Spread intensity and invasiveness of sycamore maple (Acer pseudoplatanus L.) in Lithuanian forests
iForest - Biogeosciences and Forestry, Volume 8, Issue 5, Pages 693-699 (2015)
doi: https://doi.org/10.3832/ifor0763-007
Published: Mar 19, 2015 - Copyright © 2015 SISEF
Research Articles
Abstract
The primary objectives of this study were to estimate seedling abundance, spread intensity, and invasiveness of sycamore maple (Acer pseudoplatanus L.) in Lithuanian forests. The species was introduced to Lithuania in 1802, and since then has subsequently become gradually invasive. Seedling understory abundance, colonization and dispersal were investigated in six forest blocks covering the principal sycamore distribution areas in southwestern Lithuania. Seedlings height and densities in the undestory were evaluated, and seedlings assigned to four height groups. Species invasiveness was estimated applying the Pest Plant Prioritization Process (PPPP), based on the Analytic Hierarchy Process (AHP) method. Results showed the average spread distance from the parent tree was 257 m, with a mean seedling number per hectare of 2064. Sycamore maple invasive score was 0.6426 (range: 0-1), the current relative to potential distribution rating was 0.57, and the social, environmental, and economic impact score was 0.1682. Such values were used to assess the Pest Plant Score for sycamore maple, obtaining a value (0.3537) lower than expectations (0.5). Results indicated that the species exhibits invasive properties and a rapid spread in the study area. Some implications of the above results in view of the upcoming climate change and the use of sycamore maple in Lithuanian forest plantations are discussed.
Keywords
Sycamore, Seedling spread, PPPP, Invasiveness, Pest Plant Score
Introduction
During the early 19th century, the practice of planting non-native tree species in Lithuania was initiated with the primary goal of increasing wood productivity. It was unknown at the time certain species would successfully undergo naturalization, and subsequently exhibit a widespread geographic distribution, threatening natural ecosystems. Vitousek & Walker ([36]) characterized the alterations of the community stability and structure due to non-native tree species, which successfully proliferated and became invasive community members, limiting native plant growth ([21]). Boxelder (Acer negundo L.), black locust (Robinia pseudoacacia L.), and black cherry (Prunus serotina Ehrh.) are included in the list of invasive species in Lithuania, and additional alien species are currently being considered for invasive status. One of the more problematic non-native species in Lithuania is the sycamore maple (Acer pseudoplatanus L., Sapindaceae), characterized by rapid growth (when grown on suitable sites) and potentially high timber prices ([13]). Sycamore maple is mostly a central-European, mountain species reaching its northeastern range limit in Poland. Sycamore is rarely found in pure stands covering small areas on steep slopes on limestones where it forms its own communities (Acerion pseudoplatani alliance - [17]). More frequently it grows in mixed broad-leaved forests of the Carpinion and Alno-Padion alliances ([4]). In Lithuania, the species grows in fertile moist sites and in secondary forests. Moreover, it is shade and frost tolerant ([23]), and in naturally regenerated stands often grows mixed with ash ([3]). Typically, the species begins bearing fertile seeds at approximately 20 to 30 years of age ([5], [23]).
Sycamore maple is well adapted to the current climatic conditions of central Europe, and is expected to expand beyond its current distributional limits ([19], [20]). This species has become naturalized in many countries, including Spain, Turkey, Ireland, Sweden, North and South America, India, and New Zealand ([3], [28]). In Lithuania, sycamore maple may reach up to 24 m of height and 1 m of stem diameter ([15]), and is mostly found in parks and gardens, with only few forest plantations.
The question we pose is whether sycamore maple may be considered an invasive non-native tree in Lithuania. Williamson & Fitter ([39]) estimated that only approximately 10% of naturalized plant species become invasive and produce significant economic and ecological impacts. Historical records indicated sycamore maple was introduced to the Vilnius University Botanical Garden in 1802 ([30]), and since then it readily spreads into local forest stands. Informed decisions must be made to control invasive trees in forests, which requires establishing the relative importance of each alien tree.
The principal objectives of this study were to estimate sycamore maple seedling abundance, spread intensity, and invasiveness in Lithuanian forests. Invasiveness was determined by evaluating the following questions: (i) how invasive is the species, i.e., how rapidly can it spread? (ii) what is the present and potential extent of the species? (iii) what are the social, environmental, and economic impacts of the species in Lithuania?
