 in Holambra. The soils were formed from different parent materials, from sandstone to diabase, which resulted in oxisols with medium (red-yellow oxisol) to high fertility (red oxisol). All forest remnants contained considerable litter stocks (8.1-11.8 Mg ha-1), as typically observed in other tropical forests ([50], [63]). The predominant land use around the forest remnants is agriculture in Holambra and Americana, industry and agriculture in Paulínia, while a mix of urban and agricultural areas is found in Campinas and Jaguariúna (Tab. 1).
Tab. 1 - Main characteristics (area, distance from industrial complex, land use in surrounding areas), location (geographical coordinates), soil (litter layer, type, parent material) and vegetation structure (basal area, tree density) of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil. (*): Domingos et al. ([11]); (**): A.R. Santos et al., unpublished data.
| Characteristics | Paulínia (PA) |
Holambra (HO) |
Americana (AM) |
Jaguariúna (JA) |
Campinas (CA) |
Cosmópolis (CO) |
|---|---|---|---|---|---|---|
| Area (ha) | 180 | 47 | 74 | 6 | 235 | 120 |
| Distance from industrial complex (km) |
4.5 | 9.3 | 10.0 | 10.5 | 11.2 | 12.1 |
| Geographical coordinates | 22° 44′ 58″ S 47° 05′ 55″ W |
22° 39′ 33″ S 47° 06′ 42″ W |
22° 42′ 30″ S 47° 12′ 40″ W |
22° 43′ 35″ S 47° 01′ 32″ W |
22° 49′ 22″ S 47° 06′ 17″ W |
22° 37′ 38″ S 47° 08′ 02″ W |
| Land use in surrounding areas |
Industrial/Agricultural (sugarcane) | Agricultural (citrus sugarcane) |
Agricultural (sugarcane) |
Urban/Agricultural (sugarcane) | Urban/Agricultural (sugarcane) | Agricultural (sugarcane) |
| Basal area (tree dbh >10 cm, m2 ha-1)* |
17.2 | 29.7 | 19.2 | 20.2 | 26.8 | 21.5 |
| Tree density (ind ha-1)* |
556 | 588 | 550 | 400 | 528 | 706 |
| Litter layer (Mg ha-1)** |
9.1 | 10.3 | 9.1 | 11.8 | 10.5 | 8.1 |
| Soil Type | red-yellow oxisol | red-yellow oxisol | red-yellow oxisol | red oxisol | red oxisol | red-yellow oxisol |
| Parent material | diabase sandstone |
Sandstone | siltstone rhythmites |
diabase | diabase | diabase sandstone siltstone |
Soil sampling and analyses
Soil sampling procedures were based on protocols for monitoring European forests ([7], [16], [17]), modified according to the forest physiognomy and environmental conditions found in the tropics. Two plots of 140 × 50 m were established at each sampling site (forest remnant) 100 m apart from the forest edge, totalling 14000 m2 at each site. Twenty-four soil samples were collected in each plot at 0-10, 10-20 and 20-40 cm depths in July 2010 (dry season) and January 2011 (wet season). Three composite samples per plot, layer and season (n = 12) were analyzed for each forest remnant, which were obtained by randomly mixing eight individual soil samples.
Samples were air-dried, sieved through a 2 mm sieve and analyzed for texture, soil nutrients and heavy metals, according to EMBRAPA ([12]). Soil texture (granulometry) was determined with the Boyoucos hydrometer method. The soil pH was measured in 0.01 mol L-1 CaCl2 solution (soil/CaCl2 ratio 1:2.5). Available phosphorus (P) and the exchangeable potassium (K), calcium (Ca) and magnesium (Mg) from soils were simultaneously extracted using a ion-exchange resins, following the method proposed by Raij et al. ([53]). The extraction procedure included the disaggregation of soil by shaking in water, the transfer of elements from the soil to a sodium bicarbonate treated mixture of anion and cation exchange resins, and the separation of the resin from the soil by sieving and extraction of elements from the resin. Phosphorus was determined spectrophotometrically with the molybdenum blue complex, K by the flame emission spectrometry, and Ca and Mg with the atomic absorption spectrometry. Al was extracted with a KCl solution (1 mol L-1) and determined by titration with 0.025 mol L-1 NaOH. The organic carbon (Corg) was determined by colorimetry, after organic matter oxidation with Na2Cr2O7 2H2O and H2SO4. Total nitrogen (Ntot) was determined by the Kjeldahl distillation method, after H2SO4 digestion. Extractable sulfur (S) was determined by the turbidimetric method in soil extracts prepared with Ca(HPO4)2. Extractable heavy metals (Cu, Fe, Mn and Zn) in soil extracts prepared with DTPA-TEA were determined with atomic absorption spectrometry. The cation exchange capacity (CEC = Ca+Mg+ K+Al+H), aluminum saturation (m% = [Al/ Ca+Mg+K+Al]·100), base saturation (V% = [(Ca+Mg+K)/CEC]·100) and C/N ratio were calculated.
Statistical analyses
Differences in physical and chemical soil properties among forest remnants (sampling sites) were tested by non-parametric ANOVA (Kruskal-Wallis test - α = 0.05), followed by multiple-comparison tests (Dunn’s method). Pairwise Spearman’s correlation coefficients among soil characteristics were also calculated for testing variable associations. All the above statistical analyses were performed using the software package SJHNAPMPU® ver. 11.0 (Systat Software Inc., San José, CA, USA).
Principal component analyses (PCA) using the software package PC-ORD® 6.0 (MjM Software Design, Gleneden Beach, OR, USA) and cluster analysis using the SPSS® 7.0 software (IBM, NY, USA) were performed to group similar forest remnants based on soil variables after log10 transformation (pH-CaCl2, carbon, nitrogen, C/N ratio, phosphorus, sulfur, cation exchange capacity, aluminum saturation, base saturation, copper, iron, manganese, zinc, sand and clay). Cluster analysis was perfomed using the Ward’s algorithm, and the distances between the fifteen variables were calculated by Pearson’s correlation.
Results
Soil texture (granulometry) at all depths was sandy-clay in Americana, sandy-clay-loam in Paulínia, Holambra and Cosmópolis and clay in Jaguariúna and Campinas (Tab. 2). Significant negative correlations (p < 0.001) were found between the sand content and Corg, Ntot, S and Cu concentrations in soils. Conversely, significant positive correlations were identified between the above elements and the clay content (Tab. 3).