Material and methods
Description of the study area
The Lithuanian climate is transitional and varies from maritime in the west to continental in the east ([12]). The mean annual temperature is approximately 6 ºC, and mean annual precipitation varies from 620 to 700 mm along an east-west gradient. Lithuania’s terrain is an alternation of moderate lowlands and highlands, with a maximum elevation of nearly 300 m. Alisov ([1]) classifies Lithuania in the southwest sub-area climate of the South Baltic. In general, the country lies in boreal and broadleaved forest belts, and forests cover nearly 33% of the territory. The natural geographic range of hornbeam (Carpinus betulus L.) marks the border between the predominantly broadleaved zone to the south and the coniferous zone to the north. Conifers constitute about 56.1% of the total forest area (source: State Forest Service). The main conifer species are Scots pine (about 35.3%) and Norway spruce (about 20.8%). Broadleaf trees dominate the central part of the country: birch constitutes about 22.2% of the total, followed by black alder (about 6.8%) and European aspen (about 3.8%). Other hardwoods make up 4.7% of the total forest area and consist of pedunculate oak (2.0%), common ash (1.8%) and other species (elms, hornbeam, etc.).
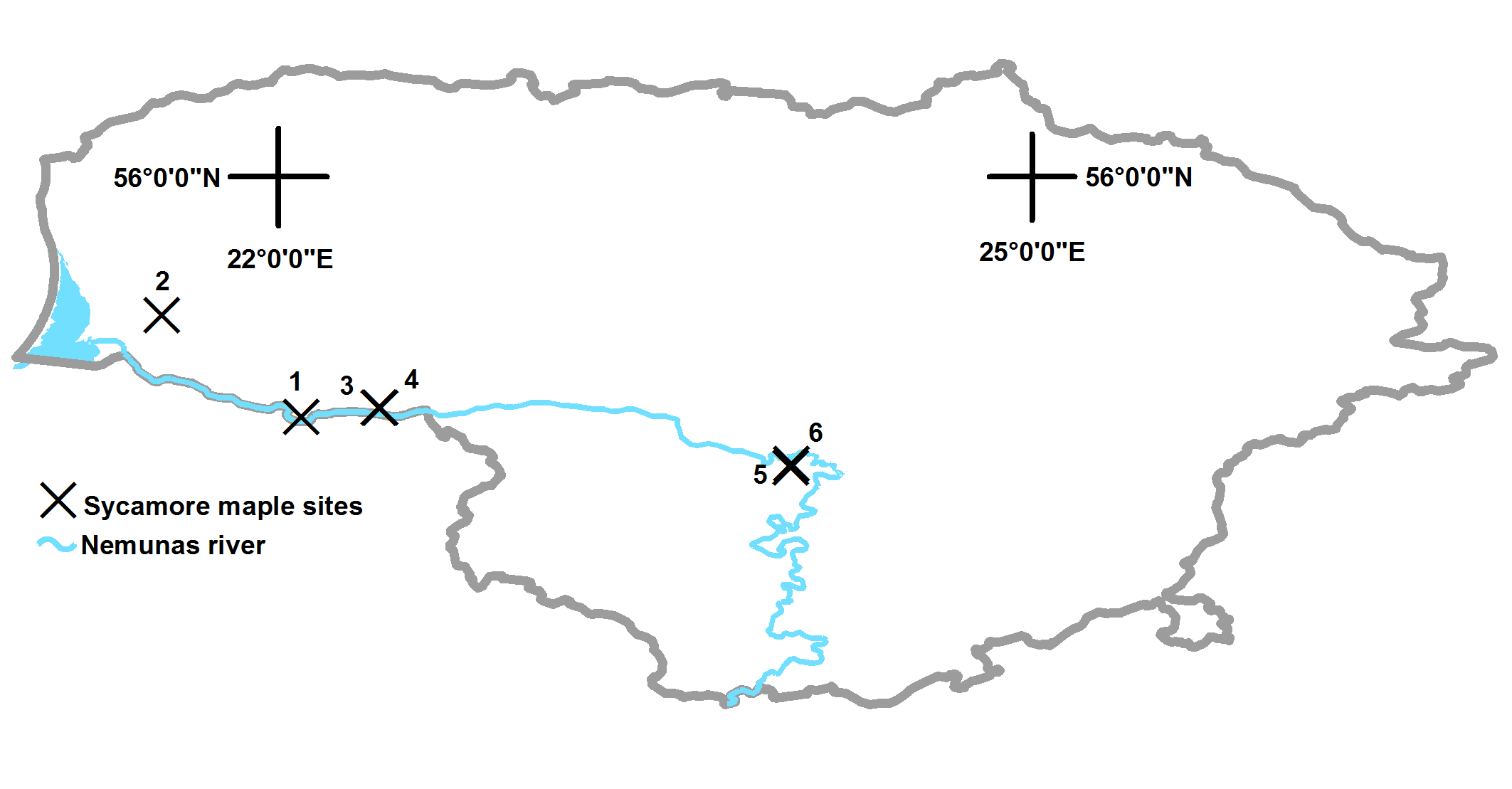
This study was conducted in six forest blocks, in south-western Lithuania (Tab. 1), differing in dominant tree species area and prevailing site type. The study area includes 40-90 year old stands. Soil fertility was obtained from the forest inventory database. The dominant tree species were Pinus sylvestris L., Acer pseudoplatanus and Fagus sylvatica L. The shrub layer was dominated by Acer pseudoplatanus, Sorbus aucuparia L. and Corylus avellana L. Acer pseudoplatanus and Fagus sylvatica are non-native to Lithuania.
Tab. 1 - Descriptive data for the sycamore maple forest blocks used in this study. (*) Site Index: humidity (N, normal; L, temporarily wet); fertility (b, poor; c, fertile); soil texture (l, light soil; p, two story soil). (SD): standard deviation.
| No | Forest enterprise, forest district |
Region | Climate type |
Latitude Longitude |
Altitude a.s.l. (m) |
Site index* |
Stand age |
No. seedlings ha-1 | Seedling spread distance (m) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | |||||||||
| 1 | Silutes, Rambyno |
West southern |
maritime | 55° 02′ 44″ 22° 05′ 35″ |
63 | Ncl | 60 | 2465 | 373 | 51 |
| 2 | Silutes, Norkaiciu |
West central |
maritime | 55° 26′ 59″ 21° 32′ 17″ |
2 | Ncl | 90 | 3446 | 291 | 59 |
| 3 | Jurbarko, Viesviles |
West southern |
maritime | 55° 04′ 52″ 22° 24′ 09″ |
24 | Ncl | 80 | 1354 | 210 | 119 |
| 4 | Jurbarko, Viesviles |
West southern |
maritime | 55° 04′ 58″ 22° 24′ 25″ |
23 | Ncl | 60 | 492 | 134 | 71 |
| 5 | Dubravos, Vaisvydavos |
Central | transitional | 54° 50′ 38″ 24° 02′ 33″ |
79 | Nbl | 60 | 2528 | 209 | 34 |
| 6 | Dubravos, Vaisvydavos |
Central | transitional | 54° 51′ 26″ 24° 02′ 40″ |
68 | Ncp | 40 | 2098 | 130 | 13 |
Seedling abundance and spread intensity
The density of sycamore maple seedlings in the understorey was evaluated in each block per unit area and then reported to hectare. Seedlings were grouped into four height groups: (1) < 0.5 m; (2) 0.5-1.5 m; (3) 1.5-3 m; (4) > 3 m. No replicates per forest block were made as the data collection was performed in a comprehensive way (see below) to forest stand in areas larger than half a hectare at each site. The spread of sycamore maple was assessed by collecting the following information:
- latitude, longitude and altitude of the most likely parent tree or group of parental (mature) trees; in almost all cases mature trees were scattered over large areas and at considerable distance each other, so seedlings dispersal was considered from the closest mature tree;
- the area covered by sycamore maple in the understory from the parent tree in four directions (N, E, S, and W): the number of seedlings along 5m-wide transects was recorded, and the average spread distance calculated based on mean values for all directions. The length of transects varied based on the seedling spread distance.
- the species composition in the canopy layer and the dominant tree species of the sampled stand;
- the relative density of the upper canopy layer on a scale 0 to 1.0, where 1.0 indicates the highest density, following the procedure adopted by the Lithuanian National Forest Inventory (2003-2007);
- the underbrush cover density was recorded as the coverage of the crown projection on the 5-m transect area (0-100%).
Statistical analysis was carried out using SAS software (SAS Institute, Inc., SAS/ STAT software Release 9.3). SAS PROC CORR was used to calculate Pearson’s pairwise correlation coefficients between seedling abundance and stand characteristics. The exact Kolmogorov-Smirnov two-sample two-sided test (SAS PROC NPAR1WAY) was performed for testing differences in seedling height classes and densities among the six sites. The same test was used in the comparison of different seedling groups by site fertility and underbrush cover. Mantel test with 10 000 permutations was carried out on a Euclidean distance matrix for similarity measures using densities of seedling height classes against a matrix of pairwise geographical distances between sites.
Invasiveness assessment
The Pest Plant Prioritisation Process (PPPP) was used to determine the invasiveness degree ([29], [38]). PPPP is a prioritization process or risk assessment based on the Analytic Hierarchy Process (AHP), which ranks weeds (or invasive plant species) by: (1) assessing plant invasiveness; (2) comparing the species’ present and potential distributions; and (3) determine the impacts of the plant on social, economic, and environmental values. The method described by Panetta & Dodd ([24]) and developed by the Biosciences Research Division of the Department of Primary Industries, Victoria, Australia ([2]) was followed in this study. Weightings of each criteria were set using an expert system relying on multi-criteria analysis ([2]). An intensity rating (score) was assigned to each species considered for each criteria listed in Tab. 2 based on the following scale: high (1), medium-high (0.75), medium (0.5), medium-low (0.25), and low (0).