Tab. 2 - Soil texture at 0-10 cm, 10-20 cm and 20-40 cm depths of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil (mean ± standard error, n = 12).
| Sampling site | Depth (cm) |
Sand (g kg-1) |
Silt (g kg-1) |
Clay (g kg-1) |
Texture |
|---|---|---|---|---|---|
| Paulínia (PA) | 0 - 10 | 698 ± 37 | 55 ± 6 | 247 ± 33 | Sandy-clay-loam |
| 10 - 20 | 684 ± 33 | 49 ± 6 | 267 ± 29 | Sandy-clay-loam | |
| 20 - 40 | 666 ± 36 | 46 ± 4 | 288 ± 33 | Sandy-clay-loam | |
| Holambra (HO) | 0 - 10 | 702 ± 20 | 81 ± 15 | 217 ± 15 | Sandy-clay-loam |
| 10 - 20 | 704 ± 19 | 79 ± 8 | 217 ± 16 | Sandy-clay-loam | |
| 20 - 40 | 700 ± 19 | 74 ± 9 | 226 ± 16 | Sandy-clay-loam | |
| Americana (AM) | 0 - 10 | 559 ± 12 | 75 ± 13 | 366 ± 18 | Sandy-clay |
| 10 - 20 | 546 ± 13 | 78 ± 15 | 376 ± 16 | Sandy-clay | |
| 20 - 40 | 521 ± 12 | 86 ± 10 | 393 ± 16 | Sandy-clay | |
| Jaguariúna (JA) | 0 - 10 | 346 ± 6 | 90 ± 2 | 564 ± 4 | Clay |
| 10 - 20 | 321 ± 6 | 77 ± 7 | 602 ± 1 | Clay | |
| 20 - 40 | 301 ± 6 | 85 ± 2 | 614 ± 4 | Clay | |
| Campinas (CA) | 0 - 10 | 372 ± 29 | 168 ± 4 | 460 ± 29 | Clay |
| 10 - 20 | 340 ± 26 | 163 ± 5 | 497 ± 29 | Clay | |
| 20 - 40 | 353 ± 28 | 171 ± 6 | 476 ± 31 | Clay | |
| Cosmópolis (CO) | 0 - 10 | 731 ± 17 | 55 ± 6 | 214 ± 13 | Sandy-clay-loam |
| 10 - 20 | 714 ± 19 | 52 ± 6 | 234 ± 13 | Sandy-clay-loam | |
| 20 - 40 | 693 ± 18 | 56 ± 7 | 251 ± 14 | Sandy-clay-loam |
Tab. 3 - Spearman’s correlation coefficients among edaphic variables in the 0-10 cm soil layer in semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil. (CEC): Cation exchange capacity = Ca+Mg+K+Al+H; (V%): Base saturation = [(Ca+Mg+K)/CEC]·100; (m%): Aluminum saturation = (Al/Ca+Mg+K+Al)·100. (*): p<0.05; (**): p<0.01; (***): p<0.001.
| Variable | Corg | Ntot | P | S | CEC | V% | m% | Cu | Fe | Mn | Zn | Sand | Clay |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Corg | 1.00 | - | - | - | - | - | - | - | - | - | - | - | - |
| Ntot | 0.86*** | 1.00 | - | - | - | - | - | - | - | - | - | - | - |