Tab. 2 - Groups and criteria weightings for determining the invasive degree.
| Group | Criteria | Notes | Criteria weightings |
Rating | Impact |
|---|---|---|---|---|---|
| Establishment | Germination requirements | Seeds germinate in spring; seeds show dormancy ([25]); germination regimes: 20ºC, continuous light, 20 d ([26]) | 0.0425 | 0.75 | 0.032 |
| Establishment requirements | Occurs under moderate canopy forest, prefers moist soil | 0.3355 | 0.75 | 0.25 | |
| Disturbance requirements | Establishes in open woodlands, forest | 0.122 | 0.75 | 0.09 | |
| Growth/competitive ability | Life form | Tree | 0.00576 | 0 | 0 |
| Allelopathic properties | Roots, leaves have moderate effect ([22]) | 0.00864 | 0.5 | 0.0043 | |
| Tolerates herbivore pressure | Roe deer browsing ([11], [37]) | 0.0456 | 0.5 | 0.0228 | |
| Normal growth rate | Moderately rapid growth. One of the fastest growing trees in Europe ([8]) |
0.018432 | 0.75 | 0.0138 | |
| Stress tolerances | Tolerates frost, fire | 0.01776 | 0.5 | 0.0089 | |
| Reproduction | Reproductive system | Reproduces by seeds | 0.005593 | 0.25 | 0.0014 |
| Propagule production | Propagule density (2064 ha-1) | 0.05474 | 1 | 0.054 | |
| Seed longevity | Seeds can be stored for 3 years at -3°C with 24-32% mc ([32], [33]) | 0.030464 | 0.25 | 0.0076 | |
| Reproductive period | Trees are long lived (350-400 years); tree produces new growth each spring | 0.012019 | 1 | 0.012 | |
| Time to reproductive maturity | Plants start flowering at 20 years of age ([23]) | 0.016184 | 0 | 0 | |
| Dispersal | Number of mechanisms | Propagules spread by wind, animals | 0.094572 | 0.5 | 0.0472 |
| How far do propagules disperse | Propagules will disperse several hundred metres | 0.189428 | 0.5 | 0.0947 | |
| Invasiveness Score | (max. = 1, min. = 0) | - | Σ | 0.6426 | |
Invasiveness can be defined as the ability to establish, reproduce and disperse within an ecosystem. To assess a plant for both invasiveness and impact, information from a variety of sources including the scientific literature, databases, online information, journal articles and other sources was collected. Such information has been then grouped under several “criteria” that are listed in Tab. 2. For each criterion, an “intensity” value was assigned based on the following ratings: high (1.0); medium-high (0.75); medium (0.5); medium-low (0.25); and low (0.0). Wherever the information on a specific criterion was lacking, rating was set to 0.5 (medium). The scored ratings were then tallied and weighting for each criterion calculated to obtain an “invasiveness score” (IS) for maple (eqn. 1):
where W g and W c are the weightings calculated for groups and criteria, respectively, and R i is the intensity rating value based on the scale reported above. The closer the invasiveness score is to 1, the more invasive the plant is.
Current and potential distributions are other major components required in the AHP. Current distribution of A. pseudoplatanus was derived from the Lithuanian Forest Inventory database, while its potential distribution was inferred based on the regional scientific literature (see references in Tab. 2). The ratio of present to potential distribution (PPD - range: 0-1) provides an indication of the stage the spreading of sycamore maple has currently reached.
Group and criteria ratings used to evaluate the species’ impacts are listed in Tab. 3. To obtain an “impact score” (I), the intensity rating scores were multiplied by the overall weighting for each criterion and summed with all the other criteria scores, obtaining a value ranging between 0 and 1.
Tab. 3 - Intensity ratings for the ratio of present to potential distribution of sycamore maple.
| Rating | Weight | Regional Rating |
|---|---|---|
| Very high | 1 | Infestations that can be eradicated with no chance of reinvasion from outside the area of control |
| High | 0.85 | Infestation(s) that can be eradicated with some chance of reinvasion |
| Medium high | 0.71 | Several small infestations beyond eradication |
| Medium | 0.57 | A large partially dispersed infestation or a few widely scattered small infestations |
| Medium low | 0.42 | Numerous large, dispersed infestations or many scattered, small infestations |
| Low | 0.28 | The majority of region is infested with some large areas still “clean” (more “clean” areas than infested) |
| Very low | 0.14 | The majority of region is infested with some small areas still “clean” (less “clean” areas than infested) |
| Extremely low | 0 | Reached full potential, but may increase in density within the infested area |
Finally, a Pest Plant Score (PPS) was obtained as follows (eqn. 2):
where IS is the invasiveness score, PPD is the ratio present-to-potential distribution, I is the impact and α, β and γ are weightings for the subcomponents drawn from the literature ([38]).
Results
Seedling abundance and spread intensity
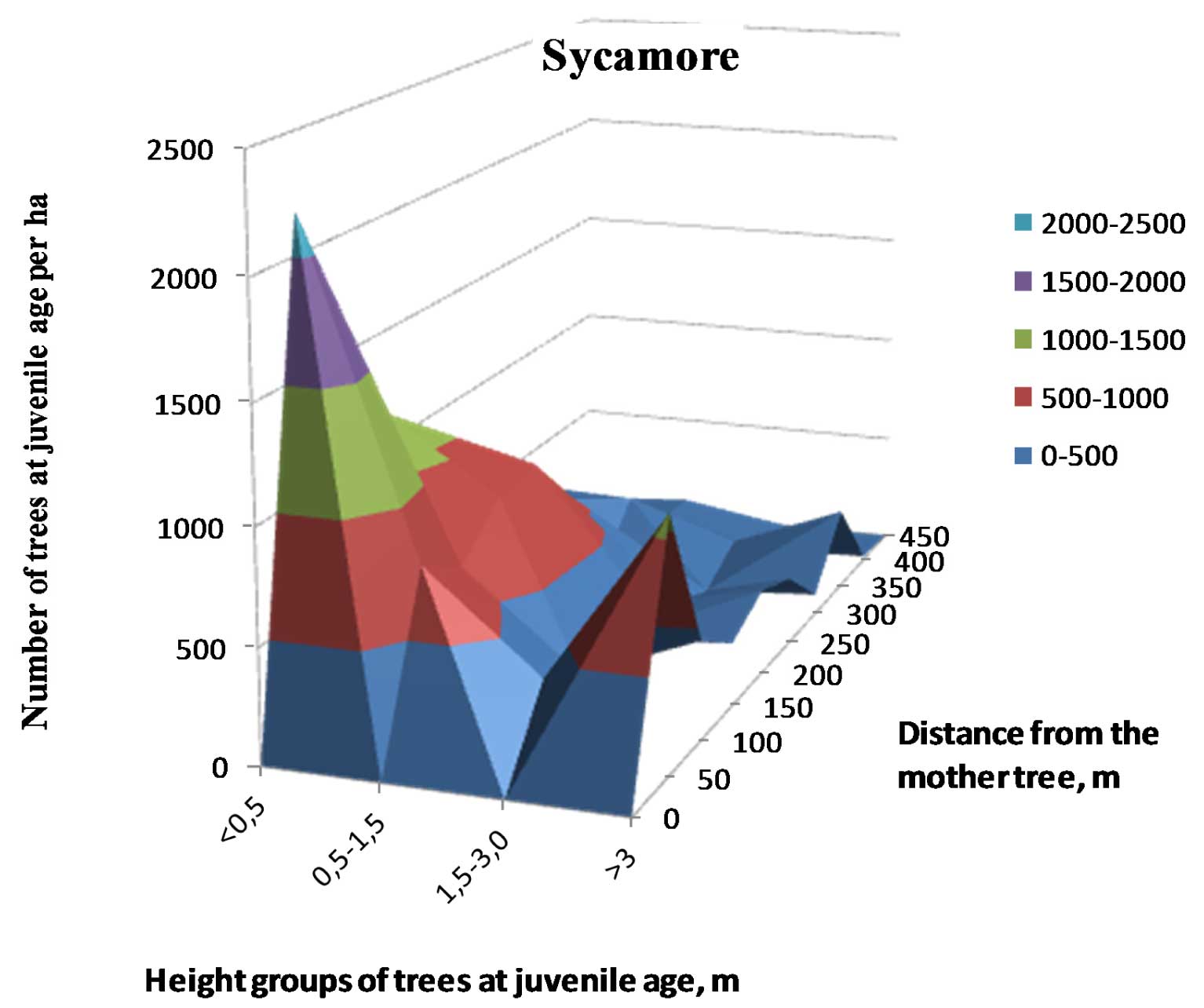
The highest sycamore maple spread density was observed in blocks dominated by birch, pine, maple, and lime. The mean number of seedlings per hectare over all study sites was 2064 ± 419 (Fig. 1), the maximum number observed was 3446 seedlings per hectare, and the minimum was 492.
Fig. 1 - A. pseudoplatanus seedling density as a function of the distance from the likely parent tree(s) by seedling height group. Density per hectare was pooled over 50m distance intervals from the source.
A significantly higher percentage of sycamore maple seedlings was detected in the first two height classes (≤0.5 m and 0.5-1.5 m) of the understory on fertile sites (p = 0.0175 and p = 0.0006, respectively). The P-values from Kolmogorov-Smirnov test for differences among the six sites for seedling height classes and densities are presented in Tab. 4. The estimates clearly show that the smallest (≤0.5 m) and the largest (>3 m) seedling classes significantly differed among sites. Mantel’s test using the total seedling quantity and the distance among sites resulted in a positive though not significant correlation (r = 0.56, p > 0.05). However, using the smallest class seedling quantity a significant result was obtained (r = 0.91, p = 0.0263), while no significant correlations were detected by the Mantel’s test carried out the largest class data and the distance among sites (r = 0.21, p > 0.05).