| P | 0.67*** | 0.65*** | 1.00 | - | - | - | - | - | - | - | - | - | - |
| S | 0.34 | 0.56*** | 0.43 | 1.00 | - | - | - | - | - | - | - | - | - |
| CEC | 0.74*** | 0.65*** | 0.67** | 0.35 | 1.00 | - | - | - | - | - | - | - | - |
| V% | -0.04 | 0.04 | 0.30 | 0.07 | -0.56*** | 1.000 | - | - | - | - | - | - | - |
| m% | -0.00 | -0.05 | -0.29 | -0.01 | 0.50*** | -0.96*** | 1.00 | - | - | - | - | - | - |
| Cu | 0.71*** | 0.69*** | 0.65*** | 0.33 | 0.43 | 0.20 | -0.26 | 1.00 | - | - | - | - | - |
| Fe | 0.29 | 0.18 | -0.06 | -0.17 | 0.65*** | -0.79*** | 0.77*** | 0.02 | 1.00 | - | - | - | - |
| Mn | -0.25 | -0.27 | 0.08 | -0.16 | 0.65*** | 0.81*** | -0.85*** | 0.10 | -0.77*** | 1.00 | - | - | - |
| Zn | 0.31 | 0.33 | 0.47 | 0.16 | -0.18 | 0.75*** | -0.75*** | 0.48 | -0.48 | 0.61*** | 1.00 | - | - |
| Sand | -0.55*** | -0.64*** | 0.47 | -0.62*** | -0.38 | 0.14 | 0.13 | -0.67*** | 0.06 | 0.07 | -0.37 | 1.00 | - |
| Clay | 0.53*** | 0.60*** | 0.44 | 0.60*** | 0.41 | 0.06 | -0.05 | 0.58*** | 0.03 | 0.43 | -0.95*** | 0.48 | 1.00 |
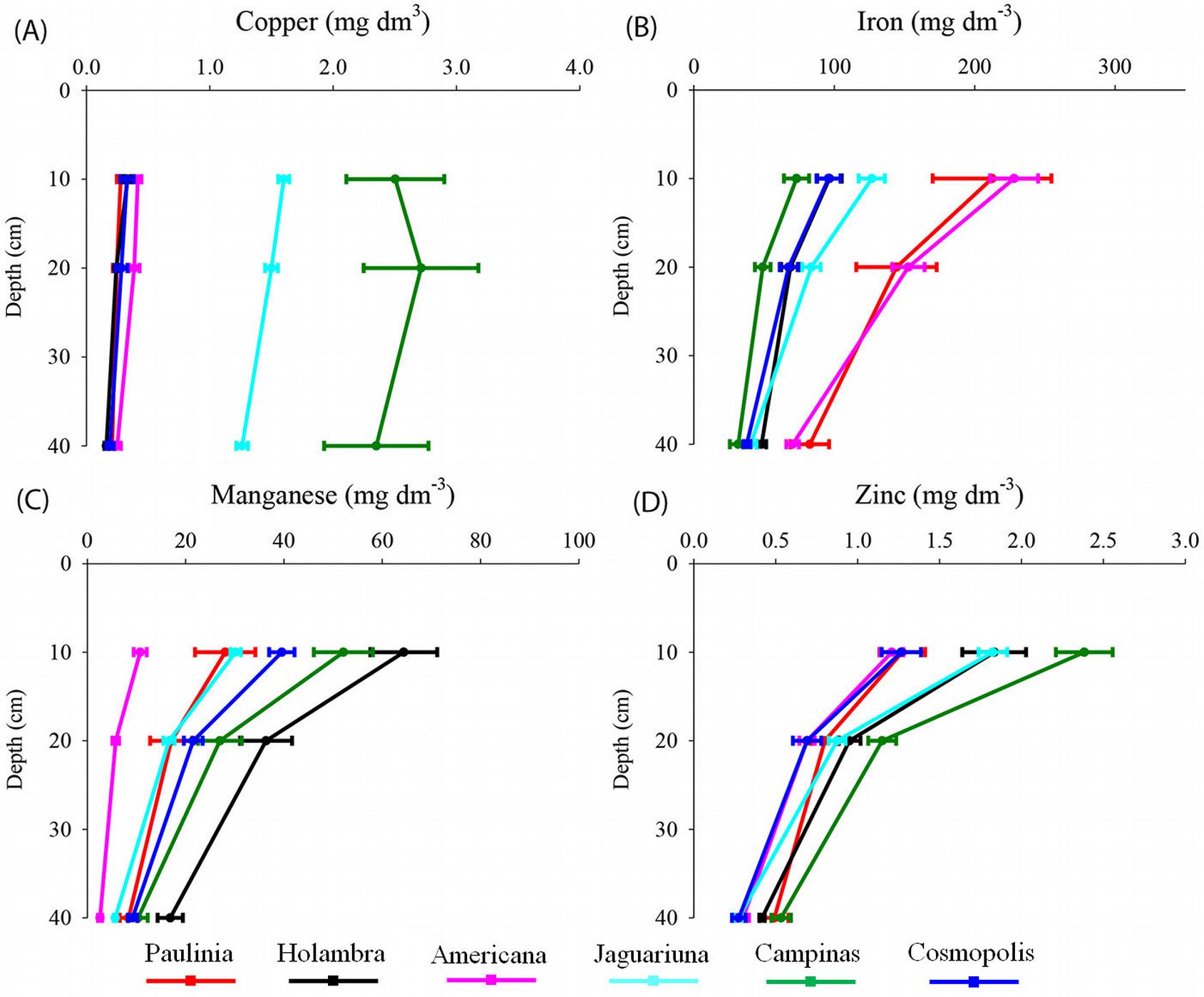
The variation with depth of soil chemical parameters are presented in Fig. 2 to 5. All elements decreased in concentration from the surface to the deeper soil layers, with the exceptions of S and Al.
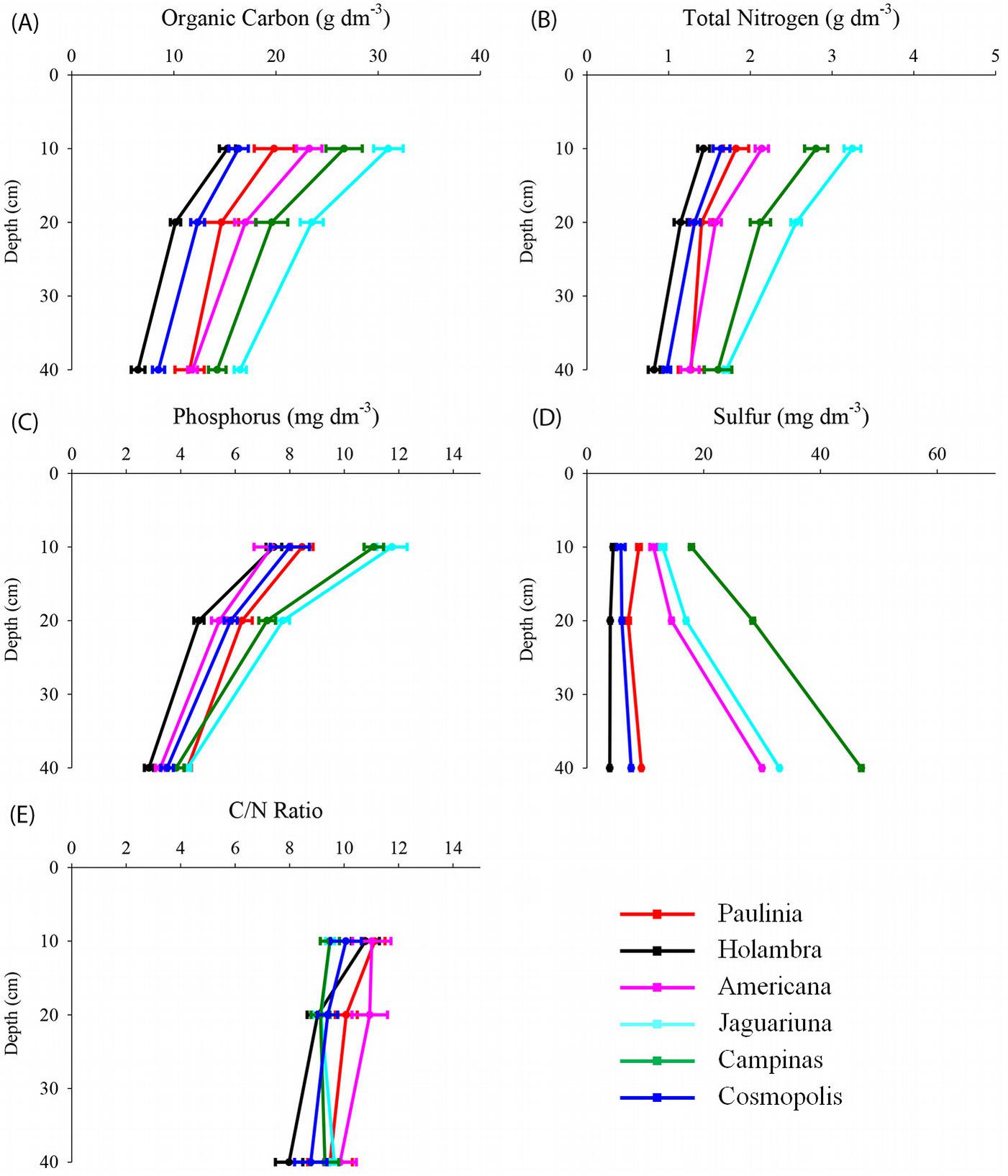
Fig. 2 - Soil organic carbon (A), total nitrogen (B), extractable phosphorus (C) and sulfur (D), and C/N ratio (E) at 0-10 cm, 10-20 cm and 20-40 cm depths of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil (mean ± standard error, n = 12).
The largest amounts of Corg, Ntot, and extractable P and S were found in soils from Jaguariúna and Campinas (p<0.05), whereas the lowest levels were found in Holambra and Cosmópolis (Fig. 2). The Corg contents varied from medium (< 15 g dm-3) to high (20 to 31 g dm-3) and were directly related to the high levels of organic matter. The Corg in soils was correlated positively with Ntot, CEC, P and Cu. The Ntot content was correlated with the same parameters, as well as with S content (Tab. 3).