Tab. 4 - P-values of the Kolmogorov-Smirnov test for differences in seedling height classes and densities among the six sites analyzed. The P-values for the first (upper row) and second (lower row) classes are reported above the diagonal, while the third (upper row) and fouth (lower row) classes are reported below the diagonal.
| Site No. | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| 1 | - | 0.2372 0.0591 |
0.1630 0.6379 |
<.0001 0.8001 |
0.0068 0.3359 |
0.0056 0.0643 |
| 2 | 0.4143 0.4143 |
- | 0.6917 0.2377 |
0.0004 0.1411 |
0.0347 0.0963 |
0.2109 0.1203 |
| 3 | 0.1126 <.0001 |
0.2011 <.0001 |
- | 0.0036 0.8107 |
0.1147 0.8208 |
0.3033 0.4507 |
| 4 | <.0001 0.0387 |
<.0001 0.0003 |
0.2001 0.0267 |
- | 0.8591 0.9582 |
0.0218 0.0851 |
| 5 | 0.1458 0.0001 |
0.2562 <.0001 |
0.9577 0.5671 |
0.1258 0.0477 |
- | 0.2985 0.1250 |
| 6 | 0.9954 <.0001 |
0.4392 <.0001 |
0.3643 0.7112 |
0.0083 0.0189 |
0.8331 0.9997 |
- |
Pearson’s correlation coefficient between the underbrush cover and the quantity of sycamore maple seedlings was not statistically significant (p > 0.05). Nonetheless, it just revealed the tendency of a negative effect/relationship of underbrush cover on the quantity of seedlings in the first two height classes, as well as a positive effect on the fourth class (the largest seedlings). Underbrush cover was divided into two groups, the first including all the values below 50% and the other including all the values above such threshold. Differences in the quantity of seedlings in each height class were then tested using the Kolmogorov-Smirnov test, revealing significant differences in all but the third height class (1.5-3 m).
The age of the parent tree was positively correlated with the density of the largest sycamore maple understorey seedlings (r = 0.83, p < 0.0001) and with the total quantity of seedlings (r = 0.51, p = 0.0354). The upper canopy layer density had positive effect on the quantities of smallest seedling group (r = 0.87, p = 0.0026) and negative on the largest group (r = -0.77, p = 0.0154). Mean seedling spread distance from the parent tree was 257 m.
Sycamore maple invasiveness
The invasiveness score obtained for sycamore maple in this study was 0.6426 (Tab. 2). The highest ratings were recorded for propagule density and the reproduction period (rating=1.0). Medium-high ratings (0.75) were obtained by germination, growth rate, and establishment and disturbance requirements. In particular, the establishment features of the species mostly contribute (58%) to increase the invasiveness score for sycamore maple.
The intensity ratings for the ratio of present to potential distribution are shown in Tab. 3. Based on data evaluation, a medium intensity rating was obtained (weight = 0.57). Indeed, sycamore maple in Lithuania has been primarily recorded along the roadsides, in the riparian corridors and near the few parent tree plantations (Fig. 2).
Criteria ratings for determining the social, environmental and economic impacts of sycamore maple are shown in Tab. 5. Based on such ratings, the impact score obtained for sycamore maple was very low (0.1682).
Tab. 5 - Group and criteria ratings for determining social, environmental and economic impacts.
| Group | No | Criteria | Comments | Rating | Impact |
|---|---|---|---|---|---|
| SOCIAL (Tourism, Visual aesthetics, Cultural sites) |
1 | To what extent does the weed restrict human access? | Would not hinder human access | 0 | 0 |
| 2 | To what level does this weed reduce the tourism/aesthetics/recreational use of the land? | Sycamore not obvious to the average visitor | 0 | 0 | |
| 3 | To what level is the plant injurious, toxic or the spines affect people? | Not toxic and no spines | 0 | 0 | |
| 4 | How much damage is done to indigenous or European cultural sites? | No damage | 0 | 0 | |
| NATURAL RESOURCES (soil, water and processes) |
5 | To what extent does this weed impact water flow within watercourses or water bodies? | No effect | 0 | 0 |
| 6 | To what extent does the weed impact water quality (i.e., dissolved O2, water temperature)? | No impact | 0 | 0 | |
| 7 | To what extent does the weed increase soil erosion? | No increase | 0 | 0 | |
| 8 | To what extent does this weed reduce the biomass of the community? (nb. biomass acting as a carbon sink). | Biomass may increase in forest gaps | 0 | 0 | |
| 9 | To what extent does the weed change the frequency or intensity of fires? | Can suppress fire in pine forests | 0 | 0 | |
| Fauna and flora / vegetation and EVCs |
10 | To what extent does this weed impact the vegetation (composition on the following): | - | - | - |
| - | a. High value EVCs | Minor displacement | 0.25 | 0.0205 | |
| - | b. Medium value EVCs | Changes some dominant spp. | 0.5 | 0.0240 | |
| - | c. Low value EVCs | Do not grow | 0 | 0 | |
| 11 | To what extent does this weed affect the structure of a vegetation community? | After long period, can affect trees, shrubs and ground cover | 0.75 | 0.0518 - |
|
| 12 | What effect does the weed have on threatened flora? | Data not available | 0.5 | 0.0304 | |
| Flora and fauna / fauna | 13 | What effect does the weed have on threatened fauna? | No threatened fauna affected | 0 | 0. |
| 14 | What effect does the weed have on non-threatened fauna spp.? | Alien moth eggs and larvae develop in leaves | 0 | 0 | |
| 15 | To what extent does this weed benefit or facilitate the establishment of indigenous fauna? | Provides some food and shelter for birds, mammals, insects | 0.75 | 0.0173 | |
| 16 | To what extent is the plant toxic or its burrs or spines affect indigenous fauna? | No effect | 0 | 0 | |
| FLORA AND FAUNA/ FAUNA /Pest Animal | 17 | To what extent does this weed provide a food source for pest animals? | Provides minimal food for pest animals | 0 | 0 |
| 18 | To what extent does this weed provide habitat/harbor for serious pests? | May provide harbor for rabbits and foxes throughout the year | 1 | 0.0168 | |
| AGRICULTURE (Quality, Quantity, Cost to Production, Effect on land use and value) |
19 | 1To what extent does this weed impact on the quantity or yield of agricultural produce? |
Little or negligible impact on quantity of yield | 0 | 0 |
| 20 | To what extent does the weed impact on agricultural quality? | Negligible impact on quality of yield | 0 | 0 | |
| 21 | To what extent does this weed affect land values? | None | 0 | 0 | |
| 22 | To what extent does this weed cause a change in land use priorities? |
No change | 0 | 0 | |
| 23 | To what extent does the presence of the weed increase the cost of harvesting? |
Little effect | 0 | 0 | |
| 24 | To what extent does this weed act as an alternative host or vector for diseases of agriculture? | Not a host | 0 | 0 | |
| Total Impact Score | - | Σ | 0.1682 | ||
In order to evaluate the relative importance of sycamore maple as invasive plant species in Lithuania, results from invasiveness (Tab. 2), distribution (Tab. 3) and impact (Tab. 5) assessments were than combined by calculating the Pest Plant Score (PPS - eqn. 3):
The obtained Pest Plant Score showed that, according to primary invasive parameters, the importance of sycamore maple as an invasive species was not high (0.3537), being lower than the average score (0.5).
Discussion and conclusion
Alien species are typically more invasive in regions climatically similar to their native environment. The northern limit for sycamore maple’s native geographic range is Poland ([4]), where climate is only slightly harsher than in Lithuania. All current climate change scenarios predict for the Baltic region an increase in temperature and rainfall by at least 15% ([18]), thus a shift northbound of the habitats suitable for sycamore maple is expected. Indeed, such species has the potential to colonize plant communities similar to those where it is currently found in Poland ([10]), such as Fraxino-Ulmetum, Pino-Quercetum, Tilio-Carpinetum and others. In the present study, the largest sycamore maple spread was observed in stands dominated by birch, pine, maple and lime, though it can act as a pioneer species colonizing meadows and abandoned pastures ([28]).
In this investigation the average distance of maple seedlings from likely parent trees exceeds 200 meters, though other studies reported that very few seedlings may be found more than 50 m apart from parent trees ([9]). The relatively high proportion of saplings up to 1.5 m in height suggests that a large part of the sycamore maple understorey is likely to reach the upper canopy layer. Small seedlings (< 50 cm) can survive for long periods (> 15 years) under dense canopies where the light intensity is as low as 1% of the full light ([14]). Such characteristics, along with the rapid juvenile growth, allow the sycamore maple to colonize neighboring stands with dense canopies, especially where the competition in the understorey layer is low ([13]).
Studies of regeneration of broadleaved species in northeastern France showed that 13 years after gap creation, the understorey species composition was dominated by Fagus sylvatica and Acer pseudoplatanus seedlings ([7]), with density for the latter species reaching 19 000 per hectare and the mean seedling height of 0.7 m. In our study sites, such high density were not reached since the scattered distribution of the A. pseudoplatanus parent trees, though the average height of seedlings was close to that reported by Collet et al. ([7]).
In our study sites, density of Norway maple (A. platanoides) or small-leaved lime (Tilia cordata) seedlings in the understorey locally reached that of sycamore maple, but with a lower mean seedling height. Moreover, their distance from parent tree was lower than that recorded for sycamore maple. Furthermore, later bud flushing of A. pseudoplatanus as compared to A. platanoides allows their seedlings to avoid spring frost ([6]).