The extractable P and S levels were similar in the soils of sampling sites in Paulínia, Holambra, Americana and Cosmópolis and were significantly higher in Jaguariúna and Campinas (p<0.05 - Fig. 2). The P content values were significantly and positively correlated with CEC and Cu concentrations (Tab. 3).
The C/N ratio (< 11/1) was low and similar in all sampling sites. Primarily in Jaguariúna and Campinas soils, such ratio indicated an optimal decomposition of the soil organic matter (Fig. 2).
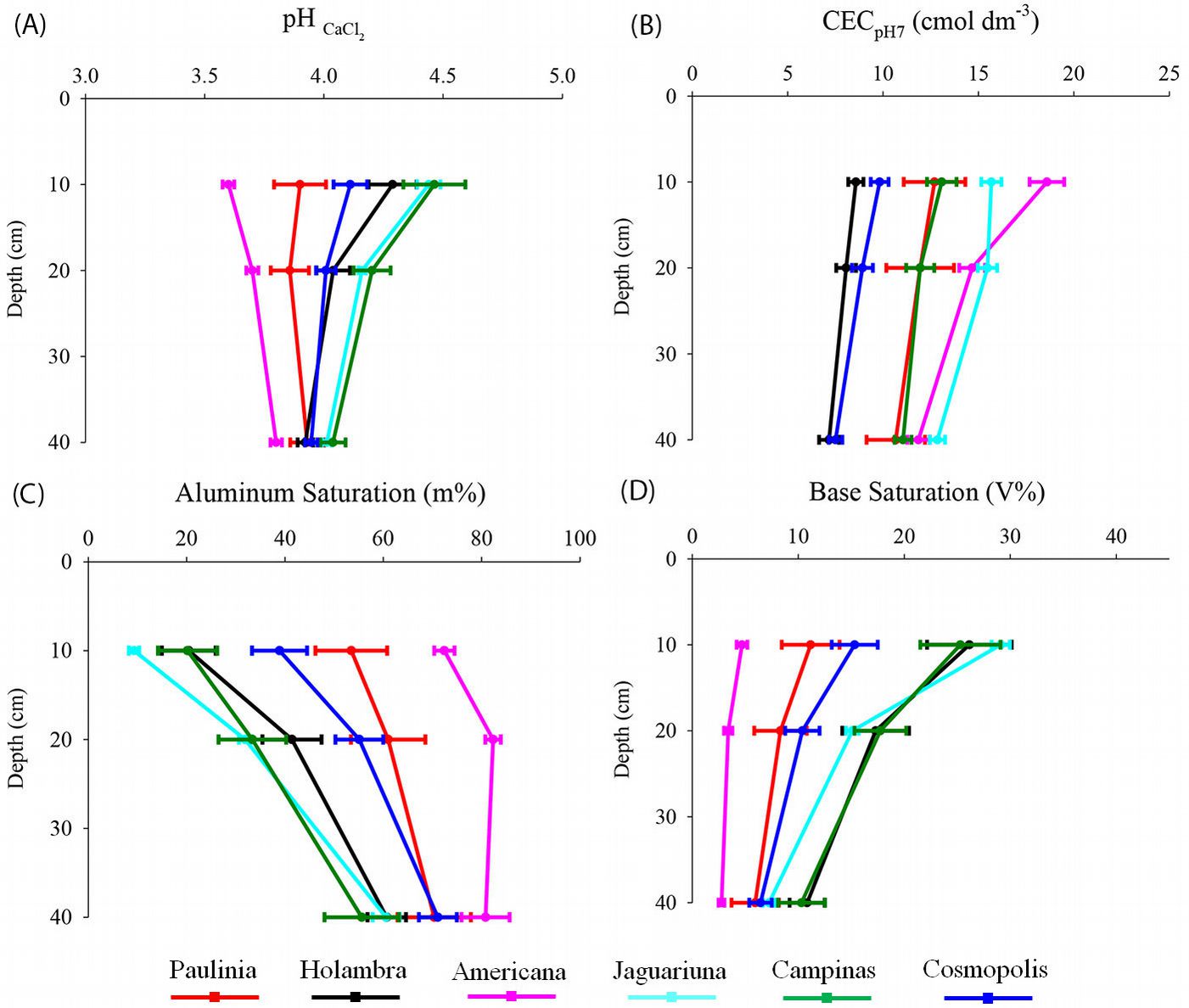
The soils were extremely acidic in all the forest remnants analyzed (pH < 4.5 - Fig. 3). However, pH values of soil samples from Americana and Paulínia (3.6-3.9) were lower than those from Jaguariúna and Campinas (4.0-4.5).
Fig. 3 - Soil pHCaCl2 (A), cation exchange capacity (CECpH7 - B), aluminum saturation (m% - C) and base saturation (V% - D) at 0-10 cm, 10-20 cm and 20-40 cm depths of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil (mean ± standard error, n = 12).