In Lithuania, the highest invasiveness among forest tree species was reported for boxelder (Acer negundo L. - [35]), with a PPS of 0.464, followed by the red oak (Quercus rubra L. - [27]). As compared to such species, sycamore maple showed an invasiveness much lower (0.354). However, its impact on the local biocoenosis should not be underestimated. Indeed, litter decomposition rate after 12 months was 30.4% for red oak ([31]) and 43.5% for sycamore maple, suggesting a greater influence of the latter species on the microbial community.
In Denmark, the invasive capacity of sycamore maple has been widely exploited since the late 1960s to achieve reliable, rapid, and inexpensive establishment of a new generation of trees in canopy gaps following conifer wind throw ([16], [34]). In Lithuania, sycamore invasion has only been observed in recent years when it started to colonize the understorey of forest stands, determining a gradual changes in tree species composition.
High timber prices and rapid growth rates have made sycamore maple economically attractive. However, our results show that sycamore maple spreads rapidly from its planted locations. Therefore, a cautious approach should be taken when establishing new sycamore maple forest plantations.
References
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Gscholar
Authors’ Info
Authors’ Affiliation
Virgilijus Baliuckas
Aleksandras Stulginskis University, Faculty of Forest Science and Ecology, Institute of Forest Biology and Silviculture, Studentu 11, LT-53361 Akademija, Kaunas district (Lithuania)
Corresponding author
Paper Info
Citation
Straigyte L, Baliuckas V (2015). Spread intensity and invasiveness of sycamore maple (Acer pseudoplatanus L.) in Lithuanian forests. iForest 8: 693-699. - doi: 10.3832/ifor0763-007
Academic Editor
Renzo Motta
Paper history
Received: Sep 06, 2012
Accepted: Feb 20, 2015
First online: Mar 19, 2015
Publication Date: Oct 01, 2015
Publication Time: 0.90 months
Copyright Information
© SISEF - The Italian Society of Silviculture and Forest Ecology 2015
Open Access
This article is distributed under the terms of the Creative Commons Attribution-Non Commercial 4.0 International (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Web Metrics
Breakdown by View Type
Article Usage
Total Article Views: 52540
(from publication date up to now)
Breakdown by View Type
HTML Page Views: 44493
Abstract Page Views: 2801
PDF Downloads: 3706
Citation/Reference Downloads: 18
XML Downloads: 1522
Web Metrics
Days since publication: 3972
Overall contacts: 52540
Avg. contacts per week: 92.59
Article Citations
Article citations are based on data periodically collected from the Clarivate Web of Science web site
(last update: Mar 2025)
(No citations were found up to date. Please come back later)
Publication Metrics
by Dimensions ©
Articles citing this article
List of the papers citing this article based on CrossRef Cited-by.
Related Contents
iForest Similar Articles
Research Articles
Seasonal dynamics of Cryptostroma corticale conidial spread
vol. 18, pp. 79-86 (online: 17 April 2025)
Research Articles
Optimum light transmittance for seed germination and early seedling recruitment of Pinus koraiensis: implications for natural regeneration
vol. 8, pp. 853-859 (online: 22 May 2015)
Research Articles
Matching seedling size to planting conditions: interactive response with soil moisture
vol. 12, pp. 220-225 (online: 25 April 2019)
Technical Reports
Nursery practices increase seedling performance on nutrient-poor soils in Swietenia humilis
vol. 8, pp. 552-557 (online: 09 December 2014)
Research Articles
Light availability influences the invasion of Teline monspessulana (L.) K. Koch in a temperate fragmented forest in Central Chile
vol. 15, pp. 411-416 (online: 19 October 2022)
Research Articles
The spread of the non-native pine tortoise scale Toumeyella parvicornis (Hemiptera: Coccidae) in Europe: a major threat to Pinus pinea in Southern Italy
vol. 11, pp. 628-634 (online: 04 October 2018)
Research Articles
Tropical seedling performance under drought: a functional trait approach for species selection in restoration
vol. 19, pp. 9-17 (online: 10 January 2026)
Research Articles
Effects of cultural treatments, seedling type and morphological characteristics on survival and growth of wild cherry seedlings in Turkey
vol. 5, pp. 283-289 (online: 17 December 2012)
Short Communications
Upscaling the estimation of surface-fire rate of spread in maritime pine (Pinus pinaster Ait.) forest
vol. 7, pp. 123-125 (online: 13 January 2014)
Research Articles
Natural spread of Verticillium wilt as effective constraint on Ailanthus altissima invasion
vol. 18, pp. 391-398 (online: 22 December 2025)
iForest Database Search
Search By Author
Search By Keyword
Google Scholar Search
Citing Articles
Search By Author
Search By Keywords
PubMed Search
Search By Author
Search By Keyword