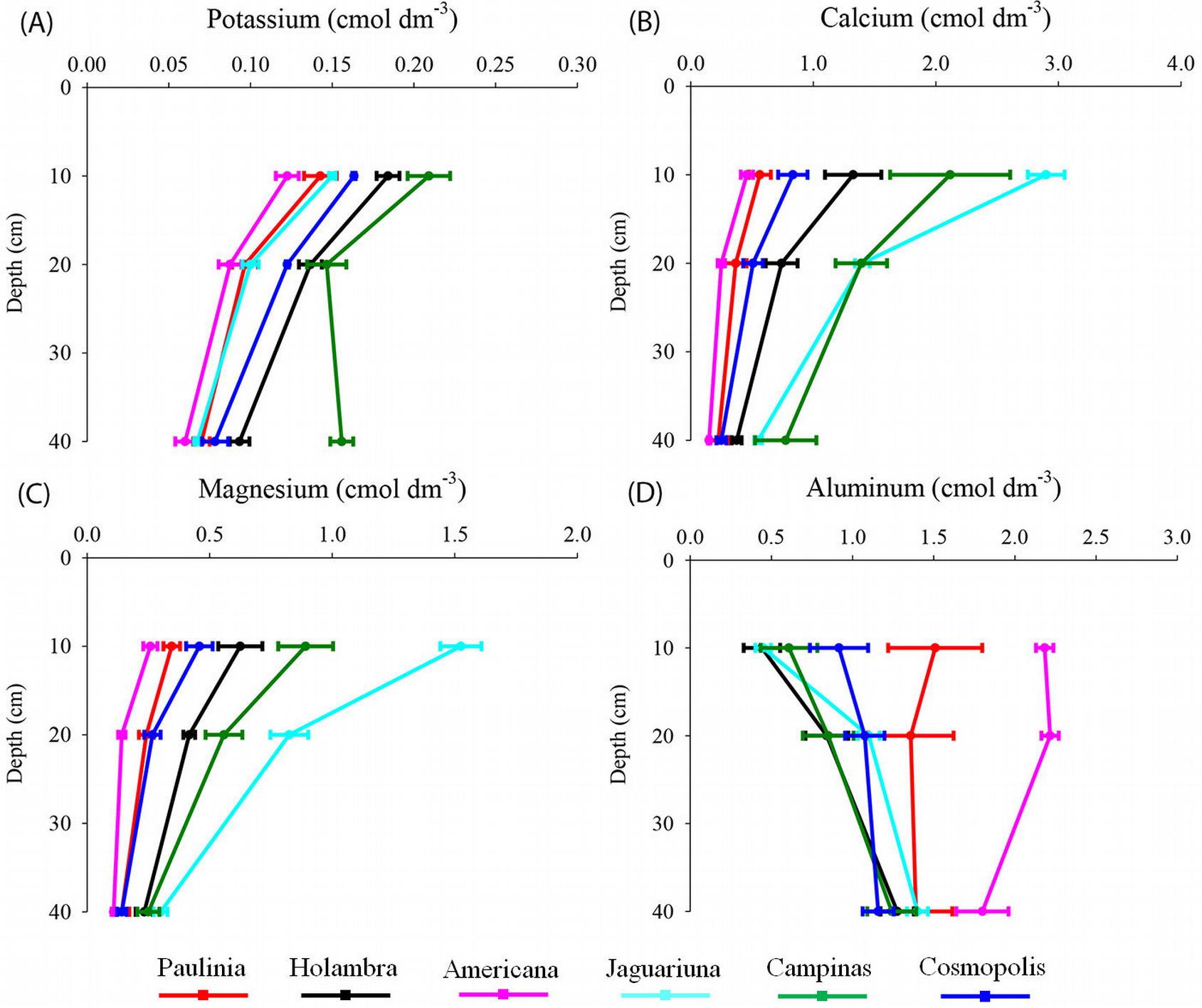
Soil samples from Jaguariúna and Campinas sites had low Al concentrations (0.5-0.6 cmol dm-3), high base saturation (28-29%), and high contents of exchangeable Ca (2.6-2.9 cmol dm-3) and Mg (0.9-1.5 cmol dm-3), available P (0.15-0.21 cmol dm-3), Corg (27-31 g dm-3) and Ntot (2.8-3.2 g dm-3) in the surface layer (Fig. 3, Fig. 4). By contrast, high Al content and low basic cations (Ca, Mg and K) were found in the Americana soil (Fig. 3, Fig. 4). Correlation analysis revealed significant negative associations between CEC and V%, V% and m% or Fe, and m% and Mn or Zn. Positive associations were found between CEC and m%, Fe or Mn, and V% and Mn or Zn (Tab. 3).
Fig. 4 - Soil exchangeable potassium (A), calcium (B), magnesium (C) and aluminum (D) at 0-10 cm, 10-20 cm and 20-40 cm depths of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil (mean ± standard error, n = 12).
The studied forest sites had different concentrations of available heavy metals (Fig. 5). The levels of Cu (5.0 mg dm-3) and Zn (2.4 mg dm-3) were significantly higher in the Campinas soil. The levels of Fe in the Americana (228 mg dm-3) and Paulínia (208 mg dm-3) soils were significantly higher. Higher levels of Mn were detected in the soil from the Holambra forest (64.4 mg dm-3). The available levels of Zn and Mn showed a significant positive correlation with each other. Levels of Mn were negatively correlated with Fe contents (Tab. 3).
Fig. 5 - Soil extractable copper (A), iron (B), manganese (C) and zinc (D) at 0-10 cm, 10-20 cm and 20-40 cm depths of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil (mean ± standard error, n = 12).
Tab. 4 summarizes the results of the correlation analysis between pH values and soil characteristics for each forest fragment studied. The soil pH in the Paulínia forest was negatively correlated with Ntot, Al, CEC, m%, Cu, Fe and sand and was positively correlated with Mn, silt and clay. In the Holambra site, the pH was negatively correlated with Al and m% and was positively correlated with K, Ca, Mg, V%, Mn and Zn. The pH values found in the forest soil from Americana were negatively associated with Corg, Ntot, C/N ratio, P, K, CEC, Cu, Fe, Mn, Zn and sand and were positively associated only with S content. In soil samples from Jaguariúna and Campinas, positive relationships were found between pH and Corg, Ntot, P, K, Ca, Mg, V%, Cu, Mn and Zn, and negative relationships were observed between pH and S, Al, m% and clay. In soils from the Cosmópolis site, K, Ca, Mg, V% and Mn values were positively correlated with soil pH, whereas and Al and m% were negatively correlated with soil pH.
Tab. 4 - Spearman’s correlation coefficients between pH and physical and chemical parameters in the 0-40 cm soil layer in semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil. (PA): Paulína; (HO): Holambra; (AM): Americana; (JA): Jaguariúna; (CA): Campinas; (CO): Cosmópolis; (CEC): cation exchange capacity = Ca+Mg+K+Al+H; (V%): base saturation = [(Ca+Mg+K)/CEC]·100; (m%): Aluminum saturation = (Al/Ca+Mg+K+Al)·100. (*): p<0.05; (**): p<0.01; (***): p<0.001.
| Parameters | PA | HO | AM | JA | CA | CO |
|---|---|---|---|---|---|---|
| Corg | -0.73*** | 0.37 | -0.76*** | 0.88*** | 0.81*** | 0.28 |
| Ntot | -0.69*** | 0.21 | -0.43 | 0.85*** | 0.76*** | 0.39 |
| C/N | -0.45 | 0.41 | -0.57*** | 0.04 | 0.18 | -0.07 |
| P | -0.46 | 0.49 | -0.56*** | 0.89*** | 0.65*** | 0.33 |
| S | -0.27 | 0.36 | 0.73*** | -0.76** | 0.12 | 0.06 |
| K | -0.01 | 0.56*** | -0.58*** | 0.92*** | 0.60*** | 0.52** |
| Ca | 0.45 | 0.87*** | -0.46 | 0.85*** | 0.74*** | 0.56*** |
| Mg | 0.40 | 0.84*** | -0.51** | 0.95*** | 0.87*** | 0.53** |
| Al | -0.79*** | -0.86*** | -0.20 | -0.94*** | -0.94*** | -0.69*** |
| CEC | -0.88*** | -0.25 | -0.84*** | 0.42 | 0.35 | -0.12 |
| V% | 0.73*** | 0.92*** | -0.12 | 0.89*** | 0.84*** | 0.60*** |
| m% | -0.63*** | -0.88*** | 0.36 | -0.95*** | -0.91*** | -0.63*** |
| Cu | -0.50*** | 0.15 | -0.76*** | 0.80*** | 0.70*** | 0.09 |
| Fe | -0.80*** | 0.11 | -0.81*** | 0.79*** | -0.23 | 0.03 |
| Mn | 0.52** | 0.77*** | -0.61*** | 0.94*** | 0.80*** | 0.51** |
| Zn | 0.11 | 0.65*** | -0.70*** | 0.85*** | 0.66*** | 0.39 |
| Sand | -0.69** | 0.42 | -0.19 | 0.56 | 0.27 | 0.11 |
| Silt | 0.66** | -0.31 | 0.18 | 0.33 | 0.06 | 0.06 |
| Clay | 0.72*** | -0.33 | 0.13 | -0.69* | -0.25 | -0.13 |
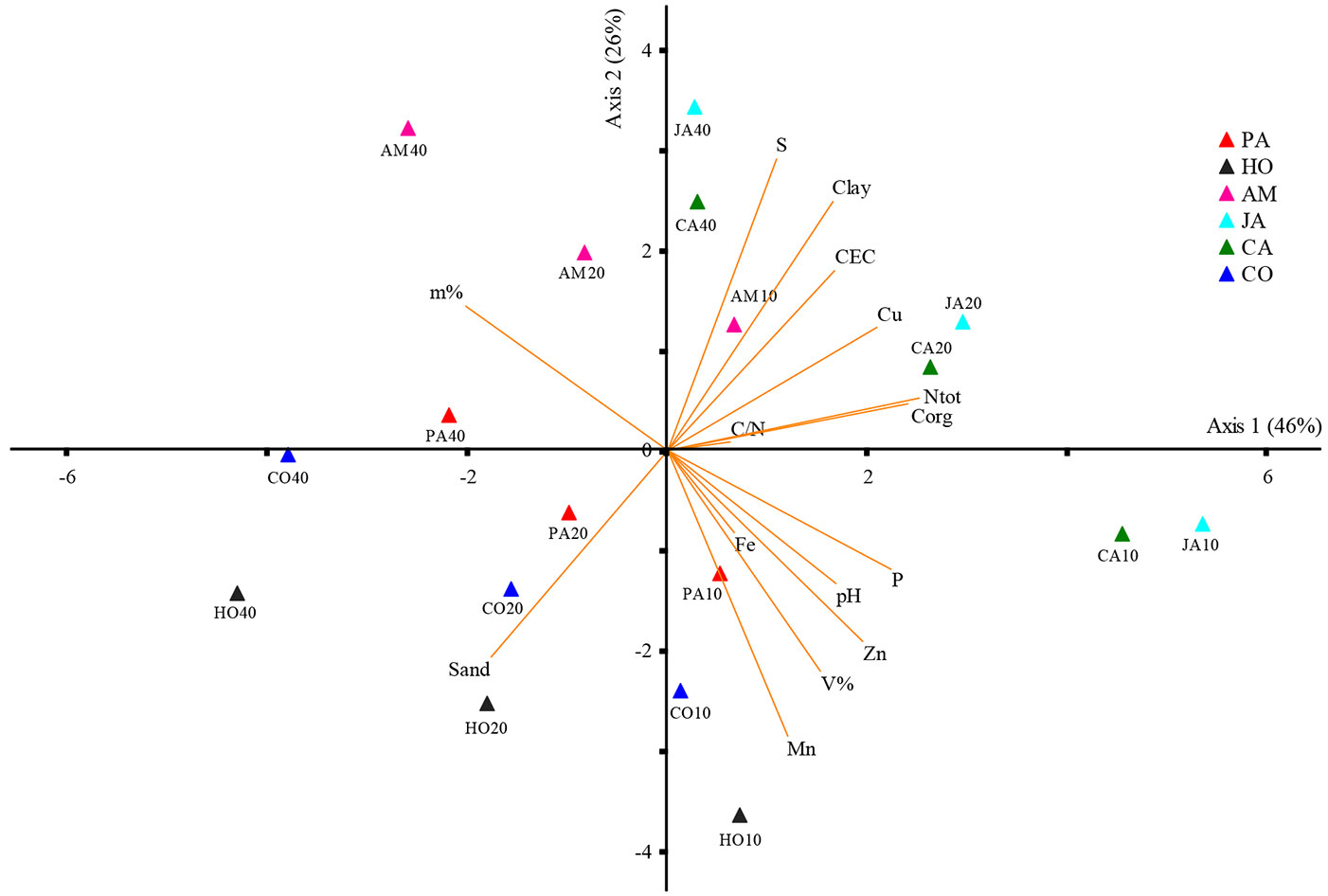
Fig. 6 and Fig. 7 show the differences in the soil physical and chemical parameters of the forest sites analyzed. The first two principal components accounted for 72% of the overall variation in the dataset (Fig. 6). The variables clay, CEC, Corg, Ntot, S, and Cu from Jaguariúna and Campinas were grouped on the positive side of the first PC axis. The second PCA group was made up by pH, V%, P, Zn and Mn (negative side of PC axis 2 - Fig. 6) for all sites in the 0-10 cm layer, except Americana. For deeper soil layers, the sand fractions from Cosmópolis and Holambra were separated on the negative side of PC axis 1 and 2. The variable m% was isolated on the positive side of PC axis 2 only for the Americana forest.
Fig. 6 - Principal component analysis (PCA) of soil characteristics from 0-10 cm (10), 10-20 cm (20) and 20-40 cm (40) depths of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil. (PA): Paulínia; (HO): Holambra; (AM): Americana; (JA): Jaguariúna; (CA): Campinas; (CO): Cosmópolis; (CEC): cation exchange capacity; (V%): base saturation; (m%): aluminum saturation.
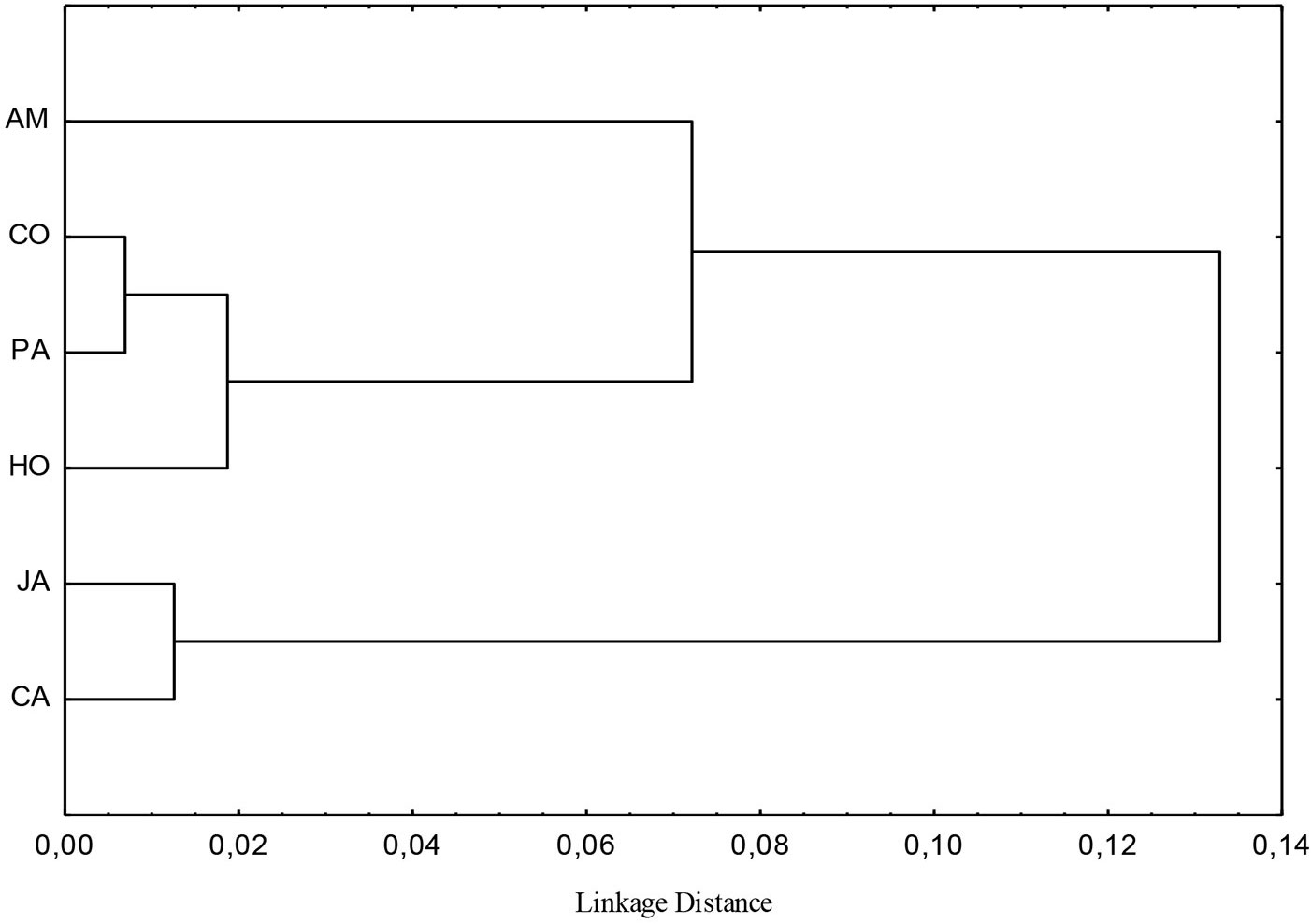
Fig. 7 - Cluster analysis on data from 0-40 cm soil layer of semideciduous Atlantic Forest remnants in the Metropolitan Campinas Region (MCR), Brazil. (PA): Paulínia; (HO): Holambra; (AM): Americana; (JA): Jaguariúna; (CA): Campinas; (CO): Cosmópolis.
The cluster analysis distinguished three groups at a 0.06 linkage distance (Fig. 7). The soil from the Americana forest site was included in the first group, whereas the soils of the Cosmópolis, Paulínia and Holambra forest remnants were included in the second group. Finally, the third group was formed by the soils from Jaguariúna and Campinas.
Discussion
Higher concentrations of nutrients in the 0-10 cm soil layer compared to those at deeper layers were observed in all the forest remnants analyzed, indicating that the soil surface likely had the conditions needed to buffer the atmospheric deposition. Additionally, organic matter content was higher in the surface layer, as commonly observed in tropical soils ([50], [63]). A substantial litter layer on the soils (Tab. 1) also explained the higher available nutrients on the soil surface.
Soil samples from sites showing a clay texture (Campinas and Jaguariúna) contained higher levels of basic cations than those with a more sandy texture (Americana, Cosmópolis, Paulínia and Holambra) where leaching was increased. Soil physical parameters have important influences on hydrological processes and plant growth ([36]). The sand and clay fractions primarily differ in the number of ion exchange sites ([38]). Therefore, the positive correlations between clay content and carbon, total nitrogen, sulfur and copper were likely due to the adsorptive properties of the fine soil particles to organic matter, as discussed by Feller & Beare ([15]).
The Corg, Ntot and the C/N ratio provide information about the chemical status of soil ([24], [35], [70], [8]). These parameters are particularly important in tropical soils, where the availability of nutrients is low and the weathering rates are high ([15]). Many nutrients are released to the soil during the mineralization of organic matter, including Ca, Mg, P, K, S and micronutrients ([55], [57]). Therefore, the high Corg and basic cation (Ca, Mg and K) contents found in the forest soils from Campinas and Jaguariúna, along with the low C/N ratio, indicated that soil mineralization and litter turnover were more rapid, which provided optimal conditions for plant growth.
Positive correlations were found between the high concentrations of Ntot, measured primarily in soil samples from Jaguariúna and Campinas (Fig. 2), and the levels of Corg, available P and S, CEC and the clay fraction (Tab. 3). These correlations suggest a direct impact on soil chemical properties of organic matter ([24]) and inputs of atmospheric nitrogen from vehicular emissions and fertilization of surrounding agricultural areas. Also, these activities likely contributed to the high levels of S in the soils of Campinas and Jaguariúna, whereas the industrial complex probably contributed to sulfur deposition in Americana and Paulínia. Indeed, in the Cosmópolis and Holambra forests which are more distant from the industrial complex, the S levels were lower, indicating that sulfur deposition decreases with increasing the distance from the emission source. In contrast to the other elements, S increased with depth, primarily in Campinas soils (17.9 g.dm-3 on the surface and 47.0 g.dm-3 in subsoil), indicating its accumulation at deeper soil layers.
Abiotic conditions in combination with atmospheric inputs results in soil acidification, which affects the development of the forest ([22], [14], [25], [2]). In the present work, the soils were extremely acidic (pH 3.6-4.5) in all forest fragments, caused by high aluminum saturation and low basic cations, as typically observed in different tropical forests ([62], [19], [68]). However, the forest soils near the industrial complex (Paulínia and Americana) were more acidic (pH = 3.6-3.9) than those located farther apart from industries near rural or urban sources (pH = 4.0-4.5), indicating that acidic deposition increases near the industrial area.
The pH and base saturation are important indicators of chemical processes in the soil and thus are key descriptors for monitoring forest changes ([7]). The low V% might have been related to the leaching of basic cations (K+, Ca2+ and Mg2+), which had a direct influence on soil acidity and increase of m% ([27], [4], [49], [32], [30], [57]), as well as on the availability of heavy metals ([67], [69], [62]). Furthermore, basic cations and organic matter buffer the soil acidity ([55], [30]). Hence, the mobilization of Al and Fe at low pH values (4.2 and 3.8, respectively), as suggested by [30], could explain the high levels of these elements in the Americana and Paulínia soils (Fig. 4 and Fig. 5).
The parent material, weathering processes and pollution inputs affect the content of heavy metals in the soil ([39], [69], [61], [6]). Soils derived from basic rocks naturally have more heavy metals (including Cu, Ni and Zn) than those derived from sandstone, siltstone or gneiss ([69], [46]). In the Campinas region, Miranda & Tomaz ([44]) reported that the soil was the main source of aluminum, while for zinc, copper and sulphur the main sources were industries and vehicle emissions. Monaci et al. ([45]) found high concentrations of aluminum, iron, copper, manganese and zinc in the particulate material originated from vehicular emissions. Therefore, the soil parent material (diabase) and the urban air pollution could both explain the high concentrations of Cu and Zn in the Campinas and Jaguariúna soils.
The PCA highlighted the direct relationship among the CEC, the Corg and the clay fraction. The variables were clearly separated under the criteria of soil acidity, nutrient availability and m%. The sand fraction in the Cosmópolis and Holambra soils was directly related to the increase in nutrient leaching to deeper soil layers, whist it was related to the Al toxicity and high acidity in the Americana site. This separation was confirmed by cluster analysis that grouped the Jaguariúna and Campinas forests by the characteristics of more optimal chemical conditions and texture (primarily because of the proportion of clay). Such conditions contrast with the Americana forest soil, which was more acidic, characterized by higher levels of Al and lower exchangeable base cations, and sandy-clay texture. Soils from the Cosmópolis, Paulínia and Holambra forests were grouped because of their similar chemical and physical conditions.
Conclusions
Remnants of the semideciduous Atlantic Forest in the MCR are growing on soils with distinct chemical and physical conditions due to the parent material and the deposition of air pollution. The optimal chemical conditions (high contents of exchangeable bases, nitrogen, phosphorus and sulfur) were found in the clay soils of Jaguariúna and Campinas, which are more affected by rural or urban pollution and less impacted by the industrial complex. These soils are expected to show the best buffering capacity against environmental pollution. However, air pollution deposition was related to the acidification of sandy soils in forest fragments more influenced by the industrial complex. Therefore, the Atlantic Forest vegetation in Paulínia, Cosmópolis, Holambra and Americana might be more susceptible to air pollution than the forest remnants located in Campinas and Jaguariúna.
Acknowledgments
The authors wish to thank Programa SISBIOTA-BRASIL (proc. CNPQ 563335/2010 and proc. FAPESP 2010/52319-2) for financial support, Área de Relevante Interesse Ecológico Mata da Santa Genebra and Cooperativa Agropecuária Holambra for providing the area for soil sampling, Coordenação Especial para Restauração de Áreas Degradadas-CERAD, Mr. Paulo R. T. Ortiz for drawing Fig. 1, and Mrs. Cristiane Aguiar Silva and Mr. Francisco Ricardo da Silva for the help in field sampling.
References
CrossRef | Gscholar
CrossRef | Gscholar
CrossRef | Gscholar
Online | Gscholar
Gscholar
CrossRef | Gscholar
Gscholar
Gscholar
Gscholar
Online | Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Andressa Ribeiro Dos Santos
Carla Zuliani Sandrin Camargo
Patricia Bulbovas
Patrícia Giampaoli
Marisa Domingos
Instituto de Botanica, Caixa Postal 68041, 04045-972 São Paulo, SP (Brazil)
Corresponding author
Paper Info
Citation
Lopes MIMS, Ribeiro Dos Santos A, Zuliani Sandrin Camargo C, Bulbovas P, Giampaoli P, Domingos M (2015). Soil chemical and physical status in semideciduous Atlantic Forest fragments affected by atmospheric deposition in central-eastern São Paulo State, Brazil. iForest 8: 798-808. - doi: 10.3832/ifor1258-007
Academic Editor
Giustino Tonon
Paper history
Received: Jan 30, 2014
Accepted: Dec 13, 2014
First online: Apr 22, 2015
Publication Date: Dec 01, 2015
Publication Time: 4.33 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2015
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 54298
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 45383
Abstract Page Views: 3266
PDF Downloads: 4164
Citation/Reference Downloads: 22
XML Downloads: 1463
Web Metrics
Days since publication: 3893
Overall contacts: 54298
Avg. contacts per week: 97.63
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
Total number of cites (since 2015): 12
Average cites per year: 1.09
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Availability and evaluation of European forest soil monitoring data in the study on the effects of air pollution on forests
vol. 4, pp. 205-211 (online: 03 November 2011)
Research Articles
Bioaccumulation of long-term atmospheric heavy metal pollution within the Carpathian arch: monumental trees and their leaves memoir
vol. 17, pp. 370-377 (online: 27 November 2024)
Research Articles
Soil stoichiometry modulates effects of shrub encroachment on soil carbon concentration and stock in a subalpine grassland
vol. 13, pp. 65-72 (online: 07 February 2020)
Research Articles
Changes in the properties of grassland soils as a result of afforestation
vol. 11, pp. 600-608 (online: 25 September 2018)
Short Communications
Linking critical thresholds for acidity to forest condition by using element contents in tree rings: consequences for the development of an integrated ecosystem assessment
vol. 2, pp. 51-53 (online: 21 January 2009)
Research Articles
Wood-soil interactions in soil bioengineering slope stabilization works
vol. 2, pp. 187-191 (online: 15 October 2009)
Research Articles
Soil fauna communities and microbial activities response to litter and soil properties under degraded and restored forests of Hyrcania
vol. 14, pp. 490-498 (online: 11 November 2021)
Research Articles
Impact of deforestation on the soil physical and chemical attributes, and humic fraction of organic matter in dry environments in Brazil
vol. 15, pp. 465-475 (online: 18 November 2022)
Research Articles
Effect of size and surrounding forest vegetation on chemical properties of soil in forest gaps
vol. 8, pp. 67-72 (online: 04 June 2014)
Research Articles
Distribution of aluminium fractions in acid forest soils: influence of vegetation changes
vol. 11, pp. 721-727 (online: 06 November 2018)
iForest Database Search
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords